Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
El hornero
versión impresa ISSN 0073-3407versión On-line ISSN 1850-4884
Hornero v.21 n.2 Buenos Aires ago./dic. 2006
Roosts and migrations of swallows
David W. Winkler
Cornell University Museum of Vertebrates, Department of Ecology and Evolutionary Biology,Cornell University. Ithaca, NY 14853, USA. dww4@cornell.edu
Abstract
Swallows of the north temperate zone display a wide variety of territorial behaviour during the breeding season, but as soon as breeding is over, they all appear to adopt a pattern of independent diurnal foraging interleaved with aggregation every night in dense roosts. Swallows generally migrate during the day, feeding on the wing. On many stretches of their annual journeys, their migrations can thus be seen as the simple spatial translation of nocturnal roost sites with foraging routes straightening out to connect them. However, swallows that must make long journeys over ecological barriers clearly fly at night as well as in the day, and many suggestions indicate that there is considerable complexity in the altitude and bearing of flights even during the day. There are especially intriguing indications that much swallow migration may take place high out of sight of ground observers with movements near the ground often associated with foraging in passage. Provided that roost sites can be reliably found, swallow migration can be extremely flexible, and there are interesting contrasts in the biogeography and phenological flexibility of swallows compared to other passerine birds. Even within the swallows, there is considerable interspecific and intraspecifc variability in the distances of their annual migrations, and we are only just beginning to understand the biological causes and consequences of this variation. The profusion of Doppler weather radar stations in the eastern United States has allowed the characterization in considerable detail of the North American distributions of Tree Swallows (Tachycineta bicolor) and Purple Martins (Progne subis) throughout the non-breeding season. Evaluating the relative roles of movements and mortality in creating these patterns remains an important challenge for further research.
Key words: diurnal, Hirundinidae, martins, migration, nocturnal, roost, swallows.
Resumen
Dormideros y migraciones de golondrinas
Las golondrinas de la zona templada de América del Norte poseen una amplia variedad de comportamientos territoriales durante la estación de cría, pero ni bien culmina la reproducción todas parecen adoptar un patrón común, alternando la alimentación diurna independiente con el agrupamiento en populosos dormideros durante la noche. Las golondrinas generalmente migran durante el día, alimentándose en vuelo; sus migraciones, entonces, pueden ser vistas como si fuesen un simple traslado entre distintos sitios que poseen dormideros nocturnos, con las rutas de alimentación conectándolos directamente. Sin embargo, las golondrinas que deben realizar largos viajes cruzando barreras ecológicas vuelan tanto de noche como de día, y hay evidencias que indican que hay una considerable complejidad en la altitud y en las características de los vuelos aún durante el día. Hay evidencias especialmente interesantes de que la mayor parte de la migración de las golondrinas puede tener lugar a una altura tal que no es advertida por los observadores en tierra, pero con movimientos cercanos al suelo a menudo asociados con la alimentación. Si los sitios con dormideros pueden ser encontrados con certeza, la migración de las golondrinas sería extremadamente flexible, y existen interesantes contrastes en la biogeografía y la flexibilidad de la fenología de las golondrinas en comparación con otros paseriformes. Entre las golondrinas mismas hay una considerable variabilidad inter e intraespecífica en la distancia de migración anual, y estamos aún empezando a entender las causas y las consecuencias biológicas de esta variación. La creciente disponibilidad de estaciones con radares climatológicos Doppler en el este de Estados Unidos ha permitido la caracterización, con un considerable detalle, de las distribuciones norteamericanas de la Golondrina Bicolor (Tachycineta bicolor) y la Golondrina Purpúrea (Progne subis) durante la estación no reproductiva. La evaluación del papel relativo que juegan los movimientos y la mortalidad en la conformación de esos patrones es un importante desafío para futuras investigaciones.
Palabras clave: diurno, dormidero, golondrinas, Hirundinidae, migración, nocturno.
Received 13 March 2006, accepted 26 December 2006
The spatial biology of swallows and martins (Hirundinidae) in the breeding season is extremely diverse, ranging from dense and large colonial aggregations to dispersed territorial breeding (Turner and Rose 1989). Most species are central-place foragers spending most of their foraging time well away from the immediate vicinity of the nest (Bryant and Turner 1982, Turner 1982, McCarty and Winkler 1999). The diversity of summer sociality has engendered a rich literature in the comparative behavioural ecology of breeding social systems (e.g., Hoogland and Sherman 1976, Møller 1987, Brown and Brown 1996, Safran 2004). The plethora of data on breeding biology and ecology (e.g., Winkler and Allen 1996, Burness et al. 2001, Hasselquist et al. 2001, Ardia et al. 2003, Czarnowski et al. 2004, Lombardo et al. 2004, Safran and McGraw 2004, Safran et al. 2005) contrasts sharply with the relatively few studies that have been published about the biology of swallows during migration and in the non-breeding season (e.g., Lyuleeva 1973, Mead and Harrison 1979, Szep 1995). My goal here is to review what we know about swallow migration and wintering biology and present hypotheses to explain some of the patterns that are beginning to appear. I must emphasize that we know far too little, and this review is thus intended to serve as an invitation to further research rather than a compendium of established facts.
Swallows as nocturnal roosters
At least in the Northern Hemisphere, hirundinids outside the breeding season tend to spend every evening in large dense roosts. These roosts can sometimes be very large (well in excess of a million birds; van den Brink et al. 2000, Burney 2002, Bijlsma and van den Brink 2005), and there are interesting differences in roosting behaviour among species. Barn Swallows (Hirundo rustica) and Tree Swallows (Tachycineta bicolor) prefer to roost in reed or cane beds usually over water (van den Brink et al. 2000, Burney 2002), occasionally using trees or wires (e.g., Moreau 1972). In contrast, the larger martins in the genus Progne prefer to roost in shrubbery or trees or on ledges under bridges and in large industrial buildings (Oren 1980, Russell and Gauthreaux 1999, Purple Martin Conservation Association 2005). All species appear to prefer to roost over water, although all will roost in their favoured substrate over dry land on occasion (e.g., Skutch 1960). Species in the genus Progne are variable in their approach to roosts, in some locations flying into the roost in small parties as sunset approaches (Russell and Gauthreaux 1999) and, in others, funnelling-in in one huge concentration (Oren 1980). By contrast, Barn, Tree and rough-winged swallows of the genus Stelgidopteryx generally enter their roosts in very large, tight and short-duration streams.
One factor determining why Progne species prefer to roost in woody vegetation and on firm structures may be that their larger mass serves as a deterrent to perching in the tips of herbaceous marsh vegetation. This may explain why Progne species are less tied to roosting over water than are the smaller species. The fact that these species enter the roost in a less organized fashion than do the smaller swallow species may also indicate that the smaller species may have had their roosting habits most strongly molded by the threat of predation. The smaller swallow species tend to come to roost some time in the hour after sundown. Our observations of Tree Swallow roosts (Burney 2002, pers. obs.) appear to be fairly typical of these smaller species (see Skutch 1960 on Stelgidopteryx species): the swallows display their normal reluctance to settle into vegetation, and the birds congregate a few hundred meters above the roost site, milling around the site in an increasingly large and dense cloud of birds. Finally, as the last daylight fades, a few courageous birds make the plunge downward into the reeds of the roost site, followed immediately by a swirling stream of birds pouring into the vegetation, with hundreds of thousands of birds settling in only a few minutes' time. Few who have witnessed these spectacles can avoid describing the descending cloud as a tornado. Both the late hour of gathering and what seems to be the birds' extreme reluctance to venture solo into the vegetation suggest that the principal selective force molding this behaviour has been the risk of predation. Observations on Barn Swallows being preyed upon by aerial raptors as they come to roost in Africa (Bijlsma and van den Brink 2005) support this interpretation. This concern about predation may also explain why the birds appear to gather higher over the roost on clearer evenings (Skutch 1960).
The truly large roosts of hirundinids occur in migratory species, with the largest roosts generally being reported in wintering areas that support birds that bred over much larger areas in the north. It remains to be seen to what extent the non-migratory or short-distance species visit roosts in the non-breeding season. Resident Southern Rough-winged Swallows (Stelgidopteryx ruficollis) appeared to join roosts of migrant Northern Rough-winged Swallows (Stelgidopteryx serripennis) in cane when they were in the vicinity, but they also roosted in smaller aggregations in the same site when the migrants were gone (Skutch 1960). Skutch (1960) also describes a small group of seven Black-capped Swallows (Notiochelidon pileata) roosting together every night after the breeding season in cold cloud forest habitat, in a burrow that was not used for nesting. He also reports that the Blue-and-white Swallow (Notiochelidon cyanoleuca) can roost year-round in and near its nest; thus, these species apparently do not join large post-breeding roosts. I have seen a White-winged Swallow (Tachycineta albiventer) pair settle down for the night with a fully volant juvenile on small branches over a running stream near Eldorado, Misiones Province, Argentina, on a date (9 March 2001) that appeared to be outside the local breeding season (Belton 1985). Roosts of 2000-10000 swallows (mostly the migrants Hirundo rustica and Riparia riparia in southern summer and Tachycineta leucorrhoa and Tachycineta meyeni in southern winter) occur regularly in the state of Rio Grande do Sul in Brazil (R Dias and C Fontana, pers. com.). And all the South American Progne species appear to occur in large roosts at some time in their annual cycles (Oren 1980, Hill 1995). Further reports on the sizes and locations of the roosts of tropical swallows and data on whether they live their more sedentary lives without visiting large nocturnal roosts, would be very interesting. Such data would help us understand to what extent nocturnal roosting aggregations are tied with migratory behaviour.
Roosts and radar
Roosts generally empty out a few minutes before sunrise the next morning, and the birds tend to leave the roost once again en masse, retracing their flight upward to even higher altitudes before flaring out on foraging flights in all directions (Skutch 1960). This upward flight, followed by the near-symmetric exodus of foraging birds in all direction, makes these large roosts of swallows apparent in the Doppler weather radar now deployed throughout the United States (Russell and Gauthreaux 1999, Burney 2002; Fig. 1). The resulting roost "ring-echoes" provide the prospect of a synthetic view of the size and seasonal distribution of swallow roosts throughout North America (Russell and Gauthreaux 1998, Russell et al. 1998, Burney 2002). These remote methods are being supplemented by organized citizen science projects in both North America (Purple Martin Conservation Association 2005) and the United Kingdom (British Trust for Ornithology 2006) from which a great deal is being learned about comparative roosting biology.
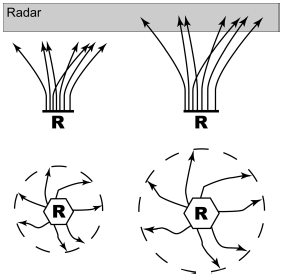
Figure 1. How a flock of swallows emerging in early morning from their roost (R) climbs into the altitudes being scanned by weather radar (above), and spreading out from there in foraging flights (below) leaves a distinctive ring echo in the radar image before it spreads out enough that the density of flying swallows becomes so low as to be undetectable by the radar.
One of the interesting aspects of roosting biology is that roost sites vary considerably in how consistently they are used from year to year. For instance, only a few coastal sites, such as the one at Icklesham, Sussex, are reliable Barn Swallow migratory stopover sites from year to year in the United Kingdom, while all others being monitored so far in this country seem to move around from year to year (Griffin et al. 2005). Similarly, a large swallow roost seems to occur every fall in the marshes of Montezuma National Wildlife Refuge near Ithaca, New York, but its precise location can vary by 5 km or so from year to year (Burney 2002, Winkler and Haber, unpublished data). As we learn more about hirundinid roosting biology, it will be interesting and of considerable conservation importance to try to understand just what makes a site attractive from year to year: it may actually be the case that unpredictability per se is an advantage in reducing the familiarity of roost sites to local predators.
Roosts and migration
Over much of their migratory and wintering ranges, hirundinid roost sites appear to have fairly consistent roost spacing. Roost sites in North America tend to be about 100 to 150 km apart, though there are certainly consecutive roost sites in prospective northbound or southbound directions that are farther apart (Fig. 2). Spacing at distances approximating 100 km is very similar to the scale of movements reported for roosts of migrant species of the genus Hirundo in South Africa (Oatley 2000). By contrast, roost sites in Britain are often much closer together, averaging as little as 25-30 km apart (Ormerod 1991), and the wave of Barn Swallows returning to Britain moves about 50 km/day (Huin and Sparks 1998). This suggests that migrating hirundinids in Britain may actually have the luxury of hopping over some roost sites in their journeys. However, even in North America, the distances between many of the roost sites are well within the range of a day's fairly leisurely flight. Flight speeds of migrating hirundinids are in the range of 40-80 km/h (Lyuleeva 1973), and even if the speeds of leisurely foraging birds are half those, most roost spacings would appear to accommodate an unhurried itinerary from site to site.
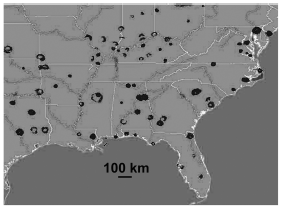
Figure 2. A compilation of weather radar data throughout the southeastern United States for an early morning in late summer. The image shows the clear signatures of roost "ring-echoes" spaced fairly regularly at about 100 km throughout the region.
These observations suggest an interesting model for swallow migration, with birds migrating through the day linking every night to the next site in their successive chain of nocturnal roost sites. Wintering swallows are reasonably seen as classical central place foragers, with birds radiating out from the roost site in early morning and most often returning to the same roost the following evening. Thus, migration for swallows may often be a simple extension of the loops of daily central place foraging into line segments of approximately the same length linking up to the next roost site (Fig. 3). It would be interesting to explore how these connections to the next roosts are made. Individual birds may set out from a roost in the morning with a memory of the next site on their migratory chain, or they may simply forage more in the preferred migratory direction and then be recruited to the next roost site by aggregating near the end of the day with birds that used the next site for their own roost the night before. These behavioural mechanisms of roost joining would also presumably illuminate the mystery of which sites are used from year to year and serve as a clue to the causes of the interannual ebb and flow of roost size.
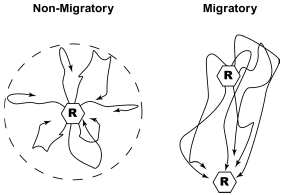
Figure 3. A simple graphical interpretation of how movements by foraging swallows between roost sites may involve very little if any increase in the distance flown per day (right) relative to birds returning to the same roost site in succeeding nights (left).
Hirundinidae migratory behaviour
This view of swallow migrations as journeys stitched together by roost sites suggests that the exertion of migration on many parts of their journeys may not differ substantially from that expended during the daily and routine foraging movements out from and back to a nocturnal central place. Thus, most movements of swallows appear to take place during the day with these fairly routine movements from one roost site to the next. Yet, no matter what the details of how roosts and migration are integrated, it is clear that not all parts of all hirundinid migrations are a simple redirection of routine foraging patterns. There are records of marked Barn Swallows covering 12000 km in 34 days (320 km/day) and 3028 km in 7 days (433 km/day; Turner 2004). These rates of movement are much greater than those likely to be achieved by birds making more leisurely movements from roost to roost. In addition, many of the longest and fastest passages made by swallows are those made by the three long-distance European swallow migrants as they cross the Mediterranean and Sahara. Like most passerines that make this trip twice annually, or die trying, it seems likely that the vast majority head south from staging areas in Spain or Italy (Rubolini et al. 2002b), then cross directly to Africa, some probably stopping to "coast" along the Saharan verge of the sea and others probably continuing directly to areas south of the Sahara (reviewed by Moreau 1961). Hirundo rustica is the most commonly seen bird crossing the Sahara, and, especially in spring, it is often seen flying against, and not uncommonly succumbing to, the northerly winds that prevail at that season (Moreau 1961). As elsewhere in their annual cycle, Hirundo species tend to fly lower than other hirundinids, probably making them more visible than Delichon and Riparia species during passage (Lyuleeva 1973). Moreau (1961) estimated that all the passerines making the non-stop trans-Saharan trip in spring must fly 50-60 h non-stop against head-winds. Recent radar evidence (Schmaljohann et al. 2007) suggests, however, that the 2100-2400 km trip between Europe and tropical Africa is not made non-stop by most European passerines that cross the Sahara and Mediterranean. Much more detail is needed on the biology of these migrants. Nevertheless, the sudden and sporadic appearance of large numbers of all three European swallow genera at low elevations during migration, together with correlations between the intensity of observed migrations and local weather, suggest the hypothesis that hirundinids under very favourable winds fly out of sight high above the ground and that they fly nearer the ground when they are faced with head winds or the need to refuel on insects en route. Flying insects are generally more abundant near the ground (e.g., Glick 1939, Taylor 1974), and it appears likely that, when insects are relatively abundant over the terrain being over-flown, hirundinids descend to lower levels (i.e., nearer the ground) to refuel, returning, once fed, to higher elevations to cover much larger distances when and where the winds are favourable. This possible alternation between higher altitude cruising and lower altitude foraging flight could also explain: (1) why migrating swallows often do not appear at migration observation stations until hours after sunrise (Lyuleeva 1973) when they presumably leave the roosts many hours earlier, and (2) why adults and juveniles appear to fly for different amounts of the day (e.g., Gatter and Behrndt 1985). Nearly exclusive high altitude foraging may also explain the extreme sparseness of observations of some common species (e.g., Delichon urbicum; Moreau 1972) on the wintering grounds.
J. Cobb (in Griffin et al. 2005) reported that Hirundo species lured down to nets near a roost site in the United Kingdom by playback of swallow song in the afternoon, well before swallows would be coming to roost, were never captured in the roost later in the day and averaged about 2 g heavier than those captured later in the evening at the same site. This seemed to happen after warm days, suggesting interesting environmental effects on this behaviour; however, it also suggests not only that high-flying birds may have greater energy stores but also that, even in the United Kingdom, there may be a diversity of migratory strategies in the Hirundo population and that some birds might even be migrating at night and making much more direct over-land progress over the United Kingdom as a result. The generality and interpretation of this result would be strengthened enormously by playback experiments well in advance of the roosting hour near hirundinid roost sites in other places.
Any journey of two days or more requires both nocturnal and diurnal migrants to abandon their preferred hours of flight, and it is clear that hirundinids fly both day and night during these long migratory passages (reviewed in Moreau 1961). It remains an open question whether and how much hirundinids fly at night outside the most extreme legs of their journeys, but it seems safe to continue to designate them as diurnal migrants.
In the Western Hemisphere, observations of enormous numbers of migrant swallows passing through Central America (Table 1; Brown and Brown 1999, Ruelas Inzunza et al. 2005), suggest that the bulk of North American swallows access wintering ranges south of the Gulf of Mexico by an overland route; but the presence of some individuals from all North American species in Caribbean islands and migrating over water in the Gulf confirm that at least some individuals attempt the over-water route to the Southern Hemisphere (Hailman 1962, Yunick 1977, Russell 2005). Given what closely related birds are able to do in the Eastern Hemisphere, it would be interesting and important to try to determine the actual proportions of birds using the overland vs. direct aquatic route over the Gulf of Mexico, especially given that north-bound birds appear to take more varied routes over land during spring migration than fall (Ruelas Inzunza et al. 2005, E Ruelas Inzunza, pers. com.).
Table 1. Counts of migratory swallows made on the coastal plain of the Gulf of Mexico at La Mancha (19°36'N, 96°22'W), Veracruz, Mexico, in the autumn of 1999 by Robert Straub, assisted by Tom Valega and Leticia Cruz. Counts were conducted approximately every other day from 24 August to 30 November, 1999. The count was made from sunrise to sunset whenever possible; however, efforts tended to be concentrated on the higher activity of the morning and afternoon flights. Data compiled by, and courtesy of, Ernesto Ruelas Inzunza of the Centro de Investigaciones Costeras de La Mancha (CICOLMA).

Biology of the migrants
Southbound Hirundo species in Europe can accumulate up to about 40% of their mass as fat (Rubolini et al. 2002a) preparing for the Mediterranean-Saharan journey, and they have different fat dynamics if heading over Gibraltar vs. Italy (Rubolini et al. 2002b). By contrast, Hirundo species in fall appear to gain only 1-2 g (<25% of body mass) on average in the United Kingdom before heading south to mainland Europe. Other hirundinids in Europe also appear to store less fat than does Hirundo rustica before heading to Africa (Cramp 1988). The dynamics of fat stores in different age and geographic classes of Hirundo rustica appears to make sense relative to the ecologies of those groups (Pilastro and Magnani 1997). However, given the potentially biased qualities of birds caught during migrations (see Hirundinidae migratory behaviour above), interpretation of the masses reported must be made cautiously. If indeed the migratory behaviours of hirundinids are actually quite heterogeneous, a careful study of how changes in body mass and individual body components relate to the migratory behaviour of individual birds could be both extremely difficult and enlightening.
The biology of moult and how it relates to hirundinid breeding and migration appears to be equally diverse. Among species, there is a gradient from species (such as Tachycineta bicolor; Stutchbury and Rohwer 1990) that complete a full moult of body feathers, rectrices and remiges before and during migration; through populations that moult body feathers as they initiate migration, stopping to complete moult of rectrices and remiges before making a long-distance migration (e.g., eastern Stelgidopteryx serripennis; Yuri and Rohwer 1997); through species moulting body feathers before and during migration and not initiating or interrupting early flight feather moult until reaching the wintering ground (apparently all trans-Saharan species: Riparia and Delichon species, Cramp 1988; Hirundo rustica, Cramp 1988, van den Brink et al. 2000, Rubolini et al. 2002b, Griffin et al. 2005). Once again, there is ample evidence of flexibility in moult schedules within species, with Western Stelgidopteryx serripennis adopting a strategy more like Tachycineta bicolor, moulting while it migrates without interruption, in contrast to the "moult migration" of the Eastern population (Yuri and Rohwer 1997).
Why do some migrate so much farther than others?
Winkler (2000) suggested that there were multiple selective reasons that have made changes in swallow nest-type less evolutionarily flexible than other aspects of the life-history, and much of the spring life history of these birds can be seen as adjustments to the mode of nesting. The swallow fauna of eastern North America is distinctive in the presence of six obligate migrant species with diverse nesting mode among them (Fig. 4). Of these, three are cavity adopters, reliant on other species or processes to create a nest-cavity in which they build a grass-nest cup. It seems more than coincidence that these three cavity-adopting species migrate the shortest distances of the lot, a feature that likely allows them to return to the breeding grounds earlier in spring to compete for and secure a nest-cavity, which, unlike the burrowing and mud-nesting species, they cannot make for themselves (see also Rubolini et al. 2005).
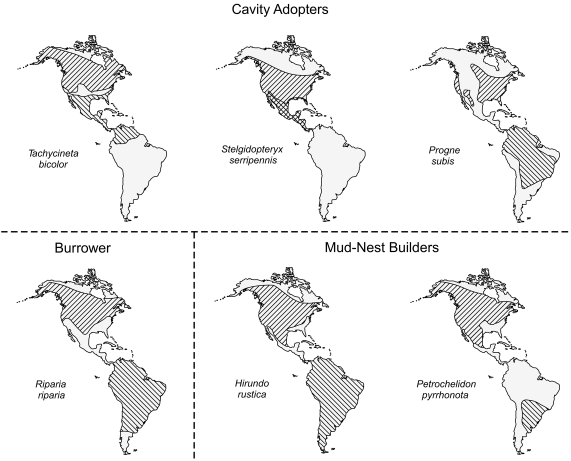
Figure 4. The breeding (right diagonal hatching) and wintering (left diagonal hatching) ranges of six eastern North American swallow species relative to the type of nest they build at their northern nesting sites.
How phylogenetically flexible is movement in swallows?
An alternative hypothesis to explain the more northerly wintering of the cavity-adopters is that patterns of movement may be nearly as difficult to evolve as are patterns of nest construction, and that all the longest-distance migrants in the New World hirundinid fauna (Riparia riparia, Hirundo rustica and Petrochelidon pyrrhonota; Fig. 4) are derived from Old World forms (Sheldon et al. 2005) that presumably colonized the Western Hemisphere relatively recently. However, available evidence in support of this phylogenetic inertia hypothesis is less than compelling. First, there are two tropical species in the New World fauna, Petrochelidon fulva and Petrochelidon rufocollaris, that appear to have derived from Petrochelidon pyrrhonota in the New World, and both are short-distance- or non-migrants. Second, Riparia riparia, together with its short-distance congener Riparia paludicola from the Old World, appears to be sister group to the genus Tachycineta and much more closely related to the endemic New World swallows than they are to other Old World forms. Thus, the short-distance or non-migratory habits of Tachycineta species and Riparia paludicola appear not to have been constrained by the habits of their long-distance close relative Riparia riparia. Finally, Hirundo rustica has begun breeding in South America, on what were solely its wintering grounds, within the past 30 years (Martínez 1983). Though the movement patterns of this small breeding population are not known, it appears extremely unlikely that these birds have retained the long-distance migratory habit of their immediate ancestors and perform a long migration all the way to North America in the austral winter (GH Huber, pers. com.). Hirundo rustica is known for the complexity of movement patterns within its large populations in both the Eastern and Western hemispheres (Moreau 1972, Phillips 1986, Cramp 1988), with some resident forms interacting during parts of the year with both long- and short-distance forms passing through. Furthermore, Hirundo rustica is closely related to a large number of other species of the genus Hirundo in sub-Saharan Africa, many of which are residents or short-distance migrants (for all phylogenetic statements in this paragraph see Sheldon et al. 2005). Finally, patterns of movement within the New World endemic hirundinids are very flexible. Though there are no long-distance migrants that go as far as Petrochelidon, Hirundo or Riparia species do in the New World, there is a great deal of diversity in distances travelled in the genera Tachycineta, Progne and Stelgidopteryx. In short, the distance of movement seems to be quite flexible in swallows (see also Turner and Rose 1989), and the patterns observed above for the summer and winter ranges of North American breeding species seem more readily explained by nest-site competition on the breeding grounds than by phylogenetic conservatism of the traits.
Flexibility within species in whether to migrate
There are at least two observations that suggest considerable flexibility in migratory scheduling within populations of hirundinids. First, many first-year male Progne subis return to the breeding grounds even though they do not generally breed their first year. This suggests that the observation of first-year males in the wintering grounds during the northern breeding season (Hill 1995) may actually be the result of a strategy by these younger birds to forego the return to breeding areas until their chances of breeding are higher (see also comment by Oren 1980). It would be interesting to investigate the properties of those birds that stay in the south their first year and compare them to those that complete the annual migratory circuit without attempting breeding on the northern end.
The other exceptional indicator of flexibility in breeding and migration is provided by those northern migrants that have begun breeding in the southern wintering grounds. Delichon urbicum has bred and produced fledglings intermittently in South Africa, with individual colonies sometimes lasting "for some years", but no sustained population has been established (McLachlan and Liversidge 1957, Maclean 1988:465). By contrast, and as mentioned above, a breeding population of Hirundo rustica in Buenos Aires Province, Argentina, colonized in the early 1980's (Martínez 1983) and is still slowly growing (P Petracci and GH Huber, pers. com.). It is not yet clear whether this population is being sustained by local production of offspring or continued recruitment from the migrant pool. In any event, these cases of re-distribution of major life events in the annual cycle suggest a flexibility in swallows that is equalled only by nomadic passerines of boreal forest or tropical deserts, and swallows appear to be the only long-distance migrant passerines with such flexibility. Just how, physiologically, this readjustment of the life history is performed is a fascinating area for on-going research.
Implications of diurnal migration
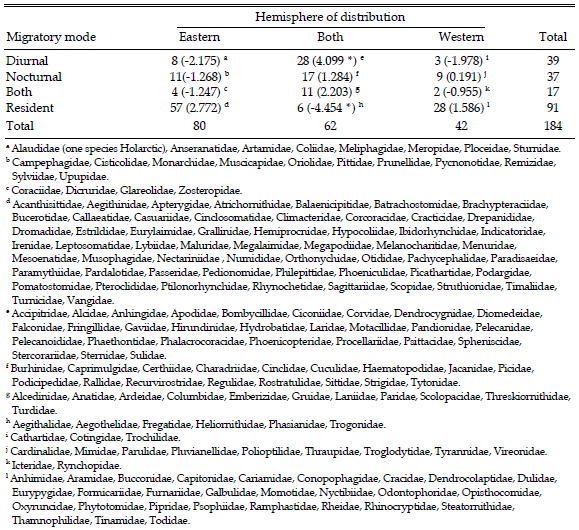
Unlike the bulk of other migrant bird species, the Hirundinidae are cosmopolitan. Few other bird groups have achieved this breadth of distribution, and the migrations of swallows seem analogous with other broadly distributed migrants, such as Apodidae, Accipitridae and Anatidae, with thriving Northern Hemisphere populations linked by migration to habitats further south in the northern winter. All of these groups also share the property of being diurnal migrants, and other aerial insectivore long-distance migrants (e.g., Tyrannidae in the Western Hemisphere and Muscicapidae in the East) are nocturnal and are limited to one hemisphere or the other in their distributions. Of course, there are numerous insectivorous diurnal migrants that migrate smaller distances and do not currently occur in both hemispheres (e.g., Meropidae, Artamidae). To test the possibility that diurnality of migration per se is associated with the broad-scale distributions of birds at the family level, I categorized each avian family into residents and migrants. I further divided the migrants into diurnal, nocturnal, or both, and recorded whether these types are distributed solely in the Eastern or Western Hemispheres or both (a much more detailed species-level treatment of migration modes world-wide is forthcoming; Farnsworth et al., unpublished data). In characterizing distributions, I ignored single species in one hemisphere that otherwise were endemic to the other in four families (Alaudidae, Troglodytidae, Timaliidae and Sylviidae). The resulting analysis (Table 2) shows, not surprisingly, that residents are much more likely to be endemic to one hemisphere or the other (i.e., there are far fewer distributed in both than expected by chance). By contrast, diurnal migrants are significantly more likely to occur in both hemispheres. Exactly how this pattern arises is unclear, but the broad array of visual cues available to diurnal migrants may lend them increased flexibility and the ability to explore routes for migratory possibilities that change with changing Earth climate and that may not be available to a more pre-programmed navigation-orientation system in nocturnal migrants. Hirundinids are clearly one of the most flexible groups both in their migratory biology and breeding schedules, and it is perhaps a bit surprising that there are so many tropical forms in this group with such limited distributions (e.g., Hirundo megaensis, Tachycineta stolzmanni, Tachycineta cyaneoviridis, Tachycineta euchrysea, Notiochelidon pileata).
Table 2. Summary of the distributions of families of birds of the world as they relate to the migratory modes of their members. For each cell, the number of families is followed (in parentheses) by the deviation of the observed cell frequency from that expected from the marginal totals. Global analysis: χ26 = 66.091, P < 0.001. The two cells that contribute the most to the observed χ2 are indicated by an asterisk. Four families with unknown major migratory mode (Acanthizidae, Eopsaltridae/Petroicidae, Chionididae and Thinocoridae) were left out of this analysis.
The way forward: migration and mortality
The exceptional flexibility of hirundinid life-cycles raises many interesting possibilities for research, many of which will become available as soon as digital telemetry tags and data-loggers become small enough to deploy on swallows. These technological developments raise the prospect of following individuals through their annual cycles and relating their personal histories and physiological states to the movements that they undergo. Such tags may also eventually lead to a refinement of our estimates of where in the life cycle most swallows die and why.
Improved information on mortality could dramatically increase our understanding of the lives of these resourceful birds and give us powerful new knowledge to help sustain their populations on Earth. It appears that most adults die in non-breeding quarters and juvenile survival is most affected by conditions on or near the breeding grounds (Szep 1995, Stokke et al. 2005). The huge preponderance of juveniles in roosts in northern latitudes (Griffin et al. 2005), their variable proportions in African roosts from year to year (van den Brink et al. 2000), and the frequent observation of dead and dying Hirundo rustica during trans-Saharan migration (Moreau 1961, 1972) all indicate that mortality can be swift and severe at times.
Understanding the causes of mortality in these long-distance migrants is a fascinating and important challenge, but it may also be extremely interesting to apply this increased knowledge to interpretation of patterns of seasonal movement. If gathering the traces of roost ring-echoes from weather radar from across North America can be automated, there is the prospect of understanding the changing distributions of a very large fraction of the world's population of at least Tachycineta bicolor through the annual cycle. When viewing the changing distributions and sizes of these roosts, it is tempting to interpret the changes in distributions as representing the movements of birds up and down the north Atlantic coast. But the survival rates of Tachycineta bicolor at our Ithaca study site are, if anything, lower than those of Hirundo rustica (unpublished data). Thus, if we attribute shifts in distribution to movement, we may forget that a large number of the birds detected in the radars will not be returning to the breeding grounds. Just as dispersal from the natal grounds confounds estimates of survival, movement and mortality may well be confounded in the non-breeding season as well. When large roosts on the north Atlantic coast disappear from the radar in autumn, some of the animals have no doubt migrated to the south, but a large proportion of them may have died instead. Short-distance facultative migrants retain the possibility of getting back to the breeding grounds earlier, but the mortality cost of doing so may be larger than the extreme costs incurred by their cousins migrating longer distances.
Acknowledgements. Many thanks to Ernesto Ruelas Inzunza, Curt Burney, Eduardo Iñigo-Elias, Rafael Dias and Carla Fontana for sharing unpublished data with me and aiding their interpretation; to Andrew Farnsworth for sharing his widespread knowledge of the migratory habits of the world's birds; to Andrew Farnsworth, Jamie Mandel, Gernot Huber, and Dave Cerasale for providing me with lots of insights and perspectives on bird migration; to Theunis Piersma and Andrew Farnsworth for valuable comments on the manuscript; and to Benjamin Shattuck and Noah Hamm for help with the figures. This material is based upon work supported by the National Science Foundation under grants No. IBN-013437 and 9207231, and a Cooperative Agreement with the National Center for Environmental Assessment, EPA (CR 829374010).
Literature Cited
Ardia DR, Schat KA and Winkler DW (2003) Reproductive effort reduces long-term immune function in breeding tree swallows (Tachycineta bicolor). Proceedings of the Royal Society of London B 270:1679-1683 [ Links ]
Belton W (1985) Birds of Rio Grande do Sul, Brazil. Part 2. Formicariidae through Corvidae. Bulletin of the American Museum of Natural History 180:1-241 [ Links ]
Bijlsma RG and van den Brink B (2005) A Barn Swallow Hirundo rustica roost under attack: timing and risks in the presence of African Hobbies Falco cuvieri. Ardea 93:37-48 [ Links ]
van den Brink B, Bijlsma RG and van der Have TM (2000) European Swallows Hirundo rustica in Botswana during three non-breeding seasons: the effects of rainfall on moult. Ostrich 71:198-204 [ Links ]
British Trust for Ornithology (2006) Swallow Roost Project. (URL: http://www.bto.org/ringing/swallowroostproject/index.htm) [ Links ]
Brown CR and Brown MB (1996) Coloniality in the Cliff Swallow. University of Chicago Press, Chicago [ Links ]
Brown CR and Brown MB (1999) Barn Swallow (Hirundo rustica). Pp. 1-32 in: Poole A and Gill F (eds) The birds of North America. Academy of Natural Sciences and American Ornithologists' Union, Philadelphia and Washington DC [ Links ]
Bryant DM and Turner AK (1982) Central place foraging by swallows (Hirundinidae): the question of load size. Animal Behaviour 30:845-856 [ Links ]
Burness GP, Ydenberg RC and Hochachka PW (2001) Physiological and biochemical correlates of brood size and energy expenditure in tree swallows. Journal of Experimental Biology 204:1491-1501 [ Links ]
Burney CW (2002) A study of swallow roosts found in the Eastern United States. MSc thesis. Cornell University, Ithaca [ Links ]
Cramp S (1988) The Birds of the Western Palearctic. Oxford University Press, Oxford [ Links ]
Czarnowski MR, Lombardo MP, Power HW and Hasselquist D (2004) Sudden energetic deficit in male tree swallows (Tachycineta bicolor) has negligible effect on immunocompetence and glucocorticosteroid levels. Integrative and Comparative Biology 44:543-543 [ Links ]
Gatter VW and Behrndt M (1985) Unterschiedliche tageszeitliche Zugmuster alter und junger Vogel am Beispiel der Rauchschwalbe (Hirundo rustica). Vogelwarte 33:115-120 [ Links ]
Glick P (1939) The distribution of insects, spiders and mites in the air. United States Department of Agriculture Technical Bulletin 673:1-150 [ Links ]
Griffin B, Clark J and Robinson RE (2005) Swallow Roost Project Newsletter. British Trust for Ornithology, Thetford [ Links ]
Hailman JP (1962) Direct evidence for trans-Caribbean migratory flights of swallows and dragonflies. American Midland Naturalist 68:430-433 [ Links ]
Hasselquist D, Wasson MF and Winkler DW (2001) Humoral immunocompetence correlates with date of egg-laying and reflects work load in female tree swallows. Behavioral Ecology 12:93-97 [ Links ]
Hill JR III (1995) Thousands of martins killed by hail in Brazilian tornado. Purple Martin Update 6:2-3 [ Links ]
Hoogland JL and Sherman PW (1976) Advantages and disadvantages of Bank Swallows (Riparia riparia) coloniality. Ecological Monographs 46:33-58 [ Links ]
Huin N and Sparks TH (1998) Arrival and progression of the Swallow Hirundo rustica through Britain. Bird Study 45:361-370 [ Links ]
Lombardo MP, Green ML, Thorpe PA, Czarnowski MR and Power HW (2004) Repeated sampling affects Tree Swallow semen characteristics. Journal of Field Ornithology 75:394-403 [ Links ]
Lyuleeva DS (1973) Features of swallow biology during migration. Pp. 219-272 in: Bykhovskii BE (ed) Bird migrations: ecological and physiological factors. John Wiley & Sons, New York [ Links ]
Maclean GL (1988) Roberts' birds of southern Africa. New Holland, London [ Links ]
Martínez MM (1983) Nidificación de Hirundo rustica erythrogaster (Boddaert) en la Argentina (Aves, Hirundinidae). Neotropica 29:83-86 [ Links ]
McCarty JP and Winkler DW (1999) Foraging ecology and diet selectivity of tree swallows feeding nestlings. Condor 101:246-254 [ Links ]
McLachlan GR and Liversidge R (1957) Roberts birds of South Africa. South African Bird Book Fund, Cape Town [ Links ]
Mead CJ and Harrison JD (1979) Sand Martin movements within Britain and Ireland. Bird Study 26:73-86 [ Links ]
Møller AP (1987) The advantages and disadvantages of coloniality in the swallow Hirundo rustica. Animal Behaviour 35:819-832 [ Links ]
Moreau RE (1961) Problems of Mediterranean-Saharan migration. Ibis 103a:373-427, 103a:580-623 [ Links ]
Moreau RE (1972) The Palearctic-African bird migration systems. Academic Press, London [ Links ]
Oatley TB (2000) Migrant European Swallows Hirundo rustica in southern Africa: a southern perspective. Ostrich 71:205-209 [ Links ]
Oren DC (1980) Enormous concentration of martins (Progne spp.) in Iquitos, Peru. Condor 82:344-345 [ Links ]
Ormerod SJ (1991) Pre-migratory and migratory movements of Swallows Hirundo rustica in Britain and Ireland. Bird Study 38:170-178 [ Links ]
Phillips AR (1986) The known birds of North and Middle America. Part I. Private edition, Denver [ Links ]
Pilastro A and Magnani A (1997) Weather conditions and fat accumulation dynamics in pre-migratory roosting Barn Swallows Hirundo rustica. Journal of Avian Biology 28:338-344 [ Links ]
Purple Martin Conservation Association (2005) Project Martin Roost (URL: http://www.purplemartin. org/roost/) [ Links ]
Rubolini D, Massi A and Spina F (2002a) Replacement of body feathers is associated with low premigratory energy stores in a long-distance migratory bird, the Barn Swallow (Hirundo rustica). Journal of Zoology 258:441-447 [ Links ]
Rubolini D, Pastor AG, Pilastro A and Spina F (2002b) Ecological barriers shaping fuel stores in Barn Swallows Hirundo rustica following the central and western Mediterranean flyways. Journal of Avian Biology 33:15-22 [ Links ]
Rubolini D, Spina F and Saino N (2005) Correlates of timing of spring migration in birds: a comparative study of trans-Saharan migrants. Biological Journal of the Linnean Society 85:199-210 [ Links ]
Ruelas Inzunza E, Hoffman SW and Goodrich LJ (2005) Stopover ecology of Neotropical migrants in central Veracruz, Mexico. Pp. 657-672 in: Ralph CJ and Rich TD (eds) Bird conservation implementation and integration in the Americas. General Technical Report PSW-GTR-191, Pacific Southwest Research Station, USDA Forest Service, Albany [ Links ]
Russell KR and Gauthreaux SA (1998) Use of weather radar to characterize movements of roosting purple martins. Wildlife Society Bulletin 26:5-16 [ Links ]
Russell KR and Gauthreaux SA (1999) Spatial and temporal dynamics of a Purple Martin pre-migratory roost. Wilson Bulletin 111:354-362 [ Links ]
Russell KR, Mizrahi DS and Gauthreaux SA Jr (1998a) Large-scale mapping of Purple Martin premigratory roosts using WSR-88D weather surveillance radar. Journal of Field Ornithology 69:316-325, 69:509 [ Links ]
Russell RW (2005) Interactions between migrating birds and offshore oil and gas platforms in the northern Gulf of Mexico. Final Report. OCS Study MMS 2005-009, Gulf of Mexico OCS Region, Minerals Management Service, United States Department of the Interior, New Orleans [ Links ]
Safran RJ (2004) Adaptive site selection rules and variation in group size of Barn Swallows: individual decisions predict population patterns. American Naturalist 164:121-131 [ Links ]
Safran RJ and McGraw KJ (2004) Plumage coloration, not length or symmetry of tail-streamers, is a sexually selected trait in North American barn swallows. Behavioral Ecology 15:455-461 [ Links ]
Safran RJ, Neuman CR, McGraw KJ and Lovette IJ (2005) Dynamic paternity allocation as a function of male plumage color in barn swallows. Science 309:2210-2212 [ Links ]
Schmaljohann H, Liechti F and Bruderer B (2007) Songbird migration across the Sahara: the non-stop hypothesis rejected! Proceedings of the Royal Society of London B 274:735-739 [ Links ]
Sheldon FH, Whittingham LA, Moyle RG, Slikas B and Winkler DW (2005) Phylogeny of swallows (Aves: Hirundinidae) estimated from nuclear and mitochondrial DNA sequences. Molecular Phylogenetics and Evolution 35:254-270 [ Links ]
Skutch AF (1960) Life histories of Central American birds, II. Family Hirundinidae. Pacific Coast Avifauna 34:265-286 [ Links ]
Stokke BG, Møller AP, Saether BE, Rheinwald G and Gutscher H (2005) Weather in the breeding area and during migration affects the demography of a small long-distance passerine migrant. Auk 122:637-647 [ Links ]
Stutchbury BJ and Rohwer S (1990) Molt patterns in the Tree Swallow (Tachycineta bicolor). Canadian Journal of Zoology 68:1468-1472 [ Links ]
Szep T (1995) Survival rates of Hungarian sand martins and their relationship with Sahel rainfall. Journal of Applied Statistics 22:891-904 [ Links ]
Taylor LR (1974) Insect migration, flight periodicity and boundary-layer. Journal of Animal Ecology 43:225-238 [ Links ]
Turner A and Rose C (1989) A handbook to the swallows and martins of the world. Christopher Helm, London [ Links ]
Turner AK (1982) Optimal foraging by the Swallow (Hirundo rustica, L). Prey size selection. Animal Behaviour 30:862-872 [ Links ]
Turner AK (2004) Family Hirundinidae (swallows and martins). Pp. 602-685 in: del Hoyo J, Elliott A and Christie DA (eds) Handbook of the birds of the world. Volume 9. Cotingas to pipits and wagtails. Lynx Edicions, Barcelona [ Links ]
Winkler DW (2000) The phylogenetic approach to avian life histories: an important complement to within-population studies. Condor 102:52-59 [ Links ]
Winkler DW and Allen PE (1996) The seasonal decline in tree swallow clutch size: physiological constraint or strategic adjustment? Ecology 77:922-932 [ Links ]
Yunick RP (1977) A Caribbean Barn Swallow recovery. Auk 94:149-150 [ Links ]
Yuri T and Rohwer S (1997) Molt and migration in the Northern Rough-winged Swallow. Auk 114:249-262 [ Links ]














