Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Medicina (Buenos Aires)
versión impresa ISSN 0025-7680versión On-line ISSN 1669-9106
Medicina (B. Aires) v.66 n.5 Buenos Aires sep./oct. 2006
Ocular lesions and experimental choline deficiency
Georgina P. Ossani, David Pelayes1, María L. Diaz 2, Nestor R. Lago1, Silvia L. Fariña1, Alberto J. Monserrat1, Jorge O. Zarate1
1Laboratorio de Investigaciones Oftalmológicas y Visuales, Centro de Patología Experimental, Departamento de Patología, Facultad de Medicina, Universidad de Buenos Aires;
2Servicio de Medicina Nuclear, Hospital Británico, Buenos Aires
Postal address: Dra. Georgina P. Ossani. Laboratorio de Investigaciones Oftalmológicas y Visuales, Centro de Patología Experimental Departamento de Patología, Facultad de Medicina, Universidad de Buenos Aires, J. E. Uriburu 950 5to piso, 1114 Buenos Aires, Argentina Fax: (54-11) 4508-3602
E-mail: georginaossani@yahoo.com.ar
Abstract
Previous studies have shown ocular haemorrhages in choline-deficient rats. The aim of this paper is to study further the relationship between ocular and renal lesions and biochemical alterations in rats fed a choline-deficient diet. Fifty one weanling male Wistar rats, were divided into two groups. Thirty one of them were fed a choline-deficient diet and the rest was fed a choline-supplemented diet ad libitum. Animals from both groups were killed between the fifth and the eighth day. Urea, creatinine and homocysteine concentrations in blood were determined. Eyes were used for light microscopy study; high resolution light microscopy and the study of the retina as "rétine a plat". Kidneys were studied by light microscopy. Choline-supplemented rats did not show ocular or renal lesion. Choline-deficient rats that showed renal lesions, tubular or cortical necrosis, did not always have ocular changes. There were no ocular changes in the only choline-deficient rat without renal lesion. The ocular changes consisted mainly in haemorrhage in both cameras and ciliary and vitreous bodies. Correlations between ocular and renal lesion (r=0.72, p<0.0001, CI 95%: 0.48-0.86); ocular lesion and creatinine (r=0.86, p<0.0001, CI 95%: 0.72-0.93) and ocular lesion and urea (r=0.70, p<0.0001, CI 95%: 0.44-0.85) were positive. Choline-deficiency induces ocular haemorrhagic lesions after the development of renal necrosis. The ocular pathology could be due to the immaturity of the ocular vasculature at this age. The hyaloid, choroid and retinal system are involved.
Key words: Choline-deficiency; Eye; kidney; Acute renal failure
Resumen
Lesiones oculares y deficiencia experimental de colina. Estudios previos han demostrado hemorragia ocular en ratas deficientes en colina. El objetivo de este trabajo es profundizar en la relación entre las alteraciones oculares, renales y bioquímicas en ratas deficientes en colina. Cincuenta y una ratas Wistar macho recién destetadas fueron divididas en dos grupos: treinta y una fueron alimentadas con una dieta colino deficiente y el resto con colina suplementada ad-libitum. Los animales de ambos grupos fueron sacrificados entre el quinto y el octavo día. Se midió la concentración de urea, creatinina y homocisteína en sangre. Los ojos fueron estudiados por microscopía de luz, microscopía óptica de alta resolución y para el estudio de la retina como retina plana. Los riñones fueron estudiados por microscopía de luz. Las ratas suplementadas con colina no mostraron lesiones oculares o renales. Las colino deficientes que mostraron lesiones renales, necrosis tubular o cortical, no siempre tuvieron cambios oculares. No se encontraron cambios oculares en la única rata deficiente en colina sin lesión renal. Los cambios oculares consistieron principalmente en hemorragia en ambas cámaras, cuerpo ciliar y vítreo. La correlación entre la lesión ocular y renal (r=0.72, p<0.0001, CI 95%: 0.48-0.86), lesión ocular y creatinina (r=0.86, p<0.0001, CI 95%: 0.72-0.93) y lesión ocular y urea (r=0.70, p<0.0001, CI 95%: 0.44-0.85) fue positiva. La deficiencia de colina induce lesiones oculares luego del desarrollo de la necrosis renal. La patología ocular podría ser debida a la inmadurez de los vasos oculares. El sistema hialoide, coroideo y retinal están involucrados.
Palabras clave: Colino-deficiencia; Ojo; Riñón; Insuficiencia renal aguda
Choline is a precursor for the synthesis of acetylcholine, it is required for the synthesis of phospholipids that constitute cell membranes and, through betaine it is a source of labile methyl groups1, 2.
Weanling rats fed a choline-deficient diet show a variety of biochemical, functional and morphologic alterations that involve different organs such as the kidneys, liver, heart, or eyes1, 2. Renal pathology is characterized by tubular or cortical necrosis3, liver alterations by fatty changes, cirrhosis and eventually cancer2, 4, and heart changes by necrosis5, 6.
As far as we know, there are only two previous papers that refer to the ocular changes as the primary interest in choline-defficient (CD) rats. Both describe the presence of haemorrhages; almost all of them situated in parts of the eye supplied by the hyaloid arterial system7,8.
The present paper deals with the ocular alterations due to choline nutritional deficiency in weanling male Wistar rats, and its correlation with renal lesions and biochemical alterations.
Materials and Methods
Fifty one weanling male Wistar rats, from the Bioterium of the Department of Pathology, School of Medicine, University of Buenos Aires, were divided into two groups. One of them (31 animals) was fed a CD ad libitum (diet 1, Table 1); the other (20 animals) was fed a choline supplemented diet (CS) ad libitum (diet 2, Table 1).
TABLE 1.- Composition of diets (g/100g)
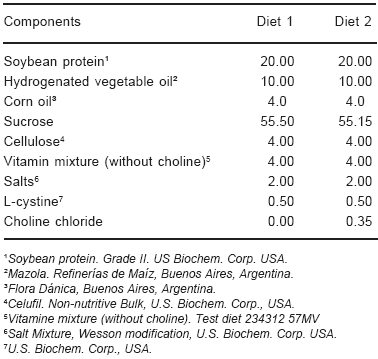
Food intake and body weight were recorded daily. Ophthalmoscopic examination was carried out from the third day until the sacrifice. All experimental procedures conformed to The Association for Research in Vision and Ophtalmology (ARVO) statement for the use of animals in ophthalmic and vision research9.
Animals from both groups were killed between the fifth and the eighth day.
Urea (Wiener Laboratory, Buenos Aires), creatinine (Wiener Laboratory, Buenos Aires) and homocysteine10 concentrations were determined in blood.
Both eyes were labelled with threads of different colours in the superior part. Then, they were removed and fixed in buffered formalin for at least seven days. Afterwards, eyes were cut horizontally. The superior right half and the inferior left half were embedded in paraffin and sections were stained with hematoxylin and eosin following standard methods for histopathological study. The inferior right half and the superior left half were embedded in Polibed and sections were stained with toluidine blue and fuchsin for high resolution light microscopy (HRLM)11 or they were used to study the retina as "rétine a plat"12 by surface light microscopy13, 14.
By the different methods previously mentioned, it was analysed the presence or absence of haemorrhage in the different parts of the eye.
For classification of ocular lesions a macroscopic score was made: 0 (normal), 1 (punctiform haemorrhage), 2 (haemorrhage in anterior chamber), 3 (haemorrhage in vitreous), 4 (2 plus 3). Data from both eyes were employed.
Both kidneys were removed, weighted, fixed in buffered formalin and embedded in paraffin. Then, sections were cut and stained with hematoxylin and eosin in order to analyse the histopathological alterations.
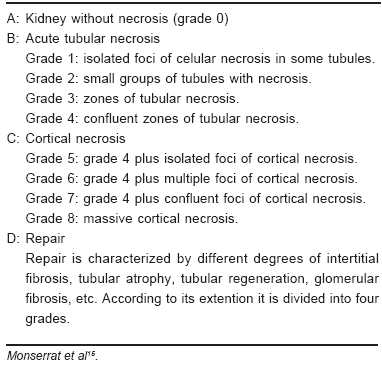
Histologic classification of renal necrosis was taken from Monserrat et al15 (Table 2).
TABLE 2.- Histologic classification of renal necrosis
Levels of urea, creatinine and homocysteine in serum were compared by one way analysis of variance (ANOVA). In the case of significant differences ANOVA was followed by the Tukey-Kramer test. Differences which resulted in p values lower than 0.05 were considered significant (S). Correlations between ocular lesion and renal lesion, creatinine and urea were made by Spearman test. The statistical evaluation was made using the program GraphPad Instat version 3.05, GraphPad Software, San Diego, California.
Results
Rats fed a CS diet did not show any ocular or renal lesions.
All rats, except for one, fed a CD diet showed renal necrosis (Table 3). The only CD rat that did not show renal lesion, did not have any ocular alteration.
TABLE 3.- Day of killing, ocular and renal lesionsin CD rats
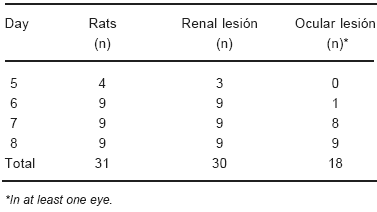
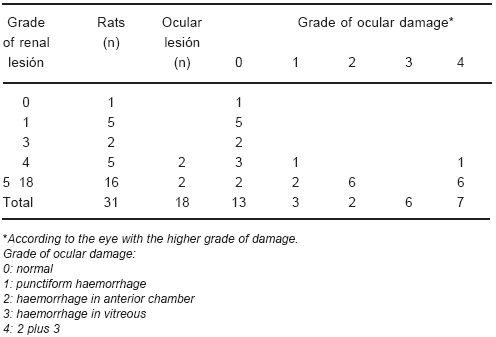
Twelve out of 31 CD rats showed renal necrosis but did not have ocular changes (Tables 3 and 4).
TABLE 4.- Grade of renal and ocular damage
The acute renal changes were grossly characterized by an increase in size and weight and purplish red discoloration. Necrosis involved mainly proximal convoluted tubules and was characterized by increased eosinophilia, pyknosis and mainly kariolysis; in more advanced stages glomerular and vascular necrosis were also observed.
Those CD rats that showed ocular lesions developed haemorrhages, most frequently in anterior chamber, posterior chamber, ciliary region and vitreous. Macroscopic appearance is shown in Figure 1.
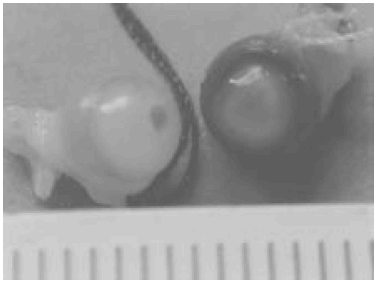
Fig. 1.- Eyes from rat killed the eighth day. One of them with severe haemorrhage and the other with punctiform haemorrhage.
Histopathological changes are observed in Figure 2 (panoramic view).
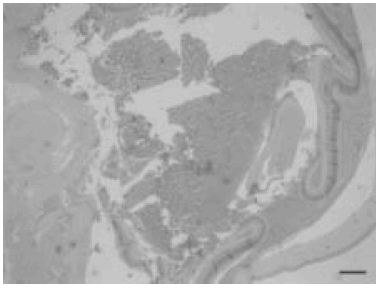
Fig. 2.- Microscopic panoramic view. Ocular haemorrhage lesions. (H-E). Scale bar, 150 µm.
There were no ocular changes in the rat without renal lesion. However, there were twelve CD rats that did not have ocular haemorrhage but did have renal necrosis (Tables 3 and 4).
In most of the animals, the grade of renal necrosis increased in relation with the day of sacrifice, the later the day the greater the lesion.
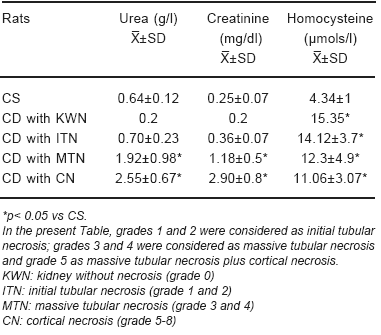
Urea and creatinine levels increased in relation with the grade of renal lesion (Table 5). Homocysteine levels were increased in CD rats despite of the grade of renal alteration; it was also increased in the rat without renal necrosis (Table 5).
TABLE 5.- Urea, creatinine and homocysteine
Rats grew adequately well during the first five days. Afterwards, CD rats lost weight, due to renal damage16.
HRLM (Figure 3) and the "rétine a plat" (Figure 4) allowed a more precise study of the ocular damage.

Fig. 3.- HRLM. Retina in CD rat with vascular proliferation, microhaemorrhage and congestion. Scale bar, 150µm.
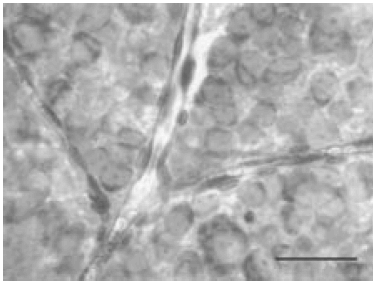
Fig. 4.- Rétine a plat in CD rat. Vascular network. Scale bar, 50µm.
There was a good relationship between ophthalmos-copic examination and ocular lesions.
Correlations between grade of ocular and renal lesions (r=0.72, p<0.0001, CI 95%: 0.48-0.86); grade of ocular lesion and day of sacrifice (r=0.87, p<0.0001, CI 95%: 0.74-0.93) grade of ocular lesion and creatinine (r=0.86, p<0.0001, CI 95%: 0.72-0.93) and grade of ocular lesion and urea (r=0.70, p<0.0001, CI 95%: 0.44-0.85) were high and positive. From the four rats killed at the fifth day, three of them showed renal damage; however, none of them displayed ocular lesion.
Discussion
The relationship between ocular and renal pathology in human beings is well known in some type of alterations17-19. Ocular neovascularization and haemorrage with associated hiperpermeability are the most common etiologies of visual loss. Neovascularization is another major etiology of visual impairment in diverse conditions such as: radiation retinopathy, sickle cells retinopathy and probably in metabolic alterations secondary to nephropathies. Most disorders associated with ocular neovascularization and haemorrage have several characteristics in common, although each disease has unique etiologies; finally all of them lead to areas of nonperfusion and the development of haemorragic changes. The information is scanty in other situations, such as the relationship between acute renal failure and ocular damage20.
The aim of this work was to study the ocular alterations occuring in the acute renal failure due to choline deficiency.
To the best of our knowledge only two papers have primary adressed the ocular alterations in choline-deficiency7, 8. Bellows and Chinn7 concluded that: 1. "Free blood was most frequently found between the anterior limiting membrane of the vitreous and the cristalline lens; 2. The ciliary processes were swollen and frequentely hemorrhagic; 3. The presence of blood in the anterior chamber was not uncommon". Burns and Hartroft8 described that the haemorrhage was located mainly between the vitreous and the crystalline lens, with extension in a few cases to the ciliary region and into the aqueous chambers. In addition they found ocular haemorrhages in 80% of weanling rats with bilateral nephrectomy but not in adult rats. On the other hand, choline-deficiency did not induce ocular haemorrhages in adult rats. The hyaloid arterial system would be the origin of the haemorrhages.
Ocular blood supply has some distinctive characteristics: 1. Retina constitutes the tissue with the greatest oxygen consumption by unit of weight; 2. Ocular blood supply is based on a triple vascular system: a: hyaloid system; b: choroid system; c: retinal system. The hyaloid artery originates from the primitive dorsal ophthalmic artery; arborization of the hyaloid artery originates the tunica vasculosa lentis. After atrophy of the vasa hyaloidea, the eye has two separate circulatory systems that have different anatomical and physiological characteristics. Outer third of the retina is nourished by the chorideal circulation and the inner two thirds receive nutrition from the retinal circulation. These facts make the eye a vulnerable tissue to the biochemical alterations that follow renal necrosis in this experimental model such a increased levels of urea and creatinine3, 21 and consumption coagulopathy15.
Data showed that ocular lesion does not occur without a previous renal lesion (Tables 3 and 4). As the renal lesion increase with time15, 22, so does the ocular lesion. This fact could explain the higher correlation between day of killing and ocular lesions than between grade of ocular and renal lesions. This indicates that some time is required to the development of the ocular lesion after the renal damage has appeared.
According to what it has been mentioned above, it is probably that through an unknown mechanism, the uremia induced by the renal lesion produces microhaemorrhages and massive haemorrhages in a place of low resistance like the eye. In this regard it is interesting to know that Burns and Hartroft8 found ocular haemorrhages in weanling rats made uremic by bilateral nephrectomy.
It is probably that consumption coagulopathy that occurs in acute renal failure due to a choline-deficient diet may contribute to the development of ocular haemorrhages15.
Another factor that could be involved in the ocular haemorrhage is arterial hypertension. Arterial hypertension has been described in adult rats deprived of dietary choline for 5 to 6 days when they were weanling and then transfered to a normal diet for a period of 4 to 7 months23. In addition, young male choline-deficient rats (65-85 g body weight) developed progresive hypertension after at least 2 weeks of the deficient diet24. However, neither Sobin and Landis25 nor Hartroft26 found arterial hypertension in wealing rats fed a choline-deficient diet; so this factor can be discarded in our study.
As shown in our study, the most frequently sites where ocular haemorrhages occurred were: anterior chamber, posterior chamber, ciliary region and vitreous. Taking into account the ocular blood supply we can state that all vascular systems are involved. This conclusion differs from those of the previous papers that assert that almost all the ocular haemorrhages where situated in sites supplied by the hyaloid system7, 8.
The hyaloid circulation is a transitory network of blood vessels responsible for maturing the embryonic and fetal lens and possible post-vitreo retinal regression of this circulation is complete by approximately the third to fourth weeks after birth in the rat. In this experiment all rats were killed between the fifth and eight day after weaning.
Homocysteine was increased in all CD rats, even in the only one without renal damage, no relationship was found between the degree of renal damage and the increase of homocysteine. Since the only CD rat without renal damage had also similar increased levels of homocysteine, it is likely that the acute renal failure was not the cause of it. It is possible that this increase was due to the choline deficiency, as this metabolite, through betaine, is one of the methyl donors for the synthesis of methionine from homocysteine2.
In summary, our results extent previous results of Bellows and Chinn7 and Burns and Hartroft8. The main differences are: the significative correlation between morphological ocular and renal lesions and concentrations of urea and creatinine in serum and ocular damage and that we have found that the hyaloid system is not the only vascular system involved, although most of the haemorrhages occurs in its territory.
Acknowledgements: This study was partially supported by grants from the University of Buenos Aires and CONICET. The technical assistance of Ana Uceda is gratefully recognized.
References
1. Niculescu MD, Zeisel SH. Diet, methyl donors and DNA methylation: interactions between dietary folate, methionine and choline. J Nutr 2002; 132: 2333S-5S.
2. Zeisel S. and Blusztajn J. Choline and human nutrition. Annu Rev Nutr 1994; 14: 269-96.
3. Courrèges C, Caruso C, Klein J, Monserrat AJ. Protective effect of menhaden oil on renal necrosis occurring in weanling rats fed a methyl-deficient diet. Nutr Res 2002; 22: 1077-89.
4. Ghoshal AK, Farber E. Biology of Disease. Choline deficiency, lipotrope deficiency and the development of liver disease including liver cancer: A new perspective. Lab Invest 1993; 68: 255-60.
5. Wilgram GF, Hartroft WS, Best CH. Dietary choline and the maintenance of the cardiovascular system in rats. Br Med J 1954; 1-5.
6. Arienti de García I, Perazzo JC, Monserrat AJ. Necrosis cardíaca y factores lipotrópicos. Medicina (Buenos Aires) 1981; 41: 556-64.
7. Bellows J, Chinn H. Intraocular hemorrhages in choline deficiency. Arch of Ophtalmology 1943; 30: 105-9.
8. Burns J, Hartroft W. Intraocular hemorrhages in young rats on choline-deficient diets. Am J Ophth 1949; 32: 79-91.
9. http://www.arvo.org/eweb/dynamicpage.aspx?site=arvo 2&webcode=AnimalsResearch, Consultado 7/2/06.
10. Ubbink J, Hayward VW, Bissbort S. Rapid HPLC assay for total homocysteine levels in human serum. J Chrom Biomed Appl 1991; 565: 441-6.
11. Hoffmann EO. High-resolution light microscopy for interpretation of renal biopsies. Pediatr Nephrol 1995; 9: 763-9.
12. Offret G, Dhermy P, Brini A, Bec P. Anatomie pathologique de l'oeil et de ses annexes. Masson & Cie Editeurs. Paris, 1974, pp 268, 272.
13. Argento C, Zárate J. Study of the lens epithelial cell density in cataractous eyes operated on with extracapsular and intercapsular techniques. J Cataract Refract Surg 1990; 16: 207-10.
14. Zárate JO. Surface Light Microscopy. XVIII Congreso Internacional de la Academia Internacional de Patología. Buenos Aires, Argentina, 1990: 108 (Abstract).
15. Monserrat AJ, Musso AM, Tartas N, Nicastro MA, Konopka HF, Arienti de García I, Sánchez Avalos JC. Consumption coagulopathy in acute renal failure induced by hypolipotropic diets. Nephron 1981; 28: 276-84.
16. Montes de Oca M, Perazzo JC, Monserrat AJ, Arrizurieta de Muchnik EE. Acute renal failure induced by choline deficiency. Structural-functional correlations. Nephron 1980; 26: 41-8.
17. Celville DJ, Savige J. Alport syndrome. A review of the ocular manifestations. Ophthalmic Genet 1997; 18: 161-73.
18. Schwartz MM, Lewis EJ, Leonard-Martin T, Lewis JB, Battle D. Renal pathology patterns in type II diabetes mellitus: relationship with retinopathy. The Collaborative Study Group 1998; 13: 2547-52.
19. Deguchi HE, Amemiya L. Two cases of uveitis with tubulointerstitial nephritis in HTLV-1 carriers. Jpn J Ophthalmol 2003; 47: 372-8.
20. Caton B, Díaz de Otazu R, Aldamiz-Achebarria M, Viguri A: Haemolytic-uraemic syndrome with thrombotic microangiopathy of the retina following cytomegalovirus infection: postmortem findings. Postgrad Med J 1993; 71: 2260-9.
21. Monserrat AJ. Injuria renal nutricional. Estudios experimentales. Tesis de doctorado. Facultad de Medicina. Universidad de Buenos Aires, 1974.
22. Arienti de García I, Konopka HF, Perazzo JC, Monserrat AJ: Patología de la médula renal en ratas deficientes en factores lipotrópicos. Medicina (Buenos Aires) 1979; 39: 49-57.
23. Hartroft WS, Best CH: Hypertension of renal origin in rats following less than one week of choline deficiency. Brit Med J 1949; 1: 423-6.
24. Kratzing CC, Wetzig GA, Ellway CP: Adrenergic mechanisms in choline-deficient rats. J Nutr 1970; 100: 781-5.
25. Sobin SS, Landis EN. Blood pressure of the rat during acute and chronic choline deficiency. Am J Physiol 1947; 148: 557-62.
26. Hartroft WS: Hypertension and renal lesions as manifestations of nutritional deficiency. Nutrition in Relation to Health and Disease, Proceedings of the 1949 Milbank Memorial Fund, 1950, pp144-65. [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ]
Received: 21-12-2005
Accepted: 14-06-2006














