Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Medicina (Buenos Aires)
versión impresa ISSN 0025-7680versión On-line ISSN 1669-9106
Medicina (B. Aires) v.67 n.5 Buenos Aires sep./oct. 2007
Erythrocyte membrane, plasma and atherosclerotic plaque lipid pattern in coronary heart disease
Natalia R. Lausada1, Sebastián Boullón2, Fernando Boullón2, Irma N. Tacconi De Gómez Dumm1
1Instituto de Investigaciones Bioquímicas de La Plata (INIBIOLP-CONICET-UNLP), Facultad de Ciencias Médicas, Universidad Nacional de La Plata;
2Servicio de Cirugía Cardiovascular, Hospital Francés, Buenos Aires
Postal address: Dra. Natalia Raquel Lausada, INIBIOLP Facultad de Ciencias Médicas, Calle 60 y 120, 1900 La Plata, Buenos Aires, Argentina Fax:(54-221) 4258989 e-mail: nlausada@atlas.med.unlp.edu.ar
Abstract
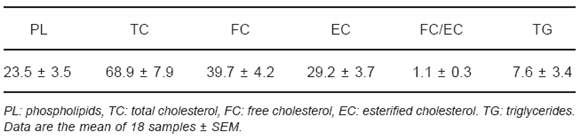
The objective was to analyze the lipid composition of the atherosclerotic plaque (AP), plasma and erythrocyte membrane (EM) in patients with advanced coronary heart disease (CHD). AP were obtained through endarterectomy in 18 patients. Ten normolipemic healthy subjects were selected to obtain the normal lipid pattern profile. Total lipids of AP and EM were determined by HPTLC, and the fatty acid profile from AP, EM and plasma using TLC-FID. The relative amount of the lipid species analyzed in AP was in line with the data in the literature [phospholipids: 23.5 mol% ± 3.5; total cholesterol 68.9 mol% ± 7.9; triglyceride 7.6 mol% ± 3.4]. Plasma and EM from CHD patients compared to controls, showed a decrease in polyunsaturated fatty acids and an increase in saturated fatty acids leading to a decrease in the unsaturation index (plasma: 1.67 ± 0.06 vs. 1.28 ± 0.03, P<0.05; EM: 2.28 ± 0.04 vs. 1.25 ± 0.010, P<0.05) and an enhancement in the saturated/unsaturated ratio (plasma: 0.35±0.02 vs. 0.52 ± 0.02, P<0.05; EM: 0.45 ± 0.01 vs. 0.83 ± 0.04, P<0.05). These data are consistent with an essential fatty acid deficiency. Total cholesterol was increased in the CHD's EM (32.3 ± 0.8 vs. 40.6 ± 2.5, P<0.05) with a decrease in phospholipid percentage (67.7 ± 0.7 vs. 59.4 ± 2.6, P<0.05) indicating an alteration in membrane fluidity. These findings suggest changes in EM lipids in CHD patients in spite of different pathological conditions such as age, smoking status and diabetes. The analysis of the lipid composition of EM could provide a useful tool to monitor the evolution of the CHD.
Key words: Coronary arteriosclerosis; Dyslipidemia; Endarterectomy; Erythrocyte membranes; Fatty acids
Resumen
Perfil lipídico de membrana de eritrocito, plasma y placa ateromatosa en la enfermedad coronaria. El objetivo fue analizar la composición lipídica de las membranas de eritrocitos (ME), plasma y placas ateromatosas (PA) en pacientes con enfermedad coronaria avanzada (ECV). Las PA fueron obtenidas de endarterectomías coronarias de 18 pacientes. Fueron seleccionados 10 sujetos sanos, normolipémicos, como grupo control. Los lípidos totales de PA y ME se determinaron utilizando HPTLC, y el perfil de ácidos grasos de las PA, ME y plasma mediante TLC-FID. La cantidad relativa de las especies lipídicas obtenidas de las PA coinciden con la literatura [fosfolípidos 23.5 mol% ± 3.5; colesterol total 68.9 mol% ± 7.9; triglicéridos 7.6 mol% ± 3.4]. En el plasma y en las ME de los pacientes con ECV se observó, comparando con los pacientes controles, una disminución de los ácidos grasos poli-no saturados acompañado de un aumento de los ácidos grasos saturados que provocó el descenso del índice de instauración (plasma: 1.67 ± 0.06 vs. 1.28 ± 0.03, P<0.05; ME: 2.28 ± 0.04 vs. 1.25 ± 0.010, P<0.05) y el incremento del cociente AG saturados/insaturados (plasma: 0.35 ± 0.02 vs. 0.52 ± 0.02, P<0.05; ME: 0.45 ± 0.01 vs. 0.83 ± 0.04, P<0.05). Estos datos indicarían una deficiencia de ácidos grasos esenciales. Se observó una elevación en los valores de colesterol total (32.3 ± 0.8 vs. 40.6 ± 2.5, P<0.05) y una disminución de los valores de fosfolípidos (67.7 ± 0.7 vs. 59.4 ± 2.6, P<0.05) en las ME de los pacientes con ECV. Estos hallazgos sugieren cambios en los lípidos de las ME en los pacientes con ECV a pesar de presentar diferencias con respecto a edad, tabaquismo y diabetes. El conocimiento del perfil lipídico de las ME podría constituirse en una herramienta útil para monitorear la evolución de la enfermedad.
Palabras clave: Arteriosclerosis coronaria; Dislipemia; Endarterectomía; Membrana de eritrocito; Ácidos grasos
The increase in the number of patients with diffuse coronary disease and poor distal portion of vessels requires sometimes the use of a complex procedure with multiple graft and endarterectomy of one or more vessels. In our population 28% of patients were revascularized using coronary endarterectomy as a necessary procedure in order to obtain complete revascularization1. The development of tools for detection and assessment of vulnerable atherosclerotic plaque (AP) in different vascular beds is an important goal in current cardiovascular research. Several studies have shown that chemical composition and morphology, rather than anatomy (degree of stenosis), determine AP instability and predict either disease progression or regression2,3. Accumulation of lipids in AP causes progressive narrowing of the arterial lumen, often followed by thrombosis and ischemia. The lipid composition of the plaque is determined by chemical analysis of completely disrupted plaques4-7. However, clinical diagnostic methods that reliably document or predict plaque development or instability have not been described yet. It has been established that monitoring atherosclerotic inflammatory activity could provide important clues for the early diagnosis in individuals with potentially unstable disease3. It is well known that many alterations in plasma lipid patterns are predictors of coronary heart disease (CHD). However, recent papers described the importance of knowing the lipid pattern composition of erythrocyte membrane (EM) such as red blood eicosapentaenoic+docosahexaenoic acids (called Omega-3 index) that could be considered as a new risk factor for death from CHD8-10. The EM lipid composition adjusts to a change in dietary fatty acids (FA) almost completely in ten days11. Recently, it has been reported that EM FA composition may also be preferred over plasma because the n-3 FA family in EM was not affected by recent food consumption8. The erythrocyte lipid pattern was also analyzed in other pathologies such as female obesity12, insulin sensitivity13, chronic alcoholics14 and diabetic retinopathy15. On the other hand, Thies et al.16 described the importance the n-3 FA that are implicated in the prevention of coronary atherosclerosis. N- 3 FA may additionally modulate atherogenesis by affecting processes of endothelial activation and cause a beneficial shift in the eicosanoid system decreasing platelet aggregation and blood pressure17-19.
In our experience we observed different macroscopic characteristics in plaques. In this multifactorial disease, this observation could belong to different stages of the same pathological condition or to dissimilar pattern of coronary disease. The aim of the present work was to analyze the lipid composition of the AP, plasma and EM in patients with CHD, and to compare plasma and EM lipid pattern with the data obtained from healthy control subjects in order to determine the lipid profile of the group of patients with advanced coronary heart disease.
Materials and Methods
The present study is a prospective, double blind trial where we included all patients with CHD who were revascularized with endarterectomy technique. Plaques were obtained from 18 patients (aged 39 to 71 years, 17 men and one woman) who were all hospitalized at the Servicio de Cirugía Cardiovascular, Hospital Francés, Buenos Aires, Argentina. We enrolled all those patients who agreed to participate. Permission for all procedures entailing patients was obtained from Hospital Francés Research Ethics Committee, and all the patients gave written informed consent. Table 1 shows patients' characteristics at study entry. All of them presented very important vascular compromise.
TABLE 1.- Patients characteristics at study entry: with coronary heart disease (CHD) and controls

Plaque samples were taken by endarterectomy procedure, then preserved in chloroform/methanol 2:1 v/v. The control group consisted of 10 age-matched healthy volunteers (4 men, 6 women) who had no history of hematological or heart diseases (see Table 1).
Isolation of EM: blood was collected in test tubes containing an anticoagulant EDTA solution ( Wiener Lab., Rosario, Argentina). Whole blood was centrifuged, the plasma was immediately separated, and packed red blood cells were washed four times with a buffered solution containing NaCl (140 mM), KCl (5 mM), NaHSO4 (1 mM), Tris buffer (10mM), pH 7.4 at 4 °C. After agitation they were kept at 4ºC for 10 min and centrifuged at 16,000 g for 15 min. This procedure was done twice, leaving a substantially hemoglobin-free pellet of EM, which was suspended in a small amount of supernatant and stored (-70 °C) until assayed.
Lipid extraction and analysis: after the endarterectomy procedure, AP was weighed and placed in a tube with chloroform- methanol (2:1 v/v in 3:1 w/v) for 24 h. Plasma, EM and AP lipids were extracted using chloroform-methanol mixture as indicated above. An aliquot from the organic phase was methylated and analyzed using a Hewlett-Packard Model 5840-A gas liquid chromatograph equipped with flame-ionation detector (Hewlett-Packard, California, USA). Another aliquot from the same phase was separated to determine phospholipid and neutral lipid content: free cholesterol; esterified cholesterol; tryglicerides. They were separated by thin layer chromatography (TLC) on Whatman high performance-TLC plates of 20 X 10 cm, using ether petroleum-ether/diethyl-ether/acetic acid (80:20:1 v/v/v). They were quantified by comparison with pure standards (1-5 µg). After staining with ferric chloride, plates were developed at 37 °C for 30 min. The plates were scanned and the densitometry quantification was performed using ID Image Analysis Software (Kodak Rochester, NY, USA)20.
Statistical analysis
The statistical package from Excel 97 (Microsoft Corporation) was used to calculate correlation coefficients according to Spearman, and to perform paired t Test and one-way ANOVAs as appropriate. Data are expressed as mean ± standard error of the mean (SEM). Significance was set at P<0.05.
Results
In Figures 1 and 2: fatty acid profile of plasma and EM from control and CHD patients are presented.

Fig. 1.- Fatty acid composition of plasma from 10 control ![]() patients. Fatty acids are identified by: number of carbon atoms in the chain is given first, value following the double point represents number of double bond (zero means saturated fatty acids); number following "n" indicates the position of the last double bond counting the double bond from the terminal methyl group. Data are the mean ± SEM expressed as % of total fatty acids. (*) Significantly different from controls at P<0.05.
patients. Fatty acids are identified by: number of carbon atoms in the chain is given first, value following the double point represents number of double bond (zero means saturated fatty acids); number following "n" indicates the position of the last double bond counting the double bond from the terminal methyl group. Data are the mean ± SEM expressed as % of total fatty acids. (*) Significantly different from controls at P<0.05.

Fig. 2.- Fatty acid composition of erythrocyte membrane from 10 control![]() patients. Identification of fatty acids as in figure 1. Data are the mean ± SEM expressed as % of total fatty acids. (*) Significantly different from controls at P<0.05.
patients. Identification of fatty acids as in figure 1. Data are the mean ± SEM expressed as % of total fatty acids. (*) Significantly different from controls at P<0.05.
The FA composition of plasma and EM showed an increase in the relative percentage of palmitic and oleic acids in both tissues and an increment of palmitoleic acid in the EM in CHD patients. A significant decrease in n-6 and n-3 families long chain polyunsaturated fatty acids (PUFAs) was noted in both plasma and EM in CHD patients when compared with controls. This result was evidenced by the decrease of the unsaturated index and the increment in the saturated/unsaturated ratio observed in CHD patients (Table 2). This table also shows indirect evidence of desaturase activities in EM. Thus, an increment in ∆9 desaturase activity was inferred through the enhancement 16:1/16:0 and 18:1/18:0 ratios. The decrease in 20:4/18:2 ratio included the ∆6 and ∆5 desaturase activities. Considering that 20:4/20:3 ratio, in which ∆5 desaturase was involved, was not statistically different in CHD patients compared to controls, it was possible to infer that in these patients there was a decrease in ∆6 desaturase activity. Figure 3 shows the FA profile of AP. The major FA were represented by palmitic, oleic and linoleic acids. Stearic and arachidonic acids corresponded to 7% and the remaining FA expressed values below 5%. When EM and AP fatty acid profile was correlated, a statistically significant positive relationship was found, Fig. 4. The Omega-3 index shows a significant decrease in CHD patients, where the control group presented normal values (up to 10%, Fig. 5).
TABLE 2.- Unsaturated index and fatty acid ratios in plasma and EM

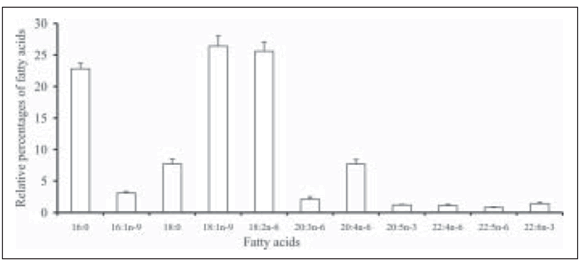
Fig. 3.- Fatty acid composition of atherosclerotic plaque from CHD patients. Identification of fatty acids as in figure 1. Data are the mean ± SEM expressed as % of total fatty acids.

Fig. 4.- Significant correlation of erythrocyte membrane (EM) and atherosclerotic plaque (AP) of total lipids fatty acid composition according to the correlation coefficient by Spearman.

Fig. 5.- Omega-3 index (%)= eicosapentaenoic + docosahexaenoic acids obtained from the fatty acid profile of erythrocyte membranes from 10 controls and 18 CHD patients. Data are the mean ± SEM. (***) Significantly different from controls at P<0.001.
The relative amount of different lipid species in EM and AP is shown in Tables 3 and 4 respectively. A significant decrease in phospholipid as well as an increase in total cholesterol levels and in total cholesterol/phospholipid ratio were found in EM of CHD patients.
Table 4 shows that total cholesterol was the most important component of the AP where it represented over 60%.
TABLA 3.- Total lipid composition in erythrocyte membranes (EM) (mol %)

TABLA 4.- Total lipid composition in the atherosclerotic plaque (AP) (mol %)
Discussion
Patients with CHD exhibited a decrease in PUFAs and an increase in saturated fatty acids (SFAs) and monounsaturated fatty acids (MUFAs) in plasma and EM, indicating an essential FA deficiency. Some investigators have expressed the "double bond" or "unsaturated index" as a crude index of membrane fluidity where the number of double bonds relative to the number of FA was compared. When microviscosity of membranes increased, the unsaturated index decreased21; in this work we observed a significant decrease in the unsaturated index and in the saturated/unsaturated ratio in red blood cells as well as in plasma, suggesting an alteration in the fluidity in the lipid bilayer of red blood cells. Other factors are believed to affect membrane fluidity, including the cholesterol to phospholipid ratio that was increased in the EM of CHD patients.
Our results showed that the EM and plasma were strongly affected in the CHD group in contrast to healthy subjects. A remarkable fact is that we found a similar lipid pattern in plasma and red blood membranes of CHD patients. We also observed a positive correlation between the AP fatty acid composition and the red blood membranes, in spite of patients differences such as age, smoking status, serum lipids, body mass index, pharmacological treatments and sample size. This correlation was not evident in plasma values. The biochemical description of total lipids obtained from AP reported in this work, is in line with that published in the literature, confirming once again the presence of high content of free cholesterol and esterified cholesterol, phospholipid and minor amounts of triglyceride in the examined plaques22, 23. We observed that free cholesterol was more abundant than esterified cholesterol. This could be due to the fact that most analyzed patients were in the end-stage artery disease. The free cholesterol/esterified cholesterol ratio is in line with the literature too, demonstrating an indirect measure of the plaque texture24. As most of the "soft plaques" were esterified cholesterol rich, the ratio of free cholesterol to esterified cholesterol could be one of the key factors for determining the stability of the fibrous cap2. Guyton et al.22 demonstrated, by chemical analysis, that the lesions that contained crystal cholesterol also showed relatively high levels of free cholesterol. Data in the li-terature have shown the mechanism by which lysosomal hydrolysis of esterified cholesterol could contribute to the accumulation of FC in cultured cells, that was exacerbated by inefficient transfer of cholesterol to extralyso-somal locations22-25.
We processed nine calcified AP, four of them belonging to patients with diabetes type II. Burke et al.26 showed that calcified lesions were more prevalent in patients with diabetes type II in comparison to non-diabetic subjects. Moreover, the inflammatory response was greater in diabetic than in non-diabetic plaques. However, in our study, patients with diabetes type II did not present differences in their AP lipid pattern profile when compared with "soft" plaques obtained from patients with diabetes type I and non-diabetic patients.
The high values of oleic and linoleic FA found in AP could show the fact that the FA esterified to cholesterol in the core contained a large amount of linoleic acid, similar to plasma lipoproteins and dissimilar from the oleic acid predominance of cholesteryl ester fatty acids resulting from lipid processing in lesion foam cell. The discrepancy in cholesteryl ester fatty acyl pattern has been confirmed in comparisons of foam cells-rich areas versus core areas within the same microdissected lesions22. The increment of arachidonic acid in AP could be ascribed to the enhancement of the lipoprotein-associated phospholipase A2 activity within the plaque considered as risk factor27.
At early asymptomatic phases of the atherosclerotic process, important chemical changes not only in serum lipids but also in EM, this would contribute to predict the disease progression. Although this is a preliminary study that does not included a large number of patients we conclude that a simple laboratory technique which determines the EM lipid pattern (FA and total lipid) composition could be a complementary tool for monitoring the progression of the CHD in a safe and valid manner to select and evaluate the effect of various intervention therapies.
Acknowledgements: The authors wish to thank M. C. P. de Stringa for her technical assistance. Supported by grants from CONICET, Argentina.
References
1. Levin R, Tolstanol A, Andreu E, Tajes H, Tanus E, Boullon F, et al. Endarterectomía coronaria. Parte 1 (resultados iniciales). Rev Argent Cardiol 1999; 67: 747-52.
2. Peng S, Guo W, Morrisett JD, Johnstone MT, Hamilton JA. Quantification of cholesteryl esters in human and rabbit atherosclerotic plaques by magic-angle spinning (13) CNMR. Arterioscler Thromb Vasc Biol 2000; 20: 2682-8.
3. Weissberg P. Mechanisms modifying atherosclerotic disease - from lipids to vascular biology. Atherosclerosis 1999;147 Suppl 1: S3-10.
4. de Korte CL, Pasterkamp G, van der Steen AF, Woutman HA, Bom N. Characterization of plaque components with intravascular ultrasound elastography in human femoral and coronary arteries in vitro. Circulation 2000; 102: 617-23.
5. Marinello E, Setacci C, Giubbolini M, et al. Lipid composition in atheromatous plaque: evaluation of the lipid three-phase percentage. Life Sci 2003; 72: 2689-94.
6. McGrath LT, Elliott RJ. Lipid analysis and fatty acid profiles of individual arterial atherosclerotic plaques. Anal Biochem 1990; 187: 273-6.
7. Romer TJ, Brennan JF, Schut TC, et al. Raman spectroscopy for quantifying cholesterol in intact coronary artery wall. Atherosclerosis 1998; 141: 117-24.
8. Harris WS, Sands SA, Windsor SL, et al. Omega-3 fatty acids in cardiac biopsies from heart transplantation patients: correlation with erythrocytes and response to supplementation. Circulation 2004; 110: 1645-9.
9. Harris WS, Von Schacky C. The Omega-3 Index: a new risk factor for death from coronary heart disease? Prev Med 2004; 39: 212-20.
10. Sands SA RK, Windsor SL, Harris WS. The Impact of age, body mass index, and fish intake on the EPA and DHA content of human erythrocytes. Lipids 2005; 40: 343-7.
11. Walker BL, Yurkowski M. Effect of cell age on erythrocyte fatty acid composition in rats on different dietary regimes. Biochem J 1967; 103: 218-24.
12. Cazzola R, Rondanelli M, Russo-Volpe S, Ferrari E, Cestaro B. Decreased membrane fluidity and altered susceptibility to peroxidation and lipid composition in overweight and obese female erythrocytes. J Lipid Res 2004; 45: 1846-51.
13. Felton CV, Stevenson JC, Godsland IF. Erythrocytederived measures of membrane lipid composition in healthy men: associations with arachidonic acid at low to moderate but not high insulin sensitivity. Metabolism 2004; 53: 571-7.
14. Parmahamsa M, Reddy KR, Varadacharyulu N. Changes in composition and properties of erythrocyte membrane in chronic alcoholics. Alcohol Alcohol 2004; 39:110-2.
15. Kulacoglu DN, Kocer I, Kurtul N, Keles S, Baykal O. Alterations of fatty acid composition of erythrocyte membrane in type 2 diabetes patients with diabetic retinopathy. Jpn J Ophthalmol 2003; 47: 551-6.
16. Thies F, Garry JM, Yaqoob P, et al. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: a randomised controlled trial. Lancet 2003; 361: 477-85.
17. De Caterina R, Spiecker M, Solaini G, et al. The inhibition of endothelial activation by unsaturated fatty acids. Lipids. 1999; 34 Suppl: S191-4.
18. von Schacky C. The role of omega-3 fatty acids in cardiovascular disease. Curr Atheroscler Rep 2003; 5: 139-45.
19. von Schacky C. Omega-3 fatty acids and cardiovascular disease. Curr Opin Clin Nutr Metab Care 2004; 7: 131-6.
20. Brenner RR, Bernasconi AM, Gonzalez MS, Rimoldi OJ. Dietary cholesterol modulates delta6 and delta9 desaturase mRNAs and enzymatic activity in rats fed a low-eFA diet. Lipids 2002; 37: 375-83.
21. Kaysen GA. Hyperlipidemia of chronic renal failure. Blood Purif 1994; 12: 60-7.
22. Guyton JR, Klemp KF. Development of the lipid-rich core in human atherosclerosis. Arterioscler Thromb Vasc Biol 1996; 16: 4-11.
23. Katz SS, Small DM. Isolation and and partial characterization of the lipid phases of human atherosclerotic plaques. J Biol Chem 1980; 255: 9753-9.
24. Lee RT, Libby P. The unstable atheroma. Arterioscler Thromb Vasc Biol 1997; 17: 1859-67.
25. Tabas I. Cholesterol and phospholipid metabolism in macrophages. Biochim Biophys Acta 2000; 1529: 164-74.
26. Burke AP, Kolodgie FD, Zieske A, et al. Morphologic findings of coronary atherosclerotic plaques in diabetics: a postmortem study. Arterioscler Thromb Vasc Biol 2004; 24: 1266-71.
27. Macphee CH. Lipoprotein-associated phospholipase A2: a potential new risk factor for coronary artery disease and a therapeutic target. Curr Opin Pharmacol 2001; 1: 121-5. [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ]
Received: 24-11-2006
Accepted: 6-07-2007














