Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Archivos argentinos de pediatría
Print version ISSN 0325-0075On-line version ISSN 1668-3501
Arch. argent. pediatr. vol.115 no.2 Buenos Aires Apr. 2017
http://dx.doi.org/10.5546/aap.2017.133
ORIGINAL ARTICLE
http://dx.doi.org/10.5546/aap.2017.eng.133
Vitamin D deficiency and insulin resistance as risk factors for dyslipidemia in obese children
Meltem Erol, M.D.a, Özlem Bostan Gayret, M.D.a, Şahin Hamilçıkan, M.D.a, Emrah Can, M.D.a and Özgül Yiğit, M.D.a
a. Department of Pediatrics, Bagcilar Training and Research Hospital, Istanbul, Turkey.
E-mail address: Emrah Can, M.D.: canemrahcan@yahoo.com
Funding: None.
Conflict of interest: None.
Received: 8-3-2016
Accepted: 10-19-2016
ABSTRACT
Introduction. Dyslipidemia is one of the major complications of obesity; vitamin D deficiency and insulin resistance are attending metabolic complications in dyslipidemic obese children.
Objective. To determine if vitamin D deficiency and insulin resistance are risk factors for dyslipidemia in obese children.
Materials and Methods. This study was conducted in the Department of Pediatrics at Bagcilar Training and Research Hospital in Istanbul, Turkey between 2014 and 2015. Obese patients whose age range was 8-14 were included in the study. The serum triglyceride, total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, fasting glucose, insulin, alanine aminotransferase, vitamin D levels were measured; a liver ultrasonography was performed. Homeostatic model assessment (HOMA-IR), was used to calculate insulin resistance.
Results. 108 obese children were included; 39 (36.11%) had dyslipidemia. The average fasting blood glucose (88.74 ± 7.58 vs. 95.31 ± 6.82; p= 0.0001), insulin level (14.71 ± 12.44 vs. 24.39 ± 15.02; p= 0.0001) and alanine aminotransferase level (23.45 ± 11.18 vs. 30.4 ± 18.95; p= 0.018) were significantly higher in the children with dyslipidemia. In the dyslipidemic obese children, the average hepatosteatosis rate and HOMA-IR level were higher; 28 (71.9%) had hepatosteatosis, 37 (94.87%) had insulin resistance; the vitamin D levels were <20 ng/ml in 69.3%. Vitamin D deficiency was significantly more common (p= 0.033). The multivariate regression analysis confirmed that the increase in the HOMA-IR level (p= 0.015) and the low vitamin D level (p= 0.04) were important risk factors for dyslipidemia.
Conclusion. Obese children in our region exhibit low vitamin D and increased HOMA-IR levels, which are efficient risk factors of dyslipidemia.
Key words: Obesity; Dyslipidemias; Isulin resistance; Vitamin D deficiency.
INTRODUCTION
Childhood obesity continues into adult life and causes serious health problems.1 Low 25-hydroxyvitamin D [25(OH)D] levels have been reported to lead to dyslipidemia and other cardio-metabolic risk factors due to or independent of obesity.2 Vitamin D deficiency is common in the Middle East and Turkey due to the style of wearing skin-covering clothing and less exposure to sun.3 Vitamin D is critical for glucose homeostasis and insulin secretion via its endocrine mechanisms;4 in addition adipose tissue plays a major role as an autocrine and paracrine organ.5 Insulin resistance is important in obesity; as a population if obesity begins at younger ages, the age of onset of type 2 diabetes mellitus also decreases.6 The literatures report that obesty prevelace increases up to 7% among 6-15 years old children. This development was associated with increased cardiometabolic risk factors.7 According to Bogalusa Heart Study childhood obesity with increased total cholesterol (TC) and low-density lipoprotein (LDL-C) levels are related with atherosclerotic plaques.8 The literature includes conflicting data as to whether vitamin D deficiency is a risk factor for the development of insulin resistance in childhood obesity. Lifestyle factors contribute to vitamin D deficiency with obesity development. Obese children are usually sedentary, less likely to play outdoors and to be exposure to sunlight. Unhealthy high caloric food might be low in vitamin D and minerals. In obese subjects vitamin D accumulates in adipose tissue, so that bioavailability of vitamin D might be low.9 The objective was to determine if vitamin D deficiency and insulin resistance are risk factors for dyslipidemia in obese children.
MATERIALS AND METHODS Study Population
This present study was conducted in the Department of Pediatrics at Bagcilar Training and Research Hospital in Istanbul, Turkey between 2014 and 2015. The inclusion criteria for the study group consisted of being obese and ages between 8-14 years. Children whose obesity was the result of a syndromic condition (i,e, Laurence-Moon Biedl syndrome or Prader-Willi) and those who had an endocrinal cause, such as Cushing's syndrome or hypothyroidism were excluded. None of the participants used medication or had a history or evidence of current metabolic, cardiovascular, respiratory, or hepatic disease. Patients taking vitamins and/or mineral supplements were excluded. Medical records were evaluated for age, gender and physical examination findings.
Anthropometric measurements
Weights and heights of children were measured in the first examination in pediatric outpatient clinic. All anthropometric measurements were performed by the same pediatrician. Weights were measured with subjects in minimal (without shoes and with light clothing) underclothes, using a standard beam balance sensitive to 0.1 kg. Heights were determined to the nearest 1 mm using a portable Seca stadiometer. Body mass index (BMI) was calculated as weight (in kilograms) divided by height (in squared meters). BMI greater than the 95th percentile for their age and gender were considered obese.10
Laboratory measurements
All blood analyses were performed on 12 hour fasting samples from both the study and control groups. Total cholesterol (TC), high density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and triglycerides (TG) were measured using the homogenous colorimetric enzyme technique (Roche, Modular-P 800). The serum 25(OH)D levels were determined using the electrochemiluminescence enzyme immunoassay method (ECLIA) (ADVIA Centaur, USADPC Co, USA). Glucose was measured by the glucose oxidase technique (Siemens ADVIA 1800) and insulin levels were analyzed with a direct chemiluminescence technique (Siemens Centaur, USA). Serum TC levels over 200 mg/dL, TG levels over 150 mg/dL, LDL-C levels over 130 mg/dL, or HDL-C levels under 40 mg/dL were accepted as dyslipidemia adapted from the Lipid
Research Clinic Pediatric Prevalence Study and NCEP guidelines for children and adolescents.11 Vitamin D deficiency was defined as a serum 25(OH)D level <20 ng/mL.3 Insulin resistance was estimated from the fasting plasma measurements using homeostatic model assessment (HOMA-IR) [(insulin (mU/L) x glucose (mmol/L)/22,5)].6 The insulin resistance criterion was a HOMA-IR >2,5 for prepubescent children.4
Liver ultrasonography
The ultrasonographic (USG) examinations of all the children were evaluated by the same radiologist in the Department of Radiology Bagcilar Training and Research Hospital. USG evidence of NAFLD was based on the bright hepatic echo pattern, increased echo attenuation, and loss of intrahepatic architecture.
Sample size
We hypothesized that in obese children 25(OH) D level <20 ng/mL was associated with dyslipidemia. Based on previous findings, we calculated that a sample size of 94 would allow us to detect a difference in D vitamin levels between the 2 groups (a= 0.05, power= 80%), the a level was set at 0.05 based on a 2-sided, 2-sample t test.
The study proposal was approved by the Ethics Committee of Bagcilar Training and Research Hospital (Approval number: 2015-355). Written informed consent was obtained from the parents of the participants.
Statistical analysis
The statistical analysis was performed with NCSS (Number Cruncher Statistical System) 2007. A paired t-test was used to calculate the difference between two parameters in the groups, and one-way ANOVA was used to calculate the difference between two parameters in groups with more than two variables in the same group and between different groups. Associations were evaluated using logistic regression analysis. Categorical data were evaluated using the Chi square test; p < 0.05 was accepted as statistically significant.
RESULTS
A total of 108 obese children were included; 39 (36.11%) had dyslipidemia. The mean age was 11.51 ± 3.32 years in the dyslipdemic obese group and 10.95 ± 3.47 years in the non dyslipdemic obese group; the male/female ratio was 14/25 in the dyslipidemic group and 30/39 in the non dyslipidemic group. No statistically significant difference was observed between the age and gender distributions of the dyslipidemic and non-dyslipidemic children (p >0,05).
The average BMI was higher in the obese children with dyslipidemia than in those without dyslipidemia (p= 0.002). The average fasting blood glucose (88.74 ± 7.58 vs. 95.31 ± 6.82; p= 0.0001), insulin level (14.71 ± 12.44 vs. 24.39 ± 15.02; p= 0.0001), ALT level (23.45 ± 11.18 vs. 30.4 ± 18.95; p= 0.018) were significantly higher in the children with dyslipidemia. The average TC level (170.13 ± 29.07; p= 0.032), LCL-C level (102.15 ± 31.76; p= 0.004), TG level (155.43 ± 54.92; p= 0001) were significantly higher and HDL-C level (44.58 ± 17.99) was significantly lower in the dyslipidemic group (Table 1). Among patients with dyslipidemia, 28 (71.9%) had hepatosteatosis, 37 (94.87%) had insulin resistance. The mean vitamin D level was significantly lower in dyslipidemic obese children (25.27 ± 15.54 vs. 17.2 ± 10.43; p= 0.005). Among patients with dyslipidemia 27 (69.23%) had vitamin D deficiency. Vitamin D deficiency was significantly higher in the children with dyslipidemia compared to those without (p= 0.033) (Table 2).
Table 1. Laboratory characteristics of dyslipidemic and nondyslipidemic groups
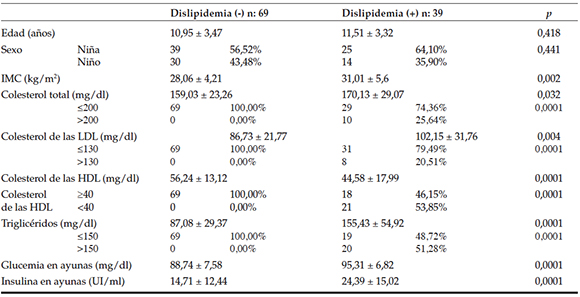
Table 2. Comparison of clinical and laboratory findings between dyslipidemic and nondyslipidemic obese children
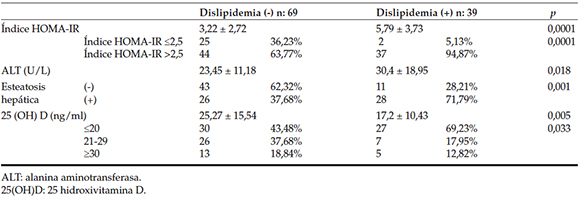
A logistic regression analysis was conducted with the BMI, Homa-IR, ALT, and vitamin D variables to determine secondary factors (excluding primary factors such as cholesterol, HDL, TG, and LDL) that affected the presence of dyslipidemia in the obese children. The HOMA-IR level (p= 0.015) and vitamin D deficiency (p= 0.04) were identified as effective secondary factors in the formation of dyslipidemia (Table 3).
Table 3. Logistic regresion analysis of BMI, HOMA-IR, ALT and 25(OH)D levels

DISCUSSION
Vitamin D is an essential hormone mainly for bone health and sustainability.12 In recent years, studies on the effects of vitamin D other than those on bone tissue have been carried out. Low 25(OH)D levels are associated with increased insulin resistance and impaired lipid profile.13 There are reviews and meta-analysis studies evaluating the relationship between the serum 25(OH)D level and metabolic syndrome and type 2 diabetes in adults.14 According to a limited amount of accessible meta-analysis information, there is a weak relationship between 25(OH)D and lipid profile in children and adolescents.15 Dyslipidemia increases proportionally with age and BMI.4 In our study, we did not observe a significant relationship between age, gender and dyslipidemia but in agreement with previous studies we did show a significant correlation with BMI. BMI is an important parameter in determining dyslipidemia and cardiovascular risk factors.16 We found that 25(OH)D deficiency was significantly more common in the obese dyslipidemic patients. However, the detection of low vitamin D levels in many studies in obese patients often showed a higher prevalence of vitamin D deficiency compared to the normal population.17 The relationship between obesity and vitamin D is controversial.18 Because vitamin D is a fat-soluble molecule, it can be withdrawn from circulation for storage in adipose tissues in obese individuals; therefore, there is actually a sufficient amount in the body, although low levels are measured in the serum.19 Because obese children are less active, they spend more time inside the home environment and therefore are not sufficiently exposed to the sun's rays, which causes insufficient synthesis in the skin.
Additionally, the bioavailability of 25(OH)D is decreased in obese children due to its sequestration in subcutaneous adipose tissue.18 Low vitamin D levels lead to obesity; thus, obesity has been shown to occur more frequently in adults with low vitamin D levels. A 3-year study that included the parameters BMI, skin thickness and waist circumference established that children with low vitamin D levels had increased adipose tissues in the upcoming years.20
Obese children and adolescents tended to exhibit increased total serum cholesterol, triglyceride and LDL levels and decreased HDL levels. Insulin resistance plays a role in these pro-atherogenic lipid changes.21 Due to the development of insulin resistance in obese children, insulin-dependent hormone-sensitive lipase and lipoprotein lipase cannot be inhibited and are involved in the pathogenesis of lipid metabolic disorders.22 The excess fatty tissue directly causes the lipotoxic effect and the release of immune cytokines by affecting insulin secretion and function.23 Due to the obesity and dyslipidemia, insulin cannot be used effectively. Compensatory hyperinsulinemia results in an increase in LDL, TG and free fatty acids and a decrease in HDL in the liver.24 In our study, serum triglyceride, LDL cholesterol level as well as HDL level were determined to be high in dyslipidemic obese children. Botella-Carretero et al.,25 reported in their study conducted on obese adults that HDL levels were low and TG levels increased in individuals with vitamin D level of 20 ng/ml. In a study performed by Dolinsky et al.,2 the presence of a contradictory relationship between vitamin D and lipid profile was reported. Kelishadi et al.,15 reported in their meta-analysis study that there was a weak but reverse relationship between vitamin D and TG. No relationship was reported in some studies.26 In a study, a reverse but not statistically significant relationship was reported between TG and vitamin D in male adolescents while a statistically significant relation was identified between TG and vitamin D in female adolescents.27 Again, in the studies in the literature, except for one study,26 the presence of a significant positive relationship between HDL level and vitamin D is reported. In the studies, a significant negative relationship is reported between LDL level and vitamin D. Only in 3 studies, a positive correlation which was not statistically significant was identified between LDL and vitamin D.28
In our study, high levels of insulin in the obese group with dyslipidemia seemed to result in a more pronounced HOMA-IR, which was consistent with the literature. Additionally, our study showed that a high HOMA-IR rate was an effective secondary factor for the development of dyslipidemia. A study involving 80 severely obese children and adolescents in the Netherlands showed cardiometabolic risk factors associated with the HOMA-IR.29 In our study, higher HOMA-IR values were detected in dyslipidemic obese children, and the number of cases with insulin resistance was found to be significantly higher in this group. This relationship was also highlighted in some previous studies.30,31
Non-alcoholic fatty liver disease is highly prevalent in obese children.32 Insulin resistance is responsible for the development of obesity-related non-alcoholic liver disease. Studies reported an incidence of hepatosteatosis in obese children of 12 to 72.9%.33 In our study, a dyslipidemic fatty liver was found in abdominal ultrasonography of 71.9% of the obese children. A high ALT level is typically accompanied by a fatty liver in childhood obesity.34 In our study, the ALT level appeared to be higher in our dyslipidemic patients than in the group of non-obese dyslipidemic patients. The study was carried out in Istanbul. Istanbul receives enough sunlight in the summer season. However, vitamin D deficiency is observed in children and adolescents in our region. They have inadequate sun exposure during winter despite living in a sunny climate. Vitamin D intake is also low in most children, due to little consumption of vitamin D food sources. The continuation of childhood vitamin D prophylaxis may be useful for the prevention of obesity and its complications. However, in a study conducted by Hirschler et al.,35 obese children with vitamin D deficiency were given vitamin D; when the group to which vitamin D was given and the group to which vitamin D was not given were compared, no change was determined in TG and
HDL concentration. This situation was attributed to nutrition and ethnic reasons. The effect of giving vitamin D to obese children with vitamin D deficiency in the treatment of dyslipidemia seems to be controversial.
The number of the patients included in our study was low. This is the weak aspect of the present study. The relation of vitamin D deficiency with dyslipidemia in obese children in the age group of 8-14 was investigated in the present study. The studies in the literature include adolescent and adult age groups. There are very few studies conducted on this age group. This is the strong aspect of the present study.
CONCLUSION
In our study, the HOMA-IR level and vitamin D deficiency were identified as effective secondary factors in the formation of dyslipidemia in obese children.
Acknowledgments
We would like to thank the patients and their families, at the Bagcilar Training and Research Hospital Department of Pediatrics, Istanbul, Turkey.
1. Park MH, Falconer C, Viner RM, Kinra S. The impact of childhood obesity on and mortality in adulthood: a systematic review. Obes Rev 2012;13(11):985-1000. [ Links ]
2. Dolinsky DH, Armstrong S, Mangarelli C, Kemper AR. The association between vitamin D and cardiometabolic risk factors in children: a systematic review. Clin Pediatr (Phila) 2013;52(3):210-23. [ Links ]
3. Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, et al. Endocrine Society. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2011;96(7):1911-30. [ Links ]
4. Gahagan S. Child and adolescent obesity. Curr Probl Pediatr Adolesc Health Care 2004;34(1):6-43. [ Links ]
5. Miraglia del Giudice E, Grandone A, Cirillo G, Capristo C, et al. Bioavailable Vitamin D in Obese Children: The Role of Insulin Resistance. J Clin Endocrinol Metab 2015;100(10):3949-55. [ Links ]
6. Al Harthi F, Huraib GB, Zouman A, Arfin M, et al. Apolipoprotein E gene polymorphism and serum lipid profile in Saudi patients with psoriasis. Dis Markers 2014;2014:39645. [ Links ]
7. Korsten-Reck U, Kromeyer-Hauschild K, Korsten K, Baumstark MW, et al. Frequency of secondary dyslipidemia in obese children. Vasc Health Risk Manag 2008;4(5): 1089-94. [ Links ]
8. Tracy RE, Newman WP 3rd, Wattigney WA, Berenson GS. Risk factors and atherosclerosis in youth autopsy findings of the Bogalusa Heart Study. Am J Med Sci 1995;310(Suppl 1):S37-41. [ Links ]
9. Roth CL, Elfers C, Kratz M, Hoofnagle AN. Vitamin D Deficiency in Obese Children and Its Relationship to Insulin Resistance and Adipokines. J Obes 2011;2011:495101. [ Links ]
10. Braga-Tavares H, Fonseca H. Prevalence of metabolic syndrome in a Portuguese obese adolescent population according to three different definitions. Eur J Pediatr 2010;169(8):935-40. [ Links ]
11. Daniels SR, Greer FR; Committee on Nutrition. Lipid screening and cardiovascular healthin childhood. Pediatrics 2008;122(1):198-208. [ Links ]
12. Hossein-nezhad A, Holick MF. Vitamin D for health: a global perspective. Mayo Clin Proc 2013;88(7):720-55. [ Links ]
13. Jorde R, Grimnes G. Vitamin D and metabolic health with special reference to the effect of vitamin D on serum lipids. Prog Lipid Res 2011;50(4):303-12. [ Links ]
14. Parker J, Hashmi O, Dutton D, Mavrodaris A, et al. Levels of vitamin D and cardiometabolic disorders: systematic review and meta-analysis. Maturitas 2010;65(3):225-36. [ Links ]
15. Kelishadi R, Farajzadegan Z, Bahreynian M. Association between vitamin D status and lipid profile in children and adolescents: a systematic review and meta-analysis. Int J Food Sci Nutr 2014;65(4):404-10. [ Links ]
16. Weisell RC. Body mass index as an indicator of obesity. Asia Pacific J Clin Nutr 2002;11(Suppl 7):681-84. [ Links ]
17. Olson ML, Maalouf NM, Oden JD, White PC, et al. Vitamin D deficiency in obese children and its relationship to glucose homeostasis. J Clin Endocrinol Metab 2012;97(1):279-85. [ Links ]
18. Mai XM, Chen Y, Camargo CA Jr, Langhammer A. Cross-Sectional and Prospective Cohort Study of Serum 25-Hydroxyvitamin D Level and Obesity in Adults. The HUNT Study. Am J Epidemiol 2012;175(10):1029-36. [ Links ]
19. Pourshahidi LK. Vitamin D and obesity: current perspectives and future directions. Proc Nutr Soc 2015;74(2):115-24. [ Links ]
20. Gilbert-Diamond D, Baylin A, Mora-Plazas M, Marin C, et al. Vitamin D deficiency and anthropometric indicators of adiposity in school-age children: a prospective study. Am J Clin Nutr 2010;92(6):1446-51. [ Links ]
21. Jago R, Harrell JS, McMurray RG, Edelstein S, et al. Prevalence of abnormal lipid and blood pressure values among an ethnically diverse population of eighth-grade adolescents and screening implications. Pediatrics 2006;117(6):2065-73. [ Links ]
22. Holland WL, Bikman BT, Wang LP, Yuguang G, et al. Lipid induced insulin resistance mediated by the proinflammatory receptor TLR4 requires saturated fatty acid-induced ceramide biosynthesis in mice. J Clin Invest 2011;121(5):1858-70. [ Links ]
23. Holland WL, Knotts TA, Chavez JA, Wang LP, et al. Lipid mediators of insulin resistance. Nutr Rev 2007;65(6 Pt 2): S39-46. [ Links ]
24. Savage DB, Petersen KF, Shulman GI. Disordered lipid metabolism and the pathogenesis of insulin resistance. Physiol Rev 2007;87(2):507-20. [ Links ]
25. Botella-Carretero JI, Alvarez-Blasco F, Villafruela JJ, Balsa JA, et al. Vitamin D deficiency is associated with the metabolic syndrome in morbid obesity. Clin Nutr 2007;26(5):573-80. [ Links ]
26. Nam GE, Kim DH, Cho KH, Park YG, et al. 25-Hydroxyvitamin D insufficiency is associated with cardiometabolic risk in Korean adolescents: the 2008-2009 Korea National Health and Nutrition ExaminationSurvey (KNHANES). Public Health Nutr 2014;17(1):186-94. [ Links ]
27. Ashraf AP, Alvarez JA, Gower BA, Saenz KH, et al. Associations of serum 25-hydroxyvitamin D and components of the metabolic syndrome in obese adolescent females. Obesity (Silver Spring) 2011;19(11):2214-21. [ Links ]
28. Williams DM, Fraser A, Sayers A, Fraser WD, et al. Associations of 25-hydroxyvitamin D2 and D3 with cardiovascular risk factors in childhood: cross-sectional findings from the Avon Longitudinal Study of Parents and Children. J Clin Endocrinol Metab 2012;97(5):1563-71. [ Links ]
29. Makkes S, Renders CM, Bosmans JE, van der Baan-Slootweg OH, et al. Cardiometabolic risk factors and quality of life in severely obese children and adolescents in The Netherlands. BMC Pediatr 2013;13(1):62. [ Links ]
30. Yoshitomi Y, Ishii T, Kaneki M, Tsujibayashi T, et al. Relationship between insulin resistance and effect of atorvastatin in non-diabetic subjects. J Atheroscler Thromb 2005;12(1):9-13. [ Links ]
31. Raitakari OT, Porkka KV, Ronnemaa T, Knip M, et al. The role of insulin in clustering of serum Lipids and blood pressure in children and adolescents. The cardiovascular risk in young finns study. Diabetologia 1995;38(9):1042-50. [ Links ]
32. Volovelsky O, Weiss R. Fatty liver disease in obese children--relation to other metabolic risk factors. Int J Pediatr Obes 2011;6(Suppl 1):59-64. [ Links ]
33. Fu JF, Shi HB, Liu LR, Jiang P, et al. Non-alcoholic fatty liver disease: an early mediator predicting metabolic syndrome in obese children? World J Gastroenterol 2011;17(6):735-42. [ Links ]
34. Boyraz M, Hatipo lu N, Sari E, Akçay A, et al. Non-alcoholic fatty liver disease in obese children and the relationship between metabolic syndrome criteria. Obes Res Clin Pract 2014;8(4):356-63. [ Links ]
35. Hirschler V, Molinari C, Maccallini G, Sanchez M, et al. Status of Dyslipidemia in Vitamin D Supplemented Argentinean Indigenous Children Versus A Non-supplemented Mixed Population Group. Cardiovasc Hematol Agents Med Chem 2015;13(2):129-36. [ Links ]











 text in
text in 


