Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista argentina de microbiología
versión On-line ISSN 1851-7617
Rev. argent. microbiol. vol.42 no.3 Ciudad Autónoma de Buenos Aires jul./set. 2010
MICROBIOLOGÍA CLÍNICA Y ENFERMEDADES INFECCIOSAS
HPV Detection and genotyping in males from the city of Córdoba, Argentina
F. Venezuela1 *, L .E. Kremer2, X. Kiguen1, C. Cuffini1
1Instituto de Virología "Dr.J. M. Vanella" U.N.C. Calle Enfermera Gordillo Gómez s/n (entre avenidas Enrique Barros y Valparaiso) Ciudad Universitaria, Córdoba (5016);
2Hospital Nacional de Clínicas-Córdoba. Calle Santa Rosa 1500, B° Alberdi, Córdoba (5000)
*Correspondence. Email: venezuelafernando@hotmail.com
ABSTRACT
A wide range of human papillomavirus (HPV) types can infect the anogenital region of males. Although there is a vast knowledge on HPV infections in women as well as on their association with cervical cancer, the study of HPV infections in males is scarce and controversial. The aim of the present work was to detect and typify HPV infections of the anogenital region in males and analyze the associated risk factors in the population studied. Anogenital samples from 37 patients (30 of whom were HIV carriers) attending the Infectology Service at the Hospital Nacional de Clínicas in Córdoba, Argentina, were studied. Nine of these patients tested HPV-positive and five out of these nine were found to have mixed infections, being 18 and 61 the most frequent genotypes. There was a significant correlation between the HPV-positive patients and those having an HPV-compatible lesion or AIDS. The present work is the first study in the city of Cordoba which contributes relevant results to the knowledge of HPV infection and to the possible implementation of measures for its prevention.
Key words: Male; Genotyping; HPV; AIDS; Anogenital; Córdoba
RESUMEN
Detección y genotipificación de VPH en varones de la ciudad de Córdoba, Argentina. Un amplio espectro de tipos de virus papiloma humano (VPH) puede infectar la zona anogenital de los varones. Si bien existe un vasto conocimiento de la infección por VPH en las mujeres y su asociación con el cáncer de cérvix, el estudio de la infección por VPH en los varones ha sido escaso y sus resultados controvertidos. El presente trabajo tuvo como objetivo detectar y tipificar infecciones por VPH en la región anogenital de varones y analizar los factores de riesgo asociados en la población estudiada. Se estudiaron muestras anogenitales de 37 pacientes (30 portadores del VIH) que asistieron al Servicio de Infectología del Hospital Nacional de Clínicas de la ciudad de Córdoba. Nueve resultaron positivas para VPH, de las que 5 correspondían a infecciones mixtas. Los genotipos de mayor frecuencia fueron el 18 y el 61. Hubo una correlación significativa entre los resultados VPH positivos en aquellos pacientes con lesión compatible o con sida. Nuestro trabajo es el primero de su tipo realizado en la ciudad de Córdoba y aporta resultados relevantes para el conocimiento de la infección por VPH y la implementación de medidas de prevención.
Palabras clave: Varón; Genotipificación; HPV; Sida; Anogenital
Human papillomavirus (HPV) infection has become one of the most important sexually transmitted diseases (13, 14). HPV causes not only benign lesions (like condylomata acuminata), but also a wide range of preneoplastic lesions and carcinoma. Although the relationship between HPV and the etiopathogeny of cervical cancer is well known, there are few studies on HPV infection in male anogenital lesions. In addition, it has been proposed that males play an important role as reservoirs and transmitting agents of HPV infection (4, 11).
During March-June 2009, in order to detect and typify HPV infections in the anogenital region of males, we studied 40 samples from 37 patients attending the Infectology Service at the Hospital Nacional de Clínicas in Córdoba, Argentina, who presented at least one of the following conditions: a) a lesion compatible with HPV in the anogenital region, b) their last sexual partner presented a lesion compatible with HPV in the anogenital region, and c) more than three sexual partners in the previous year. The average age of patients was 36.9 years (range 21-57 years).
The Infectology Service provides care to patients with different types of lesions. However, it is interesting to point out that over 80% of the patients included in this study were carriers of the human immunodeficiency virus (twenty-nine of HIV and one of HIV-HCV). In accordance with the Center for Disease Control and Prevention (CDC), 16 of the 29 HIV carriers were classified as HIV-AIDS (A3: 3; C1: 2; C2: 6 and C3: 5).
An epidemiological chart was performed for each patient and, according to the data collected, either a urethral swab/penile brushing or a brushing of the anal/perianal region was obtained, since these samples are considered optimal to evaluate the infection in the anogenital region (5). A total of 28 urethral swabs/penile brushing and 12 anal/perianal were obtained. In three of the patients reporting both passive and active sexual intercourse, both types of samples were collected. The reasons why each patient attended the Infectology Service are detailed in Table 1.
Table 1. Distribution according to the inclusion conditions (n = 37)

Of the 29 patients that attended the Infectology Service for reasons other than an HPV-compatible lesion, six presented this kind of lesion in the anogenital region and 12 stated that they had had previous HPV lesions (Table1).
Of the 14 patients who presented an HPV-compatible lesion, two presented perianal condyloma acuminata and 12 presented flat warts (four in the perianal region, three in the glans penis and five on the surface of the penis).
The samples were collected in 500 μl phosphate-buffered saline (PBS). DNA was then extracted using the commercial AccuPrep Genomic DNA Extraction Kit (Bioneer Inc., CA, USA), according to the manufacturer's instructions. A 450-bp segment, corresponding to the L1 region of the viral genome, was amplified by PCR, using the degenerate primers MY09 and MY11 (1). The product was detected by electrophoresis in agarose gel at 1.5% using an U.V. transilluminator.
The Beta globin gene was used as a DNA preservation marker. Viral genotypification was carried out by analysis of restriction fragment length polymorphism (RFLP), following the technique described by Bernard et al. (1). A statistical analysis was carried out with Epi Info™, Version 3.5.1, 2008 (http://www.cdc.gov/epiinfo/).
Of all the samples analyzed, nine were positive for HPV and belonged to patients that presented an HPV-compatible lesion (Table 3). In addition, five patients that presented an HPV-compatible lesion were HPV-negative. This could be due to the fact that the primers used in this study detected the mucosal HPV types (1). This may indicate that the lesions were caused by non-mucosal HPV types, since the β-globin gene was the one amplified in all the samples. Another hypothesis is that the lesions could have been caused by another etiologic agent, since they were classified as HPV-compatible, always under a clinical criterion.
The largest number of patients presenting lesions was observed in the compatible range of 21 to 30 years of age (57%), which correlates with the positive results for HPV (28.6%).
Several articles have reported the detection of HPV in men, whose infection rates range between 1.0% and 82.9% (3) depending on the population studied, the anatomic site of the sample, the collection method and the sensitivity of the technique for the detection of HPV (6, 7, 9).
In the present work, the percentage of patients who attended the Infectology Service presenting an HPV infection was 24.4%, a value comparable to the 33% found in the males attending a private hospital specialized in sexually transmitted diseases in the USA (15). It is worth pointing out that the methodology used, the population included and the type of sample obtained were similar in both studies.
Table 2 shows that HPV-positive patients, with more than three sexual partners in the year previous to our study, reported not having used a condom (never / sometimes) in any of their sexual encounters. If we point out that all of them had at least a high school degree, we could conclude that the educational background does not guarantee the knowledge and application of the measures to prevent a sexually-transmitted infection.
Table 2. Characteristics of HPV-positive patients
Table 3. Frequency of detection of HPV vs risk factors
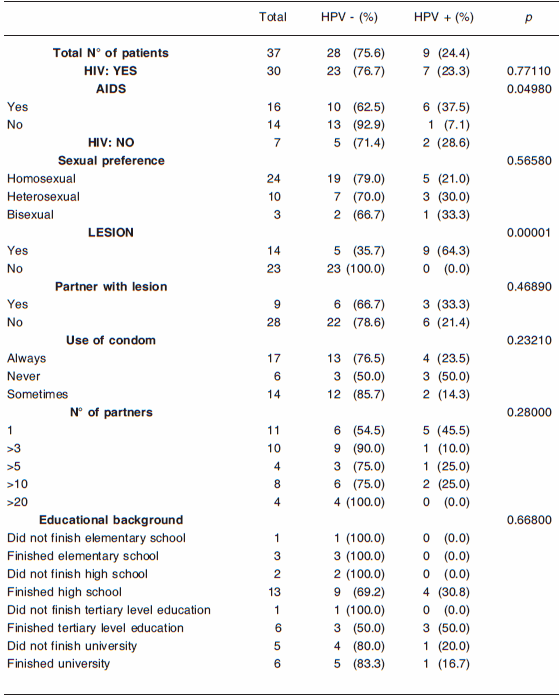
This table also shows that three out of the six patients presenting an HPV-compatible lesion who had attended the Infectology Service for other reasons tested positive. It is interesting to point out that one of them (patient No. 4) presented a mixed infection, whose genotypes belonged to the high risk group. This patient had had sexual intercourse with the opposite sex, which indicates the lack of knowledge of the population of the risks of this infection, not only for themselves, but also for their sexual partner(s). This result is very important for the purposes of determining the guidelines for the prevention of HPV infection.
Table 2 also indicates that of all the genotypes identified (6, 11, 16, 18, 26, 44, 45, 59, 61 and 68), the most frequent ones were 18 and 61, and shows that in patients with condylomata acuminata, only the low-risk HPV genotypes 61 and 11 were detected.
Of the three patients from whom both kinds of samples were taken, only one (patient n° 6) was positive (genotype 18-68) and corresponded to the sample taken from the perianal region.
Regarding the genotype frequency, no significant differences between the low-risk and the high-risk HPV types were observed. It can be noticed that every two HPV-positive patients with compatible lesions, one could have at least one infection caused by a high-risk genotype. Likewise, 80% of the patients with an AIDS diagnosis by the presence of the disease marker (C2 and C3) were positive for at least one high-risk genotype, although for HIV-carriers any genotype should be considered high risk. It should also be noted that, since the implementation of antiretroviral therapy has failed to reduce the incidence of some cancers caused by HPV (10), all the necessary precautions had been taken in the Infectology Service where this study was carried out.
The RFLP analysis of three (HIV positive) out of the seven HPV positive-patients, showed DNA bands that were not attributed to other genotypes present. Since a significant association with the presence of multiple HPV infections in HIV patients has been reported (8), this result suggests that some of them may have had co-infections with other HPV genotypes. Importantly, the two HCV-positive patients were negative for HPV detection.
HPV detection was significantly associated with two factors: AIDS and the HPV-compatible lesion, which is in agreement with the results reported by other authors (2, 12). The presence of HPV in patients with an AIDS diagnosis could be due to the immunosupression caused by HIV, which may lead to a higher degree of HPV replication, a higher viral load and, consequently, an increase in epithelial mucosal lesions (12).
In the present work, we found no significant association between HIV and HPV infections, which may be mainly due to a difference in the number of HIV carriers (n = 30) and non-carriers (n = 7) included in this study (Table 3).
The understanding of HPV infection in males is important in order to reduce transmission to women as well as for its possible association with penis and anus cancer. According to the results obtained in this study, the preventive measures in this age group (21-30 years old) should be emphasized.
Our work is the first of its kind in the city of Córdoba and contributes relevant data to the knowledge of HPV infection and to the possible implementation of measures to prevent HPV in the male population.
Acknowledgements: We thank all the patients enrolled in this study and Drs Fanny N. Cohen, Carlos M. Quinteros and Luis Allende, from the Infectology Service at the Hospital Nacional de Clínicas in Córdoba, Argentina, for their invaluable contribution to this study.
1. Bernard H, Chan SY, Manos MM, Ong CK, Villa LL, Delius H, et al. Identification and assessment of known and novel human papillomaviruses by polymerase chain reaction amplification, restriction fragment length polymorphisms, nucleotide sequence, and phylogenetic algorithms. J Inf Dis 1994; 170: 1077-85. [ Links ]
2. Chan PK, Luk AC, Luk TN, Lee KF, Cheung JL, Ho KM, et al. Distribution of human papillomavirus types in anogenital warts of men. J Clin Virol 2009; 44: 111-4. [ Links ]
3. Dunne EF, Nielson CM, Stone KM, Markowitz LE, and Giuliano A. Prevalence of HPV infection among men: a systematic review of the literature. J Infect Dis 2006; 194: 1044-57. [ Links ]
4. Franceschi S, Castellsagué X, Dal Maso L, Smith JS, Plummer M, Ngelangel C, et al. Prevalence and determinants of human papillomavirus genital infection in men. Br J Cancer 2002; 86: 705-11. [ Links ]
5. Giovannelli L, Migliore MC, Capra G, Caleca MP, Bellavia C, Perino A, et al. Penile, urethral, and seminal sampling for diagnosis of human papillomavirus infection in men. J Clin Microbiol 2007; 45: 248-51. [ Links ]
6. Grupo de estudio de VPH en hombres de Brasil-Estados Unidos-México. Human Papillomavirus infection in men residing in Brazil, Mexico, and the USA. Salud Pública Mex 2008; 50: 408-18. [ Links ]
7. Lajous M, Mueller N, Cruz-Valdéz A, Aguilar LV, Franceschi S, Lazcano-Ponce, et al. Determinants of prevalence, acquisition, and persistence of human papillomavirus in healthy mexican military men. Cancer Epidemiol Biomarkers Prev 2005; 14: 1710-6. [ Links ]
8. Müller EE, Chirwa TF, Lewis DA. Human papillomavirus (HPV) infection in heterosexual South African men attending sexual health services: associations between HPV and HIV serostatus. Sex Transm Infect. 2010; 86: 175-80. [ Links ]
9. Nielson CM, Flores R, Harris RB, Abrahamsen M, Papenfuss MR, Dunne EF, et al. Human papillomavirus prevalence and type distribution in male anogenital sites and semen. Cancer Epidemiol Biomarkers Prev 2007; 16: 1107-14. [ Links ]
10. Palefsky J. Human papillomavirus-related disease in people with HIV. Curr Opin HIV AIDS 2009; 4: 52-6. [ Links ]
11. Partridge JM, and Koutsky LA. Genital human papillomavirus in men. Lancet Infect Dis 2006; 6: 21-31. [ Links ]
12. Sirera G, Videla S, Herranz P, Corzo-Delgado J. Human papilloma virus and HIV/AIDS. Enferm Infecc Microbiol Clin 2006; 2: 40-6. [ Links ]
13. Watson R. Human Papillomavirus: Confronting the epidemic. A urologist's perspective. NJ Rev Urol 2005; 7: 135-44. [ Links ]
14. Weaver BA. Epidemiology and natural history of genital human papillomavirus infection. J Am Osteopath Assoc 2006; 106: 2-8. [ Links ]
15. Weaver BA, Feng Q, Holmes KK, Kiviat N, Lee SK, Meyer C, et al. Evaluation of genital sites and sampling techniques for detection of human papillomavirus DNA in men. J Infect Dis 2004; 15: 677-85. [ Links ]
Recibido: 06/10/09
Aceptado: 05/05/10














