INTRODUCTION
It is widely accepted that, with the exception of Amborella trichopoda Baill. (Amborellaceae), the order Nymphaeales is sister to all the remaining extant angiosperms, based on phylogenetic evidence from both plastid and nuclear DNA sequence data (Li et al., 2019; Yang et al., 2020; Zhang et al., 2020). Although not particularly species-rich (94 spp: Barbosa et al., 2018; Govaërts, 2021; Lima et al., 2021), Nymphaeales has a subcosmopolitan distribution, being widespread in lentic, or rarely lotic, freshwater habitats of all continents and regions, except the polar ones and, with few exceptions, most deserts and mountainous regions (Stevens, 2001; La-ongsri et al., 2009; Christenhusz et al., 2017). Of the three families of the order, morphologically aberrant Hydatellaceae (13 species: Govaërts, 2021) is restricted to India, Australia and New Zealand (Sokoloff et al., 2019). Nymphaeaceae, with 75 spp, is found in all continents except Antarctica, and its distribution basically corresponds to that of the order (Lima et al., 2021). Finally, Cabombaceae is native to all continents, except Antarctica and Europe, but outside the Americas its distribution seems to be restricted and localized. It only has seven accepted species (Ørgaard, 1991; Wiersema, 1997; Barbosa et al., 2018) in two genera: Cabomba Aubl. (6 species) and Brasenia Schreb. (1 species) are extant. The first genus is native to the lowlands of tropical and warm temperate regions of the Americas, from Ontario, Canada, to Río Negro, Argentina (Wiersema, 1997; Ospina & Zanotti, 2015), but C. caroliniana A. Gray has been introduced to and is considered an invasive species in other continents (particularly Asia, Australia, and Europe; CABI, 2020). Brasenia schreberi J.F. Gmel. on the other hand, has extant native populations on all continents except Europe (although there is fossil evidence of its presence there in the recent geologic past; Drzymulska, 2018) and Antarctica (Verdcourt, 1971; Cook et al., 1974; Wiersema, 1997; Fu & Wiersema, 2001; Stanley & Orchard, 2007). In the Americas this species has been reported from as far north as Alaska and Newfoundland (with populations on eastern and western North America separated by the Rocky Mountains and the Great Plains, Wiersema, 1997) to as far south as NW Venezuela and Guyana (Goebel, 1893; Wiersema in Hokche et al., 2008). In this paper we review the distribution of B. schreberi across the Neotropics and report its occurrence in Costa Rica for the first time.
MATERIALS AND METHODS
Collections examined. Examination of specimens of Brasenia that were collected in the Neotropics was conducted in the online catalogues of COL, F, MA, MO, K, NY, P, U, US, USF and VT (herbaria acronyms as per Thiers, 2021). Additional specimen images were kindly provided by the staffs of EAP, LAGU, TEFH, PSO and the herbarium of the Corporación Autónoma Regional para la Defensa de la Meseta de Bucaramanga (CDMB, not in Index Herbariorum). Additional records were obtained from the iNaturalist platform and the literature. We mapped the examined specimens (and unequivocal photos) using the R-package maps (Becker & Wilks, 2018). The collection localities were retrieved from the specimen labels. When no coordinates were given, we estimated the collection locality as precisely as we could, using the geonames.org website, or Google Earth.
Field work. Field work was carried out in the Cureña district of Sarapiquí county, Heredia province, Costa Rica, in the lagoon systems of La Marina, Copalchí and Tambor, within the limits of the Refugio Nacional de Vida Silvestre Mixto Maquenque (RNVSMM) in the Area de Conservación Huetar Norte. The Copalchí lagoon has an approximate area of 136660 m2 (Clima Pesca, 2017) and according to the Instituto Meteorológico Nacional de Costa Rica, the average atmospheric humidity in the area is 88.5%, with 3525 mm of average yearly precipitation and an average temperature of 26.6 °C. The RNVSMM is located at low elevations with the Copalchí lagoon at approximately 23 m a.s.l. The wetland vegetation is dominated by several Poaceae species, as well as Ludwigia L. sp. (Onagraceae) and Nymphaea sp. The lagoon has a maximum depth of about 2 m and the presence of the large tropical gar (Atractosteus tropicus Gill, 1863) has been noted by us. Representative specimens of Brasenia schreberi collected in the region are deposited in USJ (Thiers, 2021).
RESULTS AND DISCUSSION
Brasenia Schreb., Gen. Pl., ed. 8[a]. 1: 372. 1789. Type species. Brasenia schreberi J. F. Gmel.
Hydropeltis Michx., Fl. Bor.-Amer. (Michaux) 1: 323. 1803. Type species. Hydropeltis purpurea Michx.
Brasenia schreberi J. F. Gmel., Syst. Nat., ed. 13[bis]. 2(1): 853. 1791. TYPE: United States, New Jersey: [Warren Co.], Hope [Township?], s.d., Floyd 1 (holotype: M 0110828!).
Hydropeltis purpurea Michx., Fl. Bor.-Amer. (Michaux) 1: 324, t. 29. 1803. Brasenia peltata Pursh, Fl. Amer. Sept. (Pursh) 2: 389. 1813. Nom. superfl. Brasenia nymphoides Baill., Hist. Pl. (Baillon) 3: 82, footnote. 1871. Nom.superfl. Brasenia purpurea (Michx.) Casp., Jorn. Sci. Math. Phys. Nat. 4(16): 312. 1873. Cabomba peltata (Pursh) F. Muell., Fragm. (Mueller) 10(85): 77. 1876. Nom superfl. Probable original material: [United States:] In Aquis Carolinae, s.d., A. Michaux s.n. (P 00752297!); Amer. Sept., s.d., A. Michaux s.n. (P 00752298!); sine loc, s.d., A. Michaux s.n. (P 00675228!).
Hydropeltis pulla Salisb., Ann. Bot. [König & Sims]. 2(1): 74. 1805. TYPE: Canada, 1800, F.
Masson s.n. (BM 000574916, not seen).
Brasenia hydropeltis Muhl., Cat. Pl. Amer. Sept.: 55. 1813. Probable original material: United States, Pennsylvania, Lancaster, 1799, H. Muhlenberg 671 (LINN hs925-1!); H. Muhlenberg [?] 795 (PH 00034888!).
Plants of Brasenia schreberi grow submersed and have been described as “free-floating” but are often fixed to the bottom by their adventitious roots that emerge from the stem nodes and have well- developed root caps that act as anchors (Arber 1920). They produce “trailing” stems (Fig. 1) that can reach 3 m or more in length and creep on the surface of the mud. Leaves are alternate (occasionally appearing opposite by production of leaves from short, axillary branches) with petioles up to 80 cm long. The leaf blades are oval, entire, centrally peltate and floating (Fig. 1A), 6-12 cm long and 4-6 cm wide (Williamson & Schneider, 1993), with the adaxial surface bright green and the abaxial surface purple. All submersed parts are usually thickly coated with a clear mucilage (Fig. 1E), which is secreted by glandular trichomes. Flowers are solitary and produced from the axils of leaves on long purplish pedicels (Raciborski, 1894; Arber, 1920; Wood, 1959). In areas with cold winters, the tips of the stems develop turions that sink to the bottom, supposedly to resist the winter (but Adams [1969] found no evidence of their survival over the winter); it is unknown if similar turions are produced in tropical habitats.
The flowers of Brasenia schreberi are protogynous, to ca. 2 cm in diameter (Osborn & Schneider, 1988) with the perianth, androecium and gynoecium dark purple (Fig. 1C). The perianth is composed of six to eight purple tepals, 1-2 cm long, 0.5 cm wide (Rahayu & Magandhi, 2018) organized in two whorls. The androecium has ca. 18-36 stamens (also in two whorls; 18-51 reported by Les, 2004) and the anthers are ca. 4 mm long (Williamson & Schneider, 1993). The gynoecium has six free carpels with two ovules in each (Raciborski, 1894; Wood, 1959; but 4-18 carpels reported by Les, 2004). Anthesis is diurnal, with individual flowers emerging from the water and opening in the morning and closing and going back into the water in the afternoon, for two consecutive days (dianthesis). First-day flowers have short, undehisced stamens and elongated, papillate stigmas; second-day flowers have elongated staminal filaments with dehisced anthers. Pollination is by wind (Osborn & Schneider, 1988). Little is known about the mechanism of seed dispersal, but Hill (1900) observed that the peduncles (pedicels?) bearing fruits apparently separate and float away from the mother plant.
Nomenclatural notes. Sometimes “Rondachine” (Bosc, Encyclopédie méthodique. Agriculture. 6: 180. 1816) is considered a synonym of Brasenia, however to us it appears that it is merely a common name intended for Brasenia. Brasenia nymphoides has been considered to be another combination of Menyanthes nymphoides L. (footnote 3 in Baillon, Hist. Pl. 3: 82. 1871), however we consider this a misapplication of the name that probably stemmed from the usage of Limnanthemum peltatum Griseb., (in Gen. Sp. Gent. 348. 1838. non S. G. Gmel. Novi Comment. Acad. Sci. Imp. Petrop. 14(1): 527 (t. 17). 1770), as Grisebach included in it Thunberg’s concept of Menyanthes nymphoides, which is not that of Linnaeus: e.g. specimens labeled Limnanthemum peltatum Griseb. in L (L 1708423!) and P (P 01956399!) are Brasenia schreberi. We consider Brasenia hydropeltis as heterotypic to Hydropeltis purpurea: Muhlenberg (1813) did not cite Michaux or his specimens, instead citing the habitat of Brasenia hydropeltis as “Pens.” [Pennsylvania].

Fig. 1 Brasenia schreberi, from RNVSMM, Heredia province, Costa Rica, Acuña et al. 2217 and Jiménez et al. 5437 (both USJ). A, habitat, in a small, tropical, lowland lagoon. B, floating, peltate leaves and anthetic flowers. C, anthetic flower, with aphids on the pedicel and on the adaxial surface of some leaves. D, habit, notice the elongated petioles and the trailing stem. E, flower with tepals removed, showing androecium (partial) and gynoecium (complete). F, underside of a floating leaf covered in mucilage. Color version at http://www.ojs.darwin.edu.ar/index.php/darwiniana/article/view/988/1235
Economic importance. Brasenia schreberi has culinary importance, it is cultivated and widely consumed in China, Japan and Korea, and the roots eaten by Native Americans (Les, 2004, 2018). It has traditional medicine importance due to the high content of antioxidants in the form of polysaccharides (Xiao et al., 2016). The mucilage also has allelopathic properties (Elakovich & Wooten, 1987) and may reduce herbivory on the species (Thompson et al., 2014). The species also has been used as a decorative plant in aquaria and water gardens and is considered a pest in Missouri (Les, 2004, 2018).
Ecology. Brasenia schreberi often grows in acidic water bodies (Les, 2004; Kim et al., 2008, 2012) but could tolerate alkaline conditions as well (Les, 2018). It prefers shallow water to a depth of up to 3 meters, in areas fully exposed to the sun. Under good environmental conditions, B. schreberi can cover water bodies quickly, since new shoots form easily from its trailing stems (Rahayu & Magandhi, 2018). Its tolerance to water turbidity and its allelopathic interactions could contribute to its dominance in some localities (Les, 2018). Carps (Cyprinidae), turtles (Testudines), waterfowl (Anatidae), moose (Alces alces, Cervidae), aphids (Hemiptera), beetles (Coleoptera) and moths (Lepidoptera) are known to consume this species (Ernst & Lovich, 2009; Les, 2018; Rahayu & Magandhi, 2018).
Distribution in the Neotropical Region (Fig. 2) WEST INDIES: The species seems to be restricted to lakes, lagoons, and ponds in the lowlands of Cuba, Jamaica, and the Dominican Republic. Judging by the number of specimens examined, it appears that the species could be more common in the western part of Cuba than elsewhere in the Neotropics.
MEXICO: Brasenia can be found both in lowland and mid-elevation localities. It seems mostly restricted to the south of the Tropic of Cancer (Jalisco, Michoacán, San Luis Potosí, Veracruz, Tabasco, and Chiapas: Villaseñor, 2016; Zepeda-Gómez, 2017), but there is at least one extratropical record from Chihuahua (Lot et al., 2002).
NORTHERN CENTRAL AMERICA: The specimens from the Atlantic slope (Belize, northern Guatemala, and eastern Honduras) were all collected in lowland lagoons, while in those from the Pacific slope (southeastern Guatemala, western El Salvador, and central Honduras: Rivera, 2014) were found in middle elevation lagoons in mountainous areas.
SOUTHERN CENTRAL AMERICA: We record the presence of Brasenia schreberi for the first time in Costa Rica. We found the species growing in two sites within the RNVSMM of which one was sampled. The sampled plants were found growing on the eastern end of a small lagoon surrounded by forest patches, on the side of a dirt road. The area where Brasenia grew had dark, organic material-rich water, 80-100 cm deep. The substrate was muddy and soft, and probably with low oxygen levels, judging by the amount of gases liberated when the plants were collected. A man-made canal, probably to prevent the lagoon to overflow the dirt road, ran under the road allowing some water flow. Associated species include Annona glabra L., Blechnum serrulatum Rich., Eleocharis retroflexa (Poir.) Urb., Nymphaea sp., Rhynchospora corymbosa (L.) Britton, and Urospatha grandis Schott.
The aerial portions of stems (e.g., the flower pedicels) of plants in the wild were infested by aphids (Aphidoidea, Hemiptera). The submerged parts of the plants were heavily coated in a thick layer of mucilage, but a few plants that were collected alive, and transplanted to cultivation, have produced only small quantities of mucus 6 months after transplantation, while staying apparently healthy, producing new leaves, vegetative buds, and stolons.
Brasenia schreberi has not been previously documented in Costa Rica, even in some intensely- collected wetland localities such as Palo Verde, Caño Negro and Tortuguero. This underscores that, to effectively document the wetland flora of the country, more effort has to be invested in lesser known localities. Strangely, Brasenia was not mentioned in the recent treatment of Cabombaceae of Costa Rica by Crow (2020), albeit the species was already known from countries both north and south of Costa Rica. Although, as far as we know Brasenia schreberi has not been collected in Nicaragua, it is very likely it will be found there, as RNVSMM is adjacent to the Nicaraguan border (and the species has been collected at least three times in Honduras).
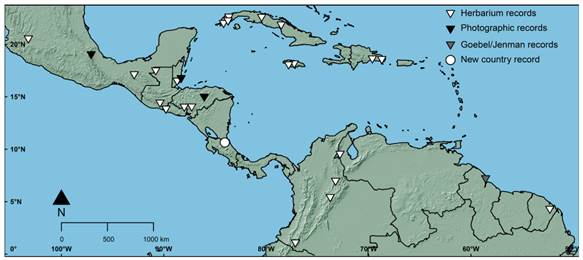
Fig. 2 Distribution of Brasenia schreberi in the Neotropics based on herbarium specimens examined in this study, trustable reports in the literature (Goebel/Jenman records), and/or unequivocal photographic records (iNaturalist and tropicos.org databases, see text for details). Color version at http://www.ojs.darwin.edu.ar/index.php/darwiniana/article/view/988/1235
COLOMBIA: We were able to examine specimens that confirm the unequivocal presence of Brasenia schreberi in Colombia, as Camargo (1969) and Schmidt-Mumm (1998) previously reported. However, the species is not cited in the recent Catálogo de las plantas y líquenes de Colombia (Bernal, 2015) and Schmidt-Mumm (1998) considered it as extinct in the Sabana de Bogotá region. Material collected more recently also indicates the species is more widespread in the country than previously thought, and very likely still persists in Colombia. Unlike the specimens of the West Indies, the Caribbean slope of Central America or the Guianas, which were all collected from the lowlands, those from Colombia grew in swamps and lagoons at intermediate or even high elevations in the Andes themselves (ca. 1250-2770 m a.s.l.). Although the species was collected as far back as the late 18th or early 19th centuries in Colombia by José Celestino Mutis and there are confirmed records from at least three Departments, only a handful of specimens have been deposited in herbaria, underlining its potentially very localized distribution in the country.
VENEZUELA: The first records of Brasenia in the country were published by Aristeguieta & Agostini (1965) for the states of Mérida and Táchira. Wiersema (in Hokche et al., 2008), also mentions its presence in Zulia (we examined a specimen from western Zulia) and Trujillo. Thus, the species seems to be restricted to the northwestern part of the country. According to the map 14 in Velásquez (1994, p. 114), it could be mostly, or even entirely, restricted to the Maracaibo Lake Basin.
THE GUIANAS: The description and illustrations in Goebel (1893: p. 232 and Tab. XXXI) can be unequivocally assigned to Brasenia schreberi. This is one of the few records from Guyana we were able to find in the literature (“Ich war aber, als ich Brasenia im Tapacooma-See in Britisch Guyana sammelte, doch überrascht, zu sehen, von welch dicker Lage von Gallerte die sämtlichen jungen Teile überzogen waren.”). However, it is uncertain if Goebel’s collection was deposited in any herbarium and the specimen, if any, has not been located (A. Fleischmann & H-J. Esser in M, pers. comm. 2021). Cramer (1979) cites that [George Samuel] Jenman collected the species in 1886, also in the Tapakuma lake, but we have not been able to locate his collections either. According to J. Wiersema (US), the species could be extirpated in the country (pers. comm. 2021).
We also examined a single French Guianan collection (several duplicates). Judging from satellite images near the inferred collection locality, it seems like the collection comes from a deeply forested lowland area, as the portion of the RN2 road from Correze and Coralie to Regina seems to be surrounded by dense lowland humid forests. These few records indicate that the species could be rare and found very sporadically in lowland lakes or lagoons surrounded by forests. Cramer (1979) indicates that the species has not been collected in Suriname.
Examined material
BELIZE. Stann Creek: Sanctuary Belize, 25-VIII-2003, Jan Meerman available in https://www.inaturalist.org/observations/32145301 (iNaturalist). Toledo: Lowland broadleaved forest. BFREE [Belize Foundation for Research and Environmental Education] Reserve, Lagoon, 45-75 m a.s.l., 15-X-2005, Whitefoord & Quiroz 106118 (MO); BFREE, 4-II-2008, Jan Meerman available in https://www.inaturalist.org/observations/32145301 (iNaturalist).
COLOMBIA. Without locality: 1783-1808, Mutis 737 (MA, US). Cundinamarca: [Prov. Ubaté], Pantanos Fúquene, 2600 [2540] m a.s.l., III.1930, Pérez Arbeláez 87 (COL, US). Nariño: Prov. Pasto, Corregimiento El Encano, Laguna de La Cocha, totoral de la Isla Corota. 2770 m a.s.l., 30-IX-2005, Benavides et al. 57 (PSO). Santander: [Prov. Metropolitana], Munic. Girón, Humedal El Pantano, 1282 m a.s.l., 16-V-2013, Anaya & Herrera 5 (CDMB), same locality and date, Anaya & Herrera 8 (CDMB).
COSTA RICA. Heredia: Cantón Sarapiquí, Cureña: Refugio Nacional de Vida Silvestre Mixto Maquenque, pequeña laguna rodeada de fragmentos boscosos, a orilla del camino a aprox. 700 m al NW del Bar Finca La Alejandra hacia Lagunas La Marina, 40 m a.s.l., 26-I-2021, Acuña et al. 2217 (USJ), same locality and date, Jiménez et al. 5437 (USJ).
CUBA. Without locality. 1860-1864, Wright 1859 (K, MO, NY, P, US). [Ciego de Ávila?]: Herradura, 28-X-1905, Cook 134 (US). Pinar del Río: Laguna en el camino a Ceja Ana de Luna, Viñales, 11-XI-1989, Urquiola et al. 5581 (NY); Laguna Jovero and vicinity, 5-7-XII-1911, Shafer 10826 (NY, US); Laguna Jovero, Remates de Guane, 5-I-1932, León 15368 (NY); Laguna Santa María, 8-IX-1910, Britton et al. 7151 (NY); La Máquina, 4-III-1943, Marie-Victorin 58220 (US, USF). Villa Clara: Mordazo, Laguna Pozo Grande, 9-VIII-1923, Ekman 17028 (NY).
DOMINICAN REPUBLIC. La Altagracia: Llano Costero, Laguna la Canasta, 14-IV-1929, Ekman 12170 (MO, U, US). [Monte Plata?]: Savannah land and lake area between Bayaguana and Guerra, 8-XI-1946, Howard & Howard 9938 (NY, P, US).
EL SALVADOR. Ahuachapán: Munic. Apaneca, Lagunita Las Ninfas, ca. 1830 m a.s.l., 28-I-1951, Fasset 28702 (MO, US); same locality, 26-XII-1998, Herrera 3794/JBL03794 (LAGU); Laguna Verde, 1829 m a.s.l., 28-XII- 1998, Herrera 3835/JBL03835 (LAGU, MO).
FRENCH GUIANA. Cayenne: Route RN2 Cayenne-Régina, 31-V-2010, González 2419 (MO, P, US).
GUATEMALA. Petén: Pozos Xan. Laguna perenne rodeada de bosque y cibal, que se inunda en época lluviosa., 50 m a.s.l., 7-VI-1997, León et al. 160 (MO). Santa Rosa: Laguna de Carrizal, ca. 1600 m a.s.l., V-1892, Heyde & Lux 3062 (MO, US).
HONDURAS. Francisco Morazán: Desvío a San Matías, Laguna El Pedregal a 10 Km N de Tegucigalpa, 1500 m a.s.l., 4-III-2010, House et al. 5398 (TEFH). La Paz: Mt. Verde, 18 Km S de Marcala, frecuente en lagunas y quebradas pantanosas, 1800 m a.s.l., 22-V-1986, Keyser 1451 (EAP). Olancho: Gualaco, 700 m a.s.l., 16-III- 2013, Vílchez et al. 570 available in http://legacy.tropicos.org/Image/100247647 (tropicos.org).
JAMAICA. Clarendon/Saint Ann: Mason River district, ca. 3 miles NW of Kellits, 22-I- 1966, Walker 292 (US). Saint Elizabeth: Holland, Vicinity of the Bamboo Avenue, Holland estate, ca. 20 m a.s.l, 4-II-1961, Proctor & Mullings 21965 (NY).
MEXICO. State unknown: Vallée de Mexico, 1875, Schaffner 483 (NY, P). Chiapas: Munic. Pueblo Nuevo Solistahuacán, large pond and adjoining marsh on the eastern side of Pueblo Nuevo de Solistahuacán, 1700 m a.s.l., 26-X-1971, Breedlove 21529 (F, MO). Jalisco: Shallow pond near Guadalajara, 26-VI-1889, Pringle 2920 (VT). Veracruz: [Munic. Huatusco?], Locality not available, 8-V-2011, César Lezama available in https://www.inaturalist.org/observations/74328085 (iNaturalist).
VENEZUELA. Zulia: [Munic. Machiques de Perijá?], Entre km. 20-30 al oeste de la carretera Machiques-La Fría, a lo largo de la vía que conduce entre la carretera y el Asentamiento Aricuaisá y el matadero, en bosque de galería y en zona de bosque en transición entre deciduo y siempre-verde., 100-200 m a.s.l., 4-V-1982, Bunting & Trujillo 11562 (MO).
COMMENTS
Brasenia schreberi has a wide but very patchy distribution in the northern Neotropics. In most countries where it has been recorded, it is known from one or a few isolated localities and by a similarly low number of herbarium records or verifiable observations. This sporadic pattern of distribution appears to be real, particularly in Colombia, Central America and the Guianas, and not an artifact that could be attributed only to the low intensity of collection efforts at national scales. Brasenia schreberi has a very distinctive morphology and in the few localities where it is present, it could be common, so it is unlikely that the species would have been overlooked over such vast expanses of its potential range if its distribution was more homogenous. Although both in Africa and globally the species is listed in the category of Least Concern by the IUCN (Gogue, 2010; Maiz-Tome, 2016), and it appears to be relatively common in temperate North America, it is considered threatened in Mexico (Zepeda Gómez, 2017), Australia (Stanley & Orchard, 2007) and Eastern Asia (Kim et al., 2012; Li et al., 2018) and it has been apparently extirpated from Guyana (Wiersema, 2007) and areas of Colombia (Schmidt-Mumm, 1998), Australia (Stanley & Orchard, 2007), China (Li et al., 2018) and South Korea (Kim et al., 2012). Sculthorpe (1967) suggested that the wide but discontinuous, “anomalous” distributions of B. schreberi and some other aquatic macrophytes could be the result of extermination from certain areas during the glacial and post-glacial periods.
While it is possible that Brasenia schreberi could eventually be found in more localities and in countries like Nicaragua, Panama or Suriname as well as possibly be rediscovered in Guyana, we expect that these potential discoveries would take place in few localities, over relatively small areas and that will reaffirm the localized and scattered distribution pattern of Brasenia in the Neotropics as a whole, as we report here. We recommend the species to be considered Vulnerable (VU) under criteria B2ab(iv) (IUCN Standards and Petitions Subcommittee, 2017). Criterion B2 was selected because, using the GEOCAT tool (Bachman et al., 2011) and a reasonable 5 x 5 km grid, we obtained an Area of Occupancy (AOO) of 725 km2. Criterion “a” was selected because of its severely fragmented geographic range (influenced by its absence from seemingly appropriate localities). Criterion “b(iv)” was selected because of the decline in the number of locations or subpopulations (as it happened in, e.g., Colombia and Guyana), hinting that the populations of B. schreberi are reduced and could be decreasing in the Neotropics.












 uBio
uBio 

