KEY POINTS
Current knowledge
• The incidence of acute kidney injury (AKI) in adults hospitalized for SARS-CoV-2, the risk factors for AKI and the impact on morbidity and mortality, varies widely according to reports in different parts of the world.
Contribution of the article to current knowledge
• The incidence of AKI in adults hospitalized for SARS-CoV-2 was 19%.
• The independent risk factors for AKI were age, chronic kidney disease, mechanical ventilation and neutrophil count at admission (the latter not described in previous studies).
• Mortality in subjects with AKI was 47.1% and was also associated with other adverse outcomes.
The outbreak of severe acute respiratory syndrome (COVID-19) due to SARS-CoV-2 has quickly turned into a global pandemic with devastating consequences1,2. The first case reported in Córdoba, Argentina was on March 3, 20203. As of May 8, 2021, more than 230 000 infected people had been reported, and more than 3.1 million in Argentina4.
Patients with COVID-19 present an eminently respira tory affectation that can trigger in its most severe forms an acute respiratory distress syndrome (ARDS), although cardiac, hematological, digestive, neurological and renal affectation has also been described5-7. The incidence of acute kidney injury (AKI) in patients hospitalized for COVID-19 varies between 0.5% and 80.3% according to the different series5-12. Direct viral cytotoxicity in kidney cells and kidney damage secondary to hypoperfusion and hypoxia due to hemodynamic instability, heart and lung damage, damage mediated by pro-inflammatory cytokines such as thrombotic microangiopathy and rhabdomyolysis, among others, have been described as possible etiologies of AKI13-15. As in AKI due to other causes, AKI secondary to COVID-19 has been associated with adverse outcomes, including worsening of other comorbidities, greater use of health care resources, and higher mortality16. Cur rent reports of mortality among subjects hospitalized for COVID-19 with AKI vary widely across different cohorts, ranging from 7 to 100%12,17-19. Factors associated with AKI and its impact on patients hospitalized for COVID-19 have not yet been evaluated in our country. Local studies are needed to better understand the AKI associated with COVID-19 and thus optimize the management of this disease and its complications.
The objectives of the study were to evaluate the inci dence, risk factors, and impact of acute renal failure in hospitalized subjects due to active SARS-CoV-2 infection in two third level hospitals in Córdoba, Argentina.
Material and methods
A retrospective cohort study was conducted. Adults who were hospitalized due to active SARS-CoV-2 infection between March 3, 2020 and October 31, 2020 in the Hospital Privado Universitario de Córdoba and Hospital Raúl Angel Ferreyra in Cordoba city were consecutively included.
Patients hospitalized for reasons other than COVID-19 and those who were referred to another institution within the first 48 hours of admission were excluded.
An active SARS-CoV-2 infection was defined as confirmed diagnosis by any of the available methods (RT-PCR, antigen or serology for SARS-CoV-2) and admission to hospital within the first 21 days from the onset of symptoms or confirmation of the diagnosis (whichever occurred first).
Demographic data, comorbidities, clinical characteristics, and complementary studies carried out at hospital admission, as well as those related to the treatment and evolution of each patient, were obtained from the electronical medical record. Each case was followed up throughout the hospitalization.
AKI and AKI stages were defined according to The Kid ney Disease: Improving Global Outcomes (KDIGO) clinical practice guidelines for acute kidney injury20. The presence of comorbidities was defined according to the record in the clinical history that the patient had this pathology and / or presented its diagnostic characteristics, according to current definitions. Based on this, arterial hypertension was defined according to American College of Cardiology/American Heart Association (ACC/AHA)21, obesity as the presence of a body mass index ≥ 30 Kg/m2, 22 and diabetes mellitus according to the criteria of American Diabetes Association23. Chronic kidney disease was defined as a structural or functional abnormality of the kidney present for more than 3 months and determined by the reduction of the glomerular filtration rate below 60 ml/ min/1.73 m2, 24, ischemic heart disease as the inadequate supply of blood to the myocardium due to obstruction of the epicardial coronary arteries and chronic heart failure as the presence of compatible clinical symptoms, with objective evidence of heart failure and that specific treatment was indicated for it25. Regarding complications, pneumonia was defined according to current diagnostic criteria, based on symptoms and the pres ence of compatible pulmonary infiltrates in diagnostic imaging methods26, acute respiratory distress syndrome according to the Berlin criteria27 and bacterial superinfection was defined as respiratory co-infection during the course of the disease or hos pital stay28. Any patient with a SOFA score ≥ 2 who also have a vasopressor requirement and an elevated lactate > 2 mmol/l (>18 mg/dl) was considered that underwent a septic shock29.
The determination of creatinine was performed by us ing the kinetic Jaffé method (Roche Diagnostics, Sussex, United Kingdom) traceable to the IDMS reference method in a Modular P autoanalyzer (normal value-NV-: 0.6-1.2 mg/dl). The calibration of the test was performed with a commercial lyophilized calibrator for automated systems. Leukocytes (NV: 4.5-9.4 k/μl), neutrophils (NV: 35-70%), lymphocytes (NV: 20-43%) and platelets (NV: 150-350 k/μl) were measured by flow cytometry (XN-3000 autoanalyzer, SYSMEX, Japan); C-reactive protein (NV < 0.6 mg/dl) and ferritin (NV: 15-250 ng/ml in men and 10-150 in women) by immunoturbidimetry (COBAS 8000- c702 autoanalyzer, ROCHE, Germany); procalcitonin (NV<0.5 ng/ml) and ultrasensitive troponin T (NV < 14 pg/ml) by electrochemiluminescence (COBAS 8000- e801 autoana lyzer, ROCHE, Germany); and D-dimer (NV < 0.5 μg/ml) by enzyme linked fluorescent assay (Mini VIDAS autoanalyzer, BIOMÉRIEUX, France). All the determinations used partici pated in internal and external quality control programs (RIQAS, United Kingdom) and surpassed the recommended analytical quality specifications (acceptable total error).
According to the institutional action protocol and taking into account the clinical criteria, the availability of drugs and the scientific evidence at each moment, the therapeutic op tions were: prophylactic heparin with enoxaparin 40 mg/day during the entire hospitalization, dexamethasone 8 mg/day for 10 days (or its equivalent in hydrocortisone), convalescent plasma 1 dose/day of 200 ml for 2 days, hydroxychloroquine sulfate 200 mg/12 h for 10 days, Lopinavir 400 mg + Rito navir 100 mg/12 h for 10-14 days, nebulized ibuprofen 50 mg/8 h until the moment of clinical improvement or up to 28 days from the beginning of its administration, ruxolitinib 15 mg/day for 14 days, tocilizumab 4-8 mg/kg/day one dose and methylprednisolone 250 mg/day for 3 days. To receive off-label prescription drugs, all patients signed an informed consent. When there was suspicion or confirmation of respi ratory bacterial superinfection ceftriaxone 2 g/day combined with azithromycin 500 mg/day for at least 5 days were the first option. Other antibiotics were used according to the infectious focus and medical criteria.
For statistical analysis, continuous variables were ex pressed as mean and standard deviation or median (M) and 25-75 percentile interquartile range (IQR) and their comparison was made with Student’s t-test or Mann-Whitney according to the variable distribution. Categorical variables were expressed as absolute frequency (n) and relative frequency (%) and were compared with chi-square test or Fisher’s exact test, accord ing to the expected frequencies. To assess the risk factors for AKI (dependent variable 1) and if it was independently associated with death (dependent variable 2), logistic regres sion was used. In the first place, univariate analyzes were per formed considering as independent variables all demographic characteristics, comorbidities, initial clinical presentation and associated complications during follow-up, and then all the variables that reached statistical significance were included in multivariate models (independent variables), except those that presented more than 20% of missing data. Then the best models were chosen according to the adjusted R2 value and the Akaike information criterion. Goodness of fit was checked using the Hosmer Lemeshow test. To assess whether AKI was independently associated with mortality, we adjusted for the rest of the complications that were associated with mortality in the univariate analysis. Adjusted odd ratio (aOR) with its 95% confidence interval (95% CI) was used as association measures. All the tests were two-tailed and a p value less than 0.05 was considered statistically significant. Statistical analysis was performed with Stata 14 statistical program (Stata-Corp. LP., College Station, TX, USA).
The study was reviewed and approved by the research committee of the Hospital Privado Universitario de Córdoba who decided that informed consent was not necessary due to the observational and retrospective design of the study, the anonymity of the data and that it did not represent any risk for the participants.
Results
During the study period, 450 adults were hospitalized due to active SARS-CoV-2 infection. Two patients were referred to another institution during the first 48 hours and were excluded from the analysis (Fig. 1). A total of 448 patients were included, with a median age of 63 (IQR 53-75) years, 63.6% were men. Seventeen % had criteria for admission to the intensive care unit (ICU) at hospital admission. The most frequent comorbidities were hyper tension (55.1%), obesity (31.7%) and diabetes (28.1%) and the most frequent clinical manifestations were fever (74.8%) and respiratory symptoms such as cough and dyspnea (Table 1). Pneumonia was evidenced in 95.8% of the patients. The incidence of AKI was 19% (85/448). Of these, 36 (42.3%) presented AKI upon admission and 49 (57.7%) developed it during hospitalization. Stage 1 AKI was observed in 50.6%, stage 2 in 20% and stage 3 in 29.4%. In this last group, 18 subjects required renal replacement therapy (RRT). The incidence of AKI among patients who remained only in general ward throughout their hospitalization was 10.8% (31/286) and among subjects who required ICU at some point during their hospitalization was 33.3% (54/162).

Table 1 Baseline characteristics and clinical presentation at the time of hospital admission of the studied population
Regarding patients with AKI, a higher percentage had criteria for admission in ICU, were predominantly males and were older, compared to those who did not present AKI (control group). As for comorbidities, the AKI group had a higher percentage of chronic kidney disease (CKD), immunocompromise, and ischemic heart disease compared to the control group. In the admission laboratory tests, subjects with AKI had higher values of total leukocytes, neutrophils, higher percentage of throm bocytopenia, and higher levels of procalcitonin, troponin T and dimer D. All these characteristics were considered risk factors and variables significantly associated with AKI in the univariate analysis. There was also a clear trend for patients with AKI to have a higher percentage of arterial hypertension and chronic heart failure as comorbidities, lower lymphocyte count at admission, and higher levels of C-reactive protein and ferritin, but these differences were not statistically significant (Table 2). There were no differences in the presence of symptoms/signs upon admission and/or pneumonia between with and without AKI groups (94.1 vs. 96.1%; p = 0.378).

Table 2 Differences between baseline characteristics and clinical presentation at hospital admission between patients with and without acute kidney injury (AKI)
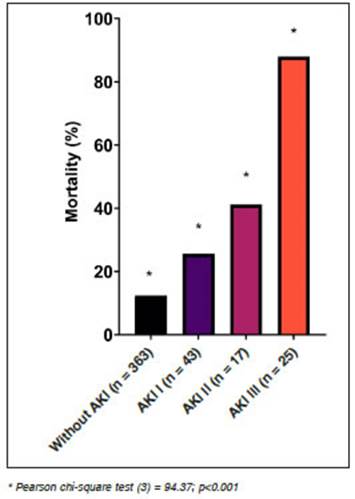
During hospitalization, 96.9% received prophylactic heparin, 85% dexamethasone or hydrocortisone, 57.6% antibiotics (47.1% azithromycin, 42.2% ceftriaxone), and 37.5% convalescent plasma. A smaller percentage of patients received: hydroxychloroquine (0.7%), ritonavir/ lopinavir (0.9%), nebulized ibuprofen (2.2%), and im munomodulatory drugs (ruxolitinib/tocilizumab, 1.1%). A higher percentage of subjects in the AKI group received piperacillin-tazobactam, Imipenem or meropenem, and convalescent plasma compared to the control group. The median length of hospital stay was 17 (IQR = 13-22) days. During follow-up, complications were bacterial su perinfection (12.3%), septic shock (8.8%), ARDS (27.9%), ICU admission (36.2%), and mechanical ventilation (MV) requirement (14.7%). Overall mortality was 19%. Patients with AKI presented a longer hospitalization, greater requirement and time of stay in the ICU, a significant positive association with bacterial superinfection, sepsis, ARDS, MV and also presented ~4-fold higher mortality compared to the control group (table 3). Mortality incidence varied according to the stage of AKI (Fig. 2). In subjects who required RRT, all were on MV and mortality was in 16 out of 18. The 2 survivors in this subgroup recovered their kidney function at the time of hospital discharge. The final multivariate model chosen to evaluate the risk factors for AKI included the variables sex, age, history of CKD, immunocompromised, neutrophil and platelet count, MV, septic shock and bacterial superinfection. The variables that were independently associated with AKI in this adjusted analysis were age (for every 10 years, aOR = 1.30, 95%CI = 1.04-1.63, p = 0.022), history of CKD (aOR= 9.92, 95%CI = 4.52-21.77, p < 0.001), blood neutrophil count (per 1000 cells increase, aOR = 1.09, 95%CI = 1.01-1.18, p = 0.037) and MV requirement (aOR = 6.69, 95%CI = 2.24-19.9, p = 0.001), (Figure 3). Among the complications found during follow-up, the only ones that were independently associated with mortality in the adjusted multivariate analysis were: AKI (aOR = 3.3, 95%CI = 1.6-6.9, p = 0.001), ARDS (aOR = 8.8, 95%CI = 3.8-20.5, p < 0.001) and MV requirements (aOR = 3.6, 95%CI = 1.2-10.4, p = 0.020).

Table 3 Medications received and associated complications in subjects with and without acute kidney injury (AKI)
Discussion
In the present study, the incidence, risk factors, and com plications associated with AKI were evaluated in subjects hospitalized in two high complexity third-level hospitals in Córdoba, Argentina.
In a meta-analysis that included more than 13000 pa tients in China, the United States and different European countries, most of them hospitalized, the incidence of AKI was 17% and approximately 5% required RRT30. These percentages were very similar to those found in our study (AKI 19%, RRT 4%) and the criteria to define AKI were exactly the same. In Argentina, a single study carried out in a public tertiary hospital in Buenos Aires used the same AKI criteria31. In that prospective cohort they included 417 COVID-19 patients admitted to the general ward. AKI oc curred in 4.8% of the patients, and 0.7% required RRT (only 3 patients, another 6 already required RRT prior to hospitalization). These percentages are much lower than those reported in our cohort and this may be mainly due to the fact that the subjects included were two decades younger, had fewer comorbidities such as hypertension (16.7 vs. 55.1%), diabetes (15.3 vs. 28.1%) and CKD (3.1 vs. 10.3%), the majority presented with a mild disease (86.8%) and a lower percentage of pneumonia (51.9% vs. 95.8%). In addition, patients with critical illness at admission were excluded in this study. Another series in Argentina reports an incidence of kidney injury of 7% but does not specify the criteria used to define AKI and both the baseline characteristics and the severity of the disease differ from that of the patients included in our cohort32. In a multicenter prospective cohort study in Argentina, which included 1909 adults with COVID-19 admitted to the ICU with invasive mechanical ventilation, the incidence of AKI (using the same definition as in our study) was 52.2%, RRT 19.5% and the presence of AKI was also one of the independent predictors of mortality33. In our series, the incidence of AKI among subjects who required ICU at some point during their hospitalization was lower (33.3%), but not all of these patients required invasive mechanical ventilation, and this was an independent risk factor for AKI demonstrated in our study. This wide range of incidence of AKI in subjects hospitalized for COVID-19 described in the literature (between 0.5 and 80.3%)5-12 could be explained by the following reasons: use of different defini tions of AKI, hospitalizations in isolation centers only for epidemiological precaution, use of different hospitalization criteria, presence of different demographic, clinical and severity characteristics in the subjects included in each study, use of different protocols for monitoring renal func tion and treatments to avoid renal hypoperfusion and the use of drugs nephrotoxic and different follow-up periods.
Currently described independent predictors of AKI in subjects with COVID-19 include age, male gender, history of obesity, diabetes, hypertension, cardiovascular disease, CKD, elevated IL-6 levels, baseline angiotensin-converting enzyme inhibitors or angiotensin II receptor blockers use, exposure vancomycin and non-steroidal anti-inflammatory drugs, septic shock, hypoxia, and MV8-10, 34,35. In Argentina, to our knowledge there are no publications on the risk factors for AKI in patients with COVID-19. In our study, in addition to age, history of CKD and MV, blood neutrophil count at admission was also identified as an independent predictor of AKI, a variable that had not been identified in previous studies that could be from great value to consider in the initial evaluation. Furthermore, in the univariate analysis male sex, immunocompromise, coronary heart disease, thrombocytopenia, and higher inflammatory markers such as procalcitonin, troponin, and dimer-D were also associated with AKI, although later in the mul tivariate adjustment lost statistical significance. And other comorbidities such as hypertension and heart failure, a lower count of blood lymphocytes, and other inflammatory markers such as C-reactive protein and ferritin, presented a clear trend of association with AKI, but without statistical significance. This may be due to the number of subjects included and statistical power.
On the other hand, patients with AKI were more fre quently treated with convalescent plasma and piperacillin tazobactam, imipenem or meropenem; these differences are probably explained by the more severe condition in this group of patients and antibiotic staging due to poor response to the initial antibiotic or established microbial resistance.
In the same meta-analysis mentioned previously, the mortality rate of patients with AKI was 52%30, a percentage similar to that found in our study (47.1%). In our cohort, as in others, AKI has been identified as an independent predictor of mortality9,33,34,36,37, and the higher the AKI stage, the greater the risk9. In addition, AKI in our series was associated with other complications such as long-term hospitalization, admission to ICU and prolonged stay in intensive care, among others.
The limitations of our study are mainly based on its retrospective nature, although in both institutions there was a protocol for action against COVID-19 which would tend to unify management. The restriction of follow-up during hospitalization could have partially underestimated the true incidence of AKI if there were patients with a short hospital stay who presented this complication after hospital discharge.
In conclusion, the determination of AKI risk factors allows the early identification of the susceptible subjects. The analysis of incidence, complications and the impact associated with AKI, such as mortality and prolongation of hospital stay, reflect the importance of early diagnosis and adequate treatment. This epidemiological analysis can be useful for planning and allocating resources in the event of a possible new outbreak.