
Fig.1: Cercidiphyllum japonicum, "ideal short shoot"; every year only a single folious leaf of exactly the same size as the original long shoot leaf is formed. Theinvestment in branch biomass is the lowest in all species studied.
ARTÍCULO INVITADO
The adaptive value of shoot differentiation in deciduous trees and its evolutionary relevance
Veit M. Dörken1 & Thomas Stützel1, 2
1 Ruhr-University Bochum, D-44780 Bochum, Germany.
2 Corresponding author: email: thomas.stuetzel@rub.de
Summary: Pronounced long shoot/short shoot differentiation is typical for deciduous angiosperm trees. It also occurs in a number of gymnosperms and very few evergreen angiosperm trees. The study of 719 angiosperm tree species (602 deciduous and 117 evergreen species) demonstrated that the deciduous condition is nearly always associated with shoot differentiation. Detailed measurements in 38 angiosperms showed that the leaf area of an entire short shoot equals the leaf area of a single long shoot leaf of the same species and individual. In the few cases where the leaf area of the short shoot is slightly larger than that of a single long-shoot leaf, the short shoot leaves shade each other and the projection of the short shoot equals the area of a single long shoot leaf. Calculations of the stem biomass needed to expose a given assimilatory surface show two interesting aspects. First, the stem biomass (dry weight) to expose leaf surface is about 10 times less in short shoots than in long shoots. Second, this biomass in long shoots and short shoots appears to be species independent. Regarding shoot structure efficiency, leaf size and shape do not matter. Some evergreen species resemble in all parameters more to deciduous species than to typical evergreen species. Phytogeographical data as well as morphological data suggest that these atypical evergreen species are derived from deciduous ancestors. As measured parameters differ markedly between all gymnosperms, except Ginkgo, and angiosperms, we suppose that the evolutionary pathway leading to shoot differentiation was different for gymnosperms and angiosperms.
Key words: Shoot differentiation; Deciduous trees; Angiosperms; Gymnosperms; Evolution; Phytogeography.
Resumen: Valor adaptativo de la diferenciación de brotes en árboles deciduos y su relevancia evolutiva. En Angiospermas arbóreas deciduas, es común encontrar un alto grado de diferenciación entre brotes largos y brotes cortos. También se presenta esta característica en un número de gimnospermas y en muy pocas angiospermas arbóreas siempreverdes. El estudio de 719 especies de angiospermas arbóreas (602 deciduas y 117 siempreverdes) demostró que la condición decidua está casi siempre asociada a la diferenciación de los brotes. Mediciones detalladas en 38 angiospermas demostraron que la totalidad del área foliar de un brote corto es semejante al área foliar de una hoja de un brote largo de la misma especie y del mismo individuo. En los pocos casos en que el área foliar del brote corto es levemente mayor que el área de una hoja de un brote largo, las hojas del brote corto se sombrean entre sí de manera que el área proyectada del brote corto se asemeja a la de la hoja del brote largo. Cálculos de la biomasa de tallo necesaria para soportar una determinada superficie asimilatoria mostraron dos aspectos interesantes. Primero, que la biomasa de tallo (peso seco) de soporte del área foliar is alrededor de 10 veces menor en brotes cortos que en brotes largos. Segundo, que esta biomasa en brotes largos y brotes cortos parece ser independiente de la especie. En cuanto a la eficiencia estructural de los brotes, el tamano y la forma de las hojas no son relevantes. Algunas especies siempreverdes se asemejan en todos sus parámetros más a las especies deciduas que a especies siempreverdes típicas. Datos fitogeográficos así como datos morfológicos sugieren que estas especies siempreverdes atípicas derivaron de ancestros deciduos. Como todos los parámetros medidos difieren notablemente entre todas las gimnospermas, excepto Ginkgo, y las angiospermas, suponemos que el camino evolutivo que condujo a la diferenciación de brotes fue diferente en gimnospermas y angiospermas.
Palabras clave: Diferenciación del eje; Árboles caducifolios; Angiospermas; Gimnospermas; Evolución; Fitogeografía.
INTRODUCTION
Long shoot/short shoot
differentiation is common in deciduous angiosperm trees. In extreme cases (e.g.
Cercidiphyllum, Fig. 1), each short shoot forms
only a single folious leaf per year which equals the long shoot leaf in size
and shape. This leads to the idea, that deciduous trees need short shoots to
replace the leaves of the previous year in the same position without the need
to invest in new stem tissue. Evergreen trees do not need this and a pronounced
shoot differentiation should be lacking there. If the idea is true, the occational
occurrence of evergreen trees with prominent shoot differentiation could probably
be regarded as an indicator for a deciduous ancestry in such species. Looking
at the recent distribution of deciduous and evergreen forests in the world,
one might assume that deciduous gymnosperms should have been more frequent prior
to the evolution of angiosperms than they are today. One might wonder whether
pronounced shoot differentiation in evergreen gymnosperms could be regarded
as an indicator for a descent from deciduous ancestors. Such an assumption is
in conflict with the present interpretation of deciduousness in gymnosperms
regarding the deciduous species as derived from evergreen ancestors (Coulter
& Chamberlain, 1982). This is also the case for angiosperms, in which deciduousness
is generally regarded as a derived feature (Axelrod, 1966; Wolfe, 1987) too.
It should however be taken into account that the arguments about primitive and
derived characters in these publications are basically morphological or typolgical.
This approach tries to interpret the rare character as derived from the frequent
character. The term "derived" is thus frequently used by morphologists with
a different meaning than in phylogenetics.
One difficulty for such an analysis is that within gymnosperms transition series
which could lead to an understanding of the evolutionary pathway of this character
can hardly be found. Within a taxon the same state is displayed and others show
the extreme opposite without any intermediates that could help towards an evolutionary
understanding (Bresinsky et al., 2008; Dallimore & Jackson ,1966;
Farjon, 1984; Kramer & Green, 1990; Krüssmann, 1983; Rehder, 1967). Many
extant gymnosperms are isolated relicts which can hardly be studied in the context
of character evolution (Jagels &Equiza, 2005; LePage et al., 2005;
Meyer, 2005; Momohara, 2005; Richter & LePage, 2005; Parakash & Kumar,
2004; Royer et al., 2003). In a first set of investigations it was therefore
tested whether shoot differentiation is really linked to the deciduous condition
and absent from evergreen trees. Then we focused on genera with evergreen and
deciduous species such as Acer, Quercus, Nothofagus and
Prunus. In a third step we checked whether the patterns in gymnosperms
coincide with those found in angiosperms.

Fig.1: Cercidiphyllum japonicum, "ideal short shoot"; every
year only a single folious leaf of exactly the same size as the original long
shoot leaf is formed. Theinvestment in branch biomass is the lowest in all species
studied.
MATERIALS AND METHODS
For the measurements, 5
year-old mature branches (Fig. 2) were cut out from the
crown of freely exposed trees in the botanical garden of the Ruhr University
Bochum (51o27´north, 7o5´east, mean temperature 10.2oC, 810 mm annual precipitation,
altitude 87-127 m). From these 5 year-old branches all current year branches
were cut off and measured in length. Frequency length curves were drawn for
all species to analyze the degree and type of shoot differentiation. Long shoot/short
shoot systems were considered, when the curve showed two distinct maxima (extreme
values), a not differentiated branching system was considered if the curve showed
only a single maximum.
All frequency length curves represent measurements from single branches. As
a botanic garden does not offer homogenous stands of many individuals of one
species, we could not pool the data from different individuals. The curves obtained
from different individuals of the same species were basically identical, but
the curve and thus also the gap between long shoots and short shoots was slightly
compressed or streched in x-direction. Thus pooling made the gap disaperaring,
dispite the fact that it was obvious and prominent in all individual measurements.
A further problem arose from the fact that we have to assign the measured shoots
to size classes to draw the curves. In comparison to long shoots, the length
variation in short shoots is very small. Using large intervals, we would not
be able to detect the variability in short shoots. Using small intervals, it
may happen that a certain size class is lacking in the measurements. This can
give the (wrong) impression of several maxima in the long shoot part of length
frequency curves. The problem could be overcome by using different intervals
or size classes for long shoots and short shoots. We decided not to use this
approach, as it may introduce prejudices in an understandable and thus not reproducable
way.
Then the leaf area of 50 long shoot leaves was measured. Long shoot leaves were
selected at random from the entire 5 year old branch. For this, the leaves were
first scanned with a standard scanner, and then the total leaf area was calculated
using the tools supplied by the analySISR software package. The same was done
with 50 short shoot leaves. In this case entire, randomly selected short shoots
were used. The arithmetic mean of the value for the short shoot leaves together
with the mean value of the number of leaves within a short shoot gives the mean
surface area of an entire short shoot. This analysis was done for a total of
38 angiosperm species, 25 of them deciduous and 13 evergreen, and 18 gymnosperms,
5 of them deciduous and 13 evergreen. The full list of all species studied is
available in Dörken (2008). Here we focus on species with typical shoot differentiation
and those species which could be regarded as misfits within their group or in
respect to our hypothesis. The bulk of further data confirming our hypothesis
has been omitted here as it supplies no additional information.
In a more detailed second analysis, the present year units of short shoots as
well as long shoots axis (without leaves) where dried in a drying oven to constant
weight to measure the dry weight. We have related the leaf area of all leaves
of a present year growth unit to the dry weight of the axis on which they are
inserted (including basal parts with cataphylls) to elucidate whether the formation
of short shoots can really reduce the investment in stem per leaf area compared
to undifferentiated shoot systems. A total of 38 angiosperms and 18 gymnosperms
(tab. 1) where analyzed in the same way.
Shoot abscission structures were analysed by SEM after fixation in FAA, dehydration
in FDA (Gerstenberger & Leins, 1978) and critical point drying, and from
serial sections using classical paraffin technique and subsequent safranin/astrablue
staining (Gerlach, 1984). Abscission structures where studied for the species
listed in tab. 2. Photographs were taken with a NIKON COOLPIX
995 in the field, with a dissection microscope ZEISS SV 11 and with a light
microscope ZEISS AXIOPLAN. Both microscopes were supplied with an OLYMPUS ColourView
IIR camera. As record system we used the analySISR software version 3.2 build
776. Multiple image alignment was done with the same software package.
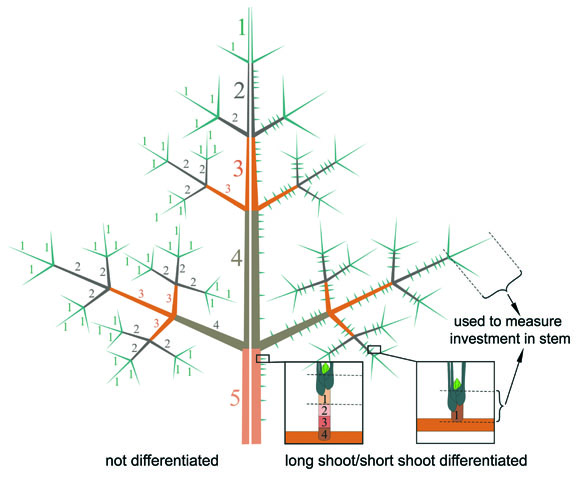
Fig 2: Not differentiated (left side) and differentiated shoot system
(right side) to measure the investment in stem biomass; only the present year
units of long shoots and short shoots have been used; numbers indicate the age
of the shoot unit; 1= present year.
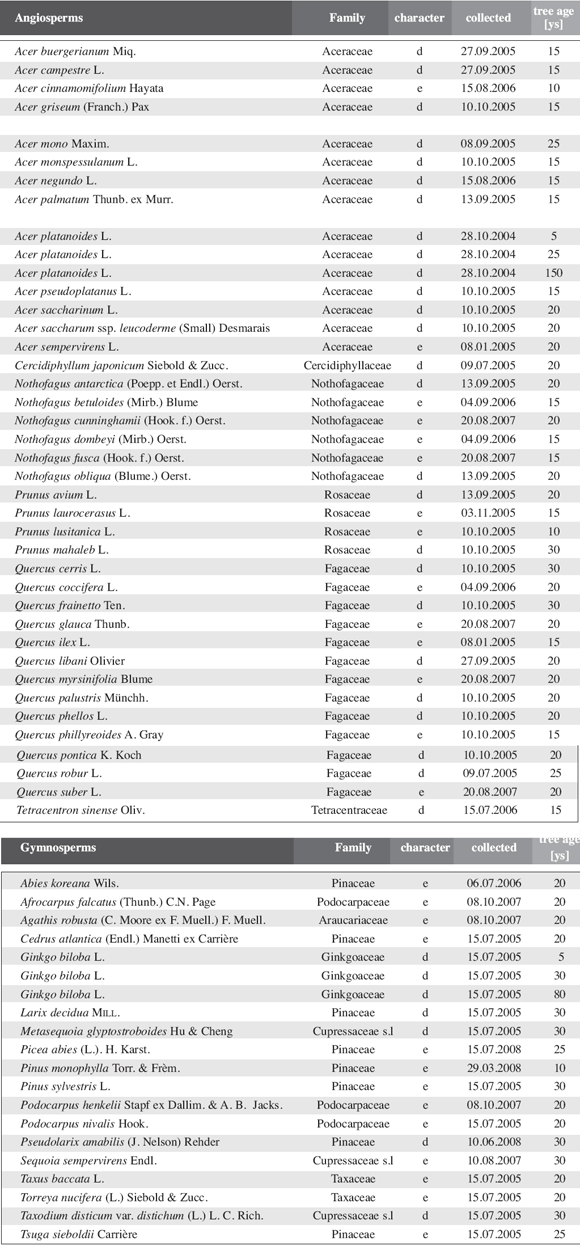
Table
1: Leaf area and dry weight of current-year growth units. For Acer
platanoides and Ginkgo biloba three individuals of markedly different
ages were used to estimate age effects. Tree age was estimated as exact data
were not available in most cases.
Table
2: Gymnosperm species for which the abscission of leaves and shoots
was studied using a dissecting microscope, by SEM or serial sectioning.
RESULTS
Length frequency diagrams
for a representative set of species are shown in Fig. 3
for deciduous species and in Fig. 4 for evergreen species.
Typically, deciduous species have differentiated shoots, with a marked short
shoot peak comprising about 80 to 90% of all shoots of the present year (Fig.
3a,b). The long shoots are much less and separated from the short shoot
peak by a pronounced gap (Fig. 3a-d). Only in some species
there is no clearly delimited length gap between short shoots and long shoots
(Fig. 3e,f). In Metasequoia glyptostroboides the
curve shows about the pattern for evergreen species gradually rising to a maximum
and then going down again. The maximum is slightly shifted to the lower end
of the curve, but is not at the lower end of the range (Fig.
3c). Nothofagus obliqua (Fig. 3f) shows more
or less the typical short shoot peak, although the short shoot peak is not placed
at the low end of the length scale and there is no distinct gap in the curve.
In typical evergreen species (Fig. 4), the length distribution
curve has only a single maximum. The peak value for the single peak in undifferentiated
systems (Fig. 4a-d) reaches about the value for long shoots
in differentiated systems and is about one dimension lower than for the short
shoot peak in differentiated systems. This single peak is placed about in the
middle of the curve if it is well developed (Fig. 4a). In
many cases there is however a broad range of values without a prominent maximum
(Fig. 4d). Sequoia sempervirens (Fig.
4c) does not really fit among the evergreen species, since it has a slight
gap in the curve. For the shorter shoots the curve is lower than for typical
deciduous species with shoot differentiation and the maximum is not at the low
end of the range. Nothofagus dombeyi (Fig. 4f)
shows a curve that resembles much more the curve for deciduous species with
a shoot length frequency curve with two peaks (Fig. 3a,b,d,e)
However, the gap is smaller, the mean length of the short shoots is longer and
there is more variation in length of short shoots than in typical deciduous
species. Evergreen species with curves similar to those for typical deciduous
species are Cedrus atlantica (Fig. 5a) and Pinus
sylvestris (Fig. 5b).
For the deciduous angiosperms species listed in (Table 3)
the leaf area was measured. The leaf area of a single long shoot leaf was then
compared to the leaf area of an entire short shoot. While the ratio between
the area of a long shoot leaf and the leaf area of an entire short shoot is
relatively constant and close to 1, exceptions are rare and range from 0.4 to
2.5. The absolute leaf areas vary considerably even within a species. These
correlations are illustrated in Fig. 6, showing that short
shoot leaves are considerably smaller and have petioles of a length that avoids
shading within a short shoot.
The length frequency curves also depend from the age of the tree (Fig.
7). This is illustrated for Ginkgo biloba (Fig.
7d-f) and Acer platanoides (Fig. 7a-c) for
a juvenile, a young but mature tree and an old tree that has reached about its
maximum height for the growth site. The absolute length of the long shoots decreases
with age and old individuals produce more or less exclusively short shoots.
The dry weight of annual growth units per leaf and leaf area is given for undifferentiated
shoot systems in Table 4. The stem biomass is not related
to the leaf weight but to the leaf area supported by the branch.
A similar calculation is presented in Table 5 for a differentiated
shoot system. The cost in biomass per cm2 leaf area is for long shoots about
the same as for the shoots of an undifferentiated shoot system. For short shoots
the cost is only between 10 and 40% (mean 17,1%) of the cost measured for long
shoots.
In respect to the relative cost in biomass per leaf area for long and short
shoots two evergreen species (Acer sempervirens and Nothofagus
dombeyi) coincide well with the bulk of deciduous species with differentiated
shoot system. This becomes even more evident comparing Nothofagus dombeyi
with other evergreen and deciduous species of Nothofagus such
as N. antarctica (Fig. 8a), N. betuloides
(Fig. 8b), N. cunninghamii (Fig.
8c)and N. fusca (Fig. 8d).
This relative costs is more or less constant for typical differentiated shoot
systems and does not depend on the size or shape of the leaves. It does not
matter in this respect whether the leaves are rather big (Acer platanoides)
or relatively small (Nothofagus antarctica) or whether they are simple
or compound (Acer negundo).
In evergreen gymnosperms without shoot differentiation, the relative cost in
shoot dry weight per shoot leaf area is very similar to the values found for
angiosperms (Table 5). In gymnosperms with shoot differentiation,
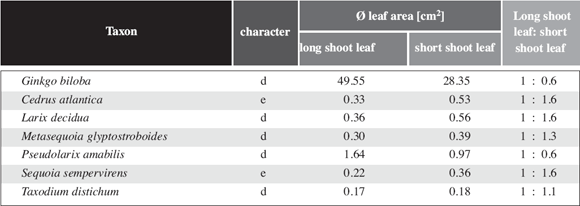
leaf size varies considerably less between long shoot leaves and short shoot
leaves than in angiosperms (Table 3). The same effect is
visualized in Fig. 6c,d. Except for Ginkgo biloba,
which is very "angiosperm like" in this respect, the leaf area of the entire
short shoot in gymnosperms with shoot differentiation is between 13.4 and 80.8
times more than the leaf area for a long shoot leaf (Table 3).
While in deciduous angiosperms any axil of a well developed long shoot leaf
bears a short shoot in the following year, in deciduous gymnosperms only a minor
part of the axils of long shoot leaves produces short shoots in the following
year. Nevertheless, in the latter group the surface area of a single short shoot
equals about the entire long shoot leaf area. Except for Ginkgo, gymnosperms
with differentiated shoots differ markedly from angiosperms with differentiated
shoots whether they are deciduous or not. This applies also for the amount of
biomass to be formed to expose a given leaf area (Table 5).
To some degree this is compensated by the fact that the number of long shoot
leaves exceeds by far the number of short shoots formed in the following year.
So in the end the relative cost per short shoot leaf area is only slightly less
than in angiosperms (see last column in Table 5).
In Metasequoia, Sequoia, Taxodium and Glyptostrobus
entire short shoots are abscised. While this occurs after one season in
three of these taxa, which are therefore deciduous, Sequoia keeps the
short shoots for more than one season and is therefore evergreen. In Sequoia
and probably also in Sequoiadendron there is however a pronounced
fall of short shoots in autumn and it needs heavy winds to blow the shed short
shoots from lower branches where they get often deposited after shedding. In
all four species short shoots are replaced from descendent serial accessory
buds (Fig. 9). In the following year the new short shoots
originate from the axils of basal bud scales of these accessory buds . In subsequent
years the renewals rise also from the axils of bud scales.
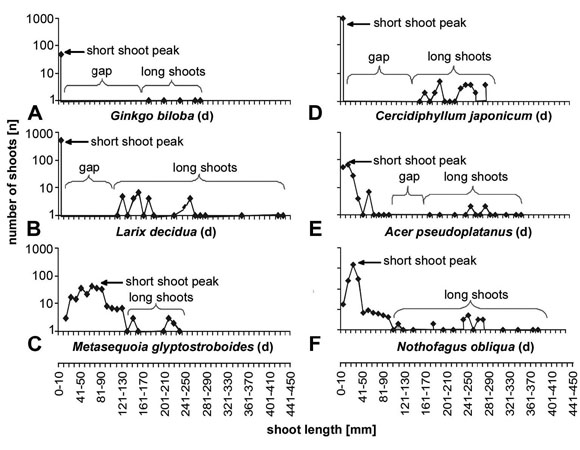
Fig. 3: Length/frequency curves for some deciduous species. Deciduous
species show generally a well developed shoot differentiation indicated by a
pronounced short shoot peak and a distinct more (fig. a,d) or less (fig. b,e)
prominent gap. The long shoots cover a rather broad range and do not display
a pronounced peak. Metasequoia differs from this pattern in having
a relatively uniform distribution curve with the maximum shifted to the lower
end of the curve. Nothofagus obliqua has a pronounced short shoot peak,
but lacks also the gap in the curve.

Fig. 4: Length frequency curves for some evergreen species. a, b, d,
e - typical evergreen species show a flat curve with a broad maximum near the
middle of the curve. Sometimes the maximum seems to be shifted to the low values
(fig.3c) but in the case of Taxus, this may be also an effect of crossings
with one of the numerous cultivars. c,f - the evergreen species Sequoia
sempervirens and Nothofagus dombeyi show curves that are very
similar to those of seasonal species.
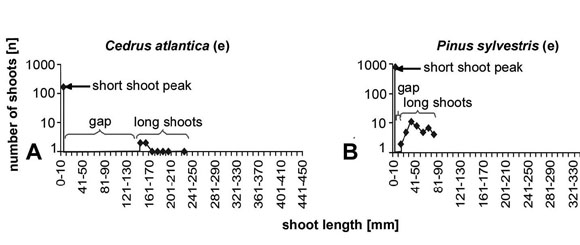
Fig. 5: The evergreen taxa Cedrus and Pinus show
length/frequency curves that are typical for seasonal angiosperms with a pronounced
short shoot peak and a gap between the short shoots and the long shoots.

Fig. 6: Long shoot leaf and entire short shoot in 1 angiosperm and
3 gymnosperm species with differentiated shoot system; a,b - the leaves of the
entire short shoot replace the long shoot leaf of the precious year; c, d -
the short shoot exceeds the leaf area of a long shoot leaf by about the factor
100.
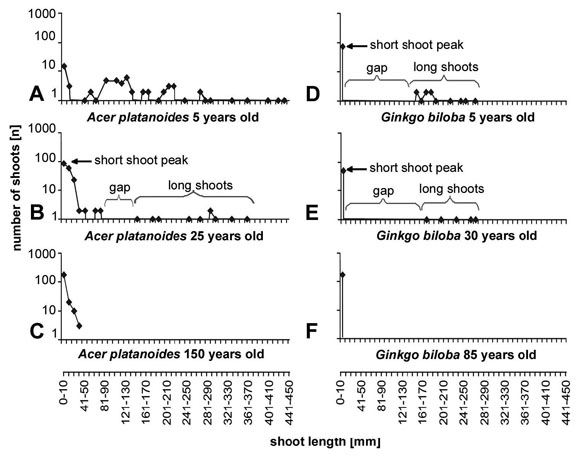
Fig. 7: Length frequency curves for tress of different age. The curves
illustrate that the shoot differentiation is most prominent in the juvenile
phase. In older stages the gap between long shoots and short shoots gets shorter
and finally disappears and the variation of the shoot length is reduced to about
what occurs in short shoots of young plants.
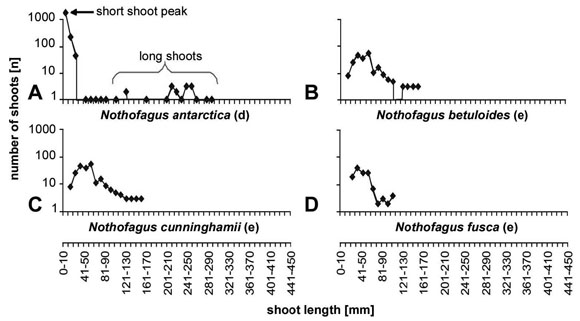
Fig. 8: Length frequency curves for different species of Nothofagus;
the deciduous N. antarctica is differentiated in long and
short shoots; in the evergreen species a shoot differentiation is lacking.
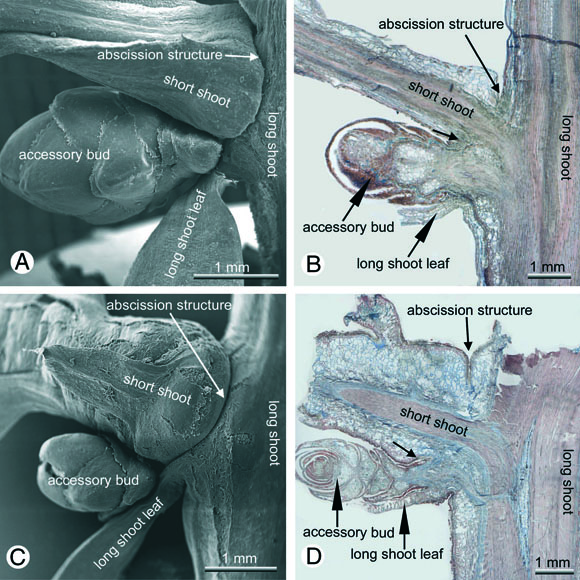
Fig. 9: Shoot abscission in Cupressaceae s.l.; a,b Metasequoia
glyptostroboides, c,d Sequoia sempervirens; from the accessory
bud the short shoot will be replaced after shedding.
Table
3: Leaf area of an individual leaf of one long shoot, leaf area of
an entire short shoot and ratio between these two leaf areas for evergreen (e)
and deciduous (d) species.

Table
4: Shoot biomass per leaf and per cm2 leaf area in some evergreen (e)
and deciduous (d) species with undifferentiated shoot system.
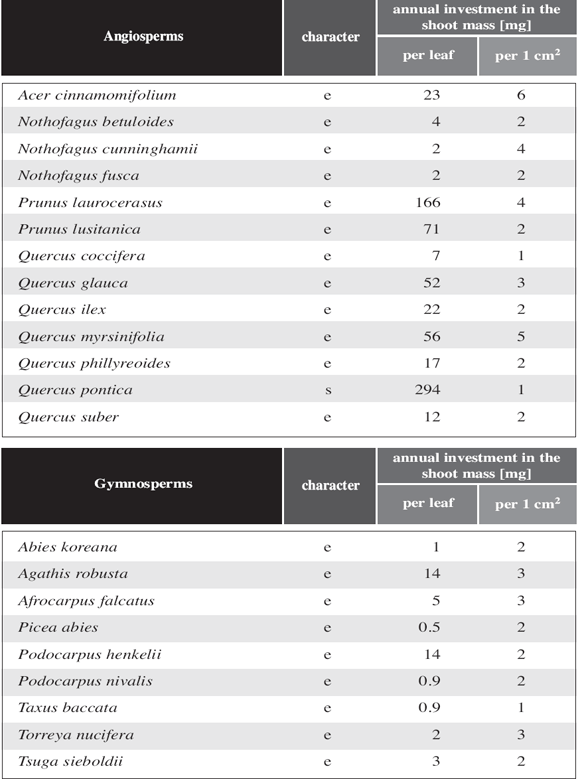
Table
5: Shoot biomass per leaf area for long shoots and relative cost per
leaf area for short shoots and cost of short shoots; evergreen (e) and deciduous
(d) species.
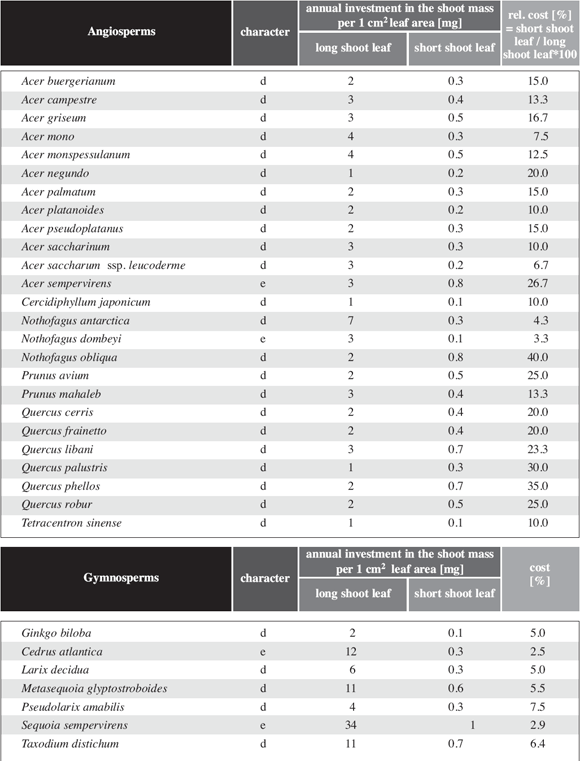
Table
6: Leaf area of long shoot leaves and short shoot leaves of some gymnosperms.
Unlike the angiosperms the leaf size is quasi identic.
DISCUSSION
There is some ambiguity
in the use of the terms long shoot and short shoots themselves. The length frequency
curve for a species with clear differentiation between long shoots and short
shoots is exemplified in Ginkgo biloba (Fig. 3a)
or Larix decidua (Fig. 3c) Both species show a
clear short shoot peak. As there are generally more than 10
times more short shoots than long shoots, the short shoot peak is very pronounced
and narrow while the long shoot maximum is much lower and this part of the curve
is much wider. In species with clear shoot differentiation, both peaks are separated
by a clear gap. In other species, the gap may be less prominent or even lacking,
it is however essential, that there are two peaks clearly separated at least
by a local minimum. This requirement is not met in Metasequoia glyptostroboides
(Fig. 3c) and only imperfectly so inNothofagus obliqua
(Fig. 3f). It seems that the terms long shoot and short
shoot in Metasequoia are not applied based on shoot length. Instead,
the persisting shoots that abscise individual needles are called long shoots
while those shoots that are abscised entirely together with the needles are
called short shoots. In Notofagus obliqua (Fig. 3f)
the terminology is applied according to the situation in other deciduous species
of Nothofagus that show a much more typical shoot differentiation.
In textbooks and diagnostic keys (e.g. Bresinsky et al., 2008) we find
however often phrases like"...has only long shoots..", in descriptions for Picea
and Abies. The contrasting character or lead in keys is in this case
"differentiated in long shoots and short shoots", e.g. for Pinus and
Larix. Looking at the length distribution curve for Abies (Fig.
4a) and Larix (Fig. 3b), one can state, that
the entire curve for Abies coincides nicely with the curve part for
long shoots in Larix in being relatively flat and broad and having
its maximum more or less in the middle instead of at the lower end. In this
case the use of the term long shoot in species without a shoot differentiation
is on a comparative basis as well. It is however an interspecific comparison
instead of the more common intraspecific comparison. The results of interspecific
comparisons depend on the species to be compared. The intraspecific comparison,
as for Larix, Cedrus and Pinus, is independent from
such effects.
For angiosperms two
results are especially surprising. First the entire short shoot replaces the
long shoot leaf rather precisely. It ranges from 1:0.5 to 1:1.5 (Table
3). Even the rare extreme values do not go farther than 1:0.4 and 1:2.5.
This is very low and covers about the variation that occurs within long shoot
leaves. The extreme values might be also an effect of the sometimes limited
availability of material. The other surprising fact is that the investment in
biomass per long shoot leaf area is about the same (ranging only from 1-7mg/cm2)
for all deciduous species studied. It appears that all species are equally well
adapted to the local situation of Bochum and that the size and shape of the
leaves has no evolutionary meaning in this respect. This should be tested by
carrying out similar studies in environments with markedly different climate.
Such studies should include at least some of the species studied here to guarantee
that the results are not affected by the selection of taxa under study. Nothofagus
in Patagonia or Tierra del Fuego might be a promising example. The invested
shoot biomass per short shoot leaf area ranges from 0.1-0.8mg/cm2.
Beogeographical data (e.g.
Romero, 1986; Tanai, 1986; Hill & Jordan, 1993; Swenson et al.,
2000; Hill, 2001; Knapp et al., 2005; Heads, 2006; Gibbs, 2006) might
indicate that Nothofagus dombeyi might be derived from a deciduous
ancestry. This would support our initial idea, that shoot differentiation in
evergreen species is generally an indicator of a deciduous ancestry. The ramification
patterns given for Nothofagus dombeyi by Puntieri et al. (2003)
fit with our findings. The author(s) study however also other species than we
did and with a different goal. A direct comparison of the data from natural
stands with our garden data was therefore not possible.The evergreen Acer
sempervirens fits into this scheme in a similar way. Spatial patterns as
well as the generell accepted taxonomic relationship suggests that the well
established shoot differentiation is the remainder of a deciduous ancestry in
this species too. In gymnosperms however the only species for which length frequency
curves as well as leaf surface and stem biomass data coincide with those for
angiosperms is Ginkgo biloba. All other gymnosperms with shoot differentiation
differ markedly from angiosperms in all these aspects.
It is therefore unlikely that the same evolutionary pathway or mode of directed
selection as in angiosperms led to differentiated shoot systems in gymnosperms.
At present we have no testable hypothesis for gymnosperms in this respect. To
understand the shoot morphology of Pinus, it might be helpful to study
the variability of shoot formation in deciduous and evergreen mature species
of Berberis. In this genus we find also long shoots with non-assimilatory
leaves (thorns) and sylleptic growth of short shoots. But while Pinus does
not show any variability that might help towards and evolutionary understanding
of this type of shoot differentiation, Berberis covers a wide range
of morphological types.
The fact that we could not prove our initial hypothesis, that in pre-angiosperm
times more gymnosperms have been deciduous and consequently recent gymnosperms
might be derived from a deciduous ancestry does not imply that this hypothesis
would necessarily be wrong. During our study we found deciduous Cupressaceae
such asMetasequoia, Taxodium and Glyptostrobus show
an abscission of entire short shoots which can be found in evergreen Cupressaceae
as well. In Sequoia sempervirens (Fig. 9c,d)
this abscission occurs together with the formation of serial accessory buds
as in the deciduous species, e.g. Metasequoia glyptostroboides (Fig.
9a,b). In all other genera and species of Cupressaceae studied (Dörken &
Stützel, in press), we found a morphologically similar abscission zone, but
abscission takes only place in an irregular mannor or even not at all. Such
a morphologically well performed but non-functional abscission structure can
probably only be understood as a reminder of a deciduous ancestry.
Therefore the idea of a more widespread deciduous ancestry in gymnosperms is
still of interest. It has however to be stated, that the morphometric data for
gymnosperms and angiosperms are so markedly different, that the adaptative pathway
to differentiated shoot systems has most likely been a different one in angiosperms
and gymnosperms, the latter one still being largely not understood.
ACKNOWLEDGEMENTS
We grateful acknowledge the support of the Botanic Gardens of Bochum, Bonn, Düsseldorf, and Dortmund, as well as the Palmengarten Frankfurt a.M. and many other Botanic Gardens. The numerous and detailed comments on an earlier version of the manuscript by an anonymous reviewer helped us to improve the manuscript significantly.
BIBLIOGRAPHY
1. AXELROD, D. I. 1966. Origin of deciduous and evergreen habits in temperate forests. Evolution 20: 1-15.
2. BRESINSKY A., C. KÖRNER, J. W. KADEREIT, G.NEUHAUS & U. SONNEWALD. 2008. Strasburger, Lehrbuch der Botanik, 36. Auflage. Spektrum, Akademischer Verlag Heidelberg.
3. COULTER, J. M. & C. J. CHAMBERLAIN. 1982. Morphology of Gymnosperms. Revised Edition. pp. 225-226. Central Book Depot, Allahabad.
4. DALLIMORE, W. &. A. B. JACKSON. 1966. A Handbook of Coniferae and Ginkgoaceae. 4th edition. Edward Arnold Publ., London.
5. DÖRKEN, V. 2008. Saisonalität und Langtrieb-/Kurztrieb-Differenzierung in Gymnospermen, ursprünglich oder abgeleitet? Dissertation zur Erlangung des Grades eines Doktors der Naturwissenschaften der Fakultät für Biologie und Biotechnologie der Ruhr-Universität Bochum. http://wwwbrs. ub.ruhr-uni-bochum.de/netahtml/HSS/Diss/Doerken VeitMartin/diss.pdf
6. DÖRKEN, V. & TH. STÜTZEL. In press. Seasonality in Cupresaceae, primitive or derived. Feddes Rep.
7. FARJON, A. 1984. Pines, Drawing and Descriptions of the Genus Pinus. E. J. Brill, Leiden.
8. GERLACH, D. 1984. Botanische Mikrotomtechnik, eine Einführung, 2. Aufl. Thieme, Stuttgart.
9. GERSTERBERGER, P. & P. LEINS. 1978. Rasterelektronenmikroskopische Untersuchungen an Blütenknospen von Physalis philadelphia (Solanaceae). Ber. Deutsch. Bot. Ges. 91: 381-387
10. GIBBS, G. 2006. Ghosts of Gondwana. Potton Publishing, Nelson, New Zealand.
11. HEADS, M. 2006. Panbiogeaography of Nothofagus (Nothofagaceae): Analysis of the main species. J. Biogeogr. 33: 1066-1075.
12. HILL, R. S. & G. J. JORDAN. 1993. The Evolutionary history of Nothofagus (Nothofagaceae). Austr. Syst. Bot. 6: 111-126.
13. HILL, R. S. 2001. Biogeography, evolution and palaeoecology of Nothofagus (Nothofagaceae): The contribution of the fosssil record. Austr. J. Bot. 49: 321-332
14. JAGELS, R. & M. A. EQUIZA. 2005. Competitive Advantages of Metasequoia in Warm Latitudes. In: LEPAGE, B. A., CH. J. WILLIAMS & H. YANG (eds.), The Geobiology and Ecology of Metasequoia, pp. 335-349. Springer, Berlin, Heidelberg.
15. KNAPP, M., K. STÖCKLER, D. HAVELL, F. DELSUC, F. SEBASTIANI & P. J. LOCKHART. 2005. Relaxed molecular clock provides evidence for long-distance dispersal of Nothofagus (southern beech). PLoS. Biol. 3: 38-43.
16. KRAMER, K. U. & P. S. GREEN. 1990. The Families and Genera of Vascular Plants, Pteridophytes andGymnosperms, edited by K. KUBITZKI. Springer Verlag, Heidelberg.
17. KRÜSSMANN, G. 1983. Handbuch der Nadelgehölze, 2. neubearbeitete Auflage. Verlag Paul Parey, Berlin und Hamburg.
18. LEPAGE, B. A., H. YANG & M. MATSUMOTO. 2005. The Evolution and Biogeographic history of Metasequoia. In: LEPAGE, B. A., CH. J. WILLIAMS & H. YANG (eds.),The Geobiology and Ecology of Metasequoia, pp. 3-114. Springer, Berlin, Heidelberg.
19. MEYER, H. W. 2005. Metasequoia in the Oligocene Bridge Creek Flora of Western North Amerika: Ecological Implications and the History of Research. In: LEPAGE, B. A., CH. J. WILLIAMS & H. YANG (eds.),The Geobiology and Ecology of Metasequoia, pp. 159-186. Springer, Berlin, Heidelberg.
20. MOMOHARA, A. 2005. Palaeoecology and History of Metasequoia in Japan, with Reference to its extinction and Survival in East Japan. In: LEPAGE, B. A., CH. J. WILLIAMS & H. YANG (eds.), The Geobiology and Ecology of Metasequoia, pp. 115-136. Springer, Berlin, Heidelberg.
21. PRAKASH, N. & M. KUMAR. 2004. Occurrence of Ginkgo Linn. in early cretaceous deposits of South Rewa Basin, Madhya Pradesh. Curr. Sci. 87: 1512-1515.
22. PUNTIERI, J. G., M. S. SOUZA, C. BRION, C. MAZZINI,& D. BARTHÉLÉMY. 2003. Axis differentiation in two south american Nothofagus species. Ann. Bot. 92: 589-599.
23. REHDER, A. 1967. Manual of cultivated trees and shrubs. 2nd edition. The Macmillian Company, New York.
24. RICHTER, S. L. & B. LEPAGE. 2005. A High-Resolution Palynological Analysis, Axel Heiberg Island, Canadian High Arctic. In: LEPAGE, B. A., CH. J. WILLIAMS & H. YANG (eds.), The Geobiology and Ecology of Metasequoia, pp. 137-158. Springer, Berlin, Heidelberg.
25. ROMERO, E. J. 1986. Fossil evidence regards the evolution of Nothofagus Blume. Ann. Missouri Bot. Gard. 73: 276-283.
26. ROYER, D. L., L. J. HICKEY & S. L. WING. 2003. Ecological conservatism in the "living fossil" Ginkgo. Palaeobiology 29: 84-104.
27. SVENSON, U., R. S. HILL, & S. MCLOUGHLIN. 2000. Ancestral area analysis of Nothofagus (Nothofagaceae) and its congruence with the fossil record. Austr. Syst. Bot. 13: 469-478.
28. TANAI, T. 1986. Phytogeographic and phylogenetic history of the genus Nothofagus BL. (Fagaceae) in the southern hemisphere. J. Fac. Sci. Hokkaido Univ., Ser. IV 21: 505-582.
29. WOLFE, J. A. 1987. Late Cretaceous-cenozoic history of deciduousness and the terminal Cretaceous event. Palaeobiology 13: 215-226.
Recibido el 8 de
abril de 2009
Aceptado el 21 de octubre de 2009.