Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista argentina de microbiología
versión impresa ISSN 0325-7541versión On-line ISSN 1851-7617
Rev. argent. microbiol. v.42 n.2 Ciudad Autónoma de Buenos Aires abr./jun. 2010
MICROBIOLOGÍA CLÍNICA Y ENFERMEDADES INFECCIOSAS
Bacterial pathogens associated with bloody diarrhea in Uruguayan children
M. I. Mota1, M. P. Gadea1, S. González1, G. González1, L. Pardo1, A. Sirok1, M. Rivas2, G. Algorta1, F. Schelotto1, G. Varela1 *
1Departamento de Bacteriología y Virología, Facultad de Medicina. Instituto de Higiene "Arnoldo Berta". Av. A. Navarro 3051 (11600) Montevideo, Uruguay;
2Servicio Fisiopatogenia, Instituto Nacional de Enfermedades Infecciosas-ANLIS "Dr. Carlos G. Malbrán". Av. Vélez Sarsfield 563 (1281) Ciudad Autónoma de Buenos Aires, Argentina.
*Correspondence. E-mail: gvarela@higiene.edu.uy
ABSTRACT
Diarrheal disease continues to be a serious health problem, especially in developing countries. Bloody diarrhea represents approximately 20-30% of all cases and has higher morbidity and mortality. Treatment with antibiotics is beneficial in cases of Shigella, Campylobacter, Yersinia and Salmonella infection, principally in those children with a higher risk of invasive disease. The aims of this study were to detect the bacterial agents associated with bloody diarrhea in children and to determine their antimicrobial susceptibility patterns. Between June 2001 and January 2008, 249 children with bloody diarrhea were studied. Shigella and Shiga toxin-producing Escherichia coli (STEC) were recovered from 48 (19.3%) and 3 (1.2%) of the total of cases, respectively. In 49 out of 249 children, in whom other enteropathogens were investigated, we recovered Campylobacter jejuni from 7 children (14.3%), Salmonella spp. from 2 (4.1%) and Aeromonas spp. from 1 (2%) in addition to Shigella from 7 children (14.3%). Thirty-four (70%) Shigella isolates showed resistance to ampicillin and 13 (27%) to trimethoprim-sulfamethoxazole. All Shigella isolates were susceptible to nalidixic acid, ciprofloxacin and ceftriaxone. Salmonella and STEC isolates were susceptible to all antibiotics assayed. Thus, the use of trimethoprim-sulfamethoxazole or ampicillin would not be appropriate for the empirical treatment of Shigella - associated diarrhea.
Key words: Bloody diarrhea; Shigella; Antibiotics
RESUMEN
Patógenos bacterianos asociados a diarrea con sangre en niños uruguayos. La enfermedad diarreica es un problema de salud importante, especialmente en los países en desarrollo. La diarrea sanguinolenta representa el 20-30% del total de casos y determina una mayor morbimortalidad. La antibioticoterapia es beneficiosa en casos de infección por Shigella, Campylobacter, Yersinia y Salmonella, principalmente en niños con riesgo elevado de enfermedad invasora. Los objetivos del presente trabajo fueron conocer las bacterias asociadas a diarrea sanguinolenta y determinar su patrón de sensibilidad a los antimicrobianos. Entre junio de 2001 y enero de 2008 se estudiaron 249 niños con diarrea sanguinolenta. Shigella y Escherichia coli productor de toxina Shiga (STEC) se recuperaron de 48 (19,3%) y 3 (1,2%) casos, respectivamente. En la subpoblación de 49 niños cuyo estudio coprobacteriológico fue más completo se aisló, además de Shigella (14,3%), Campylobacter jejuni (14,3%), Salmonella spp. (4,1%) y Aeromonas spp. (2%). Treinta y cuatro (71%) aislamientos de Shigella mostraron resistencia a ampicilina y 13 (27%) a trimetoprima-sulfametoxazol. Todos los aislamientos de Shigella fueron sensibles a ácido nalidíxico, ciprofloxacina y ceftriaxona. Los aislamientos de Salmonella y STEC fueron sensibles a todos los antibióticos ensayados. En función de estos resultados, se concluye que el uso de trimetoprima-sulfametoxazol y de ampicilina no sería apropiado para el tratamiento empírico de la diarrea asociada a Shigella.
Palabras clave: Diarrea con sangre; Shigella; Antibióticos
Diarrheal disease continues to be a health problem worldwide, especially in developing countries. In these regions, it accounts for approximately 2.5 million deaths per year in children under 5 years of age. Furthermore, acute diarrhea considerably contributes to morbidity and increases health care costs in children from industrialized countries (6). Bloody diarrhea (BD) represents approximately 20-30% of all cases, causing important inflammatory intestinal illness and, under some circumstances, producing severe complications, like sepsis, hemorrhagic colitis and hemolytic uremic syndrome (HUS) (1, 6).
The bacterial pathogens associated with BD include species of Shigella, Campylobacter, Salmonella, Escherichia coli pathotypes, especially Shiga toxin-producing Escherichia coli (STEC) and enteroinvasive E. coli (EIEC), as well as Yersinia enterocolitica. The prevalence of these agents and their antimicrobial susceptibility patterns vary among different regions (4, 12, 13).
Treatment with antibiotics is indicated in cases of Shigella infections and in special situations of Campylobacter, Yersinia and Salmonella infections, principally in those children at higher risk of invasive disease (5, 15).
However, the lack of rapid diagnostic methods for the detection of all these agents supports the implementation of empirical treatment regimens based on the epidemiological knowledge of the prevalent agents and the corresponding antimicrobial susceptibility.
The aims of this study were to detect the bacterial pathogens associated with bloody diarrhea in children and to determine their antimicrobial susceptibility patterns.
Within the framework of an institutional program aimed at the regional surveillance of STEC infections, a prospective, multicenter study was done involving health care centers in Montevideo and other cities of Uruguay. The analysis period extended from June 2001 to January 2008, 249 children with bloody diarrhea who required medical attention in these centers were studied. Stool samples were consecutively obtained and only one sample per child was processed. Each stool specimen was sent to the Department of Bacteriology and Virology in a Cary- Blair transport medium, whereas another part of the sample without transport medium was sent in a sterile plastic vial. Both were submitted in insulated, refrigerated boxes and were examined less than 12 hours after extraction. All samples were examined for the presence of fecal leukocytes, STEC and Shigella spp. Fecal leukocytes were semiquantified by microscopic examination of smears stained with methylene blue as follows: < 1 cell (+), 1-10 cells (++) and > 10 cells (+++) by high-power field. STEC detection was performed by culture and PCR using specific primers to stx1 and stx2 genes as previously described (14). Shigella species were investigated by classic procedures (3). Furthermore, between January 2005 and June 2006, at the request of the pediatrician in charge, we also studied forty-nine (20%) of these 249 children for the presence of Salmonella, Yersinia, EIEC and Campylobacter species, as described by Torres et al. (12). STEC, Salmonella and Shigella isolates were serotyped by standard procedures using commercial antisera (Difco®) and others available in our institute's collection (3). Antimicrobial disk susceptibility tests for Shigella, Salmonella and STEC isolates were done according to the Clinical and Laboratory Standards Institute guidelines (2). The following agents were tested: ampicillin (AMP), trimethoprim-sulfametho-xazole (TMP-SMX), chloramphenicol (CMP), tetracycline (TET), ciprofloxacin (CIP), nalidixic acid (NAL), gentamicin (GEM), ceftriaxone (CRO), ceftazidime (CAZ), and cefoxitin (FOX). The Fisher´s test was used to asses the association between qualitative variables.
The median age was 12 months (range, 5 days to 14 years of age), and 90% of the children were under five years old. More than 90% of the children were assisted at public health centers. No epidemiological link could be established among the 249 children studied. In the whole period of study, Shigella spp. was recovered from 48 (19.3%) children and STEC from 3 (1.2%) of the 249 cases. One child showed co-infection with two different STEC serogroups (O26 and O145). In those children (49) whose stool culture also included the search for Salmonella, Yersinia, EIEC and Campylobacter species, Shigella spp. was recovered from 7 (14.3%) children, Campylobacter jejuni from 7 (14.3%), Salmonella from 2 (4.1%), and Aeromonas spp. from one (2%) child. Fecal leukocytes were present in all Shigella-positive samples but only in 2 of the 10 children from whom another enteropathogen was isolated (p = 0.0018). Fecal leukocytes were present in 43 (89.6%) of the 48 fecal samples from which Shigella spp. was recovered; 32 of them showed ≥ 10 cells per field (+++). Ninety-four per cent of the cases of bloody diarrhea associated with Shigella were observed in the warmest months (between November and April). Thirty-seven isolates corresponded to Shigella flexneri and 11 to Shigella sonnei. Neither Salmonella dysenteriae nor Shigella boydii were recovered during this study. Twenty-one of the 37 S. flexneri strains corresponded to serotype 2a, 13 to serotype 3c and 3 to serotype 1.
Thirty out of 37 (81%) S. flexneri isolates showed resistance to AMP (17 isolates belonged to serotype 2a; 11 to serotype 3c and 2 to serotype 1) and 13 (35%) to TMP-SMX (7 isolates belonged to serotype 2a; five corresponded to serotype 3c and one to serotype 1). However, when we analyzed the periods 2001-2003 and 2004-2008, the results were 95% and 62% resistance to AMP; 48% and 18% resistance to TMP-SMX, respectively (p > 0.05). S. flexneri isolates showed eight antimicrobial resistance phenotypes and the TET, AMP, CMP resistance pattern was the most frequent (13 strains), followed by the TET, AMP, CMP, TMP-SMX resistance pattern (Figure 1). Two S. flexneri strains were susceptible to all antimicrobials tested. On the other hand, S. sonnei showed a unique AMP-resistance profile AMPR. Four out of 11 S. sonnei isolates had this profile and the other 7 isolates were susceptible to all antimicrobial agents tested. All Shigella isolates were susceptible to NAL, CIP, GEM, CRO, CAZ, FOX. STEC isolates belonged to serogroups O26 (2 strains), O111 (1 strain) and O145 (1 strain). All STEC strains carrying eae and ehxA genes. Isolates O26 carried stx genes, while strain O145 was positive for stx2 and strain O111 carried both stx1 and stx2 genes. The Salmonella strains corresponded to Salmonella enterica subsp. enterica serovar Enteritidis. The Salmonella and STEC cultures were susceptible to all antimicrobials tested. Neither EIEC nor Y. enterocolitica were recovered during this study.
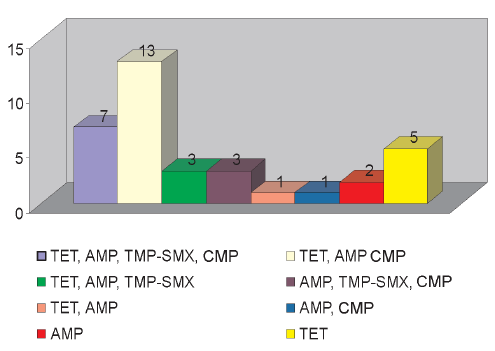
Figure 1. Antimicrobial resistance patterns in Shigella flexneri isolates.
As reported in other regions, Shigella spp. was the most frequently recovered pathogen from BD cases (11, 13). Nevertheless, when more complete stool cultures were done, C. jejuni was an important agent of BD. Therefore, in the future it would be important to perform antimicrobial susceptibility tests to know their resistance pattern. The absence of S. dysenteriae was not surprising. In Uruguay, S. dysenteriae strains are rarely found and they belong to serotype 2 (12). According to our results, the BD cases that occurred during the warmest months and showed fecal polimorphonuclear leukocytes were likely caused by Shigella spp. (p = 0.017). Serotype 2a was the most frequent among S. flexneri strains. These findings are comparable with results from Chile (9). It would be important to define antigenic determinants that should be included in a future vaccine.
As it occurs in other regions (13), we have not recovered Y. enterocolitica or EIEC in a subgroup of these 249 children. However, these results are limited due to the small sample size and probably on account of the methodology used for the detection of these agents which included only phenotypic tests. Although STEC infection represented a small proportion of all acute bloody diarrhea cases studied (1.2%), the serious associated complications, such as HUS, seem to justify the importance of searching for this agent. In this sense, 2 out of 3 children infected with STEC developed HUS during the follow-up. Salmonella isolates were susceptible to all antibiotics tested. Results obtained with Salmonella typhimurium by Macedo et al. (7) showed a marked decrease in resistance in the last years. In our country, since December 2003, the recommended drugs for empirical treatment of BD cases in children have been: oral azithromycin (5 mg/kg/day, for 5 days) for outpatients (over 6 months of age and without toxicity signs) and parenteral ceftriaxone (100 mg/kg, i/m, one dose) for inpatients (under 6 months of age or with toxicity signs). When we analized Shigella spp. strains isolated between 2001 and 2003 versus those isolated in 2004 and 2008, we observed that AMP resistance decreased from 95% to 62%, and TMP-SMX resistance from 48% to 18%. This trend may be explained by appropriate application of this therapeutic guideline; however, studies with larger number of samples are required to determine whether this difference is significant and sustained. In our country, like in other regions, the use of TMS or AMP continues to be inappropriate for the empirical treatment of BD cases. All Shigella strains recovered in this study were susceptible to NAL. This antibiotic has been the drug of choice for the last two decades in certain regions, such as Southern Africa and South Asia (15); nevertheless, physicians should anticipate increasing resistance to this drug as its use increases. In addition, Shigella strains resistant to NAL showed some degree of cross-resistance to CIP. Thus, the widespread use of NAL for treatment of shigellosis may reduce the efficacy of CIP (10). Fluoroquinolones are also recommended drugs for treating Campylobacter, Salmonella and Shigella-associated gastroenteritis (5, 15). According to the results of this study, ciprofloxacin could be used for the treatment of children with BD. However, we believe that this drug should be reserved for the treatment of severe cases for which there is no other treatment option available, taking into account the potential risk of joint damage.
Acknowledgements: This work was partially supported by grants from the Comisión Sectorial de Investigación Científica (CSIC), Universidad de la República, Montevideo, Uruguay and "Manuel Pérez" Foundation, Facultad de Medicina and we thank Lucia Isturiz for the critical reading of the manuscript.
1. Brooks JT, Shapiro RL, Kumar L, Wells JG, Phillips- Howard PA, Shi YP, et al. Epidemiology of sporadic bloody diarrhea in rural Western Kenya. Am J Trop Med Hyg 2003; 68: 671-7. [ Links ]
2. Clinical and Laboratory Standards Institute. Disk diffusion. Performance standards for antimicrobial susceptibility testing; 15th informational supplement, 2005; M100-S15. Wayne, Pa, USA. [ Links ]
3. Ewing WH. Edwards and Ewing´s, editors. Identification of Enterobacteriaceae. 4th ed. USA, Elsevier; 1980. p. 137-72. [ Links ]
4. Guerrant RL, Hughes JM, Lima NL, Crane J. Diarrhea in developed and developing countries: magnitude, special settings, and etiologies. Rev Infect Dis 1990; 12: S41-S50. [ Links ]
5. Guerrant RL, van Gilder T, Steiner TS, Thielman NM, Slutsker L, Tauxe RV, et al. Practice guidelines for the management of infectious diarrhea. Clin Infect Dis 2001; 32: 331-51. [ Links ]
6. Kosek M, Bern C, Guerrant RL. The global burden of diarrhoeal disease, as estimated from studies published between 1992 and 2000. Bull WHO 2003; 81: 197-204. [ Links ]
7. Macedo-Viñas M, Cordeiro NF, Bado I, Herrera-Leon S, Vola M, Robino L, et al. Surveillance of antibiotic resistance evolution and detection of class 1 and 2 integrons in human isolates of multi-resistant Salmonella typhimurium obtained in Uruguay between 1976 and 2000. Int J Infect Dis 2009; 13: 342-8. [ Links ]
8. Mota MI, Varela G, Gadea P, Caffer MI, Sirok A, Schelotto F. Serotipos, perfil plasmídico y antibiotipos en cepas de S. flexneri aisladas de niños menores de 5 años con diarrea sanguinolenta usuarios de los servicios de Salud Pública. Rev Med Uruguay 2005; 21: 30-6. [ Links ]
9. Prado V, Lagos R, Nataro JP, San Martin O, Arellano C, Wang, JY, et al. Population-based study of the incidence of Shigella diarrhea and causative serotypes in Santiago, Chile. Pediatr Infec Dis J 1999; 18: 500-5. [ Links ]
10. Sivapalasingam S, Nelson JM, Joyce K, Hoekstra M, Angulo FJ, Mintz ED. High prevalence of antimicrobial resistance among Shigella isolates in the United States tested by the national antimicrobial resistance monitoring system from 1999 to 2002. Antimicrob Agents Chemother 2006; 50: 49-54. [ Links ]
11. Talan D, Moran GJ, Newdow M, Ong S, Mower WR, Nakase JY, et al. Etiology of bloody diarrhea among patients presenting to United States emergency departments: prevalence of Escherichia coli O157:H7 and other enteropathogens. Clin Infect Dis 2001; 32: 573-80. [ Links ]
12. Torres ME, Pírez MC, Schelotto F, Varela G, Parodi V, Allende F, et al. Etiology of children's diarrhea in Montevideo, Uruguay: associated pathogens and unusual isolates. J Clin Microbiol 2001; 39: 2134-9. [ Links ]
13. Townes JM, Quick R, Gonzáles OY, Linares M, Damiani E, Bopp CA, et al. Etiology of bloody diarrhea in Bolivian children: implications for empiric therapy. J Infect Dis 1997; 175: 1527-30. [ Links ]
14. Varela G, Chinen I, Gadea P, Miliwebsky E, Mota MI, González S, et al. Detection and characterization of Shiga toxin-producing Escherichia coli from clinical cases and food in Uruguay. Rev Argent Microbiol 2008; 40: 93-100. [ Links ]
15. WHO. Guidelines for the control of shigellosis, including epidemics due to Shigella dysenteriae 1. - Geneva, Switzerland: World Health Organization. 2005. [ Links ]
Recibido: 20/10/09
Aceptado: 09/03/10














