Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista argentina de microbiología
versión impresa ISSN 0325-7541
Rev. argent. microbiol. vol.43 no.3 Ciudad Autónoma de Buenos Aires jun./set. 2011
ARTÍCULO ORIGINAL
Species distribution and susceptibility profile of yeasts isolated from blood cultures: results of a multicenter active laboratory-based surveillance study in Argentina
Susana Córdoba1*, Walter Vivot1, Maria E Bosco-Borgeat1, Constanza Taverna1, Wanda Szusz1, Omar Murisengo1, Guillermina Isla1, Graciela Davel1 And The Red Nacional De Laboratorios De Micologia2, Argentina
1Departamento Micología, Instituto Nacional de Enfermedades Infecciosas "Dr. C. Malbrán". Av. Vélez Sarsfield 563 (1281) Ciudad Autónoma de Buenos Aires, Argentina
* Correspondence: E-mail: scordoba@anlis.gov.ar
2Red Nacional de Laboratorios de Micología, Argentina (RNLMA): Buenos Aires City: Burkett A, Hospital General de Niños Dr. R. Gutiérrez; Tiraboschi IN, Hospital de Clínicas J. de San Martín; Pereda R, Hospital General de Niños Dr. P. Elizalde; Guelfand L, Hospital Dr. J. Fernández; Fernández A, Instituto de Cardiología y Cirugía Cardiovascular Fundación Favaloro; Schijman M, Hospital Dr. T. Álvarez; Predari S, Instituto de Investigaciones Médicas A. Lanari; Maldonado I, Hospital Alemán; Vázquez R, Clínica y Maternidad Suizo Argentina; Relloso S, Centro de Educaciones Médicas e Investigaciones Clínicas; Buenos Aires: Capece P, Hospital Nacional Dr. A. Posadas; Featherstone P, Hospital Interzonal de Agudos y Crónicos San Juan de Dios; López Moral L, Hospital General de Agudos Dr. C. Argerich; Tuduri A, Hospital Interzonal General de Agudos E. Perón; Gullo H, Hospital Zonal General de Agudos V. López y Planes; López ME, Hospital Dr. JM Ramos Mejía; Posse G, Hospital Provincial Héroes de Malvinas; Mónaco L, Hospital Zonal General de Agudos Dr. D. Paroissien; Mariñansky A, Hospital Zonal General de Agudos Dr. A. Oñativia; Trevisan V, Laboratorio Central de Salud Pública; Garbasz C, Hospital Dr. I. Pirovano; Chaco: Cech N, Hospital 4 de Junio; Giusiano G, Instituto de Medicina Regional, Universidad Nacional del Nordeste; Córdoba: Borletto N, Laboratorio Central Hospital Nacional de Clínicas; Carrizo S, Hospital Rawson; Fraenza L, Hospital Regional Pasteur; Entre Ríos: Petrussi N, Hospital San Martín de Paraná; Yoya N, Hospital D. Masvernat; Formosa: Tichellio A, Hospital Central; Jujuy: Grosso S, Hospital P. Soria; Mendoza: Pagella H, Hospital de Enfermedades Infecciosas Dr. J. Lencinas; Rosaenz L, Hospital Infantil Dr. H. Notti; Tonelli R, Hospital Central; Zanuso A, Hospital T. Schestakow; Misiones: Chade M, Facultad de Ciencias Exactas Químicas y Naturales; Neuquén: Brasili S, Hospital Provincial Castro Rendón; Salta: Fernández N, Hospital San Bernardo; Chacón Y, Hospital Señor del Milagro; San Luis: Hasuoka R, Complejo Sanitario San Luis; Chierichetti B, Policlínico Regional J. D. Perón; Santa Cruz: Villegas J, Hospital Distrital Caleta Olivia; Santa Fe: Amigot S, Centro de Especialidades Médico Ambulatorias; Nardín M, Hospital JM. Cullen; Virgolini S, Hospital de Niños Dr. O. Alassia; Colombo L, Policlínico Escuela E. Perón; Tucumán: Gorostiaga JL, Hospital ÁC. Padilla; Álvarez C, Hospital del Niño Jesús.
ABSTRACT
The Mycology Department of the Instituto Nacional de Enfermedades Infecciosas "Dr. C. Malbrán", conducted the Second National Multicenter Survey on Fungemia due to Yeasts in Argentina. The aim was to obtain updated data of the frequency of the causative species encountered and their in vitro susceptibility to seven antifungal agents. Yeast species were identified by micromorphological and biochemical studies. Antifungal susceptibility testing was performed by the reference microdilution method E.Def 7.1 of the European Committee on Antibiotic Susceptibility Testing (EUCAST). A total of 461 viable yeasts were identified. The most frequent species were: Candida albicans (38.4 %), Candida parapsilosis (26 %), Candida tropicalis (15.4 %) and Candida glabrata (4.3 %). Other uncommon species, such as Candida viswanathii (0.6 %), Candida haemulonii (0.4 %), Candida inconspicua (0.2 %) and Candida fermentati (0.2 %) were also isolated. Among the Candida spp., 5.4 % and 1.6 % were resistant to fluconazole and voriconazole, respectively. Itraconazole and caspofungin were the most efficient agents against all Candida spp. tested (MIC < 1 mg/l). For anidulafungin, 21.6 % of C. parapsilosis showed a MIC value of 4 mg/l. Fluconazole was less active against 53.1 % of Cryptococcus neoformans (MIC > 8 mg/l), 75 % of Trichosporon spp., and 100 % of Rhodotorula spp., Geotrichum candidum, Saccharomyces cerevisiae. The global percentage of mortality was 20 %. The presence of uncommon species reinforces the need for performing continuous laboratory surveillance in order to monitor possible changes, not only in the epidemiological distribution of species, but also in the resistance to antifungal drugs.
Key words: Yeasts; Fungemia; Antifungal susceptibility.
RESUMEN
Distribución de especies y perfil de sensibilidad de levaduras aisladas de hemocultivos: resultados de un estudio multicéntrico de vigilancia de laboratorio en Argentina. El Departamento Micología del Instituto Nacional de Enfermedades Infecciosas "Dr. Carlos G. Malbrán" condujo el segundo estudio multicéntrico nacional sobre funge- mias debidas a levaduras. El objetivo fue obtener datos actualizados sobre la distribución de especies y la sensibilidad in vitro frente a siete antifúngicos. Las levaduras fueron identificadas mediante el estudio de la micromorfología y la realización de pruebas bioquímicas. La determinación de la sensibilidad se realizó según el método de referencia E.Def 7.1 del European Committee on Antibiotic Susceptibility Testing (EUCAST). Se identificaron 461 levaduras. Las especies más frecuentes fueron Candida albicans (38,4 %), Candida parapsilosis (26 %), Candida tropicalis (15,4 %) y Candida glabrata (4,3 %). Se aislaron otras especies menos comunes, como Candida viswanathii (0,6 %), Candida haemulonii (0,4 %), Candida inconspicua (0,2 %) y Candida fermentati (0,2 %). Entre las especies del género Candida, el 5,4 % y el 1,6 % fueron resistentes al fluconazol y al voriconazol, respectivamente. El itraconazol y la caspofungina fueron los antifúngicos más eficaces in vitro frente a las especies de Candida evaluadas (CIM < 1 mg/l). Para la anidulafungina, el 21,6 % de los aislamientos de C. parapsilosis mostraron una CIM de 4 mg/l. El fluconazol fue menos activo para el 53,1 % de los aislamientos de Cryptococcus neoformans (CIM > 8 mg/l), el 75 % de los aislamientos de Trichosporon spp. y el 100 % de los aislamientos de Rhodotorula spp., Geotrichum candidum y Saccharomyces cerevisiae. El porcentaje de mortalidad fue del 20 %. La presencia de especies infrecuentes refuerza la necesidad de realizar la continua vigilancia de laboratorio con el fin de monitorear posibles cambios, no solo en la epidemiología de las especies causantes de fungemia, sino también en la resistencia a los antifúngicos.
Palabras clave: Levaduras; Fungemia; Sensibiliad a los antifúngicos.
INTRODUCTION
The incidence and severity of fungemia due to yeast species have increased over the past decades in hospitalized patients, and currently constitutes the predominant group of hospital-based fungal infections (1, 2, 7, 22, 24, 29, 38). In this context, Martin et al. reported that the sepsis rate due to fungal organisms increased by 207 % in the United States between 1979 and 2000, and observed that, among the yeasts, Candida albicans was the principal etiological agent (22). However, taking into account factors such as the demography of the region, the population under study and the sanitary regulations, the information gathered through studies in different countries may not be applicable to Argentinean cases (1-3, 7, 16, 24, 29, 38). Fungemia due to yeasts has a significant impact on patient outcome, and, in some studies, the associated mortality of invasive infections due Candida spp. (candidemia) has been estimated to be 15-54 % for adults and 10-15 % for neonates and children (7, 24, 38, 45). In this sense, Rodero et al. reported 30 % candidemia-associated mortality in a previous multicenter study conducted in Argentina, between 1999 and 2000, which included 36 hospitals distributed throughout nine provinces and Ciudad Autónoma de Buenos Aires (38). On the other hand, in 2006, Colombo et al. published that the crude mortality rate in Brazil accounted for 54 % of all cases (7). In both studies, the mortality values were not in concordance with those reported by authors in other countries (1, 2, 15, 16), demonstrating the need for the development of further national level studies.
Amphotericin B, azole agents and lipopeptides (echinocandins and pneumocandins) are the antifungal drugs chosen for the treatment of candidemia (26). Therefore, the presence of species with different antifungal susceptibility profiles, such as Candida species intrinsically resistant to fluconazole (C. krusei) or less susceptible to fluconazole (C. glabrata) and the cautious use of amphotericin B due to its dose-limiting nephrotoxicity, emphasize the need for therapies with different antifungal agents. Additionally, several studies have also reported a growing list of azole-resistant yeasts causing candidemia and having the capacity to develop resistance to amphotericin B (2, 3, 15, 37). Thus, it becomes necessary not only to acquire knowledge about species distribution, but also to perform continuous laboratory surveillance of the fungal infection in order to monitor incidence, characterization of emergent yeast species and detection of antifungal resistance. The reference microdilution methods E.Def 7.1 of the European Committee on Antibiotic Susceptibility Testing (EUCAST) (12) and M27-A3 of the Clinical and Laboratory Standards Institute (CLSI) (5) detect in vitro antifungal resistance; both methods produce very similar, quite equivalent MICs, indicating that methodology does not pose any obstacles to obtaining uniform standards for antifungal susceptibility testing of yeasts (39). However, these methods are expensive and very laborious, and their actual usage in hospital laboratories is limited. Thus, the participating laboratories use a disk diffusion method (Malbrán disk) (36) to monitor the susceptibility profile of Candida spp. against fluconazole. This is an available method, having easier accessibility and development, standardized by the National Reference Laboratory of Mycology of the Instituto Nacional de Enfermedades Infecciosas "Dr. C. Malbrán", which yields results comparable to those of reference methods. Therefore, as part of the Surveillance Program in Diagnosis of Fungal Infections and Antifungal Resistance, the Mycology Department conducted the Second National Multicenter Survey on yeast species isolated from blood cultures in Argentina between 2007 and 2008. The objectives were: a) to identify and determine the frequency of yeast species recovered from blood cultures and their susceptibility profile to seven antifungal agents in Argentinean hospitals and b) to determine Candida spp. susceptibility to fluconazole using the Malbrán disk diffusion method.
MATERIALS AND METHODS
Population
The study-subject population was composed of all adult and pediatric hospitalized patients of both genders who developed fungemia due to yeasts.
Candidemia was defined as one or more blood cultures positive for Candida species in patients with relevant clinical signs and symptoms (11). Candidemia that occurred 30 days after the initial case was considered to represent a new case. Other non-Candida spp. yeasts species isolated were also included in the study.
Study design
A systematic, prospective, transversal and coordinated multicenter active laboratory-based surveillance study of antifungal resistance in yeast species isolated from blood cultures was conducted. The study was developed throughout a 13-month period between June 2007 and June 2008. The Mycology Department and 47 national hospital laboratories belonging to the previously established National Laboratory Network of Mycology of Argentina (NLNMA) (10) participated in the study. The participating laboratories were distributed over 14 of the 23 Argentinean provinces and Ciudad Autónoma de Buenos Aires. These hospitals range in size from 150 to 400 beds (average = 236). Each laboratory collected both the samples and clinical data from patients (e.g. risk factors, underlying diseases, clinical symptoms, indwelling catheters, current antibiotic or antifungal therapies, and outcome) at the individual study sites. Only the isolate from the first blood culture obtained from each patient was included. All the isolates were identified by the routine methodologies used at each of the participating institutions, and the in vitro susceptibility to fluconazole against Candida spp. was tested by using the disk diffusion method (36). Then, isolates were referred to the Mycology Department for further species confirmation and antifungal susceptibility testing. Upon arrival, yeasts were subcultured in CHROMagarTM Candida medium (CHROMagar Company Ltd., Paris, France) and YM agar medium (malt extract 0.3 %, yeast extract 0.3 %, peptone 0.5 %, glucose 1 %, agar 2 %) to ensure purity and viability. All green colonies on CHROMagar were presumptively identified as C. albicans and morphology of the colony in Tobacco agar to differentiate C. dubliniensis (18) was performed. For the other yeast species the identification was confirmed by standard protocols (19). Whenever identification by these methods was not possible, a nucleic acid sequence of ribosomal DNA genes using primers ITS1 and ITS4 was carried out (32). The DNA extraction was performed according to Möller et al. (23). The yeast species were stored as suspensions in distilled sterile water at room temperature until needed.
Antifungal susceptibility testing
The minimal inhibitory concentration (MIC) was determined according to the E.Def 7.1 reference document (EUCAST) (12). For non-fermentative yeasts, such as Cryptococcus neoformans, Trichosporon spp. and Rhodotorula spp., the MIC was determined in agitated condition at 250 r.p.m. according to the technique proposed by Rodriguez-Tudela et al. (40).
Antifungal agents
Amphotericin B (AB) and flucytosine (FC) (Sigma-Aldrich Química, Argentina); anidulafungin (AN), fluconazole (FZ) and voriconazole (VZ) (Pfizer S.A., Argentina); itraconazole (IZ) (Janssen, Argentina) and caspofungin (CAS) (Merck, Co, USA) were the drugs tested and were provided as standard powders of known potency.
Quality control strains
Candida parapsilosis ATCC 22019 and C. krusei ATCC 6258 were used (12).
End points
For fluconazole and voriconazole, the interpretative break-points proposed in the E.Def 7.1 document were used (13, 14). For fluconazole, isolates were classified as susceptible (MIC of ≤ 2 mg/l), dose-dependent (MIC of 4 mg/l), or resistant (MIC of > 4 mg/l), whereas for voriconazole, they were classified as susceptible (MIC of ≤ 0.125 mg/l) or resistant (MIC of > 0.125 mg/l). Taking into account that the E.Def 7.1 does not currently include breakpoints for all the antifungal drugs tested in our study, isolates were classified by using the breakpoints proposed by the documents M27-A3 and M27-S3 of the CLSI (5, 6). For amphotericin B, the MIC end point was defined as the lowest concentration of drug that caused a prominent reduction (MIC 0 or ≥ 90 %) of growth compared with that of a drug-free growth control well. For anidulafungin, caspofungin, flucytosine and the azole drugs, the MIC end point was defined as the lowest drug concentration at which the growth of the isolates was reduced by 50 % or more compared with that of the control (MIC 2 or ≥ 50 %) (5, 6).
The MIC50, MIC90, range, geometric mean and percentage of resistance were calculated.
Disk diffusion method
Disk diffusion testing of fluconazole was used against the Candida species and was performed according to Rodero et al. (36). The 25 µg fluconazole disks (Malbrán disk) were provided by the National Institute of Biological Production, ANLIS "Dr. C. Malbrán" (10). Agar plates (90 mm diameter) containing Mueller- Hinton (Difco Laboratories Argentina)-Methylene Blue (Sigma-Aldrich Química, Argentina) with 2 % glucose at a depth of 4.0 ± 1 mm were used. The agar surface was inoculated by using a swab dipped in a cell suspension adjusted to reach the turbidity of a 0.5 McFarland standard scale (1-5 x 106 CFU/ml). The plates were incubated at 35 °C and read after 18 to 24 h. Zone diameter end points were read visually and measured with a ruler. The inhibition zone was considered up to the limit of colonies of normal size. According to the zone diameter interpretative criteria previously published by Rodero et al. (36), this test allows the categorization of the isolates into: susceptible (≥ 16 mm), dose-dependent (9 to 15 mm), and resistant (≤ 8 mm).
Quality control strains
Candida parapsilosis ATCC 22019 and C. krusei ATCC 6258 were used.
Database
A database was designed in order to record the epidemiological, clinical and laboratory data collected from patients.
Statistical analysis
The Pearson χ2 was used to compare categorical variables. Variables recognized as statistically significant in univariate analysis (defined as a p value of < 0.05) were used. Average and percentage were respectively used to describe quantitative and qualitative variables. Data were analyzed using the R-2.11.1 software for Windows (www.r-project.org). MIC determined by the reference method was correlated with inhibition zone diameters (in millimeters) around fluconazole disks. To obtain correlation results (r values), a linear regression analysis by the least-squares method (Pearson's correlation coefficient; MS Excel software) was performed by plotting zone diameters against their respective MIC end points (after log transformation). The percentage of agreement between both methods was also determined.
Discrepancies between methods were considered very major errors when the reference method categorized the organism as resistant, but the diffusion method categorized it as susceptible (falsely susceptible). Major errors occurred when the reference method categorized the isolate as susceptible, but the diffusion method categorized it as resistant (falsely resistant). Minor errors occurred when the reference method categorized an organism as susceptible or resistant and the diffusion method categorized it as susceptible dose dependent (or intermediate) or the reference method categorized it as susceptible dose-dependent (or intermediate) and the diffusion method categorized it as susceptible or resistant.
RESULTS
A total of 461 viable yeasts were recovered from blood cultures and identified. The isolates were obtained from 457 patients, four of whom exhibited infections due to two species: C. albicans + C. parapsilosis; C. albicans + C. lusitaniae; C. parapsilosis + Trichosporon spp. and C. tropicalis + C. parapsilosis.
Among the 420 Candida isolates, C. albicans accounted for 38.4 % (n = 177) of the isolates, whereas non-C. albicans species for 52.7 % (n = 243) of isolates, represented mainly by C. parapsilosis 26.0 %; (n = 120), C. tropicalis 15.4 %; (n = 71) and C. glabrata 4.3 %; (n = 20). Other uncommon Candida species, such as C. pelliculosa and C. guilliermondii 1.5 %; (n = 7 each), C. dubliniensis 0.9 %; (n = 4), C. lusitaniae and C. viswanathii 0.6 %; (n = 3 each), C. haemulonii and C. krusei 0.4 %; (n = 2 each), and C. famata, C. fermentati, C. inconspicua, C. kefyr 0.2 %; (n = 1 each) were also isolated. In addition, 41 (8.9 %) isolates were identified as other yeast non-Candida species: 32 (6.9 %) as Cryptococcus neoformans, 4 (0.8 %) as Trichosporon spp., 2 (0.4 %) as Rhodotorula spp. and Saccharomyces cerevisiae each, and 1 (0.2 %) as the yeast-like fungus Geotrichum candidum.
The fungal isolates were most frequently found in males (male-female ratio = 255: 202). Patients < 1 and > 45 years old comprised the population most susceptible to fungemia due to yeasts 24 %; (n = 110) and 39.6 %; (n = 181), respectively). Among the 177 yeast species isolated from pe- diatric units (age range 0-15 years), the distribution was as follows: C. albicans, 43.5 %; (n = 77), C. parapsilosis 33.3 %; (n = 59), C. tropicalis 13.5 %; (n = 24), C. pelliculosa 2.2 %; (n = 4), C. dubliniensis 0.5 %; (n = 1), C. viswanathii 1.1 %; (n = 2), C. lusitaniae 1.1 %; (n = 2), C. famata 0.5 %; (n = 1), C. inconspicua 0.5 %; (n = 1), Rhodotorula spp. 1.1 %; (n = 2), S. cerevisiae 1.1 %; (n = 2), and Trichosporon spp. 1.1 %; (n = 2). Candida glabrata was frequently isolated in adult patients (> 45 years). C. neoformans isolates were recovered from 32 middle-aged patients (age range 20-44 years) (Table 1).
Table 1. Distribution by age range and gender of 461 yeast strains obtained from blood cultures from 457 patients
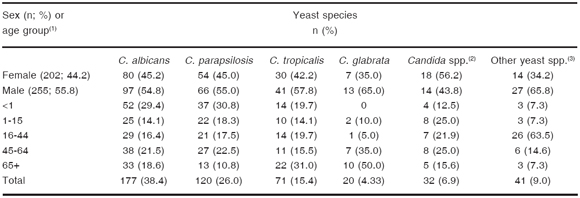
(1)Age groups expressed in years. (2)Candida spp.: 7 C. guilliermondii; 7 C. pelliculosa; 4 C. dubliniensis; 3 C. lusitaniae; 2 C. haemulonii; 3 C. viswanathii; 2 C. krusei; 1 C. famata; 1 C. fermentati; 1 C. kefyr; 1 C. inconspicua. (3)Other yeast species: 32 C. neoformans, 1 G. candidum, 2 Rhodotorula spp., 2 S. cerevisiae, 4 Trichosporon spp.
A statistically significant association with C. albicans candidemia cases was observed in antibiotic therapy (87.0 %) (p = 0.001), mechanical ventilation (43.5 %) (p = 0.000), and total parenteral nutrition (38.4 %) (p = 0.009) (Table 2).
Table 2. Patients' clinical characteristics by Candida species isolated

(1)Patient category and concomitant risk factors were not mutually exclusive (patients could have one or more characteristics within a category). PC: peripheral catheter. (2)Candida spp.: 7 C. guilliermondii; 7 C. pelliculosa; 4 C. dubliniensis; 3 C. lusitaniae; 2 C. haemulonii; 3 C. viswanathii; 2 C. krusei; 1 C. famata; 1 C. fermentati; 1 C. kefyr; 1 C. inconspicua. (3)p < 0.05. (4)Non-transplantation.
The MICs value of Candida spp. tested are summarized in Table 3. Overall, the yeasts tested were susceptible to AB (MIC ≤ 1mg/l), with the exception of one C. parapsilosis isolate (MIC 2 mg/l), and two of Trichosporon spp. (MIC 2 and 4 mg/l). Among Candida species, 20 % of C. glabrata, followed by 4.2 % of C. tropicalis, 2.5 % of C. parapsilosis, 42.8 % of C. pelliculosa and 28.5 % of C. guilliermondii were resistant to fluconazole. On the other hand, caspofungin exhibited a broad-spectrum activity against Candida species and was more active in vitro than anidulafungin, MIC < 0.015 - 2 mg/l versus < 0.015 - 4 mg/l respectively. For anidulafungin, C. parap- silosis 21.6 %, (n = 26) was the only species that showed a MIC value of 4 mg/l. For azole drugs, independently of the species tested, itraconazole was the most effective drug in vitro (MIC ≤ 1 mg/l), while for fluconazole, 53.1 %, (n = 17) of C. neoformans exhibited high values (MIC 8-128 mg/l) followed by 75 %, (n = 3) of Trichosporon spp. (MIC 8 mg/l), 100 % each, of Rhodotorula spp. (MIC 32-128 mg/l), G. candidum (MIC 32 mg/l) and S. cerevisiae (MIC 8 mg/l).
Table 3. Candida spp., MIC values and percentage of resistance of antifungal agents tested
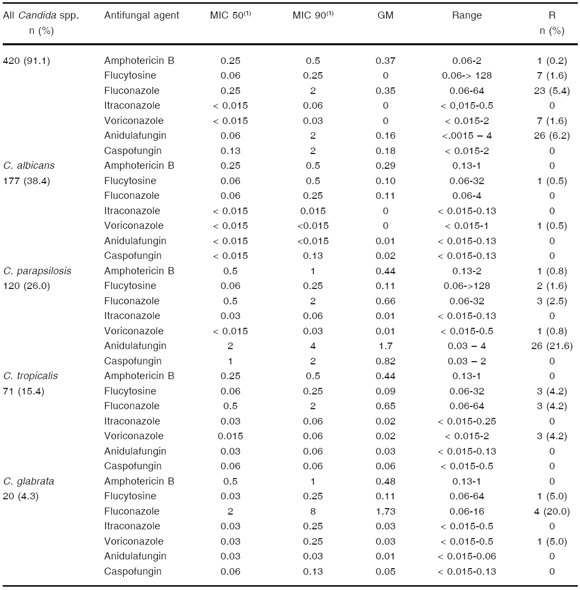
MIC: Minimal inhibitory concentration. (1)MIC at which 50 % and 90 % of isolates tested, respectively, are inhibited. GM: geometric mean; R: resistant.
Disk diffusion
The correlation coefficient (linear regression analysis) between the MIC results and the corresponding inhibition zone diameters, in millimeters, for the 420 Candida spp. evaluated was r = 0.73. Overall, an agreement of 97.1 % was observed between the microdilution standard method and the disk diffusion method, and only 12 (2.8 %) very major errors were detected (3 C. pelliculosa, 2 C. glabrata, 2 C. guilliermondii, 1 C. famata, 1 C. fermentati, 1 C. haemulonii, 1 C. tropicalis, and 1 C. parapsilosis).
Outcome
Eighty patients died while still showing blood cultures positive to Candida species: C. albicans (27), C. parapsilosis and C. tropicalis (19 each), C. glabrata (7), followed by C. guilliermondii (2) and C. dubliniensis, C. haemulonii, C. kefyr, C. krusei, C. pelliculosa, C. viswanathii (1 each). Infections due to C. neoformans were mainly associated with HIV/AIDS, 81.2 %, (n = 26); in addition, 26.9 %, (n = 7) of them, and 1/6 of non-HIV/AIDS-related cases had fatal outcomes. Other species isolated from fatal infections were Trichosporon spp. (3), and Rhodotorula spp. (1).
Regarding treatment and outcome, 75.7 %; (n = 346) cases received some antifungal treatment, but a total of 24.3 %; (n = 111) cases were never treated. Fluconazole was the drug most frequently used as primary treatment 49.4 %; (n = 171), followed by amphotericin B 48.2 %; (n = 167 cases). The global percentage of mortality was 20 %, whereas the mortality percentage in patients who received antifungal therapy versus those who died without treatment was 16.7 % and 30.1 %, respectively (p = 0.002).
DISCUSSION
The incidence and severity of bloodstream infections due to yeast species have increased in hospitalized patients, and to date, constitute an unsolved problem. Therefore, we conducted the Second National Multicenter Survey on Fungemia due to Yeasts in Argentina. This report provides updated data on the frequency of the causative species encountered and their susceptibility to antifungal agents in Argentinean hospitals between 2007 and 2008. In our study, we observed differences with authors' reports from other countries (2, 15, 16, 25, 29). In this sense, C. albicans remained the most common species causing candidemia (38.3 %), followed by C. parapsilosis (26 %), C. tropicalis (15.4 %) and C. glabrata (4.3 %). We observed an increase in the frequency of C. glabrata isolates compared with data from Rodero et al., (2.6 % vs. 4.3 %) in a previous National Multicenter Study (38). In contrast, in the United States, Germany and Venezuela (2, 15, 25, 29), C. glabrata was the second, probably due to the extended use of prophylactic therapies with fluconazole in these countries. In Argentina, the broad use of fluconazole in prophylaxis is still not a common practice; however, it is important to note that 20 % and 5 % of C. glabrata isolated were resistant to fluconazole and voriconazole, respectively. Also, the percentage of fluconazole resistance was higher than that reported by other authors (7, 25). This is a point to be taken into account in relation to the therapeutic management of patients and the use of azole drugs, due to their ability to develop secondary resistance after prolonged pre-exposure with azole agents.
In the present study, 38.3 % (n = 177) of fungemia cases occurred in pediatric patients, 33.3 % of which were due to C. parapsilosis, occupying the second place after C. albicans in agreement with data presented by Pfaller et al. (29). However, Santos et al., (41) reported that C. parapsilosis was the most frequent species isolated from candidemia in pediatric patients. Such discrepancy in species distribution could be possibly explained by the different geographical regions among pediatric hospitals and the individual patient characteristics (15, 16). The prevalence of C. parapsilosis as the causal agent of candidemia should be taken into account in order to prevent horizontal transmission due to deficiencies in catheter handling and infection control procedures, as previously reported (1, 15, 29, 38). It should be necessary to focus on preventable control techniques such as careful hand hygiene and adequate catheter placement and care in preventing C. parapsilosis infection (42).
Interestingly, we recovered only two C. krusei isolates (0.4 %), in contrast with those reported in series from the United States (2-4 %) (16, 29) and Europe (4 %) (1). The epidemiological distribution and the low frequency of C. krusei isolation in our series is difficult to explain; a possible reason is that the patients did not receive fluconazole in prophylaxis, reducing the risk of the appearance of selective pressure (43). Other Candida species less frequently isolated were C. guilliermondii, C. pelliculosa and C. dubliniensis. Several reports indicate the small proportion of these species causing candidemia ~1 to 3 % and the low susceptibility that some isolates exhibit to fluconazole and candins (20, 28, 31). In our survey, 28.5 %, (n = 2) C. guilliermondii, 42.8 %, (n = 3) C. pelliculosa and 25 %, (n = 1) C. dubliniensis showed MIC values of 8 mg/l for fluconazole; while all of them were quite susceptible to anidulafungin and caspofungin. In relation to the candins tested, our findings are similar to those reported by Pfaller et al. in a Global Surveillance Study that included 5,346 invasive (bloodstream or sterile site) Candida spp. isolates. However, in that work, 12.5 %, (n = 5) and 7.5 %, (n = 3) C. guilliermondii isolated in Latin America exhibited MIC values of 4 mg/l and ≥ 8 mg/l against anidulafungin and caspofungin, respectively (27). Thus, we highlight the importance of correctly identifying these yeast species and the knowledge of their susceptibility profile taking into account the possible disagreement with other works published.
Yeast species were identified according to standard procedures (19). However, identification to species level is not always possible and molecular techniques are needed. In our survey, using molecular procedures, we identified yeast species uncommonly isolated from blood cultures such as Candida viswanathii and C. fermentati, which were not correctly identified based on the conventional methods used (19, 20). To our knowledge, this is the first time that both species are isolated from blood cultures in Argentina. Among the other yeast species, C. neoformans was prevalent and was associated with HIV/AIDS patients. Rhodotorula spp., Trichosporon spp., S. cerevisiae and G. candidum were also isolated as agents causing fungemia. The correct identification of this group of yeasts is mandatory due to the low response, both in vivo and in vitro, to antifungal drugs that some isolates commonly exhibit (4, 9, 17, 35). In agreement, in our study, the Rhodotorula spp., Trichosporon spp., and C. neoformans isolates were less susceptible to fluconazole (MIC ≥ 8 mg/l), and most susceptible to itraconazole and voriconazole in vitro.
Overall, the Candida species tested were susceptible to amphotericin B (MIC ≤ 1 mg/l) in vitro, with the exception of 1 C. parapsilosis (MIC = 2 mg/l). It is important to note that the reference methods available (M27-A and E.Def 7.1) failed to detect resistance to amphotericin B, probably due to the narrow MIC range obtained. Some works suggest the use of Antibiotic Medium 3 to obtain a more reliable detection of resis- tant isolates (34, 43). However, this is a questioned recommendation since it has been observed that the medium varies in composition depending on the lot, and thus the results are not reproducible or reliable (21). Other susceptibility tests, such as time kill curves and minimal fungicidal concentration, should be useful to detect resistance to this polyene, as we have observed in previous studies with C. neoformans tested by time kill curves (8, 35).
In a large study against 8,803 clinical isolates of Candida spp., Pfaller et al., reported that primary resistance to flucytosine is very uncommon among Candida spp. (30). Flucytosine is not commercially available in Argentina, thus, it is not used in treatment. However, we studied the in vitro profile taking into account that flucytosine is recommended in combination with amphotericin B as initial therapy of cryptococcal meningitis (33). We observed that only 6.25 %, (n = 2) of C. neoformans isolates tested exhibited MIC values of 16 mg/l. This finding could be considered for patients showing clinical failure to therapy with amphotericin B alone or fluconazole. In our survey, among Candida species, 1.6 %, (n = 7) isolates showed MIC values of > 32 mg/l, although no patient had previously received flucytosine as treatment, probably due to primary resistance to this drug.
Itraconazole was highly effective against C. krusei and C. glabrata. These species are commonly less susceptible to fluconazole, although itraconazole is neither used nor recommended in candidemia treatment. Interestingly, researchers from Europe and Latin America found a high percentage of resistance against itraconazole in Candida spp. (27.6 % and 17.6 %, respectively) (2, 25).
The EUCAST has recently published the breakpoints to fluconazole and voriconazole for C. albicans, C. parapsilosis and C. tropicalis (13, 14). Thus, the E.Def 7.1 proposed that MIC to fluconazole ≤ 2, 4 and > 4 mg/l corresponds to susceptible, dose-dependent and resistant isolates, respectively. These breakpoints are clearly lower than those proposed in the M27-A3 document, whose catego- ries are ≤ 8, 16-32 and ≥ 64 mg/l, respectively. In our study, according to the new breakpoints, we observed that the E.Def 7.1 categorized a large number of isolates as resistant to fluconazole. In relation to voriconazole, we also detected 1.6 % (n = 7) resistant isolates among Candida spp. tested.
Caspofungin and anidulafungin act by inhibition of 1,3-ß-D-glucan synthesis in the fungal cell wall, have fungicidal activity against most species of Candida, includ- ing azole-resistant species, and are recommended for candidemia treatment (26). In the M27-S3 document, it is considered that MIC values ≤ 2 mg/l are predictive of susceptibility (6). Thus, in our study, the Candida spp. (n = 420) tested against caspofungin and anidulafungin were highly susceptible. For anidulafungin, C. parapsilosis 21.6 % (n = 26) was the only species that showed MIC values of 4 mg/l; this finding has also been previously reported (29).
The Mycology Department is the National Reference Laboratory of Mycology of the Instituto Nacional de Enfermedades Infecciosas "Dr. C. Malbrán" and provides a standardized fluconazole disk to the National Hospitals all over the Argentinean territory in order to monitor the sus- ceptibility profile of Candida spp. against this drug (36). Thus, in the present study, the participant laboratories used the Malbrán disk. Overall, an agreement of 97.1 % was observed between the microdilution standard method and the disk diffusion method. It is important to note that most discrepancies were found in uncommon species; in these cases, the MIC determination is recommended. Therefore, the Malbrán disk could be used in laboratories to explore susceptibility to fluconazole in most frequent Candida spp. This diffusion method for susceptibility testing of Candida spp. isolates is inexpensive, reliable and reproducible. However, when the inhibition zone diameter is ≤ 15 mm, it is advisable to test the isolate by the reference microdilution method (36).
With regard to the treatment and outcome, 346 pa- tients received some antifungal treatment. Fluconazole and amphotericin B were the drugs more commonly used, as recommended in the Guidelines for Candi- demia Treatment (26). Caspofungin and voriconazole alone or in combination were also used. In spite of this, a fatal outcome was observed in 92 cases (20 %), 58 of which had received some antifungal treatment, and the remaining 34 had never been treated. In our study, the percentage of mortality was lower than that reported by Rodero et al. (30 %) in a previous Multicenter National Study (38), and than that found in studies from other countries (1, 7, 15, 16, 29). A point to remark is that the mortality percentage in patients who received antifungal therapy versus patients who died without treatment was 16.7 % vs. 30.1 % (p = 0.002).
It is also important to point out that sanitary regulations have changed in Argentina during the last decades in order to provide better health care. In this sense, the present study was performed with the collaboration of the laboratories that belong to the NLNMA, where the professionals are qualified to give an early diagnosis, to correctly identify the agents causing fungemia, and to accurately decide on the appropriate treatment. This has contributed to the low mortality observed in the present work. Thus, the data obtained reveal the current situation of yeasts fungemia in our country, and this information should be a potential tool to be considered as a guide to optimize and choose the best rational treatment.
Finally, it is important to point out that resistance to antifungal drugs remains low and restricted to a few isolates. It should be necessary to focus on the pres- ence of uncommon species causing candidemia, such as C. fermentati, C. haemulonii, C. inconspicua and C. viswanathii since some of them exhibit low susceptibility to antifungal drugs (20, 29, 37). Thus, we highlight the necessity to develop continuous laboratory surveillance in order to monitor possible changes, not only in the epidemiological distribution of species but also in the epidemiological resistance to antifungal drugs.
Acknowledgements: We express our gratitude to Lucia Irazu for her assistance in the statistical analysis of the results.
REFERENCES
1. Almirante B, Rodríguez D, Park BJ, Cuenca-Estrella M, Planes AM, Almela M, Mensa J, Sanchez F, Ayats J, Gimenez M, Saballs P, Fridkin SK, Morgan J, Rodriguez-Tudela JL, Warnock DW, Pahissa A, and the Barcelona Candidemia Project Study Group. Epidemiology and Predictors of Mortality in Cases of Candida Bloodstream Infection: Results from Population-Based Surveillance, Barcelona, Spain, from 2002 to 2003. J Clin Microbiol 2005; 43: 1829-35. [ Links ]
2. Borg-von Zepelin M, Kunz L, Rüchel R, Reichard U, Weig M, Gross U. Epidemiology and antifungal susceptibilities of Candida spp. to six antifungal agents: results from a surveillance study in Germany from July 2004 to August 2005. J Antimicrob Chemother 2007; 60: 424-8. [ Links ]
3. Bruder-Nascimento A, Camargo CH, Sugizaki MF, Sadatsune T, Montelli AC, Mondelli AL, Bagagli E. Species distribution and susceptibility profile of Candida species in a Brazilian public tertiary hospital. BMC Res Notes 2010; 3: 1-5. [ Links ]
4. Chagas-Neto TC, Chaves GM, Melo AS, Colombo A. Bloodstream infections due to Trichosporon spp.: species distribution, Trichosporon asahii genotypes determined on the basis of ribosomal DNA intergenic spacer 1 sequencing, and antifungal susceptibility testing. J Clin Microbiol 2009; 47: 1074-81. [ Links ]
5. Clinical and Laboratory Standards Institute. Reference method for broth dilution antifungal susceptibility testing of yeasts. Approved standard. Third Edition. CLSI document M27-A3. Clinical and Laboratory Standards Institute, Wayne, Pennsylvania 19087-1898 USA, 2008. [ Links ]
6. Clinical and Laboratory Standards Institute. Reference method for broth dilution antifungal susceptibility testing of yeasts; Third Informational Supplement. CLSI document M27-S3. Clinical and Laboratory Standards Institute, Wayne, Pennsylvania; 2008. [ Links ]
7. Colombo AL, Nucci M, Park BJ, Nouér SA, Arthington-Skaggs B, da Matta DA, Warnock D, Morgan J. Epidemiology of candidemia in Brazil: a Nationwide Sentinel Surveillance of Candidemia in Eleven Medical Centres. J Clin Microbiol 2006; 44: 2816-23. [ Links ]
8. Córdoba S. Métodos alternativos de laboratorio para la detección de cepas de Cryptococcus neoformans resistentes a los antifúngicos. Tesis de Doctorado en Bacteriología Clínica e Industrial. 2007. INEI ANLIS "Dr. Carlos G. Malbrán" y Universidad Nacional de La Plata. [ Links ]
9. da Cunha MM, dos Santos LP, Dornelas-Ribeiro M, Vermelho AB, Rozental S. Identification, antifungal susceptibility and scanning electron microscopy of a keratinolytic strain of Rhodotorula mucilaginosa: a primary causative agent of onychomycosis. FEMS Immunol Med Microbiol 2009; 55: 396-403. [ Links ]
10. Davel G, Canteros CE. Epidemiological status of mycoses in the Argentine Republic. Rev Argent Microbiol 2007; 39: 28-33. [ Links ]
11. De Pauw B, Walsh TJ, Donnelly JP, Stevens D, Edwards JE, Calandra C, Pappas PG, Maertens J, Lortholary O, Kauffman CA, Denning DW, Patterson TF, Maschmeyer GR, Bille J, Dismukes WE, Herbrecht R, Hope WW, Kibbler CC, Jan Kullberg B, Marr KA, Munoz P, Odds FC, Perfect JR, Restrepo A, Ruhnke M, Segal BH, Sobel JD, Sorrell TC, Viscoli C, Wingard JR, Zaoutis T, Bennett JE. Revised Definitions of Invasive Fungal Disease from the European Organization for Research and Treatment of Cancer/Invasive Fungal Infections Cooperative Group and the National Institute of Allergy and Infectious Diseases Mycoses Study Group (EORTC/MSG) Consensus Group. Clin Infect Dis 2008; 46: 1813-21. [ Links ]
12. EUCAST definitive document E.Def 7.1: method for the determination of broth dilution MICs of antifungal agents for fermentative yeasts. Clin Microbiol Infect 2008; 14: 398-405. [ Links ]
13. EUCAST technical Note on fluconazole. European Committee on Antimicrobial Susceptibility Testing-Subcommittee on Antifungal Susceptibility Testing (EUCAST-AFST). Clin Microbiol Infect 2008; 14: 193-5. [ Links ]
14. EUCAST technical Note on voriconazole. European Committee on Antimicrobial Susceptibility Testing-Subcommit- tee on Antifungal Susceptibility Testing (EUCAST-AFST). Clin Microbiol Infect 2008; 14: 985-7. [ Links ]
15. Hajjeh RA, Sofair AN, Harrison LH, Lyon M, Arthington-Skaggs BA, Mirza SA, Phelan M, Morgan J, Lee-Yang W, Ciblak MA, Benjamin LE, Thomson Sanza L, Huie S, Fah Yeo S, Brandt ME, Warnock DW. Incidence of bloodstream infections due to Candida species and in vitro susceptibilities of isolates collected from 1998 to 2000 in a population-based active surveillance program. J Clin Microbiol 2004; 42: 1519-27. [ Links ]
16. Horn DL, Neofytos D, Anaissie E, Fishman JA, Steinbach WJ, Olyaei AJ, Marr KA, Pfaller MA, Chang CH, Webster KM. Epidemiology and Outcomes of Candidemia in 2019 Patients: Data from the Prospective Antifungal Therapy Alliance Registry. CID 2009; 48: 1695-703. [ Links ]
17. Kalkanci A, Sugita T, Arikan S, Yucesoy M, Ener B, Otag F, Kiraz N, Kustimur S, Sancak B, Evci C, Emektas G. Molecular identification, genotyping, and drug susceptibility of the basidiomycetous yeast pathogen Trichosporon isolated from Turkish patients. Med Mycol 2010; 48: 141-6. [ Links ]
18. Khan ZU, Ahmad S, Mokaddas E, Chandy R. Tobacco agar, a new medium for differentiating Candida dubliniensis from Candida albicans. J Clin Microbiol 2004; 42: 4796-8. [ Links ]
19. Kurtzman CP. ed. Kurtzman CP. and Fell JW. The Yeasts: A Taxonomic Study, 4th edition, Elsevier. 1998. [ Links ]
20. Lockhart SR, Messer SA, Pfaller MA, Diekema DJ. Identification and Susceptibility Profile of Candida fermentati from a Worldwide Collection of Candida guilliermondii Clinical Isolates. J Clin Microbiol 2009; 47: 242-4. [ Links ]
21. Lozano-Chiu M, Nelson PW, Lancaster M, Pfaller MA, Rex JH. Lot-to-lot variability of antibiotic medium 3 when used for susceptibility testing of Candida isolates to amphotericin B. J Clin Microbiol 1997; 35: 270-2. [ Links ]
22. Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 2003; 348: 1546-54. [ Links ]
23. Möller EM, Bahnweg G, Sandermann H, Geiger HH. A simple and efficient protocol for isolation of high molecular weight DNA from filamentous fungi, fruit bodies, and infected plant tissues. Nucleic Acids Res 1992; 20: 6115-6. [ Links ]
24. Morgan J, Meltzer MI, Plikaytis BD, Sofair AN, Huie-White S, Wilcox S, Harrison LH, Seaberg EC, Hajjeh RA, Teutsch S. Excess mortality, hospital stay, and cost due to candidemia: a case-control study using data from population-based candidemia surveillance. Infect Control Hosp Epidemiol 2005; 26: 540-7. [ Links ]
25. Panizo MM, Reviákina V, Dolande M, Selgrad S. Candida spp. in vitro susceptibility profile to four antifungal agents. Resistance surveillance study in Venezuelan strains. Med Mycol 2009; 47: 137-43. [ Links ]
26. Pappas PG, Kauffman CA, Andes D, Benjamin DK, Calandra T Jr., Edwards JE Jr., Filler SC, Fisher JF, Kullberg BJ, Ostrosky-Zeichner L, Reboli AC, Rex JH, Walsh TJ, Sobel JD. Clinical Practice Guidelines for the Management of Candidiasis: 2009 Update by the Infectious Diseases Society of America. CID 2009; 48: 503-35. [ Links ]
27. Pfaller MA, Boyken L, Hollis RJ, Kroeger J, Messer SA, Tendolkar S, Diekema DJ. In vitro Susceptibility of Invasive Isolates of Candida spp. to Anidulafungin, Caspofungin, and Micafungin: Six Years of Global Surveillance. J Clin Microbiol 2008; 46: 150-6. [ Links ]
28. Pfaller MA, Diekema DJ, Jones RN, Sader HS, Fluit AC, Hollis RJ, Messer SA, and the SENTRY Participant Group. International Surveillance of Bloodstream Infections Due to Candida Species: Frequency of Occurrence and In Vitro Susceptibilities to Fluconazole, Ravuconazole, and Voriconazole of Isolates Collected from 1997 through 1999 in the SENTRY Antimicrobial Surveillance Program. J Clin Microbiol 2001; 39: 3254-9. [ Links ]
29. Pfaller MA, Diekema DJ. Epidemiology of Invasive Candidiasis: a Persistent Public Health Problem. Clin Microbiol Rev 2007; 20: 133-63. [ Links ]
30. Pfaller MA, Messer SA, Boyken L, Huynh H, Hollis RJ, Diekema DJ. In vitro activities of 5-fluorocytosine against 8,803 clinical isolates of Candida spp.: global assessment of primary resistance using National Committee for Clinical Laboratory Standards susceptibility testing methods. Antimicrob Agents Chemother 2002; 46: 3518-21. [ Links ]
31. Pfaller, MA, Diekema DJ, Mendez M, Kibbler C, Erzsbet P, Chang SC, Gibbs DL, Newell VA, and the Global Antifungal Surveillance Group. Candida guilliermondii, an opportunistic fungal pathogen with decreased susceptibility to fluconazole: geographic and temporal trends from the ARTEMIS DISK Antifungal Surveillance Program. J Clin Microbiol 2006; 44: 3551-6. [ Links ]
32. Pincus DH, Orenga S, Chatellier S. Yeast identification -past, present, and future methods. Med Mycol 2007; 45: 97-121. [ Links ]
33. Powderly WG. Current approach to the management of cryptococcal infections. J Infect 2000; 41: 18-22. [ Links ]
34. Rex JH, Cooper CR Jr., Merz WG, Galgiani JN, Anaissie EJ. Detection of amphotericin B-resistant Candida isolates in a broth-based system. Antimicrob Agents Chemother 1995; 39: 906-9. [ Links ]
35. Rodero L, Córdoba S, Cahn P, Soria M, Lucarini M, Davel G, Kaufman S, Canteros C, Guelfand L. Time-kill curves for Cryptococcus neoformans isolated from patients with AIDS. Med Mycol 2000; 38: 201-7. [ Links ]
36. Rodero L, Córdoba S, Vivot W, Campo M, Corfield P, Olguín C, Cuirolo A, Soria M, Guelfand L, Canteros CE, Davel G, Red WHONET. Disk diffusion method for fluconazole susceptibility testing of Candida spp. isolates. Rev Argent Microbiol 2006; 38: 155-63. [ Links ]
37. Rodero L, Cuenca-Estrella M, Córdoba S, Cahn P, Davel G, Kaufman S, Guelfand L, Rodríguez-Tudela JL. Transient Fungemia Caused by an Amphotericin B-Resistant Isolate of Candida haemulonii. J Clin Microbiol 2002; 40: 2266-9. [ Links ]
38. Rodero L, Davel G, Soria M, Vivot W, Córdoba S, Canteros CE, Saporiti A y Participantes del Grupo EMIFN. Multicenter study of fungemia due to yeasts in Argentina. Rev Argent Microbiol 2005; 37: 189-95. [ Links ]
39. Rodriguez-Tudela JL, Donnelly JP,.Pfaller MA, Chryssantou E, Warn P, Denning DW, Espinel-Ingroff A, Barchiesi F, Cuenca-Estrella M. Statistical Analyses of Correlation between Fluconazole MICs for Candida spp. Assessed by Standard Methods Set Forth by the European Committee on Antimicrobial Susceptibility Testing (E.Dis. 7.1) and CLSI (M27-A2) J Clin Microbiol 2007; 45: 109-11. [ Links ]
40. Rodríguez-Tudela JL, Martín-Diez F, Cuenca-Estrella M, Rodero L, Carpintero Y, Gorgojo B. Influence of shaking of antifungal susceptibility testing of Cryptococcus neoformans: a comparison of NCCLS Standard M27-A medium, buffered yeast nitrogen base, and RPMI-2 % glucose. Antimicrob Agent Chemoter 2000; 44: 400-4. [ Links ]
41. Santos PE, Córdoba S, Carrillo-Munoz A, Rodero L, Rubeglio E, Soria M. Epidemiología de las fungemias en un hospital pediátrico de alta complejidad. Rev Iberoam Micol 2010; 27: 200-2. [ Links ]
42. Sarvikivi E, Lyytikainen O, Soll DR, Pujol C, Pfaller MA, Richardson M, Koukila-Kähkölä P, Luukkainen P, Saxén H. Emergence of fluconazole resistance in a Candida parapsilosis strain that caused infections in a neonatal intensive care unit. J Clin Microbial 2005: 43: 2729-35. [ Links ]
43. Slavin MA,Sorrell TC, MarriottD, ThurskyKA, Nguyen Q, Ellis DH, Morrissey CO, Chen SC; Australian CandidemiaStudy, Australasian Society for Infectious Diseases. Candidemia in adult cancer patients: risks for fluconazole-resistant isolates and death. J Antimicrob Chemother 2010; 65: 1042-51. [ Links ]
44. Wanger A. Mills K. Nelson PVV, Rex JH. Comparison of Etest and National Committee for Clinical Laboratory Standards broth macrodilution method for antifungal susceptibility testing: enhanced ability to detect amphotericin B-resistant Candida isolates. Antimicrob Agents Chemother 1995; 39: 2520-2. [ Links ]
45. Zaoutis TE, Argon J, Chu J, Berlin JA. Walsh TJ. Feudtner C. The epidemiology and attributable outcomes of candidemia in adults and children hospitalized in the United States: a propensity analysis. Clin Infect Dis 2005; 41: 1232-9. [ Links ]
Recibido: 11.'01/11
Aceptado: 5/07/11














