Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista argentina de microbiología
versión On-line ISSN 1851-7617
Rev. argent. microbiol. vol.42 no.3 Ciudad Autónoma de Buenos Aires jul./set. 2010
MICROBIOLOGÍA INDUSTRIAL Y AMBIENTAL
Enterotoxigenic gene profiles of Bacillus cereus and Bacillus megaterium isolates recovered from honey
A. C. López, A. M. Alippi*
Unidad de Bacteriología, Centro de Investigaciones de Fitopatología, Facultad de Ciencias Agrarias y Forestales, Universidad Nacional de La Plata, cc 31, calle 60 y 119, (1900) La Plata, Argentina.
*Correspondence. E-mail: alippi@biol.unlp.edu.ar
ABSTRACT
One hundred and thirty two Bacillus cereus and 52 Bacillus megaterium isolates from honeys were evaluated for the presence of genes encoding enterotoxin HBL, enterotoxin-T, cytotoxin K and the NHE complex, respectively. The relationship between hemolytic and coagulase activity and its correlation with the presence of the four mentioned enterotoxins was determined by principal component analysis (PCA). PCA in B. cereus revealed a positive correlation among free coagulase, hemolysis and the presence of genes hblA, hblB, hblC, hblD (HBL complex) and bceT (enterotoxin-T), but no correlation with the clumping factor (bound coagulase) and the presence of sequences of the NHE complex. On the other hand, PCA in B. megaterium showed a high positive correlation between coagulase (bound and free) and the haemolytic activity but no correlation in relation to the presence of genes of the HBL complex, cytotoxin K, enterotoxin T and the NHE complex. To our knowledge, this is the first report of the detection of cytotoxin K and of the NHE complex genes in B. megaterium. The relationship between the coagulase activity and the presence of virulence factors has not been described before in the genus Bacillus, being this work the first report of this correlation. Interestingly, the presence of the cytK gene was almost independent of the presence of the rest of virulence factors herein analyzed both in B. cereus and B. megaterium populations. Our results suggest that honey could be a possible vehicle for foodborne illness due to the presence of toxigenic B. cereus and B. megaterium strains containing different virulence factors.
Key words: Bacillus cereus; Honey; Hemolysis; Coagulase activity; Enterotoxins; Bacillus megaterium; Virulence factors
RESUMEN
Búsqueda de factores de virulencia en cepas de Bacillus cereus y de Bacillus megaterium aisladas de miel. Se evaluaron 132 aislamientos de Bacillus cereus y 52 de Bacillus megaterium provenientes de mieles de distintos orígenes geográficos para investigar la presencia de secuencias de ADN relacionadas con genes de virulencia y su posible correlación con la actividad hemolítica y coagulasa. Con respecto a los genes de virulencia, se analizaron por PCR secuencias de ADN de los genes nhe (A, B y C), HBL (A, B, C, D), cytK y bceT. La relación entre las variables fue evaluada mediante un análisis de componentes principales, donde se encontró que los aislamientos de B. cereus mostraron una correlación positiva entre actividad de coagulasa (coagulasa libre) y presencia de los genes del complejo HBL y bceT, mientras que en B. megaterium se halló una alta correlación positiva entre actividad de coagulasa (libre y fija) y actividad hemolítica, pero no se observó correlación significativa entre la presencia de genes de virulencia y dichas actividades. Este estudio constituye el primer registro de la presencia de los genes cyt K y NHE en cepas de B. megaterium y el primer trabajo que analiza la relación entre la actividad de coagulasa y la presencia de genes de virulencia en B. cereus y B. megaterium. La presencia del gen cytK en ambas especies resultó totalmente independiente del resto de los factores de virulencia analizados. Nuestros hallazgos sugieren que la miel podría vehiculizar enfermedades transmisibles por alimentos debido a la presencia de cepas de B. cereus y B. megaterium potencialmente tóxicas.
Palabras clave: Bacillus cereus; Miel; Hemólisis; Actividad de coagulasa; Enterotoxinas; Bacillus megaterium; Factores de virulencia
INTRODUCTION
Honey quality is influenced by microorganisms, particularly spore-forming bacteria and yeast (35). Aerobic mesophylic spore-forming bacteria of the genera Paenibacillus, Bacillus and Brevibacillus are commonly found in honey (5, 35). Several Bacillus species, including Bacillus cereus (sensu lato group), Bacillus megaterium, Bacillus coagulans, Bacillus subtilis, and Bacillus licheniformis, have been frequently isolated from honeys (4, 5, 14, 15). The presence of Bacillus species was reported, in Argentinean honeys, being B. cereus (28%) and B. megaterium (14%) the most prevalent ones (4, 5, 24, 25).
The Bacillus cereus group (also called B. cereus sensu lato) consists of six closely related species: B. cereus, Bacillus anthracis, Bacillus thuringiensis, Bacillus mycoides, Bacillus pseudomycoides, and Bacillus weihenstephanensis (22, 39), being B. cereus (sensu stricto) a common food-poisoning organism and the most prevalent one in industrial processes. It is classified into hazard category 2 and has been associated with two forms of food poisoning, named emetic and diarrheal diseases, as well as with non-intestinal pathologies (12, 19). B. cereus (sensu stricto) produces four main enterotoxins, named hemolysin BL (HBL), enterotoxin-T, non-hemolytic enterotoxin complex (NHE) and cytotoxin K, and has been involved in some human infectious processes (16, 21, 31). Hemolysin BL (HBL) is a protein complex of three distinct protein subunits: B, L1 and L2, which is considered one of the most important virulence factor of B. cereus (6). All three parts are required for biological effects and together possess hemolytic, cytotoxic, dermonecrotic, and vascular permeability activities, including fluid accumulation in ligated rabbit ileal loops (7). None of the individual subunit components have toxic activity. Enterotoxin-T is a simple component toxin with biological activities related to diarrheal enterotoxins, such as fluid accumulation and permeability in rabbit ileal loops and cytotoxic activities (3). NHE is a three-component enterotoxin responsible for the diarrheal food-poisoning syndrome. NHE is composed of three genes (nheA, nheB and nheC), that constitute one operon, being all the three components necessary for cytotoxic activity (23). Cytotoxin K (cytK) causes a diarrheal syndrome with necrotic, hemolytic and cytotoxic effects on the intestinal ephithelium (13) and it was first characterized in B. cereus strains that caused a severe poisoning outbreak (26). In addition, virulence of the socalled emetic strains is related to cereulide (2, 11), a thermostable cyclic peptide responsible for fatal cases of foodborne infections (9). B. megaterium is another gram positive spore-forming bacterium that produces several enzymes and antibiotic-like compounds and has been found in diverse habitats including honey (1, 25, 28, 29, 38). Taylor et al. (36) reported the production of a novel heatstable toxin in B. megaterium strain F98/3079, while the presence of diarrheagenic genes bceT (enterotoxin-T), hblC, hblA, and hblD (HBL complex) was detected in one out of two B. megaterium strains tested by Rowan et al. (34). On the other hand, hemolytic and coagulase activities were observed in 77% and 74% of B. megaterium isolates from honeys (25). Coagulase activity is most often associated with pathogenic staphylococci or yersiniae and is not ordinarily reported as a property of the Bacillus species. Because coagulase may be correlated with virulence of certain organisms (10), it seemed appropriate to investigate the matter further. The objectives of the present study are: (i) to document the existence of coagulase activity in B. cereus isolates from honeys and (ii) to assess the distribution of homologous sequences of gene components encoding for the HBL complex, enterotoxin- T, cytotoxin K and the NHE complex in B. cereus and B. megaterium populations and its correlation with coagulase and hemolytic activities.
MATERIALS AND METHODS
Strain collection
A collection of 132 B. cereus and 52 B. megaterium strains isolated from honey samples from different geographical areas in Argentina was used for this study. In addition, isolates of both species from honeys from Italy, USA, Mexico, Brazil and France and reference strains were also studied. Further details of these organisms are available in López and Alippi (24, 25).
All the isolates were grown in tryptic soy agar (TSA) at 30 °C, maintained on TSA at 4 °C and stored in tryptic soy broth (TSB) plus 20% glycerol v/v at -80 °C.
Hemolytic and coagulase activities
Data for the hemolytic activity and production of a discontinuous hemolytic pattern on blood agar plates were detailed in previous studies (24, 25). The 132 B. cereus strains were evaluated for coagulase activity in rabbit plasma as reported by López and Alippi (25). Briefly, B. cereus cultures were grown in brain heart infusion broth (BHI) (Merck) for 24 h at 36 ± 1 °C. The tube procedure for determination for both free and bound coagulase was done by mixing 100 μl of broth culture with 300 μl rabbit plasma (Britania) in a sterile screw-capped tube. Tubes were incubated at 36 ± 1 °C and observed at various time intervals for clot formation. The degree of coagulation was noted as complete lack of clot formation, partial or incomplete clot formation, or complete or firm clot formation. The slide procedure was used to determine bound coagulase enzyme (clumping factor) by mixing with a wooden stick a single bacterial colony from a 24 h culture in BHI agar in saline in a clean microscope slide with one drop of rabbit plasma. In positive cases, a visible clumping of cells appeared within 20 seconds. Results from B. megaterium strains (n = 52) were detailed previously (23, 24).
Detection of enterotoxin genes in B. cereus and B. megaterium by PCR
Total genomic DNA was isolated from 24h-cultures grown on TSA using the rapid procedure described previously by López and Alippi (23). All strains were screened for the presence of genes encoding enterotoxin-T (bceT) (18), cytotoxin K (cytk) (11) and hemolysin BL (hblA, hblB, hblC, hblD) (18, 21, 33) and non hemolytic enterotoxin (nheA, nheB, nheC) (16) complexes by PCR.
PCR conditions for primers B f/r; B' f/r, L-1 f/r; L-2 f/r and ET f/r were carried out in a final volume of 25 μl as previously reported (21), while PCR conditions for CKF2/CKR5 for the detection of cytotoxin K, and NAF/R, NBF/R and NCF/R primers for the detection of the NHE complex were conducted in 50 μl volumes according to Ehling-Schulz and co-workers (11) and Guininebretiere et al. (16), respectively. B. cereus F4430/73 strain with well-characterized toxin profiles (16) was used as positive control in all PCR runs.
DNA amplifications were performed in a thermal cycler (Mastercycler personal; Eppendorf Hamburg, Germany). Amplicons were analyzed by 1.6% (W/V) agarose gel, in TBE buffer, stained with SYBR® Safe (Invitrogen, Argentina) for 2 h at 80 V using 100 bp ladder (Promega) as a molecular weight marker. Gels were photographed on the visi-blue transilluminator (Safe imagerTM) using a digital image capture gel documentation system (Digi Doc-it, UVP, v. 1.1.25).
Principal component analysis (PCA)
The correlation among the presence of DNA sequences corresponding to the genes hblA, hblB, hblC, hblD, nheA, nheB, nheC, cytK, and bceT; and hemolytic and coagulase activity was evaluated by principal component analysis (PCA). Results of hemolysis and production of a discontinuous hemolytic activity pattern were coded as 1 (hemolytic activity) or 0 (lack of hemolytic activity). Results for coagulase activity were coded as 1 (coagulase activity) or 0 (lack of coagulase activity). In addition, results of PCRs for the presence of the four enterotoxins listed in Tables 1 and 2 were ranked as 1 (PCR positive) or 0 (PCR negative). Data were not normalized and software MSVP 3.1 (Kovach
Computing Services) was used to analyze the data set by performing PCA. Plots of the first two principal components scores represent the best 2D representation of natural variance in the data. In biplots vector variables represent the positive direction of the variable axes, therefore variables forming an acute angle are positively correlated, whereas those forming an obtuse angle are negatively correlated and right angles indicate uncorrelated variables. All the variable vectors meet at the centroid, which represents the means for all the variables (27).
Table 1. Screening of virulence genes and coagulase and haemolytic activities of Bacillus cereus isolates from honey.
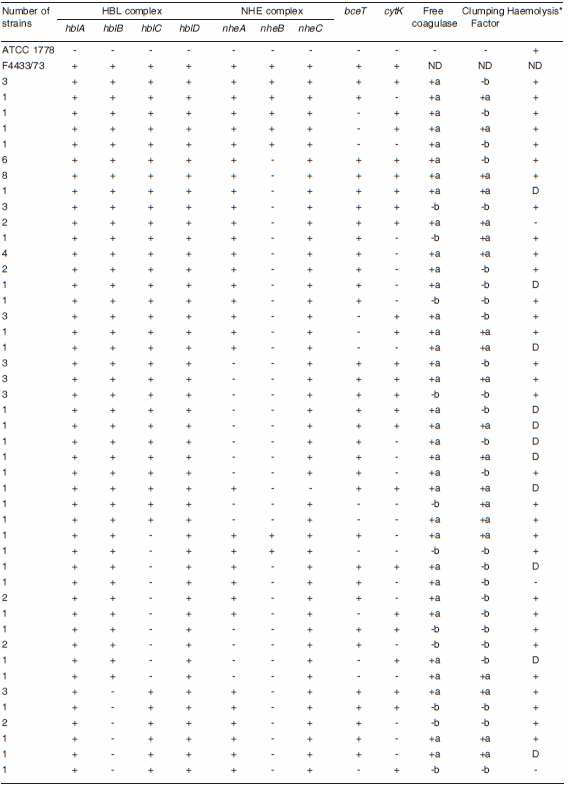

ND: not determined; +a: coagulase activity; -b: lack of coagulase activity; + PCR: product of the expected size was observed; -: no PCR product was observed; D: production of discontinuous haemolytic pattern on blood agar plates. Ø: number of strains that share the same genotypic and phenotypic characteristics listed here; *: data obtained from previous results (23).
Table 2. Screening of virulence genes and coagulase and haemolytic activities of Bacillus megaterium isolates from honey.
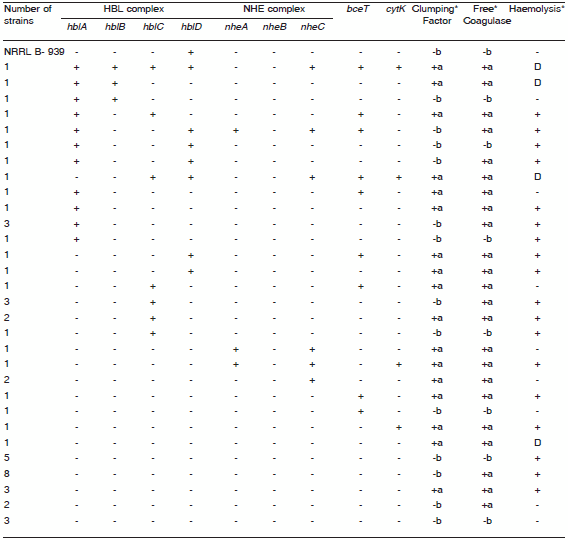
ND: not determined; +a: coagulase activity; -b: Lack of coagulase activity; + PCR: product of the expected size was observed; -: no PCR product was observed; D: production of discontinuous haemolytic pattern on blood agar plates. Ø: number of strains that share the same genotypic and phenotypic characteristics listed here. * data obtained from previous results (24).
RESULTS
Hemolytic and coagulase activities
Data for the hemolytic activity in B. cereus showed that 95% of isolates from honey presented hemolytic activity, and within these strains, 11% produced a discontinuous hemolytic pattern (Tables 1 and 3). In the case of B. megaterium, hemolytic activity was shown by 77% of isolates, and within this group, 10% produced a discontinuos hemolytic pattern (Table 2). These results are summarized in Tables 1, 2, and 3 and previous studies (24, 25). All the B. cereus strains that presented a discontinuous hemolytic pattern presented at least 3 target genes for the HBL complex (Table 1), and in the case of B. megaterium, at least 2 target genes (Table 2) with the exception of the Bm2 strain.
Table 3. Comparison of results obtained with our B. cereus collection with those reported by others authors regarding haemolytic activity, production of a discontinuous haemolytic pattern, and detection of enterotoxin genes by PCR.

*Only positive results are listed here. ND: not determined
Coagulase activity was observed as clot formation in 106 strains (80%) of B. cereus and among the positive strains, 57 (54%) presented the clumping factor (Table 1) while in the case of B. megaterium, coagulase activity was observed as clot formation on 74% of strains out of a total of 53 strains and among the positive strains, only 49% presented the clumping factor (Table 2).
Detection of enterotoxin genes in B. cereus and B. megaterium by PCR
In B. cereus isolates (n = 132) the presence of the hblA gene encoding for the B part of the HBL complex was detected in 110 strains (83%), the gen hblB encoding for the B' part in 75 strains (57%), the gen encoding for the L1 part (hblD) in 109 strains (83%) and the gene encoding for the L2 part (hblC) in 84 strains (64%), respectively. Two percent of the isolates presented the L1 and L2 components (Lytic components of the HBL complex), while 4% of the isolates showed B and B' components (binding components of HBL complex). In addition, all four HBL genes (hblA, hblB, hblC and hblD) were detected in 55 strains (42%) (Tables 1 and 3 and Figure1A). The results of the PCR analysis of the NHE complex revealed that 47 strains (56%) yielded the expected nheA-PCR product of 755 bp; 35 (12%) strains yielded the expected nheC-PCR product of 683bp, but only 13 strains (10%) presented the expected nheB-PCR product of 743 bp (Tables 1 and 3 and Figure 1C). The presence of genes encoding for the enterotoxin-T was observed in 96 strains (73%) of our collection and the presence of the CytK-PCR product of 421 bp was detected in 53% of the 133 strains tested (Table 1, Figures 1B and 1 A, respectively).
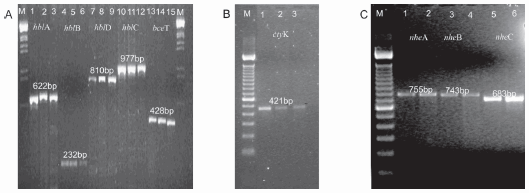
Figure 1. A) PCR patterns obtained with the various PCR primers sets for the detection of the enterotoxin HBL complex and enterotoxin- T and various B. cereus and B. megaterium strains. Lanes (from left to right): M: molecular size marker λ Eco Hind III Promega; 1 to 3: primer pair B, strains Bc20, Bc21and Bc37; 4 to 6: primer pair B', strains Bc 20, Bc 21 and Bc37; 7 to 9: primer pair L-1, strains Bc20, Bc21 and Bc37; lanes 10 to 12: primer pair L-2, strains Bc20, Bc21 and Bc37; 13 to15: primer pair ET, strains Bc20, Bc21 and Bc37; M: molecular size marker l ecoHindIII, Promega. B) PCR products obtained with the primers sets targeting the cytotoxin K gene (CK F2 and CK R5) in B. megaterium strains. Lanes (from left to right): M: molecular size marker 100 bp ladder Promega, Argentina; lanes 1 to 3: Bm1, Bm8 and Bm9. C) PCR patterns obtained with the various PCR primers sets for the detection of the enterotoxin NHE complex in B. cereus strains. Lanes (from left to right): M: molecular size marker λ Eco Hind III Promega; 1 to 2: primer pair nheA, strains Bc30 and Bc80; 3 to 4: primer pair NheB, strains Bc84 and Bc100; 5 to 6: primer pair NheC, strains Bc 101 and Bc32.
In our B. megaterium collection, results for the HBL complex showed that the presence of part B was observed in 13 strains (27%), of part B' in 3 strains (6%), of part L1 in 12 strains (23%) and of part L2 in 3 strains (6%). Only one strain (Bm8) presented all four components of the HBL complex and was correlated with the presence of a discontinuous hemolytic pattern (Table 2 and Figure 1A). The presence of the bceT gene encoding for enterotoxin- T was detected in 9 strains (17%) (Table 2 and figure 1B). Variable results were obtained for B. megaterium in relation to the NHE complex, the presence of the nheA gene was observed in 3 strains (6%), the nheC gene in 7 strains (13%) (Table 2 and Figure 1C), and none of the isolates presented the nheB gene (Table 2).
Principal Component Analysis
The relationship among hemolytic and coagulase activities and the presence of the four enterotoxins studied was determined by PCA. The PCA of 132 B. cereus strains isolated from honeys revealed that the total variance of the system was defined by the first 3 principal components, which accounted for 76%, 5% and 3% of the variance, respectively. Biplot analysis (PC1 vs. PC2) showed a positive correlation (p = 0.0001) among free coagulase, hemolysis and the presence of genes hblA, hblB, hblC, hblD (HBL complex) and bceT (enterotoxin-T) but no correlation with the clumping factor (bound coagulase) (Figure 2A). Seventeen per cent of the strains presented the 4 genes of the HBL complex, and within this group, 71% showed coagulase activity (free coagulase) (Figure 2A) but there was no correlation between genes belonging to the NHE complex and those of the HBL complex. Biplot of PC1 vs. PC3 showed that the presence of the cytK gene was almost independent of the presence of the rest of virulence factors herein analyzed (Figure 2B).

Figure 2. Biplots for the entire data set of 133 B. cereus isolates from honey. (A) PC1 vs. PC2. For visual clarity, strains positive for all enterotoxins and free coagulase activity are included in a circle. (B) PC1 vs. PC3. The percentages of the variation explained by principal components are indicated in parentheses.
The PCA for 53 strains of B megaterium showed that the percentages of the variation were explained by the first 2 principal components that accounted for 62% and 9%, respectively. Biplots showed high positive correlation (p = 0.0001) between coagulase and hemolytic activities (Figure 3). Seventeen strains presented both activities (the strains have been placed inside a ring for visual clarity) (Figure 3). However, no correlation was observed between the presence of enterotoxin genes hbl (A, B, C, D), cytK, bceT and nhe and these activities (Figure 3).

Figure 3. Biplots for the entire data set of 52 B. megaterium isolates from honey. PC1 vs. PC2. A circle has been placed around strains positive for coagulase (bound and free coagulase) and hemolytic activities. The percentages of the variation explained by principal components are indicated in parentheses.
DISCUSSION
According to previous studies on our B. cereus collection isolated from honeys (24), data for the hemolytic activity showed that 95% of isolates presented hemolysis, and within these strains, 11% of them produced a discontinuous hemolytic pattern (Table 3). In the case of B. megaterium, hemolytic activity was shown by 77% of isolates, and within this group, 10% produced a discontinuous hemolytic pattern (25). According to Beecher and Wong (6), the presence of a discontinuous hemolytic pattern is usually correlated with the presence of hemolysin BL in B. cereus. Another study by Pruβ et al. (32), found that all but one of the strains within the B. cereus group that were positive for hblA exhibited hemolytic activity (Table 3). Thaenthanee et al. (37) found that among all tested B. cereus strains carrying the three hbl genes, more than 60% displayed a discontinuous hemolytic pattern (Table 3). These results were correlated with those herein observed where 57% of the strains showing a discontinuous hemolytic pattern presented the 4 components of the HBL complex and the rest (43%), at least 3 components (Tables 1 and 3). In the case of B. megaterium, the strains showing discontinuous hemolytic pattern presented at least 2 target genes (Table 2) with the exception of the Bm2 strain. Coagulase activity was observed as clot formation in 106 B. cereus strains (80%) and among the positive strains, 57 (54%) presented the clumping factor. Our previous studies on the B. megaterium collection from honeys showed that coagulase activity was observed as clot formation in 74% of strains out of a total of 53 strains and, among the positive strains, only 49% presented the clumping factor (25). The coagulase activity has been broadly studied in Staphylococcus aureus and is considered the most important virulence factor in this species (10, 20). In vitro assays for testing the ability to clump plasma via the activity of the clumping factor (bound coagulase) (CF) and coagulase (free coagulase) has not been described in B. cereus.
The presence of the hblA gene encoding for the B part of the HBL complex, the gen hblB encoding for the B' part, the gen encoding for the L1 part (hblD), and the gene encoding for the L2 part (hblC) was detected in 83%, 57%, 83%, and 64% of the isolates tested, respectively (Table 3). Two percent of the isolates presented the L1 and L2 components of the HBL complex, while 4% of the isolates showed B and B' components (Tables 1 and 3). In addition, all three HBL genes were detected in 42 % of strains (Tables 1 and 3). In relation to the NHE complex, 56% of strains yielded the expected nheA-PCR product; 12% the expected nheC-PCR product, but only 10% presented the expected nheB-PCR (Tables 1 and 3). The high distribution of these genes within B. cereus populations isolated from honey samples are in agreement with results obtained with B. cereus strains isolated from different sources (8, 16, 21, 34, 40) and are summarized in Table 3. The presence of genes encoding for the enterotoxin-T was observed in 73% of our strains (Tables 1 and 3) while other workers (16) (21) have reported that most of the B. cereus strains from food-poisoning outbreaks carried the bceT genes in a proportion of 91% and 57% of positive strains, respectively (Table 3). The presence of the cytK-PCR product of 421 bp was detected in 53% of our collection (Tables 1 and 3), while Guinebretiere et al. (16) found that 92% of strains carried the cytK gene (Table 3).
In our B. megaterium collection, the presence of part B, part B', part L1 and part L2 was detected on 27%, 6%, 23% and 6% of the strains and only one strain (Bm8) presented all four components of the HBL complex that was correlated with the presence of a discontinuous hemolytic pattern. The presence of the bceT gene encoding for the enterotoxin-T was detected in 9 strains (17%). Rowan et al. (34) found that one B. megaterium strain out of a total of 2 analyzed presented genes associated with the HBL complex and Bcet. In relation to the NHE complex, the presence of the nheA and nheC genes was observed in 36% and 13% of strains, respectively, while none of the isolates presented the nheB gene.
Phelps and McKillip (30) detected by PCR individual enterotoxin genes within the Bacillaceae family in some but not all the strains tested that were shown to produce enterotoxins. In addition, Pruβ et al. (32) determined the prevalence of the HBL complex in all species of the B. cereus group, where most of the strains studied carried hblA, but its presence was not related to a certain species or a specific environment. These studies emphasize the need to design assays for Bacillus species that do not rely on a single virulence determinant.
The PCA in our B. cereus collection revealed a positive correlation among free coagulase, hemolysis and the presence of genes hblA, hblB, hblC, hblD (HBL complex) and bceT (enterotoxin-T), but no correlation with the clumping factor (bound coagulase) and the presence of sequences of the NHE complex. This lack of correlation was also observed by Minnaard et al. (27) in B. cereus strains isolated from different sources. On the other hand, PCA in B. megaterium showed a high positive correlation between coagulase (bound and free) and hemolytic activity but no correlation in relation to the presence of genes of the HBL complex, cytotoxin K, enterotoxin T and NHE complex.
Its interesting to point out that the presence of the cytK gene was almost independent of the presence of the rest of virulence factors analyzed here, both in B. cereus (Figure 2B) and B. megaterium populations.
In conclusion, our results show the associations among free coagulase activity and the presence of other virulence factors as specific DNA sequences of enterotoxins and/or hemolytic activity in B. cereus populations isolated from honey samples. It is important to point out that this study is the first report of the detection of genes of cytotoxin K (cytK) and the genes of the non-hemolytic enterotoxin complex (NHE) in B. megaterium and the first screening of virulence factors in B. cereus and B. megaterium isolates from honey.
In addition, an association between coagulase and hemolytic activities was observed in B. megaterium that did not correlate with the presence of specific DNA sequences of enterotoxins. The potential risk of enterotoxins produced by B. megaterium has been noted in honey and their significance in other foods remains to be determined.
Until now, honey is not known to have been involved in outbreaks of disease cause by B. cereus, but our results suggest that honey could be a possible vehicle for foodborne illness. Further studies involving cell infection assays with the collection of B. cereus and B. megaterium from honey on human enterocytes are needed in order to corroborate the correlation between the presence of virulence genes and biological activity.
Acknowledgements: This research was supported by CIC (Comisión de Investigaciones Científicas de la Provincia de Buenos Aires, Argentina) and ANPCyT (Argentina). AMA is a Career Investigator of CIC and ACL is a Career Investigator of CONICET. We wish to thank Dr. Marie-Helene Guinebretiere for providing the strain F4430/73 of B cereus.
1. Alfoldi L. La production induite de megacine en milieu synthetique. Induced production of megacin in synthetic medium. Ann Inst Pasteur 1957; 94: 474-84. [ Links ]
2. Agata N, Ohta M, Arakawa Y, Mori M, Sobe M. A novel dodecadepsipeptide, cereulide, is an emetic toxin of Bacillus cereus. FEMS Microbiol Lett 1995a; 129: 17-20. [ Links ]
3. Agata N, Ohta M, Arakawa Y, Mori M. The bceT gene of B. cereus encodes an enterotoxic protein. Microbiology 1995b; 141: 983-8. [ Links ]
4. Alippi AM. Detection of Bacillus larvae spores in Argentinean honeys by using a semi-selective medium. Microbiología SEM 1995; 11: 343-50. [ Links ]
5. Alippi AM, Reynaldi FJ, López AC, De Giusti MR, Aguilar OM. Molecular epidemiology of Paenibacillus larvae larvae and incidence of American Foulbrood in Argentinean honeys from Buenos Aires Province. J Apic Res 2004; 43: 135-43. [ Links ]
6. Beecher DJ, Wong ACL. Identification of hemolysin BL-producing B. cereus by a discontinuous hemolytic pattern in blood agar. Appl Environ Microbiol 1994; 60: 1646-51. [ Links ]
7. Beecher DJ, Schoeni JL, Wong ACL. Enterotoxic activity of hemolysin BL from Bacillus cereus. Infect Immun 1995; 63: 4423-8. [ Links ]
8. Borge GI, Skeie M, Sorhaaug T, Langsrud T, Granum PE. Growth and toxin profiles of Bacillus cereus isolated from different food sources. Int J Food Microbiol 2001; 69: 237-46. [ Links ]
9. Dierick K, Van Coillie E, Swiecicka I, Meyfroidt G, Devlieger H, Meulemans A, et al. Fatal family outbreak of Bacillus cereus-associated food poisoning. J Clin Microbiol 2005; 43: 4277-9. [ Links ]
10. Dinges MM, Orwin PM, Schlievert M. Exotoxins of Staphylococcus aureus. Clin Microbiol Rev 2003; 13: 16-34. [ Links ]
11. Ehling-Schulz M, Guinebretiere MH, Monthan A, Berge O, Fricker M, Svensson B. Toxin gene profiling of enterotoxic and emetic Bacillus cereus. FEMS Microbiol Letters 2006; 260: 232-40. [ Links ]
12. European Commission. Harmonization of safety criteria for minimally processed foods. In: Martens T, Luchetti A, editors. Inventory Report FAIR Concerted Actions. Brussels, Belgium. FAIR CT, 1997, p. 1-45. [ Links ]
13. Fagerlund A, Ween O, Lund T, Hardy SP, Granum PE Genetic and functional analysis of the cytK family of genes in Bacillus cereus. Microbiology 2004; 150: 2689-97. [ Links ]
14. Gilliam M. Microbiology of pollen and bee bread: the genus Bacillus Apidologie 1979; 10: 269-74. [ Links ]
15. Gilliam M, Valentine DK. Bacteria isolated from the intestinal contents of foraging workers honey bees, Apis mellifera: the genus Bacillus. J Invert Pathol 1976; 28: 275-6. [ Links ]
16. Guinebretiere MH, Broussolle V, Nguyen-The C. Enterotoxigenic profiles of food-poisoning and food-borne Bacillus cereus Strains. J Clin Microbiol 2002; 40: 3053-6. [ Links ]
17. Granum PE. Bacillus cereus and its toxins. J Appl Bacteriol Symposium 1994; (Suppl 76): 61S-66S. [ Links ]
18. Granum PE, Andersson A, Gayther C, Giffel M, Larsen H, Lund T, et al. Evidence for a further enterotoxin complex produced by Bacillus cereus. FEMS Microbiol Letters 1996; 14: 145-9. [ Links ]
19. Granum PE, Lund T. Bacillus cereus and its food poisoning toxins. FEMS Microbiol Letters 1997; 157: 223-8. [ Links ]
20. Guerrant RL, Lima AA, Davidson F. Nausea, vomiting and noninflamatory diarrhea. In: Mandel DG, Bennet JE, Dolin R, editors. Principles and Practice of Infectious Diseases. Philadelphia, USA, Churchil Livingstone, 2000, p. 1126-36. [ Links ]
21. in't Veld PH, Ritmeester WS, Delfgou-van Asch EHM, Dufrenne JB, Wernars K, Smit E, van Leusden FM. Detection of genes encoding for enterotoxins and determination of the production of enterotoxins by HBL blood plates and immunoassays of psychrotrophic strains of Bacillus cereus isolated from pasteurised milk. Int J Food Microbiol 2001; 64: 63-70. [ Links ]
22. Jensen GB, Hansen BM, Eilenberg J, Mahillon J. Minireview. The hidden lifestyles of Bacillus cereus and relatives. Environ Microbiol 2005; 5: 631-40. [ Links ]
23. Lindbäck T, Fagerlund A, Rødland MS, Granum PE. Characterization of the Bacillus cereus NHE enterotoxin. Microbiology 2004; 150: 3959-67. [ Links ]
24. López AC, Alippi AM. Phenotypic and genotypic diversity of Bacillus cereus isolates recovered from honey. Int J Food Microbiol 2007; 117: 175-84. [ Links ]
25. López AC, Alippi AM. Diversity of Bacillus megaterium isolates cultured from honeys. LWT - Food Sci Technol 2009; 42: 212-9. [ Links ]
26. Lund T, De Buyse ML, Granum PE. A new cytotoxin from Bacillus cereus that may cause necrotic enteritis. Mol Microbiol 2000; 38: 254-61. [ Links ]
27. Minnaard J, Delfederico L, Vasseur V, Hollmann A, Rolny I, Semorile L, et al. Virulence of Bacillus cereus: a multivariate analysis. Int J Food Microbiol 2007; 116: 197-206. [ Links ]
28. Padgham J L, Sikora RA. Biological control potential and modes of action of Bacillus megaterium. Crop Protection 2007; 26: 971-7. [ Links ]
29. Pelletier A, Sygusch J. Purification and characterization of three chitosanase activities from Bacillus megaterium P1. Appl Environ Microbiol 1990; 56: 844-8. [ Links ]
30. Phelps RJ, McKillip JL. Enterotoxin production in natural isolates of Bacillaceae outside the Bacillus cereus group. Appl Environ Microbiol 2002; 68: 3147-51. [ Links ]
31. Pirttijarvi TSM, Andersson MA, Scoging AC, Salkinoja- Salonen MJ. Evaluation of methods for recognising strains of the Bacillus cereus group with food poisoning potential among industrial and environmental contaminants. Syst Appl Microbiol 1999; 22: 133-44. [ Links ]
32. Prub BM, Dietrich R, Nibler B, Martlbauer E, Scherer S. The hemolytic enterotoxin HBL is broadly distributed among species of the Bacillus cereus group. Appl Environ Microbiol 1999; 65: 5436-42. [ Links ]
33. Ryan PA, Macmillan JD, Zilinskas BA. Molecular cloning and characterization of the genes encoding the L1 and L2 components of haemolysin BL from Bacillus cereus. J Bacteriol 1997; 179: 2551-6. [ Links ]
34. Rowan NJ, Deans K, Anderson JG, Gemmell CG, Hunter Sl, Chaithong T. Putative virulence factor expression by clinical and food Isolates of Bacillus spp. after growth in reconstituted infant milk formulae. App Environ microbiol 2001; 67: 3873-81. [ Links ]
35. Snowdon JA, Cliver DO. Microorganisms in honey. International Food Microbiol 1996; 31: 1-26. [ Links ]
36. Taylor JM, Sutherland AD, Aidoo KE, Logan NA. Heat-stable toxin production by strains of Bacillus cereus, Bacillus firmus, Bacillus megaterium, Bacillus simplex and Bacillus licheniformis. FEMS Microbiol Letters 2005; 242: 313-7. [ Links ]
37. Thaenthanee S, Wong ACL, Panbangred W. Phenotypic and genotypic comparisons reveal a broad distribution and heterogeneity of hemolysin BL genes among Bacillus cereus isolates. Int J Food Microbiol 2005; 105: 203-12. [ Links ]
38. Vary PS, Biedendieck R, Fuerch T, Meinhardt F, Rohde M, Deckwer WD, Jahn D. Bacillus megaterium - from simple soil bacterium to industrial protein production host. Appl Microbiol Biotechnol 2007; 76: 957-67. [ Links ]
39. Vilas-Bôas GT, Peruca APS, Arantes OMN. Biology and taxonomy of Bacillus cereus, Bacillus anthracis, and Bacillus thuringensis. Can J Microbiol Review 2007; 53: 673-87. [ Links ]
40. Zahner V, Cabral DA, Régua-Mangia AH, Rabinovitch L, Moreau G, McIntosh D. Distribution of genes encoding putative virulence factors and fragment length polymorphisms in the vrrA gene among Brazilian isolates of Bacillus cereus and Bacillus thuringiensis. Appl Environ Microbiol 2005; 71: 8107-14. [ Links ]
Recibido: 19/01/10
Aceptado: 18/05/10














