Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista argentina de microbiología
versión impresa ISSN 0325-7541versión On-line ISSN 1851-7617
Rev. argent. microbiol. v.39 n.4 Ciudad Autónoma de Buenos Aires oct./dic. 2007
Biofilm formation by Stenotrophomonas maltophilia isolates from device-associated nosocomial infections
B. Passerini De Rossi1, M. Calenda1, C. Vay2, M. Franco1*
1 Cátedra de Microbiología, Facultad de Farmacia y Bioquímica, Universidad de Buenos Aires. Junín 956 (1113) Ciudad Autónoma de Buenos Aires. Argentina;
2 Laboratorio de Bacteriología Clínica, Departamento de Bioquímica Clínica, Hospital de Clínicas José de San Martín, Facultad de Farmacia y Bioquímica, Universidad de Buenos Aires. Córdoba 2150 (1113) Ciudad Autónoma de Buenos Aires. Argentina.
*Correspondence. E-mail: mfranco@ffyb.uba.ar
ABSTRACT
Medical devices are often colonized by bacteria which may cause severe infections. The aim of this work was to evaluate biofilm formation by S. maltophilia isolates from device-associated nosocomial infections. The 13 local isolates exhibited different capacities of biofilm formation on hydrophilic and hydrophobic surfaces. All isolates formed strong biofilms in polystyrene microplates, while strong, moderate or weak biofilms were detected in borosilicate (BS) or polypropylene (PP) tubes. The proficiency of biofilm formation was better evaluated by the level of crystal violet staining expressed relative to the final culture density. The microscopic analysis of biofilms formed on glass coverslips revealed the presence of a matrix of exopolysaccharides and microcolonies typical of biofilm architecture. Isolates with increased adhesion to BS showed larger microcolonies. According to our results, twitching correlated well with attachment to the three abiotic surfaces tested, while swimming only showed a slight correlation with biofilm formation on PP. Poor correlation was observed between cell surface hydrophobicity and biofilm formation. One of the highest biofilm-producing isolates adhered to urethral catheters of different materials, and exhibited an increased resistance to oxidative stress, one of the common stresses encountered by bacteria during the infection process due to the immune response.
Key words: Stenotrophomonas maltophilia; Biofilms; Swimming; Twitching; Hydrophobicity; Stress oxidative
RESUMEN
Formación de biopelículas por aislamientos de Stenotrophomonas maltophilia recuperados de infecciones nosocomiales asociadas al uso de dispositivos médicos. El objetivo de este trabajo fue evaluar la formación de biopelículas por parte de aislamientos de Stenotrophomonas maltophilia. Los 13 aislamientos locales evaluados mostraron diferente capacidad de formar biopelículas en superficies hidrofílicas e hidrofóbicas. Todos ellos formaron biopelículas fuertes en microplacas de poliestireno (PS), mientras que se observaron biopelículas fuertes, moderadas o débiles en tubos de borosilicato (BS) o polipropileno (PP). La medida del cristal violeta unido a la biopelícula expresada en función de la densidad final de los cultivos permitió una mejor evaluación de la eficiencia de formación de biopelículas. El análisis microscópico de biopelículas formadas sobre cubreobjetos mostró la presencia de una matriz de exopolisacáridos y microcolonias típicas de la arquitectura de las biopelículas. Los aislamientos con mayor adhesión a BS mostraron microcolonias de mayor tamaño. La motilidad asociada a superficies ( twitching) presentó buena correlación con la adhesión a BS, PP y PS, mientras que la motilidad asociada a flagelos solo correlacionó ligeramente con la adhesión a PP. La correlación entre la hidrofobicidad de la superficie bacteriana y la formación de biopelículas fue escasa. Uno de los aislamientos productores de biopelículas fuertes evidenció capacidad de adhesión a catéteres uretrales de diferentes materiales y mayor resistencia al estrés oxidativo.
Palabras clave: Stenotrophomonas maltophilia; Biopelículas; Motilidad; Twitching; Hidrofobicidad; Estrés oxidativo
INTRODUCTION
The increasing use of indwelling medical devices has led to a concomitant increase in the incidence of devicerelated infections. Bacteria colonizing medical devices grow as biofilms, constituted by a surface-associated community of cells that is embedded in a matrix of exopolysaccharides (EPS). Biofilms exhibit phenotypic characteristics that are distinct from planktonic organisms, including increased resistance to host immune defences and antimicrobial compounds (9).
In Pseudomonas aeruginosa, an important source of device-associated nosocomial infections, flagella, type IV pili and the alginate matrix have been implicated in biofilm formation (21). P. aeruginosa uses extracellular quorum-sensing (QS) signals to coordinate biofilm formation and development (4). It has been shown that microcolony formation is dependent on type IV pili-mediated twitching activity, a QS dependent motility (8). In addition, QS regulates the production of virulence factors such as elastase (23) and the oxidative stress response (13).
Stenotrophomonas maltophilia (formerly Pseudomonas and Xanthomonas maltophilia) is a widespread environmental Gram-negative bacillus that has become a nosocomial pathogen of increasing importance (5, 19). The putative virulence factors of S. maltophilia have not been defined yet. Extracellular enzymes including DNase, RNase, fibrinolysin, lipases, hyaluronidase, protease, and elastase may play a role in the pathogenesis of S. maltophilia- associated infection (2). A major extracellular alkaline serine protease that degrades several human proteins from serum and connective tissues, has recently been described (30). Infection occurs principally in immunocompromised patients and in patients exposed to invasive devices and/or broad spectrum antibiotics (5, 19). The most common site of isolation of S. maltophilia in hospitalized patients is the respiratory tract, being pneumonia associated with mechanical ventilation and the use of nebulizers. S. maltophilia can also cause catheterrelated bacteraemia, septicaemia and urinary tract infections. The ability of S. maltophilia to survive and multiply within intravenous infusates and in dialysis fluids and to adhere to catheters may contribute to the pathogenesis of hemodialysis and intravenous line-related infections (5, 19). Treatment of infections caused by this opportunistic pathogen can be difficult because of its intrinsic multiresistance to antibiotics, including carbapenems, and the presence of class 1 integrons described in trimethoprimsulfamethoxazole resistant isolates from nosocomial infections in Buenos Aires, Argentina (1). Furthermore, treatment may also be complicated by biofilm formation. S. maltophilia is able to adhere to glass and to several types of plastic materials including polyvinyl chloride and Teflon, some of which are used in the fabrication of medical devices (17, 19). De Oliveira-Garcia et al. (6, 7) have recently characterized flagella and fimbriae 1 (SMF-1) produced by S. maltophilia, suggesting that these surface determinants can be involved in biofilm formation.
The aim of this work was to evaluate the ability of local S. maltophilia isolates, from device-associated nosocomial infections, to form biofilms on different surfaces, and to evaluate their motility, cell surface hydrophobicity, and expression of potential virulence/survival factors.
MATERIALS AND METHODS
Bacterial isolates
S. maltophilia isolates used in this study were recovered from 13 patients with device-associated nosocomial infections, between January 2004-April 2005, at Hospital de Clínicas José de San Martín. Their sources and clinical manifestations are outlined in Table 1. Isolates were identified as S. maltophilia by conventional bacteriological tests. Identification was confirmed by using API 20NE strip (bioMérieux, Italy) according to the manufacturer's instructions. Isolates were kept frozen at -20 °C in 15% glycerol.
Table 1. Sources, clinical manifestations and biocodes of S. maltophilia isolates

Assays for biofilm formation
Preparation of inocula: S. maltophilia cultures were incubated on Trypticase soy agar (TSA, Oxoid Ltd, Basingstoke, Hampshire, England) for 24 h. Isolated colonies were inoculated into Trypticase soy broth (TSB, Oxoid), incubated overnight on a rotary shaker (200 rpm) and then cultures were diluted with fresh TSB to contain 5x107 to 1x108 cells/ml on the basis of the OD546. Standardized suspensions were used to inoculate TSB medium (1/100 dilution) for the following biofilm assays. Unless otherwise stated, all bacterial cultures were incubated at 35 °C. All assays were done at least three times using fresh samples each time.
1-Microplate assay
The microplate assay was performed as previously described (21) with the following modifications. Briefly, 200 ml of the inoculated TSB medium were added to the wells of sterile flatbottom 96-well polystyrene (PS) microtiter plates (TPP, tissue culture test plate, Switzerland) and incubated for 24 h and 48 h. Total cell biomass was estimated by measuring the OD546 using a plate reader (Metertech 960). Then, cultures were decanted and wells were washed with distilled water in order to remove non adherent cells, and 200 ml of 0.01% (w/v) Crystal Violet (CV, Mallinckrodt, Chemical Works, New York, USA) were added to the wells for 30 min. The stained biofilms were rinsed with distilled water and extracted with 200 µl of 95% ethanol. The amount of biofilm was quantified by measuring the OD546 of dissolved CV using the plate reader. Each isolate was assayed in octuplicate. Uninoculated medium controls (ODc) were included. The cutoff was defined as three standard deviations above the mean ODc (3). Each isolate was classified as follows: weak biofilm producer OD £ 2xODc, moderate biofilm producer 2xODc < OD £ 4xODc, or strong biofilm producer OD > 4xODc (28). The levels of CV staining were also expressed relative to the final cell density measured prior to biofilm assay (OD546 CV/OD546 growth ratio) (29).
2-Tube assay
Biofilm formation assay in tubes was performed as previously described (22) with the following modifications. Two mililiters of the inoculated TSB medium were incubated statically in borosilicate (BS) or polypropylene (PP) tubes for 24 and 48 h. For each condition, 4 tubes were processed. Two tubes were vortexed before determining the OD546 to estimate total cell biomass by using a Metrolab 1700 spectrophotometer. The other 2 tubes were aspirated, washed with distilled water and stained with 2 ml of CV (0.01%) for 30 min. The biofilm was visualized and classified as strong, moderate, weak or negative, and quantified by adding 2 ml of ethanol and measuring the OD546 of dissolved CV.
3-Biofilm formation on glass coverslips
Sterile microscope coverslips were aseptically placed into Petri plates along with 15 ml of the inoculated TSB and incubated for 24 and 48h. The coverslips were removed, rinsed with distilled water and stained with CV (0.1%), acridine orange (Sigma Chemical, St. Louis, MO, 30 mg/ml) or calcofluor (Fluorescent brightener 28, Sigma, 0.1%) for 10 min (20, 31). Then, coverslips were rinsed, mounted to the microscope slides and examined with a Zeiss Axiophot microscope (Carl Zeiss, Oberkochen, Germany) at LANAIS MIE, Instituto de Biología Celular y Neurociencia "Profesor E. De Robertis", Facultad de Medicina, UBA, Argentina. All images were obtained with a X40 lens objective. Acridine orange was detected using a standard fluorescein filter set (510/20 band-pass filter), and calcofluor emissions were detected using a DAPI filter set (395/97 bandpass filter).
4-Biofilm formation in urethral catheters
The ability of Sm13 to form biofilms was evaluated in the following urethral catheters: Nelaton red rubber (Lab. Barcat), Nelaton silicone (Silmag), and Koler PVC, Type K-93 (Deplamed SRL) as described by Joyanes et al. (16). Four catheters of each material were cut under sterile conditions into segments of 2 cm in length and incubated stationary in 4 ml of inoculated TSB for 24 h. Then, catheter segments were removed, washed, placed in a tube containing 3 ml of PBS, sonicated for 45 s at 45 Khz (Ultrasonic processor XL, Heat Systems) and vortexed for 30 s. The number of adhered cells was determined by viable counts on TSA. The number of cells in the biofilms was calculated as CFU/cm2. Colonized surfaces were also visualized by staining with CV (0.1%).
Presence of extracellular polymeric substances
Detection of a matrix of EPS was also carried out by qualitatively assessing the level of calcofluor or Congo red binding of colonies grown for 48h on LB agar supplemented with 50 µg/ml calcofluor or 40 mg/ml Congo red (Fluka AG, Buchs, Switzerland) (27). Fluorescence of the cells on calcofluor agar plates was observed under a 366 nm UV light source, and colony color was used to judge Congo red binding.
Motility Assays
Swimming, swarming and twitching agar plates were used to test different types of cell motility as described by Rashid and Kornberg (24). Briefly, plates were inoculated with bacteria from an overnight culture in LB agar with a sterile toothpick. Swimming and swarming plates were incubated at 30 °C for 12-14 h and 48 h respectively. Twitching plates, incubated at 35 °C and also at 30 °C (8) for 48 h, were examined for a haze zone of growth at the interface between the agar and the Petri plate. Then, the agar was removed, the plate was stained with CV (01%) for 30 min and twitching activity was measured in milimeters (8). The plates were also microscopically examined. Motility assays were carried out in triplicate.
Cell surface hydrophobicity
Cultures grown statically in TSB for 48 h were harvested by centrifugation for 5 min at 13.000 rpm. Cell pellets were resuspended in PBS, centrifuged and washed twice. The OD546 of cell suspensions in fresh PBS was adjusted to 0.3 for microbial adhesion to hydrocarbon (MATH) test, and to 1.2 for the salt aggregation test (SAT). Hydrophobicity assays were carried out in triplicate.
1 - MATH test. Four-ml samples of a cell suspension (initial OD, ODI) were transferred to individual test tubes containing 0.45 ml of xilene (Merk). The tubes were vortexed for 1 min and left to stand for 30 min to allow phase separation. Then the OD546 of the aqueous phase was measured (final OD, ODF). Surface hydrophobicity was expressed as a percentage according to the following formula: [1- (ODF /ODI)] x 100 (16).
2 - SAT assay: Fifteen µl of bacterial suspension were mixed with 15 ml of increasing concentrations (0.05-4.00 M) of ammonium sulfate solution. The mixture was gently agitated for 1 min on a glass slide. The lowest concentration of ammonium sulfate at which aggregation of bacteria occurred was regarded to be characteristic for the cell-surface hydrophobicity (18).
3 - Replica method. Rosenberg's method (25) was used for screening of bacterial colonies for cell surface hydrophobicity. Briefly, TSA cultures were replicated onto the polystyrene lids of Petri plates, washed with tap water and stained with CV (0.1%). Adherence of bacterial isolates to PS correlates with cell surface hydrophobicity.
Sensitivity to oxidative stress
1) Sensitivity to hydrogen peroxide was determined by the disk assay described by Hassett et al. (12) with the following modifications. S. maltophilia 48 h TSB-grown cultures were normalized for number of organisms by diluting with fresh TSB to provide an OD546 of 1.0, and 100-ml aliquots were spread on TSA plates. Sterile Whatman filter paper disks (7mm diameter) were impregnated with 3 µl of 12% H2O2 and placed in triplicate on each plate. Sensitivity to H2O2 was determined as zones of growth inhibition surrounding each disk after incubation at 35 °C for 24 h. Experiments were performed in triplicate.
2) H2O2 challenge conditions (11). Overnight S. maltophilia cultures , incubated with shaking, were diluted to contain 5x107 to 1x108 cells/ml, and H2O2 (30%) solution was added to a final concentration of 20 mM. The reaction mixtures were incubated at 35 °C in a shaker. After 30 min, samples were taken for plate counting on TSA. The level of survival was calculated as the percentage of the number of CFU after treatment relative to the number of CFU at zero time, just prior to H2O2 exposure. All experiments were done three times with duplicate plating.
Protease and lipase assays
Exoenzyme production was tested by streaking isolates on appropriate indicator plates. The proteolytic activity was visualized on nutrient broth casein agar plates (Nutrient agar, Difco, supplemented with 1% skim milk powder) and the lipolytic activity on tributyrin agar base containing 1% glycerol tributyrate (both Merck) (15). Quantification was done by placing a 40µl volume of S. maltophilia cultures (OD546 of 1.0) in 7.2 mm diameter wells previously cut into the appropriate agar plate. The plates were incubated for 48 h and the diameter of the zones of hydrolysis was measured. Protease and lipase activities were also evaluated in supernatants. Cultures were centrifuged for 5 min at 13.000 rpm in a microcentrifuge and the supernatant fractions were sterilized by filtration. A 40 µl volume of the supernatant was assayed as described above. Experiments were performed in duplicate.
Statistical Analysis
Statistical analyses were conducted using one-way analysis of variance (ANOVA) followed by the pair comparison of Tuckey's test. A significance level of 0.05 or 0.01 was used.
RESULTS AND DISCUSSION
Identification of S. maltophilia isolates
Thirteen gram-negative, non-glucose fermenting and oxidase-negative isolates were identified as S. maltophilia by conventional bacteriological methods. All the isolates were DNase and gelatinase producers and exhibited biochemical profiles typical of S. maltophilia when examined by the API20NE system. Three biocodes were determined based on the ability to reduce nitrate to nitrite and to assimilate citrate (Table 1).
Biofilm formation on different abiotic surfaces
S. maltophilia isolates were screened for biofilm formation in borosilicate (BS) and polypropylene (PP) tubes and in polystyrene (PS) microplates. Firstly, total cell biomass was estimated. More than 50% of the isolates showed higher ODs at 48 h than at 24 h ( P < 0.05), and the OD546 increasing was greater in BS and PP tubes (Figure 1) than in microplates (data not shown). At 48 h, ODs ranging from 0.7 to 1.4 were detected in tubes and microplates, and Sm9, Sm11, Sm14, Sm20 and Sm26 showed ODs < 1.0. Secondly, the amount of biofilm was quantified by measuring the OD546 of dissolved CV. The same time-dependent adherence profile was observed for all the isolates on BS and PS, with a maximum level reached at 24 h, and no substantial increase in adherence beyond this period except for Sm11 on BS ( P < 0.05) and Sm27 on PS ( P < 0.01) (Figure 2A and 2B). This profile was also observed for 8 isolates on PP, but for the other 5 isolates, biofilm attachment increased by 48 h ( P < 0.05) (Figure 2A and 2B). For all surfaces, time-dependent adherence profiles were independent of the growth rate. Bacterial attachment, determined by CV staining, measures the total biofilm biomass, including adherent bacteria and slime (EPS). The increment of CV values by 48 h could reflect an increment of adherent cells and/or in EPS production.

Figure 1. S. maltophilia growth in borosilicate tubes. Standardized overnight cultures of S. maltophilia isolates were diluted 100- fold in TSB and incubated in borosilicate (BS) tubes, statically at 35 °C. Bacterial growth was determined by OD546 at 24 h and 48 h. Error bars represent standard deviations of the mean from one representative experiment.

Figure 2. S. maltophilia biofilm formation on different surfaces evaluated by crystal violet staining. Isolates were cultured at 35 °C in TSB in borosilicate and polypropylene tubes, and polystyrene microtiter plates for (A) 24 h and (B) 48 h. After incubation, planktonic cells were removed and attached cells were stained with crystal violet (CV). The amount of biofilm was quantified by measuring the OD546 of CV dissolved in ethanol. Results represent the mean and standard deviation of a representative experiment.
Since the level of attachment for some isolates increased over time, we decided to compare biofilm formation on different surfaces at 48 h of incubation. Each isolate was classified as weak 0.2 < OD546 ≤ 0.4, moderate 0.4 < OD546 ≤ 0.8, or strong biofilm producer OD546 > 0.8 on PS (28). We propose to use the same ranges for classifying biofilms formed in BS and PP tubes, which correlate with the visual score. All the isolates formed strong biofilms on PS, while differences in biofilm formation ability were observed in BS and PP tubes (Figure 2B). On BS, 11 isolates formed strong biofilms, while Sm9 and Sm10 produced weak and moderate biofilms respectively. On PP, 9 isolates formed strong biofilms while Sm9, Sm11, Sm16 and Sm19 formed moderate biofilms, and no weak biofilms were detected (Figure 2B). Visual inspection of biofilm formation in tubes has been widely used as a basic screening method for slime-producing bacteria. Interestingly, weak biofilms were visualized as a ring of cells adhered to the BS tube wall at the air-liquid interface, while moderate and strong biofilms coated the bottom and walls of the BS and PP tubes (data not shown).
In the microplate test, statistical analysis of the mean CV values of the isolates classified as strong biofilm pro- ducers allowed us to distinguish three significantly different groups ( P < 0.01) with higher (Sm13 and Sm18), intermediate (9 isolates) or lower (Sm9 and Sm10) levels of CV staining. Conversely, in the tube test, only isolates belonging to the very low or very high end of the biofilm formation spectrum could be differentiated. These results reflect the fact that the microplate test is superior to the tube test in terms of accuracy (28). Even though the microplate assay is the technique of choice for further studies, the tube assay revealed that all the isolates can form biofilms on hydrophilic (BS) and on hydrophobic (PP) surfaces, and that Sm13 and especially Sm19 adhered better to BS than to PP (P < 0.05). It has been proposed that adherence of S. maltophilia to BS is promoted by the positive charge of the bacteria at physiologic pH, probably originated by proteins located in the outer membrane (17).
Figure 3 shows the levels of CV staining relative to the final culture density, a relationship used to avoid variations due to differences in bacterial growth. This relationship reflects that Sm14, Sm20 and Sm26 (ODs growth < 1.0) were as efficient in producing biofilms as Sm13 and Sm18, which showed the highest CV values on PS, BS and PP (Figure 2B). In contrast, Sm9, which also exhibited low growth values, presented the lowest OD546 CV/ OD546 growth ratio on BS and PP. Besides, Sm10, with the highest final culture density, showed the lowest ratio on PS and BS (Figure 3). These results show that biofilm formation and final culture density do not correlate, and are in agreement with previous reports on Listeria monocytogenes (10).

Figure 3. S. maltophilia biofilm formation on different surfaces evaluated by crystal violet staining relative to the final culture density. Isolates were grown in borosilicate and polypropylene tubes, and polystyrene microtiter plates for 48 h, and assayed for biofilm-forming capacity as described in Figure 2. The levels of CV staining are expressed relative to the final culture density (OD546 CV/OD546 growth ratio). Bars represent averages of a representative experiment.
Biofilm formation on glass coverslips.
Biofilm formation was also evaluated microscopically at 24 h and 48 h. For some isolates, as the incubation time extended, the number of attached bacteria increased throughout the abiotic surface. Figure 4 shows micrographs from Sm10 and Sm13 biofilms at 48 h of incubation in TSB. Light microscopy examination of Sm10 cells attached to BS coverslips and stained with CV showed the presence of individual bacteria and small clusters of cells (microcolonies), which did not coalesce to form a monolayer. In contrast, Sm13 presented extended areas of the glass surface covered with large microcolonies, three-dimensional bacterial structures typical of biofilms (Figure 4A). Similar results were obtained when S. maltophilia cells stained with acridine orange were examined with epifluorescence microscopy (Figure 4B). For the other isolates, biofilm formation on coverslips also correlated with the adherence to BS described above. When stained with acridine orange, cells and microcolonies fluoresced orange, suggesting an elevated RNA content and high physiological activity. Cells that fluoresced green, which are presumed to have reduced RNA content and low activity, were not observed. Fluorescence microscopy of samples stained with calcofluor white, that binds to β-linked polysaccharides such as cellulose and chitin, showed that the cells attached to BS were embedded within a blue fluorescent material (Figure 4C). The presence of an EPS matrix was detected in all isolates, and Sm10 and Sm20 formed more compact structures that resembled cotton wool.

Figure 4. S. maltophilia biofilms formation on glass coverslips. Sm10 (upper panel) and Sm13 (lower panel) biofilms formed on borosilicate coverslips were stained with (A) crystal violet and examined with light microscopy, or were stained with (B) acridine orange or with (C) calcofluor and examined with epifluorescence microscopy. Biofilms, grown in TSB for 48 h, were viewed at 400x magnification. CV and acridine orange staining show that Sm13 forms larger microcolonies than Sm10. Calcofluor stains cells as discrete points while EPS is visible as a continuous sheet. Sm10 forms more compact structures that resembled cotton wool.
Extracellular polymeric substances were detected by assessing the level of calcofluor or Congo red binding of colonies as described in Materials and Methods. According to the described fluorescence microscopy results, all the isolates fluoresced under long-wave UV light on calcofluor agar plates. Congo red stains starch, amylase, cellulose and polysaccharides containing contiguous b- (1![]() 4)-linked D-glucopyranosyl units or β -(1
4)-linked D-glucopyranosyl units or β -(1![]() 3)-Dglucans and has been used to detect EPS constituting the extracellular materials of biofilms (31). Isolates screened for their ability to bind Congo red on agar plates showed red colonies. Further studies are needed to know the real nature of the EPS matrix of S. maltophilia.
3)-Dglucans and has been used to detect EPS constituting the extracellular materials of biofilms (31). Isolates screened for their ability to bind Congo red on agar plates showed red colonies. Further studies are needed to know the real nature of the EPS matrix of S. maltophilia.
Biofilm formation in urethral catheters
Indwelling urethral catheters are highly used in modern medicine, unfortunately providing access to bacteria. Sm13, one of the highest biofilm-producing isolates, was selected for assessing its ability to adhere to rubber, silicone and PVC urethral catheters. After 24 h of incubation, Sm13 showed high adhesion capability to rubber (3.99 ± 0.91 x 106 CFU/cm2), silicone (2.95 ± 0.21 x 106 CFU/cm2), and PVC (3.78 ± 0.41 x 106 CFU/cm2), with no differences being found among the three biomaterials ( P < 0.001). Sm13 biofilms, visualized by staining with CV, were observed all along the clear surfaces of silicone and PVC catheters.
Swimming and twitching motilities
S. maltophilia isolates, inoculated in defined solid media used to detect motility, showed the ability to move via swimming and twitching, but we could not detect swarming motility under the experimental conditions used in this study. On semi-solid agar plates the zones of swimming ranged from 8 to 28 mm. Statistical analysis of the mean diameter of the swimming zones allowed to distinguish three significantly different groups ( P < 0.01) with slightly impaired, intermediate or increased swimming activity (Table 2).
Table 2. Motility and cell surface hydrophobicity of S. maltophilia isolates
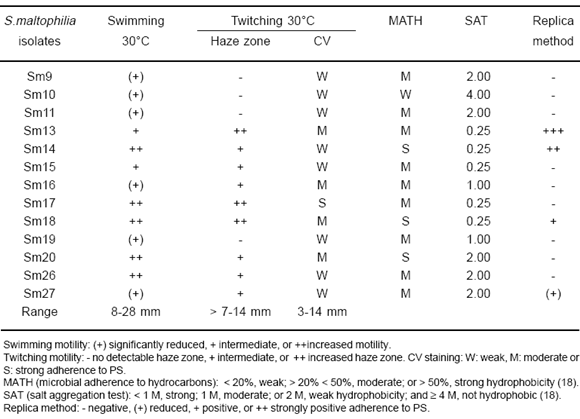
On twitching plates, 9 isolates formed a haze zone of growth at the interface between the agar and the Petri plate, surrounding the surface colony (Table 2). After staining with CV, the diameter of the twitching zones ranged from 3 to 14 mm, and 3 significantly different groups ( P < 0.01) were defined on the basis of statistical analysis (Table 2). Isolates with diameters < 7 mm did not present a haze zone of growth. CV staining also revealed weak, moderate or strong adherence of S. maltophilia isolates to PS (Table 2). The observation of the stained cells area on the PS plate demonstrated striking differences between the adherence patterns of S. maltophilia isolates. Most of them displayed an expanding donut-shaped adherent zone, indicating that only the outer side of the twitching area was attached, whereas for other isolates (Sm13, Sm17 and Sm20) cells remained adherent to the PS surface. Microscopic analysis of twitching motility revealed the leading edge of the twitching zone to be made up of rafts of monolayered cells moving away from the colony edge. Behind these rafts, an arrangement of multilayered cells and microcolonies were seen for moderate and strong adherent isolates, confirming a bacterial adherence typical of biofilms (data not shown).
When twitching activity was assayed at 35 °C, the diameters were generally smaller than those observed at 30°C and only 4 isolates presented a haze zone of twitching. Thus, for future studies we propose to detect this activity at 30 °C.
Figure 5 shows that twitching activity correlated well with biofilm formation, evaluated by CV staining, on the three tested surfaces ( r2 > 0.70). On the other hand, swimming motility only showed a slight correlation with biofilm formation on PP ( r2 = 0.53), but not on BS and PS ( r2 < 0.35) (data not shown). These results are in accordance with reports that correlated S. maltophilia twitching activity with biofilm formation in PS microtiter plates (14), and suggested a role of flagella in adherence to abiotic surfaces (6).

Figure 5. Correlation between twitching motility and biofilm formation on different surfaces: (A) polypropylene, B) borosilicate and C) polystyrene. Biofilms, grown for 48 h in TSB in tubes and microplates, were quantified by measuring the OD546 of CV. Twitching motility was measured in milimeters after 48-h incubation at 30 °C as described in Materials and Methods. Trends are shown as a straight line, with the corresponding correlation coefficient ( r2).
Cell surface hydrophobicity
The cell surface hydrophobicity of S. maltophilia isolates was assayed by means of bacterial aggregation in ammonium sulfate, as well as by affinity of the bacteria to xylene, and to PS surfaces using a replica assay (Table 2). The MATH test revealed that all isolates presented moderate or strong hydrophobicity, except Sm10, while SAT values allowed to classify isolates as strong, moderate, weak or not hydrophobic. On the other hand, the replica method identified 4 isolates that adhered to PS. Thus, as earlier reported (18, 26), evaluation by a single method seemed not to reliably determine the surface hydrophobicity of isolates. Differences in size, number, and/or distribution of bacterial surface hydrophobins may explain the observed variation among different assays. Nevertheless, for some isolates, we observed a good correlation among the three assays. Sm10 showed the least hydrophobic surface, whereas Sm13, Sm14 and Sm18 were the most hydrophobic isolates. Furthermore, for these isolates hydrophobicity correlated with the proficiency of biofilm formation based on OD546 CV/OD546 growth ratios. Hydrophobicity of bacterial cell surfaces has been considered to affect several biological phenomena such as attachment of bacteria to host tissues and adhesion of bacteria to solid surfaces (26). A recent report suggests that SMF-1 fimbriae, a surface hydrophobic determinant, is involved in S. maltophilia biofilm formation and adherence to cultured mammalian cells (7). However, other authors have not observed a relationship between bacterial adherence and bacterial surface hydrophobicity (16).
Expression of potential virulence/survival factors
Sensitivity to oxidative stress
S. maltophilia isolates tested for their sensitivity to H2O2, using a zone of inhibition assay, showed diameters ranging from 14 to 21 mm. On the basis of statistical analysis, 3 significantly different groups were defined, which showed increased resistance (14-16 mm) (Sm13), intermediate resistance (>16-18 mm) (Sm17, Sm18 and Sm 26), or reduced resistance to hydrogen peroxide (>18 mm) (9 isolates, including Sm10). Then, we examined the ability of representative isolates of these 3 groups to survive after 30-min challenge with 20 mM H2O2. There was no clear difference in survival to H2O2 between Sm10 (0.09% ± 0.06) and Sm18 (0.41% ± 0.12), whereas the level of survival of Sm13 was significantly higher (3.08% ± 0.77) ( P < 0.05). Thus, Sm13 showed an increased resistance to oxidative stress, one of the common stresses encountered by bacteria, especially during the infection process due to reactive oxygen species produced by phagocytic cells.
Protease and lipase activities
All isolates were lipase and protease producers, exoenzymes that could play a role in the pathogenesis of S. maltophilia-associated infections (2). Proteolytic activity was detected in cultures and in cell-free culture supernatants, whereas lipolytic activity was only detected in cultures. S. maltophilia cultures showed zones of lipolysis ranging from 11 to 17 mm, and Sm14 and Sm18 showed the highest values. This technique could be used to compare the lipolytic activity of isolates at equivalent cell densities. On casein agar plates, cultures showed zones of proteolysis ranging from 14 to 21 mm, and supernatants exhibited values from 9 to 18 mm. Sm13, Sm19 and Sm 26, showed the highest values in cultures and supernatants, whereas, 2 isolates showed high proteolytic activity in cultures but reduced activity in supernatants. These results suggest that S. maltophilia may produce various kinds of proteases, some of them detectable in supernatants under the conditions tested.
We did not observe correlation between exoenzyme production or sensitivity to oxidative stress and the proficiency of biofilm formation.
In summary, the results presented in this work show that 13 local S. maltophilia isolates, from device-associated nosocomial infections, exhibited different capacities of in vitro biofilm formation on hydrophilic and hydrophobic surfaces. Furthermore, we could not find any correlation between biofilm formation and final culture density. Isolates with increased adhesion to BS showed larger microcolonies in biofilms formed on glass coverslips, and microscopic analysis suggest that EPS is likely involved.
In our experience, the microplate test is superior to the tube test in terms of accuracy, and since S. maltophilia isolates adhered strongly to PS, it will be the technique of choice for further studies. However, the tube assay is an alternative method to test different surfaces. We propose to use the CV optical density ranges, described for classifying biofilm producer isolates on PS microplates, also for BS and PP tubes. The proficiency of biofilm formation among the isolates was best evaluated by using the OD546 CV/OD546 growth ratio.
According to our results, twitching motility correlated well with attachment to the three abiotic surfaces tested, whereas swimming only showed a slight correlation with biofilm formation on PP. The effect of hydrophobicity on biofilm formation was only observed in some isolates. Interestingly, one of the highest biofilm-producing isolates, adhered to different urethral catheters, and exhibited an increased resistance to oxidative stress.
Further studies are needed to better understand the biofilm formation mechanisms of this emerging nosocomial pathogen and to develop strategies for biofilm control. In this sense, we are currently using the described methodology to evaluate the influence of iron and calcium availability on S. maltophilia biofilm formation.
Acknowledgements: This work was financed by grant B082 from UBACYT. We thank Dr. Juan José López-Costa, LANAIS MIE, for help provided with microscopy. LANAIS MIE. Instituto de Biología Celular y Neurociencia "Profesor E. De Robertis". Facultad de Medicina, UBA, Argentina.
REFERENCES
1. Barbolla R, Catalano M, Orman B, Famiglietti A, Vay C, Smayevsky J, Centron D, Pineiro S. Class 1 integrons increase trimethoprim-sulfamethoxazole MICs against epidemiologically unrelated Stenotrophomonas maltophilia isolates. Antimicrob Agents Chemother 2004; 48: 666-9.
2. Bottone E, Reitano M, Janda J, Troy K, Cuttner J. Pseudomonas maltophilia exoenzyme activity as correlate in pathogenesis of ecthyma gangrenosum. J Clin Microbiol 1986; 24: 995-7.
3. Christensen G, Simpson W, Younger J, Baddour L, Barret F, Melton D, Beachey E. Adherence of coagulase-negative staphylococci to plastic tissue culture plates: a quantitative model for the adherence of staphylococci to medical devices. J Clin Microbiol 1985; 22: 996-1006.
4. Davies D, Parsek M, Pearson J, Iglewski B, Costerton J, Greenberg E. The involvement of cell-to-cell signals in the development of a bacterial biofilm. Science 1998; 280: 295-8.
5. Denton M., Kerr K. Microbiological and clinical aspects of infection associated with Stenotrophomonas maltophilia. Clin Microbiol Rev 1998; 11: 57-80.
6. de Oliveira-Garcia D, Martinez M, Dall'Agnol M, Rosales M, Azzuz A, Girón J. Characterization of flagella produced by clinical strains of emerging opportunistic pathogen Stenotrophomonas maltophilia. Emerg Infect Dis 2002; 8: 918-23.
7. de Oliveira-Garcia D, Dall'Agnol M, Rosales M, Azzuz A, Alcántara N, Martinez M, Girón J. Fimbriae and adherence of Stenotrophomonas maltophilia to epithelial cells and to abiotic surfaces. Cell Microbiol 2003; 5: 625-36.
8. Deziel E, Comeau Y, Villemur R. Initiation of biofilm formation by Pseudomonas aeruginosa 57RP correlates with emergence of hyperpiliated and highly adherent phenotypic variants deficient in swimming, swarming and twitching motilities. J Bacteriol 2001; 183: 1195-204.
9. Donlan R, Costerton J. Biofilms: survival mechanisms of clinically relevant microorganisms. J Clin Microbiol Rev 2002; 15: 167-93.
10. Djordjevic D, Wiedmann M, McLandsborough L. Microtiter plate assay for assessment of Listeria monocytogenes biofilm formation.Appl Environ Microbiol 2002; 68: 2950-8.
11. Elkins J, Hassett D, Stewart P, Schweizer H, McDermott T. Protective role of catalase in Pseudomonas aeruginosa biofilm resistance to hydrogen peroxide. Appl Environ Microbiol 1999; 65: 4594-600.
12. Hassett D, Schweizer H, Ohman D. Pseudomonas aeruginosa sodA and sodB mutants defective in manganeseand iron-cofactored superoxide dismutase activity demonstrate the importance of the iron-cofactored form in aerobic metabolism. J Bacteriol 1995; 177: 6330-37.
13. Hassett D, Ma J, Elkins J, McDermott T, Ochsner U, West S, et al. Quorum sensing in Pseudomonas aeruginosa controls expression of catalase and superoxide dismutase genes and mediates biofilm susceptibility to hydrogen peroxide. Mol Microbiol 1999; 34: 1082-93.
14. Huang T, Somers E, Wong A. Differential biofilm formation and motility associated with lipopolysaccharide/exopolysaccharide- coupled biosynthetic genes in Stenotrophomonas maltophilia. J Bacteriol 2006; 188: 3116-20.
15. Huber B, Riedel K, Hentzer M, Heydorn A, Gotschlich A, Givskov M et al. The cep quorum-sensing system of Burkholderia cepacia H111 controls biofilm formation and swarming motility. Microbiology 2001; 147: 2517-28.
16. Joyanes P, Pascual A, Martínez-Martínez L, Hevia A, Perea J. In vitro adherence of Enterococcus faecalis and Enterococcus faecium to urinary catheters. Eur J Clin Microbiol Infect Dis 2000; 19: 124-7.
17. Jucker B, Harms H, Zehnder A. Adhesion of the positively charged bacterium Stenotrophomonas (Xanthomonas) maltophilia 70401 to glass and teflon. J Bacteriol 1996; 178: 5472-9.
18. Krepsky N, Rocha Ferreira R, Ferreira Nunes A, Casado Lins U, Costa e Silva Filho F, de Mattos-Guaraldi A, Nettodos Santos K. Cell surface hydrophobicity and slime production of Staphylococcus epidermidis Brazilian isolates. Curr Microbiol 2003; 46: 280-6.
19. Looney W. Role of Stenotrophomonas maltophilia in hospital- acquired infection. BJBS 2005; 62: 145-54.
20. Neu T, Kuhlicke U, Lawrence J. Assessment of fluorochromes for two-photon laser scanning microscopy of biofilms. Appl Environ Microbiol 2002, 68: 901-9.
21. O'Toole G, Kolter R. Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development. Mol Microbiol 1998; 30: 295-304.
22. O'Toole G, Pratt L, Watnick P, Newman D, Weaver V, Kolter R. Genetic approaches to study of biofilms. Methods Enzymol 1999; 310: 91-109.
23. Pearson J, Pesci E, Iglewski B. Pearson J, Pesci E, Iglewski B. Roles of Pseudomonas aeruginosa las and rhl quorumsensing systems in control of elastase and rhamnolipid biosynthesis genes. J Bacteriol 1997; 179: 5756-67.
24. Rashid M, Kornberg A. Inorganic polyphosphate is needed for swimming, swarming, and twitching motilities of Pseudomonas aeruginosa. Proc Natl Acad Sci U S A 2000; 97: 4885-90.
25. Rosenberg M. Bacterial adherence to polystyrene: a replica method of screening for bacterial hydrophobicity. Appl Environ Microbiol 1981; 42: 375-7.
26. Rosenberg M, Doyle RJ. Microbial cell surface hydro-phobicity: history, measurement and significance. In: Rosenberg M, Doyle RJ, editors. Microbial cell surface hydrophobicity. Washington D.C., ASM Press, 1990, p. 1-30.
27. Solano C, Garcia B, Valle J, Berasain C, Ghigo J, Gamazo C, Lasa I. Genetic analysis of Salmonella enteritidis biofilm formation: critical role of cellulose. Mol Microbiol 2002; 43: 793-808.
28. Stepanovic S, Cirkovic I, Ranin L, Svabic-Vlahovic M. Biofilm formation by Salmonella spp. and Listeria monocytogenes on plastic surface. Lett Appl Microbiol 2004; 38: 428-32.
29. Tenorio E, Saeki T, Fujita K, Kitakawa M, Baba T, Mori H, Isono K. Systematic characterization of Escherichia coli genes/ORFs affecting biofilm formation. FEMS Microbiology Letters 2003; 225: 107-14.
30. Windhorst S, Frank E, Georgieva D, Genov N, Buck F, Borowski P, Weber W. The major extracellular protease of the nosocomial pathogen Stenotrophomonas maltophilia. J Biol Chem 2002; 277: 11042-9.
31. Yi K, Rasmussen A, Gudlavalleti S, Stephens D, Stojiljkovic I. Biofilm formation by Neisseria meningitidis. Infect Immun 2004; 72: 6132-8. [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ]
Recibido: 18/12/06 - Aceptado: 17/08/07














