Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Medicina (Buenos Aires)
versión impresa ISSN 0025-7680versión On-line ISSN 1669-9106
Medicina (B. Aires) v.68 n.2 Ciudad Autónoma de Buenos Aires mar./abr. 2008
Sex hormone binding globulin decrease as potential pathogenetic factor for hirsutism in adolescent girls
Graciela Cross1, Karina Danilowics1, Martha Kral1, Anne Caufriez2 , Georges Copinschi2, Oscar D. Bruno1
1División de Endocrinología, Hospital de Clínicas, Facultad de Medicina, Universidad de Buenos Aires;
2Laboratory of Physiology, School of Medicine, Université Libre de Bruxelles, Brussels, Belgium
Postal address: Dr. Oscar D. Bruno, División Endocrinología, Hospital de Clínicas, UBA, Av. Córdoba 2351, 5° piso, 1120 Buenos Aires, Argentina Fax: (54-11) 4805-0631 e-mail: divendhcli@intramed.net
Abstract
We investigated 252 non-obese female subjects aged 13-39 years to evaluate if an exaggerated descent of sex hormone binding globulin (SHBG) levels during adolescence can play a role in the development of hirsutism. Body hair was assessed according to Ferriman and Gallwey (FG), with a stringent criterion of normality of < 4. In 13-14 years girls, SHBG and free testosterone (FT) levels were similar in "hirsute" girls (FG > 4) and controls (FG < 4, regular menstrual cycles, no acne). In 15-18 years girls, SHBG values were lower in "hirsute" girls, FT levels were similar in both groups, FG correlated inversely with SHBG. In 19-39 yr women, FT levels were higher in "hirsute" subjects, SHBG values were similar in both groups, FG correlated positively with FT. Lowest SHBG values were observed at 15-18 years, but the slope of the decrease from 1314 years values was greater in the "hirsute" group. FT values increased progressively with age, but the increase was greater in the "hirsute" group. Those results suggest an important role of SHBG decrease in adolescence vs. a more accentuated testosterone increase in adults, as factors conditioning the development of hirsutism in these two different periods of life.
Key words: Hirsutism; Puberty; SHBG; Testosterone; Adolescence
Resumen
Disminución de la globulina transportadora de hormonas sexuales como factor patogénico de hirsutismo en la adolescencia. Se investigaron 252 mujeres con peso normal, de 13 a 39 años de edad, para evaluar si un descenso exagerado en los niveles de la globulina transportadora de hormonas sexuales ("sex hormone binding globulin"; SHBG) puede tener un rol en el desarrollo de hirsutismo. Este signo fue evaluado con la escala de Ferriman y Gallwey (FG), empleando un criterio riguroso de normalidad < 4. En niñas de 13-14 años, tanto SHBG como la testosterona libre ("free testosterone"; FT) fueron similares en niñas "hirsutas" (FG > 4) y controles (FG < 4, ciclos menstruales regulares, sin acné). En adolescentes de 15-18 años, los valores de SHBG fueron menores en las "hirsutas", los niveles de FT fueron similares en ambos grupos y el índice de FG correlacionó inversamente con SHBG. En las mujeres de 19-39 años, los niveles de FT fueron mayores en las "hirsutas", los valores de SHBG fueron similares en ambos grupos y FG correlacionó positivamente con FT. Los valores más bajos de SHBG se observaron entre 15 y 18 años, pero la pendiente de disminución a partir de los valores de 13-14 años fue mayor en el grupo de "hirsutas". Los valores de FT se incrementaron progresivamente con la edad, pero el aumento fue mayor en el grupo de "hirsutas". Estos resultados sugieren un rol importante del descenso de SHBG en la adolescencia vs. un incremento más acentuado de los niveles de testosterona en las adultas, como factores que condicionan el desarrollo del hirsutismo en esos dos diferentes periodos de la vida.
Palabras clave: Hirsutismo; Pubertad; SHBG; Testosterona; Adolescencia
Hyperandrogenism is one of the most frequent endocrine disorders in young women1. Hirsutism, acne, seborrhea, androgenic alopecia, menstrual disturbances and/or ovulatory dysfunction as isolated signs, or in different combinations, are possible clinical expressions of this syndrome. Other endocrine and metabolic alterations, including insulin resistance and obesity, are frequently associated with hyperandrogenic syndrome in polycystic ovary syndrome2.
The development of androgenic features may result from androgen overproduction and/or from increased androgen action in specific tissues. Androgen actions depend in particular on their bioavailability at cellular level, thus on circulating sex hormone-binding globulin (SHBG) levels3, 4. Indeed, androgenic features correlate better with free testosterone and bioavailable testosterone (i.e. non SHBG bound testosterone) than with total testosterone levels4. We have previously reported that SHBG level is a sensitive marker of hirsutism in adolescent girls5.
The aim of the present study was to evaluate if an exaggeration in the normal descent of circulating SHBG levels during puberty and adolescence can play a role in the development of hirsutism.
Materials and Methods
During routine school medical examination at the Carlos Pellegrini school and at the Universidad de Buenos Aires, Buenos Aires, Argentina, blood samples were obtained from 252 post-menarcheal (cut-off time: one year) female, non-obese subjects, in a prospective study design. The protocol was approved by the ethical review committee of the Hospital de Clínicas, University of Buenos Aires, and a written informed consent was obtained from each participant. Subjects with overt neurological, renal, hepatic or thyroid disease, personal history of psychological illness, subjects receiving any kind of medication (including hormonal contraception), and subjects with a body mass index (BMI) > 24 kg/m2 were excluded from the study.
During the visit, information regarding the duration and the regularity of the menstrual cycles as well as the date of the last menses was recorded for each subject. Physical examination included a standardized evaluation of body hair according to the scoring scale of Ferriman and Gallwey (FG score)6. For each site, hair growth was rated from 0 (absence of terminal hair) to 4 (complete and heavy cover). A stringent FG cutoff value (upper limit) of 4 was used to characterize a normal control group. In all subjects, blood samples were obtained at 07:00-10:00 h. In menstruating subjects, blood samples were drawn either before day 11 or after day 18 of the menstrual cycle in order to avoid the periovulatory period; the proportion of samples obtained in follicular and luteal phases (60/40%) was similar for all groups studied (see below). Serum was kept at -20 °C until assay. Testosterone was measured by RIA with a lower limit of sensitivity of 0.02 nmol/l (50 mg/ml) and a mean interassay coefficient of variation of 13%7. SHBG was determined by a commercially available IRMA (Farmos Diagnostica, Finland) with a lower limit of sensitivity of 0.5 nmol/l (4.5 µg/dl) and a mean interassay coefficient of variation of 5.3%. Free testosterone (FT) levels were calculated according to Södergard formula8, 9.
A total of 252 subjects, aged 13-39 yr (mean 19 yr), with a BMI of 20.8 ± 0.1 kg/m2 (mean ± SEM) were included in the study. Their menarcheal age averaged 12.2 ± 0.1 yr. Subjects were divided into three age groups according to their chronological age: 13-14 yr; 15-18 yr; 19-39 yr (referred to as adult group). Within each age group, we compared data obtained in normal control subjects (regular menstrual cycles 26-34 days long, no acne, no seborrhea, FG score < 4) and in subjects with a FG score > 4 (referred to as hirsute subjects). Subjects with a FG score < 4 but with irregular menstrual cycles and/or acne or seborrhea (n = 107) were excluded from group comparisons, but were included in a global analysis of the relationship of FG score to biological variables.
Statistical calculations were performed by non-parametric tests using the StatviewSE+ software (Abacus concepts, Berkeley, CA, USA) for Macintosh computers. All group values are expressed as means ± SEM.
Results
Relationship of FG score to biological variables
In subjects 13-14 yr old, no relationship (Spearman test) was evidenced between FG score and circulating SHBG or androgen levels (P < 0.89; n = 48). In 15-18 yr old girls, FG score was inversely correlated with SHBG levels (r = -0.42; P < 0.004; n = 49) but there was no significant relation between FG score and androgen levels (P < 0.10). In contrast, in adult women (19-39 yr old), FG score was positively associated with total testosterone (r = 0.18; P < 0.03; n = 155) and free testosterone (r = 0.23; P = 0.005; n = 155) while there was no relationship between FG score and SHBG levels (P > 0.30).
Androgen and SHBG levels in hirsute subjects and normal controls
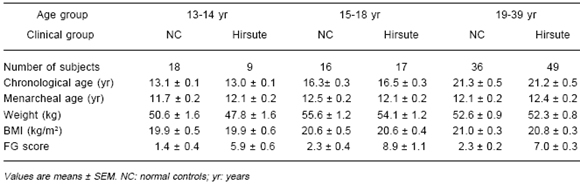
Pertinent clinical data are summarized in Table 1. For each age group, chronological age, menarcheal age, weight and BMI were similar in hirsute subjects and normal controls.
TABLE 1.- Clinical characteristics of each group of subjects
Androgen and SHBG values (means ± SEM) for each age group are shown in Table 2. In 13-14 yr old girls, circulating androgen and SHBG values were similar in hirsute subjects and normal controls. In 15-18 yr old girls, SHBG levels were significantly lower in hirsute subjects than in normal controls, averaging 40.1 ± 3.8 nmol/l and 55.3 ± 4.8 nmol/l, respectively (P < 0.03), while androgen values were similar in both groups. In contrast, in adult women, SHBG values were only slightly, but not significantly, lower in hirsute subjects than in normal controls (54.6 ± 3.4 nmol/l vs. 64.4 ± 5.4 nmol/l; P > 0.20), while free testosterone levels were significantly elevated in the hirsute group (P < 0.03).
TABLE 2.– Comparison of SHBG and androgen values between normal controls and hirsute subjects in each age group

Age-related changes in SHBG levels are further illustrated in Fig.1. A biphasic curve was observed, both in normal and hirsute groups, with lowest SHBG levels found in 15-18 yr girls. The decrease from the values recorded in the 13-14 yr age group was statistically significant in normal (P < 0.03) and in hirsute (P < 0.001) subjects. However, SHBG levels were significantly lower in the hirsute group at 15-18 yr (P <0.03). The magnitude of this decrease was far more pronounced in hirsute girls than in normal controls, averaging 49% and 21%, respectively.
In both normal and hirsute groups, total testosterone levels were similar in the 13-14 and the 15-18 yr age groups (P = 0.30), but increased significantly thereafter. In contrast, free testosterone values increased progressively with age, both in normal and hirsute groups, as illustrated in Fig. 1. However, the magnitude of this increase was considerably lower in normal than in hirsute girls, averaging 53% and 127%, respectively.

Fig. 1.- Plasma levels of SHBG (left panel) and free testosterone (right panel) in normal controls and hirsute subjects in the three age groups. Values shown are means ± SEM. * P < 0.03 vs. age-matched controls (Wilcoxon signed-rank test).
Discussion
Although hirsutism is the most frequent clinical feature of androgen excess, not all patients with hirsutism have overt evidence of androgen excess. Androgen effects depend on their availability at cellular level in specific tissues, and clinical signs of hyperandrogenism correlate better with free than with total testosterone3,4, as observed in adult women in the present study. Thus, the regulation of SHBG levels appears to be an important factor in the possible expression of hyperandrogenism. This study confirms that SHBG level could be a sensitive marker of hirsutism in adolescent girls.
In our study, we found a significant negative correlation between SHBG levels and FG score in adolescent but not in prepubertal or adult women whereas in the last, FG score positively correlated with testosterone concentrations. Those findings point to the eventual role that decreased levels of SHBG could play in the development of hirsutism during adolescence.
Sex hormone binding globulin levels have been shown to decrease from infancy to late prepuberty in healthy girls, and it has been suggested that this change could play a role in pubertal development through an increase in free fractions of sexual steroids10. In a longitudinal study of puberty in girls, a slow but significant decrease in SHBG levels was reported from 8-10 yrs of age to after 15 yrs of age11. On the other hand, insulin resistance occurs at the onset of puberty in normal children12, inducing compensatory hyperinsulinemia13. Insulin resistance returns to near prepubertal levels by the end of puberty14. It has been suggested that in normal prepubertal and pubertal girls, the GH/IGF-1 axis and insulin resistance might be involved in the mechanism of adrenarche in prepubertal girls15. A strong relationship has been evidenced in non-obese women with polycystic ovary syndrome between insulin sensitivity and SHBG levels16, and it has been suggested that SHBG level could be a marker of insulin secretion and/or insulin action17. The above mentioned normal decrease in insulin resistance together with an increased production of estrogens could explain why the differences observed in SHBG levels during adolescence are not seen in adult women.
The present study confirms the decrease of SHBG from early to late puberty: a decrease of circulating SHBG levels from the 13-14 yr age group to the 15-18 yr age group was observed both in normal and in hirsute girls. However, this decrease was more than twice as pronounced in our normal weight hirsute subjects than in our normal controls, suggesting that an exaggeration of the normal descent of circulating SHBG levels during puberty and adolescence could play a role in the development of hirsutism, possibly by inducing slight, and sometimes undetectable, elevations of free testosterone. Because of the design of the study (one single blood sample in non-fasted subjects during routine school medical examination), the relationship between insulin and SHBG could not be investigated in the present study.
In our subjects, free testosterone levels increased progressively with age, both in normal and in hirsute subjects, but the magnitude of this increase was much higher in hirsute girls, resulting in higher free testosterone values in hirsute than in normal adults.
In conclusion, the present results strongly suggest a major, though subtle, role for the decrease of SHBG in adolescence as against a more accentuated increase of testosterone in adults, as factors conditioning the development of hirsutism signs in these two different periods of life.
Acknowledgements: We thank Dr. Fabienne Mestrez for valuable collaboration, and the volunteers for generously participating in this study. This work was supported in part by research grants from the Secretary of Science and Technology, Universidad de Buenos Aires, from the Belgian Fonds de la Recherche Scientifique Médicale, and from the Université Libre de Bruxelles.
This study was presented in part at the 11th International Congress of Endocrinology, Sydney, Australia, October-November 2000.
1. Azziz R, Sanchez LA, Knochenhauer ES, et al. Androgen excess in women: experience with over 1000 consecutive patients. J Clin Endocrinol Metab 2004; 89: 453-62. [ Links ]
2. Dunaif A. Insulin resistance and the polycystic ovary syndrome: mechanism and implications for pathogenesis. Endocr Rev 1997; 18: 774-800. [ Links ]
3. Rosner W. Plasma steroid-binding proteins. Endocrinol Metab Clin North Am 1991; 20: 697-720. [ Links ]
4. Carlstrom K, Gershagen S, Rannevik G. Free testosterone and testosterone/SHBG index in hirsute women: a comparison of diagnostic accuracy. Gynecol Obstet Invest 1987; 24: 256-61. [ Links ]
5. Caufriez A, Arebalo-Cross, G, Kral, M, et al. A decrease in sex hormone-binding globulin is a sensitive marker of hirsutism in adolescent girls. Abstracts of the 74th Annual Meeting of the Endocrine Society, 1992, p 278. [ Links ]
6. Ferriman D, Gallwey JD. Clinical assessment of body hair growth in women. J Clin Endocrinol Metab 1961; 21: 1440-7. [ Links ]
7. Delbeke D, Lejeune-Lenain C. Simultaneous radio-immunoassay measurement of testosterone (T) and dihydrotestosterone (DHT) without chromatography. Ann Biol Clin (Paris) 1982; 40: 579-84. [ Links ]
8. Sodergard R, Backstrom T, Shanbhag V, Carstensen H. Calculation of free and bound fractions of testosterone and estradiol-17 beta to human plasma proteins at body temperature. J Steroid Biochem 1982; 16: 801-10. [ Links ]
9. Belgorosky A, Escobar ME, Rivarola MA. Validity of the calculation of non-sex hormone-binding globulin-bound estradiol from total testosterone, total estradiol and sex hormone-binding globulin concentrations in human serum. J Steroid Biochem 1987; 28: 429-32. [ Links ]
10. Belgorosky A, Rivarola MA. Progressive increase in nonsex hormone-binding globulin-bound testosterone and estradiol from infancy to late prepuberty in girls. J Clin Endocrinol Metab 1988; 67: 234-7. [ Links ]
11. Apter D, Bolton NJ, Hammond GL, Vihko R. Serum sex hormone-binding globulin during puberty in girls and in different types of adolescent menstrual cycles. Acta Endocrinol (Copenh) 1984; 107: 413-9. [ Links ]
12. Amiel SA, Sherwin RS, Simonson DC, Lauritano AA, Tamborlane WV. Impaired insulin action in puberty. A contributing factor to poor glycemic control in adolescents with diabetes. N Engl J Med 1986; 315: 215-9. [ Links ]
13. Caprio S, Plewe G, Diamond MP, et al. Increased insulin secretion in puberty: a compensatory response to reductions in insulin sensitivity. J Pediatr 1989; 114: 963-7. [ Links ]
14. Moran A, Jacobs DR, Jr., Steinberger J, et al. Insulin resistance during puberty: results from clamp studies in 357 children. Diabetes 1999; 48: 2039-44. [ Links ]
15. Guercio G, Rivarola MA, Chaler E, Maceiras M, Belgorosky A. Relationship between the growth hormone/insulin-like growth factor-I axis, insulin sensitivity, and adrenal androgens in normal prepubertal and pubertal girls. J Clin Endocrinol Metab 2003; 88: 1389-93. [ Links ]
16. Cibula D, Skrha J, Hill M, et al. Prediction of insulin sensitivity in nonobese women with polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87: 5821-5. [ Links ]
17. Pugeat M, Cousin P, Baret C, Lejeune H, Forest MG. Sex hormone-binding globulin during puberty in normal and hyperandrogenic girls. J Pediatr Endocrinol Metab 2000; 13 (Suppl 5): 1277-9. [ Links ]
Received: 7-5-2007
Accepted: 23-11-2007














