Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista argentina de microbiología
versión impresa ISSN 0325-7541
Rev. argent. microbiol. vol.46 no.4 Ciudad Autónoma de Buenos Aires dic. 2014
ORIGINAL ARTICLE
Response of ligninolytic macrofungi to the herbicide atrazine: dose-response bioassays
Respuesta de macrohongos ligninolíticos al herbicida atrazina: bioensayos dosis-respuesta
Wilberth Chan Cupula*, Gabriela Heredia Abarcaa, Refugio Rodríguez Vázquezb, Dulce Salmonesa, Rigoberto Gaitán Hernándeza, Enrique Alarcón Gutiérrezc
aInstituto de Ecología A. C. (INECOL), Xalapa, Veracruz, Mexico
bDepartamento de Biotecnología y Bioingeniería, CINVESTAV-IPN, Mexico City, Mexico
cInstituto de Biotecnología y Ecología Aplicada (INBIOTECA), Universidad Veracruzana, Xalapa, Veracruz, Mexico
* Corresponding author.
E-mail address: wilberth_20@hotmail.com (W.C. Cupul).
Received November 15, 2013
Accepted October 23, 2014
Abstract
The effect of atrazine concentrations on mycelial growth and ligninolytic enzyme activities of eight native ligninolytic macrofungi isolated in Veracruz, México, were evaluated in a semi-solid culture medium. Inhibition of mycelial growth and growth rates were significantly affected (p = 0.05) by atrazine concentrations (468, 937, 1875, and 3750 mg/l). In accordance with the median effective concentration (EC50), Pleurotus sp. strain 1 proved to be the most tolerant isolate to atrazine (EC50 = 2281.0 mg/l), although its enzyme activity was not the highest. Pycnoporus sanguineus strain 2, Daedalea elegans and Trametes maxima showed high laccase activity (62.7, 31.9, 29.3 U mg/protein, respectively) without atrazine (control); however, this activity significantly increased (p < 0.05) (to 191.1, 83.5 and 120.6 U mg/protein, respectively) owing to the effect of atrazine (937 mg/l) in the culture medium. Pleurotus sp. strain 2 and Cymatoderma elegans significantly increased (p < 0.05) their manganese peroxidase (MnP) activities under atrazine stress at 468 mg/l. The isolates with high EC50 (Pleurotus sp. strain 1) and high enzymatic activity (P. sanguineus strain 2 and T. maxima) could be considered for future studies on atrazine mycodegradation. Furthermore, this study confirms that atrazine can increase laccase and MnP activities in ligninolytic macrofungi.
Keywords
Herbicide tolerance; Laccase; Manganese peroxidase; Oxidative stress; White-rot fungi.
© 2013 Asociación Argentina de Microbiología. Published by Elsevier España, S.L. All rights reserved.
Resumen
Se evaluó el efecto de diferentes concentraciones de atrazina sobre el crecimiento micelial y la actividad enzimática de ocho macrohongos ligninolíticos aislados en Veracruz, México. La inhibición del crecimiento micelial y la tasa de crecimiento diaria fueron significativamente (p < 0,05) afectadas por todas las dosis de atrazina (468, 937, 1875 y 3750 mg/l) adicionadas al medio de cultivo. De acuerdo con la concentración efectiva media (CE50), Pleurotus sp. cepa 1 fue el aislamiento más tolerante a la atrazina (CE50 = 2281 mg/l), aunque sus actividades enzimáticas no fueron altas. Pycnoporus sanguineus cepa 2, Daedalea elegans y Trametes maxima mostraron actividades altas de lacasa (62,7, 31,9 y 29,3 U mg/proteína, respectivamente) en ausencia de atrazina (control); estas actividades se incrementaron (p < 0,05) significativamente (191,1, 83,5 y 120,6 U mg/proteína, respectivamente) en presencia de atrazina (937 mg/l) en el medio de cultivo. Pleurotus sp. cepa 2 y Cymatoderma elegans incrementaron significativamente (p < 0,05) sus actividades de manganeso peroxidasa (MnP) bajo la concentración de 468 mg/l de atrazina. Los aislamientos con alta CE50 (Pleurotus sp. cepa 1) y alta actividad enzimática (P. sanguineus cepa 2 y T. maxima) podrían ser considerados para futuros estudios en la micodegradación de atrazina. Además, el presente estudio confirma que la atrazina puede incrementar las actividades lacasa y MnP en macrohongos ligninolíticos.
Palabras clave
Tolerancia a herbicidas; Lacasa; Manganeso peroxidasa; Estrés oxidativo; Hongos de la pudrición blanca.
© 2013 Asociación Argentina de Microbiología. Publicado por Elsevier España, S.L. Todos los derechos reservados.
Introduction
During the last century, farming and agricultural activities released many persistent and toxic chemical pesticides into the environment including insecticides, fungicides, nematicides, rodenticides and herbicides21. They comprise a variety of molecules possessing different properties that confer some degree of environmental persistence and mobility, as well as different toxic, carcinogenic, mutagenic and teratogenic potentials10,17,21. Such substances may also affect the endocrine systems of non-targeted organisms, including humans38.
The use of herbicides in Mexico increased strongly in the last decade, where according to recent studies more than 45% of pesticides marketed were herbicides, trazine being the most frequently used7. The use of atrazine is not regulated and is widely used in Mexican agriculture, where the application rates range from 0.1 to 4 kg/ha/year. At these application rates, the annual consumption of atrazine in Mexico has been estimated to be around 1078 tons/year with an annual consumption increase of 10%18,19.
Atrazine (2-chloro-4-ethylamino-6-isopropylamino-s-triazine) is a selective herbicide that belongs to the s-triazine family, which contains an aromatic hexameric and symmetrical ring, constituted by three carbon and three nitrogen atoms in alternate positions18. Atrazine is used to control broadleaf and grass weeds in corn, sorghum, sugar cane, coffee crops and conifer reforestation. Atrazine kills susceptible plants by binding to the quinone protein in photosystem II and inhibits photosynthetic electron transport36.
Despite its apparent biodegradability, atrazine has led to the contamination of terrestrial ecosystems and can be found and measured in ground and surface waters in many countries7,30. The removal of atrazine from the environment is an ecological responsibility, and finding a safe and economical method is both a major concern for land management agencies and a challenge to science13. Bioremediation with microbial organisms is one approach. In the last two decades, the use of ligninolytic macrofungi and their enzymes in the mycoremediation of environmental contaminants has become a promising solution40. This fungal ability to bioremediate is generally attributed to the production of extra-cellular ligninolytic enzymes such as laccase, manganese peroxidase (MnP) and lignin peroxidase (LiP), which are substrate non-specific enzymes and are able to degrade a wide range of recalcitrant compounds including several herbicides that are structurally related to lignin3-5,9,39.
The first step to establish future studies of atrazine degradation through mycoremediation in contaminated sites is finding tolerant strains to atrazine28. The selection through dose-response bioassay is considered a way to identify these strains, which could be used in subsequent studies on soil mycoremediation8,29. With this in mind, we proposed the hypothesis that tolerant strains will not show mycelial growth inhibition and reduction in their enzyme activities upon exposure to the herbicide, therefore they will obtain a high mean effective concentration (EC50). To corroborate this hypothesis, the goals of this work were to assess mycelial growth rates and ligninolytic enzyme activities under atrazine stress in eight native ligninolytic macrofungi isolated from Veracruz, Mexico.
Materials and methods
Fungal isolation and identification
Carpophores of ligninolytic macrofungi were collected in the center of Veracruz State, Mexico and registered in the herbarium XAL of the Instituto de Ecologia (INECOL A. C.) situated in Xalapa, Veracruz, Mexico (Table 1). In order to obtain the strains, 0.5-1 cm2 fragments from the carpophores were washed in ethanol (70%) for 1 min, then in sodium hypochlorite (50%) for 3 min and finally in sterile distilled water for 3 min. The washed and disinfected fragments were placed on potato-dextrose agar plates (PDA, Bioxon, USA), supplemented with chloramphenicol (20 mg/l; Sigma, st. Louis, MO, USA) in order to prevent bacterial contamination, and benomyl (3 mg/l; Biesterfeld Co., USA), which inhibited mold growth. Carpophores that gave rise to the isolates were identified according to their morphologic characteristics following taxonomic keys and specialized literature14,27,29,33,37.
Table 1 Overview of strains of ligninolytic macrofungi used
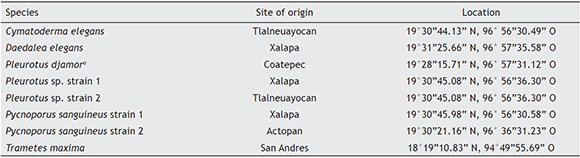
a - The strain was isolated and identified in a previous study by Salmones et al.34.
Dose-response bioassays
"Desyerbal 500™" (ANALJASA, Guadalajara, Mexico), a commercial formulation of atrazine with a concentration of 500 g/l of active ingredient was used. Potato dextrose agar was sterilized at 121 °C for 15 min. Atrazine was added to sterile PDA when it was cooled at 40 ± 2 °C, and then the PDA with atrazine was emptied out to Petri dishes (90 mm Ø). The final concentrations of atrazine in PDA plates were 0 (control), 468, 937, 1875 and 3750 mg/l. The highest concentration of atrazine corresponds to the field application rate (1.5 l of "Desyerbal 500™" dissolved in 200 l of water). One disc (6 mm Ø) from a 7-day-old culture of each strain was inoculated into the center of PDA plates containing different concentrations of atrazine. All plates were incubated in the dark at 25 °C and 75 ± 5% of relative humidity. Four replicates of each atrazine concentrations and a control (without atrazine) were used.
The colonies were photographed daily from the rear of the plates with a Kodak digital camera (Kodak, New York, USA) at 20 cm between the camera and the plates. The area of mycelium growth was calculated using digital photography and the ImageJ software31 based on the scale 1 cm2 = 358.6 pixels. The dose-response bioassays finished when the control mycelia filled the Petri dishes or up to 15 days of growth3.
Growth characteristics
From the colony areas measured, specific growth charac-teristics were calculated: the percentage of mycelial growth inhibition, as the percentage of Inhibition = [(C-T)/C]*100, where C and T are the mycelial area inside the control plates and the mycelial area in the treatment plates, respectively. Average Daily Growth Rate (DGR), was defined as DGR (cm2/d) = [Σ (R1-R0) / (T1-T0)] / n, where n is the number of evaluations, R1 and R0 were the colony areas (in cm2) at time T1 and T0 (in days), respectively2,3. The effective concentrations inhibiting mycelial growth rate by 50% (EC50) were calculated by Probit analysis12.
Enzymatic and protein assay
Enzyme activities were determined spectrophotometrically using a Lambda 3A UV/VIS spectrophotometer (Perkin-Elmer, Massachusetts, USA) at room temperature (25-27 °C). The enzyme extracts were performed removing five mycelial discs (6 mm Ø) from fungal colonies established in the dose-response bioassays. Mycelial discs were placed in test tubes containing 5 ml of sodium acetate buffer 100 mM (pH 4.5). Test tubes were shaken for 1 min at 2200 rpm in a minishaker (IKA-MS2 Gemini, Apeldoorn, Netherland), and then were incubated 15 min at 30 °C. Finally, the test tubes were cen-trifuged for 10 min at 7500 rpm and the supernatant was taken to the determination of the enzymatic activity and total protein content.
Laccase activity was determined by measuring the oxidation of ABTS [2, 2′-azino-bis (3-ethylbenzthiazoline-6-sulfonic acid)] in a reaction mixture (1 ml) containing 100 µl of ABTS (0.5 mM, Sigma, St. Louis, MO, USA), 800 µl of acetate buffer (100 mM, pH 4.5) and 100 µl of enzyme extract. Absorbance changes in the presence of the enzyme were monitored for 5 min at 420 nm (ε = 3.6 × 104/M/cm). One unit of laccase activity was defined as the amount of enzyme required to oxidize 1 µmol ABTS per minute per mg of protein under the assay conditions24.
Manganese peroxidase activity was determined at 610 nm (ε = 4460/M/cm) using the methodology described by Kuwahara et al.22. The reaction mixture contained: the enzyme extract (700 µl), phenol red 0.2% (50 µl), sodium lactate 0.5 mM (50 µl), egg albumin 0.1% (50 µl), manganese sulfate 2 mM (50 µl) and hydrogen peroxide 2 mM (50 µl). The reaction was carried out in sodium succinate buffer 20 mM at pH 4.5 (50 µl). The reactions occurred for 5 min and were stopped by the addition of NaOH 2N (50 µl). One enzyme unit was defined as 1 µmol of product formed per minute per mg of protein under the assay conditions.
The protein content of the extracts was estimated according to the Bradford method. Bovine serum albumin was used as a standard at known concentrations (0, 0.0062, 0.0125, 0.025, 0.05, 0.1 and 0.2 mg/l). The standard curve was: y = 0.1615c - 0.0125 (y = OD595, c = protein concentration in mg/l, R2 = 0.999).
Statistical analysis
Assumptions of normality and homoscedasticity were tested on all data. The results of daily growth rate, inhibition and enzymatic activities were analyzed by one way analysis of variance and the least significant difference (LSD) multiple comparison test, using the GraphPad software15. The EC50 of each strain was obtained by Probit analysis using SAS 8.1 35.
Results
Mycelial growth inhibition
In all strains the percentage of mycelial growth inhibition increased with an increase in atrazine concentration in the culture medium. However, none of the strains were inhibited at 100% (Fig. 1). The atrazine concentration of 468 mg/l did not inhibit mycelial growth of Daedalea elegans (DF = 3/12, F = 975.3, p < 0.0001) and Pleurotus sp. strain 1 (DF = 3/12, F = 148.4, p < 0.0001). Only Pleurotus sp. strain 1 (DF = 3/12, F = 148.4, p < 0.0001) did not show inhibition in its mycelial growth at the concentration of 937 mg/l of atrazine. With 1875 mg/l of atrazine: Pleurotus sp. strain 1, P. sanguineus strain 2 and T. maxima showed the lowest inhibition percentage (51.8, 53.0 and 55.3%, respectively). Pleurotus sp. strain 2 and C. elegans achieved the lowest mycelial growth inhibition (DF = 7/24, F = 17.1, p < 0.0001) at the highest atrazine concentration (3750 mg/l) with 66.7 and 68.6%, respectively.

Figure 1 Effect of atrazine concentrations on mycelial growth inhibition (%) of eight ligninolytic macrofungi cultivated on potato-dextrose agar. Means with the same letter are not significantly different from each other (n = 4, mean ± SEM, LDS test, a < 0.05).
Daily growth rate
For most isolates, the daily growth rate (DGR) decreased when the concentration of atrazine was increased in the culture medium, except for Cymatoderma elegans (0.6 cm2 /day; DF = 4/15, F = 39.4, p < 0.0001) and Pleurotus sp. strain 1 (1.6 cm2/day; DF = 4/15, F = 56.1, p < 0.0001). These strains showed a higher DGR when compared with the control in the presence of 468 mg/l of atrazine (Fig. 2). Only T. maxima maintained the same DGR in the control (6.9 cm2/day; DF = 4/15, F = 458.4, p < 0.0001) and under 468 mg/l of atrazine (6.8 cm2/day). Among the tested isolates, significant differences were found in DGR in the control treatments (without atrazine); T. maxima (DF = 7/24, F = 377.3, p < 0.0001) achieved the highest DGR with 6.9 cm2/day, and Pleurotus sp. strain 1 and C. elegans showed the lowest DGR with 0.4 and 0.3 cm2/day, respectively.
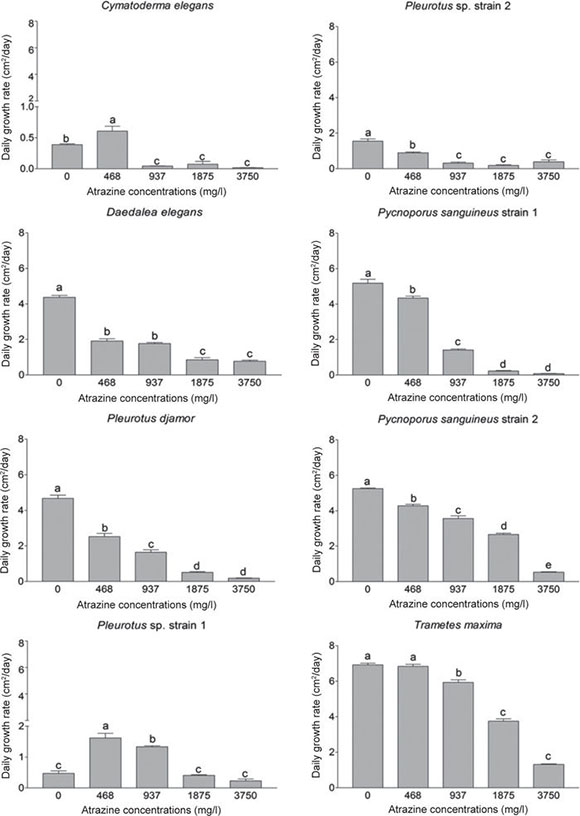
Figure 2 Daily growth rate (cm2/day) of ligninolytic macrofungi in potato-dextrose agar contaminated with different concentrations of atrazine. Means with the same letter are not significantly different from each other (n = 4, mean ± SEM, LDS test, a < 0.05).
Median effective concentration (EC50)
The EC50 ranged from 447.3 to 2281.0 mg/l of atrazine in the studied strains (Table 2). In accordance with the overlapping of confidence intervals, Pleurotus sp. strain 1 (p < 0.0001) showed the highest EC50 with 2281.0 mg/l of atrazine, therefore it was the most tolerant fungus of all studied strains. Trametes maxima and P. sanguineus achieved a CE50 of 1532.0 and 1311.0 mg/l of atrazine, both strains showed considerable tolerance to atrazine. On the other hand, the strains with the lowest EC50 (p < 0.0001) were P. sanguineus strain 1, Pleurotus sp. strain 2 and P. djamor with 706.1, 624.1 and 447.3 mg/l of atrazine, respectively. These isolates were the most susceptible to the herbicide.
Table 2 Median effective concentration (EC50) of atrazine in eight ligninolytic macrofungi
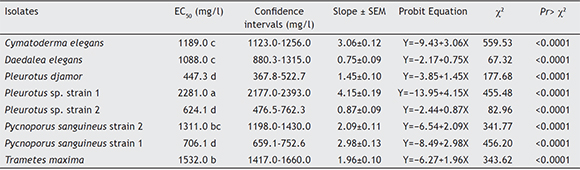
In accordance with the overlap of the confidence intervals the means with the same letter are not significantly different from each other (a = 0.05).
Effect of atrazine concentrations on laccase and MnP activities
Laccase activity varied owing to the effect of atrazine concentration in all studied strains. Pycnoporus sanguineus strain 2 showed higher (DF = 7/16, F = 21.9, p < 0.0001) laccase activity in the control (62.4 U mg/protein) compared with the remaining isolates. However, this activity increased significantly (DF = 4/10, F = 14.0, p < 0.0004) from 62.4 to 191.9 U mg/protein (205.8%) by the effect of atrazine (937 mg/l) in the culture medium (Fig. 3). Also, when T. maxima, D. elegans and C. elegans were grown under 937 mg/l of atrazine, their laccase activities increased significantly (p < 0.0001) by 311% (29.3 to 120.6 U mg/protein), 161% (31.9 to 83.5 U mg/protein) and 153% (8.2 to 20.8 U mg/protein), respectively. In the remaining strains: Pleurotus djamor, Pleurotus sp. strain 2, P. sanguineus strain 1 and Pleurotus sp. strain 1 showed the highest (p < 0.0001) laccase activity at the highest concentration of atrazine, with increases of 726% (1.5 to 12.8 U mg/protein), 238% (2.8 to 9.5 U mg/protein), 3886% (0.15 to 6.1 U mg/protein) and 51% (1.4 to 2.1 U mg/protein), respectively.
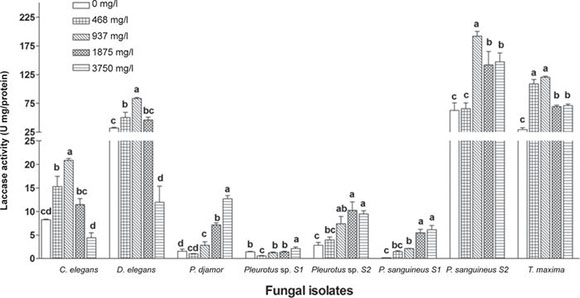
Figure 3 Laccase activity of ligninolytic macrofungi under atrazine concentrations. Means with the same letter in each isolate are not significantly different from each other (n = 4, mean ± SEM, LDS test, a < 0.05). The terms S1 and S2 correspond to strain 1 and strain 2, respectively.
Only Pleurotus sp. strain 2, C. elegans and P. sanguineus strain 1 showed MnP activity (Fig. 4). In Pleurotus sp. strain 2, MnP activity was significantly stimulated by the presence of atrazine in the culture medium, MnP activity was not detected in the control (without atrazine), however, a significantly higher activity (DF = 4/14, F = 41.4, p < 0.0001) was found at 937 mg/l of atrazine with 4.3 U mg/protein. Cymatoderma elegans achieved high MnP activity due to the effect of atrazine concentrations and showed significant increases, above 130% (0.6 to 1.4 U mg/protein) in the four tested concentrations; meanwhile the control obtained the lowest MnP activity (0.6 U mg/protein). Pycnoporus sanguineus strain 1 showed less MnP activity than Pleurotus sp. strain 2 and C. elegans; however, the atrazine concentrations had negative effects on MnP activity by Pycnoporus sanguineus strain 1.

Figure 4 MnP activity of ligninolytic macrofungi under atrazine concentrations. Means with the same letter in each isolate are not significantly different from each other (n = 4, mean ± SEM, LDS test, a = 0.05). The terms S1 and S2 correspond to strain 1 and strain 2, respectively.
Discussion
Mycelial growth was inhibited by the atrazine concentrations at different rates in the studied strains; however, none reached 100% inhibition under all tested doses. The lowest dose of atrazine (468 mg/l) did not cause inhibition in D. elegans and Pleurotus sp. strain 1. The concentration of 937 mg/l of atrazine did not inhibit the growth of Pleurotus sp. strain 1. These results suggest that moderate concentrations of atrazine are not toxic to these strains. In a previous study Nwachukwu and Osuji28 showed 8, 11 and 14% inhibition in the mycelial growth of Lentinus subnudus in soil extract agar supplemented with 5, 10 and 20 mg/l of atrazine, respectively. These inhibition percentages were low, which could be due to the low atrazine concentrations used in Nwachukwu and Osuji´s study. In another study, Ribas et al.32 assessed the susceptibility of Agaricus subrufescens (basidiomycete) to atrazine and found high inhibition rates of mycelial growth in PDA supplemented with 10 (18.8%), 20 (31.3%), 30 (43.8%), 40 (50%) and 50 (48%) mg/l of atrazine. The concentrations used by Ribas et al.32 and Nwachukwu and Osuji28 were lower compared with the concentrations used in the present study (from 468 to 3750 mg/l), which corresponded to field application rates.
The study of growth rates of ligninolytic macrofungi is important in order to understand their capacity for colonization and their potential to settle on a particular substrate. However, several pollutants have a negative effect on the growth rate of ligninolytic macrofungi27. In previous works, Casale and Hart5 reported a 69% reduction in the rate of growth for ascomycete Sclerotinia sclerotiorum when exposed to 500 mg/l atrazine in the culture medium. Furthermore, the daily growth rate of filamentous microfungi was negatively affected by exposure to atrazine; some susceptible genera are Trichoderma, Penicillium and Aspergillus7.
On the other hand, the daily growth rate of Pleurotus sp. strain 1 and C. elegans was stimulated by low concentrations of atrazine (468 mg/l). This physiological response has been previously reported in Microsporum fulvum (hyphomycete)1, when cultivated in Sabouraud agar supplemented with atrazine concentrations ranging from 5 to 50 mg/l. However, the increase in the daily growth rate by atrazine in basidiomycetes has not been previously reported. In an earlier study, Maciel et al.23 reported an increase in the mycelial growth rate of Trametes sp. in malt extract agar supplemented with 0.1 mM (24.1 mg/l) of picloram (an herbicide). In this case, the increase in mycelial growth rate in fungi by herbicides could be related to the auxin-like action16,23.
The parameter used to determine fungi sensitivity or tolerance to atrazine was the median (EC50) effective concentration42. Based on this concept, Pleurotus sp. strain 1 was the most tolerant fungus to atrazine, presenting the highest EC50. There is little information about the EC50 of atrazine in ligninolytic macrofungi; therefore it is difficult to compare our results to other references. Only Nwachukwu and Osuji28 calculated the EC50 of atrazine in L. subnudus (EC50 at 15 °C = 20 mg/l and EC50 at 25 °C = 11.9 mg/l), which was smaller than that achieved in this work in all tested strains (from 706.1 to 2281.0 mg/l). The available information only mentions that atrazine exposure in fungal organisms results in the growth inhibition of some ligninolytic macro- and filamentous micro-fungi6.
In addition, xenobiotics affect the synthesis and activity of fungal enzymes11,25. Laccase is one of the main enzymes produced by ligninolytic macrofungi, which plays an important role in fungal morphogenesis, lignin degradation and fungal plant-pathogen/host interaction3. Under the culture conditions used in this study all fungal strains produced laccase as the main ligninolytic enzyme. However, there was an increase in laccase activity for all tested strains due to the effect of the atrazine concentrations. In earlier works, Mougin et al.25 added several xenobiotics (among them atrazine), in a Trametes versicolor culture. Atrazine increased the laccase activity of T. versicolor by 369% (0.61 U/ml) and 443% (0.87 U/ml) when compared to the control (0.13 Ug/g). Bastos and Magan5 found high laccase activity (19.9 U/g soil) in T. versicolor growing in soil contaminated with atrazine at 0.9 and 2 µg/g, in comparison to soil without atrazine (9.0 U/g soil).
With regard to MnP activity, only C. elegans and P. sanguineus strain 1 showed MnP activity under the culture conditions. Atrazine concentrations in PDA increased MnP activity in C. elegans; however, in P. sanguineus strain 1 cultures resulted in a reduction. However, a particular physiological response was observed for Pleurotus sp. strain 2, which did not show MnP activity in the control treatment (without atrazine), but produced MnP activity in the presence of atrazine. The increase in laccase and MnP activities could be linked to oxidative stress on ligninolytic fungi generated by atrazine. This response is considered a defense mechanism for ligninolytic fungi26,40-42. Nevertheless, this response is not a general rule. Recently, Maciel et al.20 found a reduction in laccase activity of Ganoderma lucidum due to the effect of picloram. The latter is unusual because, in general, the addition of xenobiotics to the culture of ligninolytic fungi enhances laccase activity5,23,25. However, we must not overlook that individual species could respond dissimilarly to different pesticides.
Under natural conditions, fungi are usually not limited in growth by low atrazine concentrations in the environment30. However, in locations with an extremely high atrazine content an obvious effect on fungal growth has been observed. It should be noted that the impact of atrazine on fungal physiology is not only limited to inhibition or alteration of mycelial growth. In particular, the activities of extracellular enzymes, such as laccase and MnP, were enhanced with atrazine concentrations. For this reason, more research in the field of fungal ecology for atrazine-polluted habitats could prove helpful.
Based on the results, we conclude that Pleurotus sp. strain 1 was the most tolerant fungus to atrazine, as it did not show mycelial growth inhibition due to atrazine at either 468 or 937 mg/l concentrations, thus achieving the highest EC50; however, it did not show high laccase activity and did not enhance to a great extent its enzyme activity by atrazine stress. Nevertheless, there were other fungal isolates such as D. elegans, P. sanguineus strain 2 and T. maxima, which obtained a moderate EC50 and high enzyme activity, principally laccase; therefore, we reject the proposed hypothesis. On the other hand, the most susceptible fungus was Pleurotus djamor. Atrazine stress increased laccase activity for all the strains. The increase in MnP activity caused by the effect of atrazine occurred to a lesser extent than increases in laccase activity. This study demonstrated, for the first time, the ability of C. elegans to produce laccase and MnP, including its ability to increase enzyme activity by atrazine stress. Overall, we propose the use of native macrofungi such as D. elegans, P. sanguineus strain 2 and T. maxima for future studies in atrazine mycodegradation.
Ethical disclosures
Protection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that no patient data appear in this article.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.
Funding
The publication of this study was supported by research grant CB 169124 from the National Council of Science and Technology (CONACyT) of Mexico.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Acknowledgements
The first author thanks Dr. Gastón Guzmán and Biol. Florencia Ramiréz Guillen for their valuable assistance in the identification of specimens.
1. Alvarez DP, Luque AG, Gamberale ME. Resistencia de cepas de Microsporum fulvum a herbicidas preemergentes. Cienc Suelo. 1990;8: 25-9. [ Links ]
2. Baldrian P, Gabriel J. Intraspecific variability in growth response to cadmium of the wood-rotting fungus Piptoporus betulinus. Mycologia. 2002;94: 428-36. [ Links ]
3. Baldrian P. Fungal laccases - occurrence and properties. FEMS Microbiol Rev. 2006;30: 215-42. [ Links ]
4. Baldrian P. Increase of laccase activity during interspecific interactions of white-rot fungi. FEMS Microbiol Ecol. 2004;50: 245-53. [ Links ]
5. Bastos A, Magan N. Trametes versicolor: potential for atrazine bioremediation in calcareous clay soil, under low water availability conditions. Inter Biodeter Biodegr. 2009;63: 389-94. [ Links ]
6. Casale W, Hart P. Influence of four herbicides on carpogenic germination and apothecium development of Sclerotinia sclerotiorum. Phytopathology. 1986;76: 980-4. [ Links ]
7. Cejudo -Espinosa E, Ramos-Valdivia AC, Esparza-García F, Moreno-Casasola P, Rodriguez-Vazquez R. Short-term accumulation of atrazine by three plants from a wetland model system. Arch Environ Contam Toxicol. 2009;56: 201-8. [ Links ]
8. Colla L , Lazzarotto A, Lima M, Bertolin T, Vieira A. Isolation and screening of fungi to bioremediation from triazine herbicide contaminated soil. Ciênc Agrotec. 2008;32: 809-13. [ Links ]
9. Da Silva C, Oliveira A, Marques SC, Bracht A, Peralta RM. Effect of the herbicides bentazon and diuron on the production of ligninolytic enzyme by Ganoderma lucidum. Int Biodeter Biodegr. 2010;64: 156-61. [ Links ]
10. Silva Mdos S, Cocenza DS, Grillo R, de Melo NF, Tonello PS, de Oliveira LC, Cassimiro DL, Rosa AH, Fraceto LF. Paraquatloaded alginate/chitosan nanoparticles: preparation, characterization and soil sorption studies. J Hazard Mater. 2011; 190: 366-74. [ Links ]
11. Filazzola M, Sannino F, Rao M, Gianfreda L. Effects of various pollutants and soil-like constituents on laccase from Cerrena unicolor. J Environ Qual. 1999;28: 1929-38. [ Links ]
12. Finney DJ . Probit analysis. Third Edition. Cambridge, Cambridge University Press; 1971. [ Links ]
13. Fragoeiro S. Use of fungi in bioremediation of pesticides. Ph. D. dissertation. United Kingdom, Cranfield University; 2005. [ Links ]
14. Gilbertson RL, Ryvarden L. North American polypores. Volume 2. Megasporoporia-Wrigtoporia. Oslo, Fungiflora; 1987. p. 434-885. [ Links ]
15. GraphPad InStat. GraphPad Software. La Joya, California, USA: Software in CD; 2000. [ Links ]
16. Guo X, Zou X, Sun M. Effects of phytohormones on mycelial growth and exopolysaccharide biosynthesis of medicinal mushroom Phellinus [corrected] linteus. Bioprocess Biosyst Eng. 2009;32: 701-7. [ Links ]
17. Hai FI, Modin O, Yamamoto K, Fukushi K, Nakajiam F. Pesticide removal by a mixed culture of bacteria and white-rot fungi. J Taiwan Inst Chem E. 2012;43: 459-62. [ Links ]
18. Hansen A, Treviño Q, Márquez H, Villada M, González L, Guillén R, Hernández A. Atrazina: un herbicida polémico. Rev Inter Contam Ambient. 2013;29: 65-84. [ Links ]
19. Islas M, Villagómez J, Madariaga A, Castro J, González C, Acevedo A. Bioremediation perspectives using autochthonous species of Trichoderma sp. for degradation of atrazine in agricultural soil from the Tulancingo Valley, Hidalgo, Mexico. Trop Subtrop Agroecosyst. 2013;16: 265-76. [ Links ]
20. Joshi N, Gupta D. Soil mycofloral responses following the exposure to 2, 4-D. J Environ Biol. 2008;29: 211-4. [ Links ]
21. Kalia A, Gosal S. Effect of pesticide application on soil microorganism. Arch Agron Soil Sci. 2011;57: 569-96. [ Links ]
22. Kuwahara K, Glenn J, Morgan M, Gold M. Separation and characterization of two extracellular H2O2-dependent oxidases from ligninolytic cultures of Phanerochaete chrysosporium. FEBS Lett. 1984;169: 247-50. [ Links ]
23. Maciel G, Dorneles F, Sá-Nakanichi A, Haminiuk C, Castoli R, Comar J, Bracht A, Peralta M. Response of Ganoderma lucidum and Trametes sp. to the herbicide picloram: Tolerance, antioxidants and production of ligninolytic enzymes. Pestic Biochem Physiol. 2013;105: 84-92. [ Links ]
24. More SS, P S R, K P, M S, Malini S, S M V. Isolation, purification and characterization of fungal laccase from Pleurotus sp. Enzyme Res 2011;2011: 248735. [ Links ]
25. Mougin C, Kollmann A, Jolivalt C. Enhanced production of laccase in the fungus Trametes versicolor by the addition of xenobiotics. Biotechnol Lett. 2012;24: 139-42. [ Links ]
26. Nikolaou E, Agrafioti I, Stumpf M, Quinn IS. Alistair. Phylogenetic diversity of stress signaling pathways in fungi. BMC Evol Biol. 2009;9: 1-18. [ Links ]
27. Núñez M, Ryvarden L. East Asian polypore 2. Polyporaceae s. lato. Synopsis Fungorum. 2001;14: 170-522. [ Links ]
28. Nwachukwu EO, Osuji JO. Bioremedial degradation of some herbicides by indigenous white rot fungus, Lentinus subnudus. J Plant Sci. 2007;2: 619-24. [ Links ]
29. Parker R. Cymatoderma elegans var. Llamellatum. Australas Mycologist. 1999;18: 18-20. [ Links ]
30. Przybulewska K, Sienicka K. Decomposition of atrazine by microorganisms isolated from long-term herbicide experiment soil. Ecol Chem Eng S. 2008;15: 491-9. [ Links ]
31. Rasband W. ImageJ version 1.33. National Institutes of Health, Bethesda, MD, U.S.A. 2005 [accessed 10 Jan 2012]. Available from: http: //rsb.info.nih.gov/ij/. [ Links ]
32. Ribas LC, de Mendonça MM, Camelini CM, Soares CH. Use of spent mushroom substrates from Agaricus subrufescens (syn. A. blazei, A. brasiliensis) and Lentinula edodes productions in the enrichment of a soil-based potting media for lettuce (Lactuca sativa) cultivation: Growth promotion and soil bioremediation. Bioresour Technol. 2009;100: 4750-7. [ Links ]
33. Ryvarden L. Genera of polypores: nomenclature and taxonomy. Synopsis Fungorum. 1991;5: 1-363. [ Links ]
34. Salmones D, Gaitán-Hernández R, Pérez R, Guzmán G. Estudios sobre el género Pleurotus. VIII. Interacción entre crecimiento micelial y productividad. Rev Iberoam Micol. 1997;14: 173-6. [ Links ]
35. SAS. The SAS system for Windows 8e. Cary, NC, USA: Software in CD; 2000. [ Links ]
36. Sene L, Converti A, Ribeiro S, Garcia S. New aspect on atrazine biodegradation. Braz Arch Biol Technol. 2010;53: 487-96. [ Links ]
37. Welden A. The genus Cymatoderma (Thelephoraceae) in the Americas. Mycologia 1960;52: 856-76. [ Links ]
38. Fan W, Yanase T, Morinaga H , Gondo S, Okabe T, Nomura M, Komatsu T, Morohashi K, Hayes TB, Takayanagi R, Nawata H. Atrazine-induced aromatase expression is SF-1 dependent: implications for endocrine disruption in wildlife and reproductive cancers in humans. Environ Health Perspect. 2007;115: 720.7. [ Links ]
39. Xiao P, Mori T, Kondo R. Biotransformation of the organochlorine pesticide trans-chlordane by wood-rot fungi. N Biotechnol. 2011;29: 107-15. [ Links ]
40. Zhang Y, Meng D, Wang Z, Guo H, Wang Y. Oxidative stress response in two representative bacteria exposed to atrazine. FEMS Microbiol Lett. 2012;334: 95-101. [ Links ]
41. Zhao Y, Li J, Chen Y, Hang H. Response to oxidative stress of Coriolus versicolor induced by exogenous hydrogen peroxide and paraquat. Ann Microbiol. 2009;59: 221-7. [ Links ]
42. Zhu G, Huang F, Feng L, Qin B, Yang Y, Chen Y, Lu X. Sensitivities of Phytophthora infestans to Metalaxyl, Cymoxanil, and Dimethomorph. Agric Sci China. 2008;7: 831-40. [ Links ]














