Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Medicina (Buenos Aires)
Print version ISSN 0025-7680
Medicina (B. Aires) vol.74 no.2 Ciudad Autónoma de Buenos Aires Apr. 2014
ARTÍCULO ESPECIAL
Rheumatoid arthritis associated interstitial lung disease: a review
Deborah Assayag, Joyce S. Lee, Talmadge E. King, Jr.
Division of Pulmonary, Critical Care, Allergy and Sleep Medicine; Department of Medicine, University of California San Francisco (UCSF), San Francisco, CA, USA
Postal address: Dr. Deborah Assayag, University of California San Francisco (UCSF), 505 Parnassus Avenue, Room M1093, San Francisco, CA, 94143-0111.
Phone: 001 415-502-2361 e-mail: Deborah.Assayag@ucsf.edu
Abstract
Rheumatoid arthritis is a common inflammatory disease affecting about 1% of the population. Interstitial lung disease is a serious and frequent complication of rheumatoid arthritis. Rheumatoid arthritis associated interstitial lung disease (RA-ILD) is characterized by several histopathologic subtypes. This article reviews the proposed pathogenesis and risk factors for RA-ILD. We also outline the important steps involved in the work-up of RA-ILD and review the evidence for treatment and prognosis.
Key words: Rheumatoid arthritis; Interstitial lung disease; Pathogenesis; Risk factors
Resumen
La artritis reumatoide asociada a la enfermedad intersticial pulmonar: una revisión. La artritis reumatoide es una enfermedad inflamatoria común, que afecta cerca del 1% de la población. La enfermedad pulmonar intersticial es una complicación frecuente y seria de la artritis reumatoide y esta asociación está caracterizada por diferentes subtipos histológicos. Este artículo analiza la patogénesis y los factores de riesgo de esta asociación. Además señala los esfuerzos realizados y las evidencias para el diagnóstico y tratamiento.
Palabras clave: Artritis reumatoide; Enfermedad intersticial pulmonar; Patogénesis; Factores de riesgo
Rheumatoid arthritis (RA) is a common systemic inflammatory disorder of unknown etiology. It is characterized by symmetric arthritis and synovial inflammation leading to progressive joint erosion and eventually, deformity1.The world-wide prevalence of RA ranges from 0.5% to 1%, while the incidence is estimated to be 30 cases per 100,000 people per year, and varies based on gender, population and ethnicity2, 3.In the United States, there is some evidence that the incidence and the prevalence of RA has been increasing over the past 20 years4.
Women are more commonly affected than men. This gender imbalance has been observed across different ethnic populations, as well as in familial studies5. There are important clinical differences between men and women with RA. Men have later RA disease onset. They are more likely to have positive anti-cyclic citrullinated peptide (CCP) antibodies, and higher titers. Men are more likely to have a history of smoking and a higher prevalence of extra-articular manifestations6. One explanation for this difference between men and women is genetic variability causing increased susceptibility to autoimmunity6. In addition, there may be hormonal factors contributing to this increased susceptibility. Early menarche and irregular menses are associated with increased risk of RA, while breast feeding seems to be protective against the development of RA7. Men with RA have been shown to have lower levels of bio-available testosterone, with low levels of luteinizing hormone. This suggests that low testosterone levels may play a role in the pathogenesis of RA8.
The etiology of RA is unknown. Known genetic susceptibility are thought to predispose to RA, which can be triggered by environmental exposures such as smoking and air pollutants9.Different genetic loci have been found to be significantly associated with presence of RA in genome wide association studies and replications cohorts10,11. Presence of HLA-DR alleles within the major histo-compatibility complex are strongly associated with RA and can account for about one third of the genetic susceptibility to the disease12.
The disease burden of RA is significant. Patients with RA have lower wages, more missed workdays, lower employment rates and higher limitations at work and home compared to healthy adults. Even after adjusting for age, sex, gender, income and education, people with RA were 53% less likely to be employed compared to those without RA13.
The disease burden is even greater in patients who develop extra-articular manifestations of RA, which affects approximately 40% of patients14. Pulmonary involvement is a common extra-articular feature of RA. The chest manifestations are varied and include pleuritis and pleural effusions, airways disease such as bronchiolitis obliterans, rheumatoid nodules and interstitial lung diseases (ILD)15.
Interstitial lung diseases are a group of diseases characterized by fibrosis and inflammation of the pulmonary interstitium. Interstitial lung disease can be secondary to RA, but can also be caused by the medications used to treat RA. Several anti-inflammatory and biologic drugs have been associated with the development of ILD16. The focus of this review will be RA associated ILD (RA-ILD), including the epidemiology and pathogenesis, work-up and treatment, and prognosis of RA-ILD.
Rheumatoid arthritis associated interstitial lung disease
Epidemiology
The prevalence of RA-ILD has been reported to be between 4 and 50%17-21. The variability in the reported prevalence can be attributed to the method of detection used in each study. Studies in which a higher proportion of subjects are found to have ILD are those in which the lung involvement is identified on computed tomography (CT) scan. Computed tomography scanning has been shown to be highly sensitive for detecting ILD and can identify patients with asymptomatic and subclinical disease. One study identified 33% of subjects with early RA to have abnormal CT scans, but only 6% had an abnormal chest radiograph22. A significant number of those subjects have mild abnormalities that are subclinical. While some of these patients have progression of RA-ILD, others have disease that never becomes clinically significant22, 23.
The 30-year cumulative incidence of developing clinically significant RA-ILD in cohorts of RA subjects is between 6 and 8%14, 24.One study looking at the prevalence of ILD in U.S. decedents with RA found a prevalence of RA-ILD of 6.6% at the time of death25. Results of this study also suggested that the age-adjusted mortality rates from RA-ILD have been increasing over the past 25 years in women, despite decreasing mortality rates from RA alone. Patients with RA-ILD have a median survival after ILD diagnosis of 2.6 years, which translates to a threefold higher risk for death than for RA patients without ILD24.
Etiology and pathogenesis
The etiology of RA-ILD is unknown. It is likely that most patients have some underlying genetic predisposition to fibrosis that is triggered by some form of injury to the lung. Some of the genetic polymorphisms that have been studied in RA have been studied in patients with RA-ILD. Specifically, certain polymorphisms of the human leukocyte antigen (HLA)-DRB shared epitope have been associated with increased risk of ILD (e.g. HLA-DRB1*1502), while others appear to be protective against the development of ILD (e.g. HLA-DRB1*04, DQB1*04 and DRB1*16)26.One study investigated the association between the MUC5B polymorphism, a polymorphism associated with idiopathic pulmonary fibrosis (IPF), and the presence of RA-ILD, but found no significant association27.
A new mouse model for RA has been developed with a single abnormality in the skg gene. These SKG mice develop a spontaneous autoimmune CD4+T cell mediated illness that resembles RA joint disease andits extra-articular manifestations28. They develop cellular and fibrotic interstitial pneumonia when subjected to cigarette smoke and bleomycin, while normal mice do not29. This suggests that both genetic predisposition and lung injury with inflammation are required for the development of ILD in RA.
Risk factors
Risk factors have been identified in the development of RA-ILD. Some risk factors are patient-specific while others are related to the RA itself. ILD has been shown to be more common in males compared to females in some studies21, 30,but not in others31, 32.Older age is also associated with increased risk of ILD in RA. One study found that for each 10-year increase in age, the likelihood of having ILD increased by 64%32. In another study, age above 65 years increased the risk of ILD more than four-fold31.
Smoking is a risk factor for the development of RA33.Risk of developing RA increases with increasing numbers of pack-years smoked9. It is also an important modifiable risk factor for the development of ILD in this population. Cigarette smoking was found to be associated with presence of radiographic interstitial abnormalities as well as with reduced pulmonary function, both diffusion capacity for carbon monoxide (DLCO) and forced vital capacity (FVC)34.
There is also data to suggest that the severity of RA may be related to the development of ILD. Presence of erosive joint disease, high levels of erythrocyte sedimentation rate (ESR) and presence of rheumatoid nodules are risks factors for the development of ILD24, 32.In one study, high titers of rheumatoid factor (>100 IU/mL) significantly increased the risk of RA-ILD, while there was a trend towards high levels of anti-CCP antibodies and RA-ILD31. A recent preliminary study showed that higher levels of a variety of specific anti-citrullinated peptide antibodies and an expandedrepertoire of these antibodies were present in patients with RA-ILD withlung function abnormalities35.This suggested a link between autoimmunity against citrullinated proteins and RA-ILD.
Diagnosis
Usually, RA-ILD is diagnosed in patients with RA presenting with symptoms suggestive of lung disease, such as dyspnea (on exertion or at rest) or cough. They are often found to have pulmonary function impairment or abnormal chest imaging. Rarely, RA-ILD will be diagnosed on surgical lung biopsy.
Other causes of lung disease in RA should be excluded. The differential diagnosis for lung disease includes drug toxicity and infection. Drugs that have been associated with ILD in RA include disease-modifying agents such as methotrexate, leflunomide, gold, and cyclophosphamide, as well as biologics, includinganti-TNF-alpha agents.[36, 37]Infection, especially atypical infections (e.g., Mycobacterium species, Pneumocystis jirovecii), should also be ruled out38.
Subtypes of RA-ILD
Several histopathologic subtypes of ILD can be observed in patients with RA. The most common subtypes are the usual interstitial pneumonia pattern (UIP) and the non-specific interstitial pneumonia pattern (NSIP)39,40. Other patterns of ILD seen in RA include organizing pneumonia (OP), lymphocytic interstitial pneumonia (LIP), desquamative interstitial pneumonia (DIP), and diffuse alveolar damage (DAD).
There is increasing data to suggest that RA-ILD patients with a UIP pattern may have a distinct clinical phenotype and natural history compared to the other histopathologic phenotypes observed in RA-ILD, primarily those with NSIP (Table 1). RA-ILD patients with the UIP pattern tend to be older, more likely to be male, and have a more significant smoking history21,30,39-42. In contrast, those without the UIP pattern tend to be younger, more likely to be women, and have a less significant smoking history. What is most compelling is the observed survival difference between RA-ILD patients with and without a UIP pattern. Patients with RA-ILD and a UIP pattern appear to have a worse survival compared to those without a UIP pattern41,43. In addition, the RA-ILD patients with UIP pattern have a similar clinical phenotype and natural history seen in patients with IPF, a disease characterized by idiopathic UIP pattern.
TABLE 1.- Differences between UIP and non-UIP in RA-ILD

Abbreviations: UIP, usual interstitial pneumonia; CCP, cyclic citrullinated peptide; PFT, pulmonary function tests.
Work-up of RA-ILD
The steps involved in the work-up of patients with RA-ILD are summarized in Table 2.
TABLE 2.- Work-up for rheumatoid-arthritis related interstitial lung disease
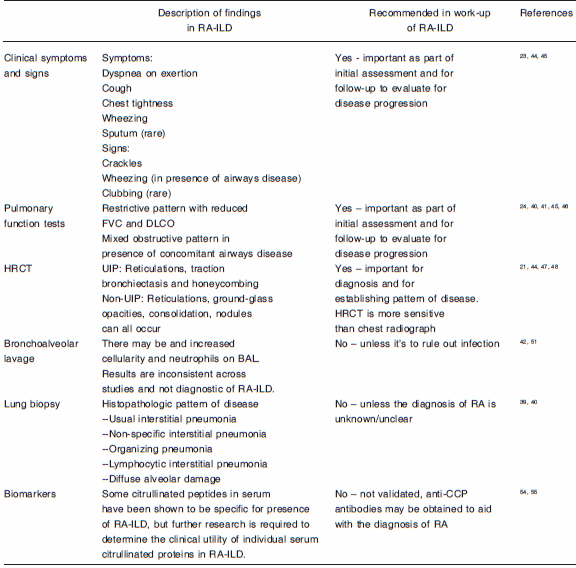
Abbreviations: RA-ILD, rheumatoid arthritis associated interstitial lung disease; FVC, forced vital capacity; DLCO, diffusion capacity for carbon monoxide; HRCT, high resolution computed tomography of the chest; UIP, usual interstitial pneumonia; CCP, cyclic citrullinated peptides.
Clinical symptoms and signs: Clinical symptoms suggesting RA-ILD are non-specific. Dyspnea, usually on exertion, is the most frequently reported symptom. Because of physical limitations from articular disease, exertional dyspnea may not be apparent in the early stages of the disease. Cough, sputum production, wheezing and chest pain can also be reported44, 45.A subset of patients can have subclinical disease, which is apparent on CT scan, but patients are clinically asymptomatic46.Patients who have abnormal pulmonary function tests are more likely to report respiratory symptoms45.
Pulmonary function tests: About one third of all RA patients will have pulmonary function abnormalities45.Pulmonary function tests (PFTs) in RA can be used as screening tools for early RA-ILD and a method to assess for disease progression. Patients most commonly have a restrictive pattern on their PFTs, like in other ILDs. A mean FVC between 60 and 70% of predicted has been reported in cohort studies of RA-ILD, with a reduced DLCOof40 to 60% of predicted values40, 41.
Computed tomography imaging: High resolution CT scan is essential in evaluating RA-ILD.Figure 1 illustrates different radiographic abnormalities observed. Most studies assessing CT abnormalities in RA-ILD did not differentiate between the different subtypes of ILD. The main abnormalities observedare reticulations and ground glass opacities21, 44, 47.When severe and extensive, interstitial fibrosis leads to parenchymal architectural distortion, traction bronchiectasis, and honeycombing48. Nodules that are centrilobular or perilymphatic can also be seen in RA-ILD, either alone, or with reticulations47.
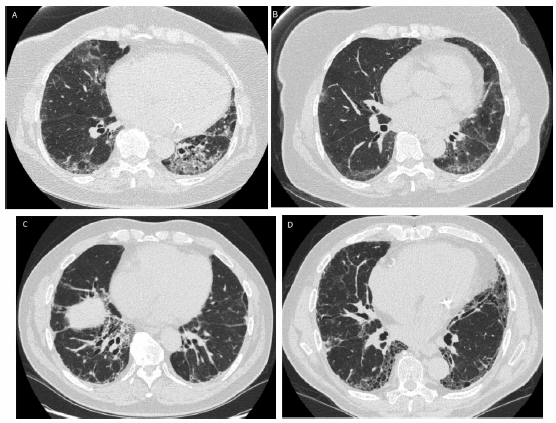
Fig. 1.- CT abnormalities in patients with RA-ILD.Figure illustrates different patterns and abnormalities that can be seen on chest computed tomography: a. Bilateral, basilar predominant ground glass opacities and consolidation; b. Ground glass opacities with reticulations; c. Bilateral reticulations and traction bronchiectasis; d. Honeycombing with reticulations, consistent with the usual interstitial pneumonia pattern
The UIP pattern in RA-ILD shares the same definition as in the idiopathic interstitial pneumonias (IIPs). It is characterized by presence of subpleural, basilar predominant reticulations, traction bronchiectasis, and honeycombing with absence of nodules, ground glass opacities or consolidation49.Similar to the IIP literature, the radiologic pattern ofa definite UIP pattern on CT scan has been shown to be highly specific and moderately sensitivefor the UIP pattern on histopathology in patients with RA-ILD50 (Fig. 1).
Bronchoscopy and bronchoalveolar lavage:There is little role for bronchoscopy and bronchoalveolar lavage (BAL) in the diagnostic algorithm of RA-ILD. One study found increased cellularity in BAL compared to normal controls, but not when compared with RA patients without ILD51. In another study, BAL showed an increase in neutrophil percentage, but this correlated poorly with extent of disease on CT scan42.BAL can be useful in patients who develop acute or subacute respiratory worsening in the context of RA-ILD in order to rule out opportunistic infection.
Surgical lung biopsy:Surgical lung biopsy as a diagnostic tool is generally not performed in the context of connective tissue diseases (CTD), unless the diagnosis of CTD is unclear or not established. In contrast to the IIPs, where histopathologic subtype is critical to the management of the ILD, ascertaining histopathologic subtype in CTD-ILD, including RA-ILD, is not done. This is primarily because management and treatment of RA-ILD is irrespective of histopathologic phenotype at this time.
Biomarkers: At this time, no biomarker has been shown to be sensitive or specific to RA-ILD disease activity or disease progression. KL-6 (Krebs von den Lungen-6)is a glycoprotein expressed by type-II pneumocytes and epithelial cells. It is a serum indicator for pulmonary fibrosis that has been evaluated in RA-ILD. It was shown to be correlated with reticular opacities and honeycombing on CT scan52. However, KL-6 has poor sensitivity in RA-ILD53.
Protein citrullination is thought to be associated with extra-articular manifestations of RA, including RA-ILD54.Serum citrullinated proteins Hsp90 isoforms a and β were found to be highly specific for RA-ILD in a cohort of RA patients, but poorly sensitive55. This suggests that they may be biomarkers for RA-ILD, although correlation with disease activity and severity has not yet been established.
Treatment
The management of RA-ILDin patients with subclinical disease remains unclear. It is not known whether treatment will alter the course of the disease in these patients.The mainstay of treatment in RA-ILD is immunosuppression. First line therapy is prednisone at high doses, for prolonged duration. Observational studies suggest that there may be benefit in using other forms of immunosuppression, such as steroid sparing agents, in the treatment of RA-ILD.In a cohort of 40 patients with RA-ILD, the addition of disease-modifying anti-rheumatic drugs (DMARDS) to tapering doses of prednisone was associated with improvement in baseline FVC. The DMARDS used by treating physicians in this study included methotrexate, leflunomide and azathioprine56.There was no difference in pulmonary function improvement between the different agents used. In a subgroup analysis, patients with a lower fibrosis score on CT were the ones who had a response to treatment. In this study, patients with RA-UIP had a higher mean fibrosis score. In another cohort of patients, thepresence of ILD was associated with an increasedodds of developing leflunomide induced pneumonitis and ILD (OR 8.2)57.
Anti-TNF alpha agents have been used in RA for several years to treat articular disease. There were reports of increased risk of developing ILD in RA patients using these biologic agents58. However, several studies have since demonstrated that these agents do not cause increased risk of developing RA-ILD36, 59.
No study thus far has looked at response to treatment according to histopathologicsubtype of RA-ILD. Lung transplant in end stage RA-ILD is being performed in some centers, but no long-term data has been published.
Prognosis
Patients with RA-ILD have an increased mortality compared to patients with RA but no evidence of ILD60.Median survival ranges between 3 and 8 years from time of diagnosis of RA-ILD40, 41, 43.Older age, male sex and greater impairment in pulmonary function at diagnosis have been associated with worse prognosis32, 37, 41. In one study, greater RA disease severity was associated with an increased hazard of death in RA-ILD37.Presence of rheumatoid factor and high LDH have also been shown to confer a poorer prognosis61.
Patients with RA-ILD may experience bouts of acute exacerbations (AE) similar to those reported in other patients with ILD, especially those with RA-ILD and a UIP pattern (RA-UIP)62, 63.The etiology of AE in RA-ILD is unknown. These events are associated with older age at ILD diagnosis, a UIP pattern on HRCT and methotrexate usage. Acute exacerbations are also associated witha worse prognosis in RA-ILD63.
As previously mentioned, the underlying histopathologic and/or radiologic phenotype may inform prognosis in RA-ILD. In general, the extent of honeycombing and severity of traction bronchiectasis appear to be independently associated with increasing mortality in fibrotic ILD associated with CTD, especially in patients with RA-ILD64.Furthermore, studieshave demonstrated that RA-ILD patients with a UIP pattern have a worse prognosis compared to those without UIP41, 43, 63. In one of those studies, the survival in patients with RA-UIP was similar to patients with IPF41.However, another study demonstrated that when IPF and RA-UIP patients are matched for age, sex, smoking and baseline lung function, RA-UIP patients havea longersurvival timecompared to IPFpatients (median survival, 53 vs. 41 months in RA-UIP vs IPF,respectively, p = 0.015)62.Another study showed no difference in survival between the UIP and NSIP forms of RA-ILD, but this may be due to limited sample size. Regardless, survival in patients with fibrotic ILD wasreduced compared to non-fibrotic RA-ILD in this study40.
ILD is a frequent complication of RA and is associated with increased morbidity and mortality. There are distinct histopathologicsubtypes of RA-ILD, which appear to have different clinical phenotypes and natural histories.At this time, it remains unclear if there is benefit to classifying patients by their histopathologic subtype. However, if the pathobiology of UIP in RA-ILD is more similar to that of IPF than in non-UIP forms of RA-ILD, it may be that we are harming a subset of RA-ILD patients with our generalized approach to treatment of RA-ILD65. Further research investigating the differences between those with and without UIP pattern in RA-ILD are needed as it may shift our current paradigm of RA-ILD management and treatment to be more in line with what has been established in the idiopathic interstitial pneumonias.
Conflict of interest: The authors report no conflict of interest related to this manuscript
1. McInnes IB, G. Schett, The pathogenesis of rheumatoid arthritis. N Engl J Med 2011; 365: 2205-19. [ Links ]
2. Spector TD. Rheumatoid arthritis. Rheum Dis Clin North Am 1990; 16: 513-37. [ Links ]
3. Gabriel SE. The epidemiology of rheumatoid arthritis. Rheum Dis Clin North Am 2001; 27: 269-81. [ Links ]
4. Myasoedova E, Crowson CS, Kremers HM, Therneau TM, Gabriel SE. Is the incidence of rheumatoid arthritis rising?: Results from Olmsted County, Minnesota, 1955-2007. Arthritis & Rheumatism 2010; 62: 1576-82. [ Links ]
5. Barragán-Martínez C, Amaya-Amaya J, Pineda-Tamayo R, et al.Gender differences in Latin-American patients with rheumatoid arthritis. Gender Medicine 2012; 9: 490-510.e5. [ Links ]
6. Jawaheer D,Lum RF, Gregersen PK, Criswell LA. Influence of male sex on disease phenotype in familial rheumatoid arthritis. Arthritis Rheumatism 2006; 54: 3087-94. [ Links ]
7. Karlson EW, Mandl LA, Hankinson SE, Grodstein F. Do breast-feeding and other reproductive factors influence future risk of rheumatoid arthritis?: Results from the Nurses' Health Study. Arthritis Rheumatism 2004; 50: 3458-67. [ Links ]
8. Tengstrand B, Carlström K, Hafström I. Bioavailable testosterone in men with rheumatoid arthritis-high frequency of hypogonadism. Rheumatology 2002; 41: 285-9. [ Links ]
9. Hoovestol R. Mikuls TR. Environmental exposures and rheumatoid arthritis risk. Curr Rheumatol Rep 2011; 13: 431-9. [ Links ]
10. Plenge RM, Padyukov L, Remmers EF, et al. Replication of putative candidate-gene associations with rheumatoid arthritis in >4,000 samples from North America and Sweden: association of susceptibility with PTPN22, CTLA4, and PADI4. Am J Hum Genet 2005; 77: 1044-60. [ Links ]
11. Eyre S, Bowes J, Diogo D, et al. High-density genetic mapping identifies new susceptibility loci for rheumatoid arthritis. Nat Genet 2012; 44: 1336-40. [ Links ]
12. Deighton CM, Walker DJ, Griffiths ID, Roberts DF,et al.The contribution of HLA to rheumatoid arthritis. Clin Genet 1989; 36: 178-82. [ Links ]
13. Sullivan PW, Ghushchyan V, Huang XY, Globe DR. Influence of rheumatoid arthritis on employment, function, and productivity in a nationally representative sample in the United States. J Rheumatol 2010; 37: 544-9. [ Links ]
14. Turesson C, O'Fallon WM, Crowson CS, Gabriel SE, Matteson EL. Extra-articular disease manifestations in rheumatoid arthritis: incidence trends and risk factors over 46 years. Ann Rheum Dis 2003; 62: 722-7. [ Links ]
15. Tanoue LT. Pulmonary manifestations of rheumatoid arthritis. Clin Chest Med 1998; 19: 667-85. [ Links ]
16. American Thoracic Society/European Respiratory Society International Multidisciplinary Consensus Classification of the Idiopathic Interstitial Pneumonias. Am J Respir Crit Care Med 2002; 165: 277-304. [ Links ]
17. Richman NC, Yazdany J, Graf J, Chernitskiy V, Imboden JB.Extraarticular manifestations of rheumatoid arthritis in a multiethnic cohort of predominantly Hispanic and Asian patients. Medicine (Baltimore) 2013; 92: 92-7. [ Links ]
18. Norton S, Koduri G, Nikiphorou E, Dixey J, Williams P, Young A. A study of baseline prevalence and cumulative incidence of co-morbidity and extra-articular manifestations in RA and their impact on outcome. Rheumatology 2013; 52:99-110. [ Links ]
19. Dawson JK, Fewins HE, Desmond J, Lynch MP, Graham DR.Fibrosingalveolitis in patients with rheumatoid arthritis as assessed by high resolution computed tomography, chest radiography, and pulmonary function tests. Thorax 2001; 56: 622-627. [ Links ]
20. Zrour SH, Touzi M, Bejia I, et al. Correlations between high-resolution computed tomography of the chest and clinical function in patients with rheumatoid arthritis: Prospective study in 75 patients. Joint Bone Spine 2005; 72: 41-7. [ Links ]
21. Zou YQ, Li YS, Ding XN, Ying ZH. The clinical significance of HRCT in evaluation of patients with rheumatoid arthritis-associated interstitial lung disease: a report from China. Rheumatology Intern 2012; 32: 669-73. [ Links ]
22. Gabbay E, Tarala R, Will R, Carroll G, Adler B, Cameron D, Lake FR. Interstitial lung disease in recent onset rheumatoid arthritis. Am J Respir Crit Care Med 1997; 156: 528-35. [ Links ]
23. Gochuico BR, Avila NA, Chow CK, et al. PRogressive preclinical interstitial lung disease in rheumatoid arthritis. Arch Int Med 2008; 168: 159-66. [ Links ]
24. Bongartz T, Nannini C, Medina-Velasquez YF, et al. Incidence and mortality of interstitial lung disease in rheumatoid arthritis: a population-based study. Arthritis Rheumatism 2010; 62: 1583-91. [ Links ]
25. Olson AL, Swigris JJ, Sprunger DB, et al. Rheumatoid arthritis-interstitial lung disease-associated mortality. Am J Resp Critical Care Med 2011; 183: 372-8. [ Links ]
26. Furukawa H, Oka S, Shimada K, et al. Association of human leukocyte antigen with interstitial lung disease in rheumatoid arthritis: a protective role for shared epitope. PLoS One 2012; 7: e33133. [ Links ]
27. Johnson C. Exploration of the MUC5B polymorphism frequency in rheumatoid arthritis interstitial lung disease. Am J Respir Crit Care Med 2013; 187: A5982. [ Links ]
28. Sakaguchi, S. SKG mice, A new genetic model of rheumatoid arthritis. Arthritis Res Ther 2003; 5(Suppl 3): 10. [ Links ]
29. Keith RC, Powers JL, Redente EF, et al. A novel model of rheumatoid arthritis-associated interstitial lung disease in SKG mice. Exp Lung Res 2012; 38: 55-66. [ Links ]
30. Aubart F, Crestani B, Nicaise-Roland P, et al. High levels of anti-cyclic citrullinated peptide autoantibodies are associated with Co-occurrence of pulmonary diseases with rheumatoid arthritis. J Rheumatol 2011; 38: 979-82. [ Links ]
31. Mori S, Koga Y, Sugimoto M. Different risk factors between interstitial lung disease and airway disease in rheumatoid arthritis. Respir Med 2012; 106: 1591-9. [ Links ]
32. Koduri G, Norton S, Young A, et al. Interstitial lung disease has a poor prognosis in rheumatoid arthritis: results from an inception cohort. Rheumatology 2010; 49: 1483-9. [ Links ]
33. Bergström U, Jacobsson LT, Nilsson JÅ, Berglund G, Turesson C. Pulmonary dysfunction, smoking, socioeconomic status and the risk of developing rheumatoid arthritis. Rheumatology 2011; 50: 2005-13. [ Links ]
34. Saag KG, Kolluri S, Koehnke RK, et al. Rheumatoid arthritis lung disease: Determinants of radiographic and physiologic abnormalities. Arthritis Rheumatism 1996; 39: 1711-9. [ Links ]
35. Giles JT, Danoff SK, Sokolove J, et al. Association of fine specificity and repertoire expansion of anticitrullinated peptide antibodies with rheumatoid arthritis associated interstitial lung disease. Ann Rheum Dis 2013 (Epub date 2013/05/30). [ Links ]
36. Picchianti Diamanti A, Germano V, Bizzi E, Laganà B, Migliore A.Interstitial lung disease in rheumatoid arthritis in the era of biologics. Pulm Med 2011; 2011/931342. [ Links ]
37. Dixon WG, Hyrich KL, Watson KD, et al. Influence of anti-TNF therapy on mortality in patients with rheumatoid arthritis-associated interstitial lung disease: results from the British Society for Rheumatology Biologics Register. Ann Rheumatic Dis 2010; 69: 1086-91. [ Links ]
38. Hamblin MJ, Horton MR. Rheumatoid arthritis-associated interstitial lung disease: diagnostic dilemma. Pulm Med 2011; 2011: 872120. [ Links ]
39. Lee HK, Kim DS, Yoo B, et al. Histopathologic pattern and clinical features of rheumatoid arthritis-associated interstitial lung disease. Chest 2005; 127: 2019-27. [ Links ]
40. Solomon JJ, Ryu JH, Tazelaar HD, et al., Fibrosing interstitial pneumonia predicts survival in patients with rheumatoid arthritis-associated interstitial lung disease (RA-ILD). Respir Med 2013; 107: 1247-52. [ Links ]
41. Kim EJ, Elicker BM, Maldonado F, et al. Usual interstitial pneumonia in rheumatoid arthritis-associated interstitial lung disease. Euro Respir J 2010; 35: 1322-28. [ Links ]
42. Biederer J, Schnabel A, Muhle C, Gross WL, Heller M, Reuter M. Correlation between HRCT findings, pulmonary function tests and bronchoalveolar lavage cytology in interstitial lung disease associated with rheumatoid arthritis. Eur Radiol, 2004; 14: 272-80. [ Links ]
43. Tsuchiya Y, Takayanagi N, Sugiura H, et al. Lung diseases directly associated with rheumatoid arthritis and their relationship to outcome. Euro Respir J 2011; 37: 1411-7. [ Links ]
44. Skare TL, Nakano I, Escuissiato DL, Batistetti R, Rodrigues Tde O, Silva MB.Pulmonary changes on high-resolution computed tomography of patients with rheumatoid arthritis and their association with clinical, demographic, serological and therapeutic variables. Rev Bras Reumatol 2011; 51: 325-30, 336-7. [ Links ]
45. Pappas DA, Giles JT, Connors G, Lechtzin N, Bathon JM, Danoff SK. Respiratory symptoms and disease characteristics as predictors of pulmonary function abnormalities in patients with rheumatoid arthritis: an observational cohort study. Arthritis Res Ther 2010; 12: R104. [ Links ]
46. Chen J, Shi Y, Wang X, Huang H, Ascherman D. Asymptomatic preclinical rheumatoid arthritis-associated interstitial lung disease. Clin Dev Immunol 2013; 2013: 406927. [ Links ]
47. Tanaka N, Kim JS, Newell JD, et al. Rheumatoid arthritis-related Lung Diseases: CT Findings1. Radiology 2004; 232: 81-91. [ Links ]
48. Bilgici A, Ulusoy H, Kuru O, Celenk C, Unsal M, Danaci M. Pulmonary involvement in rheumatoid arthritis. Rheumatol Int 2005; 25: 429-35. [ Links ]
49. Raghu G, Collard HR, Egan JJ, et al. An Official ATS/ERS/JRS/ALAT Statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med 2011; 183: 788-824. [ Links ]
50. Assayag D, Elicker BM, Urbania TH, et al. Rheumatoid arthritis-associated interstitial lung disease: radiologic identification of usual interstitial pneumonia pattern. Radiology 2014; 270: 583-8. [ Links ]
51. Perez T, Farre JM, Gosset P, et al. Subclinical alveolar inflammation in rheumatoid arthritis: superoxide anion, neutrophil chemotactic activity and fibronectin generation by alveolar macrophages. Euro Respir J 1989; 2: 7-13. [ Links ]
52. Kinoshita F, Hamano H, Harada H, et al. Role of KL-6 in evaluating the disease severity of rheumatoid lung disease: comparison with HRCT. Respir Med 2004; 98: 1131-7. [ Links ]
53. Nakajima H, Harigai M, Hara M, et al. KL-6 as a novel serum marker for interstitial pneumonia associated with collagen diseases. J Rheumatol 2000; 27: 1164-70. [ Links ]
54. Alexiou I, Germenis A, Koutroumpas A, Kontogianni A, Theodoridou K, Sakkas LI. Anti-cyclic citrullinated peptide-2 (CCP2) autoantibodies and extra-articular manifestations in Greek patients with rheumatoid arthritis. Clin Rheumatol 2008; 27: 511-3. [ Links ]
55. Harlow L, Rosas IO, Gochuico BR, et al. Identification of citrullinated Hsp90 isoforms as novel autoantigens in rheumatoid arthritis-associated interstitial lung disease. Arthritis Rheumatism 2013; 65: 869-79. [ Links ]
56. Rojas-Serrano J, González-Velásquez E, Mejía M, Sánchez-Rodríguez A, Carrillo G.Interstitial lung disease related to rheumatoid arthritis: evolution after treatment. Reumatol Clin 2012; 8: 68-71. [ Links ]
57. Sawada T, Inokuma S, Sato T, et al. Leflunomide-induced interstitial lung disease: prevalence and risk factors in Japanese patients with rheumatoid arthritis. Rheumatology 2009; 48: 1069-72. [ Links ]
58. Perez-Alvarez R, Perez-de-Lis M, Diaz-Lagares C, et al. Interstitial lung disease induced or exacerbated by TNF-targeted therapies: analysis of 122 cases. Semin Arthritis Rheumatism 2011; 41: 256-64. [ Links ]
59. Herrinton LJ, Harrold LR, Liu L , et al. Association between anti-TNF-a therapy and interstitial lung disease. Pharmacoepidemiology Drug Safety 2013; 22: 394-402. [ Links ]
60. Young A, Koduri G, Batley M, et al. Early rheumatoid arthritis study group. Mortality in rheumatoid arthritis. Increased in the early course of disease, in ischaemic heart disease and in pulmonary fibrosis. Rheumatology 2007; 46: 350-7. [ Links ]
61. Lee H, Choe JY, Kim SK, et al., Important prognostic factor in rheumatoid arthritis patients with interstitial lung disease is not usual interstitial pneumonia pattern but interstitial lung disease extent on chest high-resolution computed tomography. Annual Scientific Meeting of the American College of Rheumatology and Association of Rheumatology Health Professionals 2012, Washington, DC United States 2012; 64:S528. [ Links ]
62. Song JW, Lee HK, Lee Et Al CK, et al. Clinical course and outcome of rheumatoid arthritis-related usual interstitial pneumonia. SarcoidosisVasc Diffuse Lung Dis 2013; 30: 103-12. [ Links ]
63. Hozumi H, Nakamura Y, Johkoh T, et al., Acute exacerbation in rheumatoid arthritis-associated interstitial lung disease: a retrospective case control study. BMJ Open 2013; 3: e003132. [ Links ]
64. Walsh SL, Sverzellati N, Devaraj A, Keir GJ, Wells AU, Hansell DM. Connective tissue disease related fibrotic lung disease: high resolution computed tomographic and pulmonary function indices as prognostic determinants. Thorax 2013; 69: 216-22. [ Links ]
65. Idiopathic Pulmonary Fibrosis Clinical Research Network, Raghu G, Anstrom KJ, King TE, Jr., Lasky JA, Martinez FJ. Prednisone, azathioprine, and N-acetylcysteine for pulmonary fibrosis. N Engl J Med 2012; 366: 1968-77. [ Links ]
66. Inui N, Enomoto N, Suda T, Kageyama Y, Watanabe H, Chida K.Anti-cyclic citrullinated peptide antibodies in lung diseases associated with rheumatoid arthritis. Clin Biochem 2008; 41: 1074-7. [ Links ]
67. Park IN, Kim DS, Shim TS, et al. Acute exacerbation of interstitial pneumonia other than idiopathic pulmonary fibrosis. Chest 2007; 132: 214-20. [ Links ]
Recibido: 24-II-2014
Aceptado: 12-III-2014














