Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista argentina de microbiología
Print version ISSN 0325-7541On-line version ISSN 1851-7617
Rev. argent. microbiol. vol.50 no.2 Ciudad Autónoma de Buenos Aires June 2018
http://dx.doi.org/10.1016/j.ram.2017.04.012
ORIGINAL ARTICLE
https://doi.org/10.1016/j.ram.2017.04.012
Validation of a liquid chromatography/tandem mass spectrometry method for the detection of aflatoxin B1 residues in broiler liver
Validación de una técnica para detectar aflatoxina B1 en hígado de pollo por cromatografía líquida de alta performance acoplada a espectrómetro de masas en tándem
Alejandra P. Magnolia,b, María L. González Pereyraa,b, María P. Mongeb,c, Lilia R. Cavaglieria'b,*, Stella M. Chiacchierab,c
a Departamento de Microbiología e Inmunología, Facultad de Ciencias Exactas, Físico, Químicas y Naturales, Universidad Nacional de Río Cuarto, Río Cuarto, Córdoba, Argentina
b Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Buenos Aires, Argentina
c Departamento de Química, Facultad de Ciencias Exactas, Físico, Químicas y Naturales, Universidad Nacional de Río Cuarto, Río Cuarto, Córdoba, Argentina
Received 23 November 2016; accepted 26 April 2017
Available online 14 November 2017
*Corresponding author.
E-mail address: lcavaglieri@exa.unrc.edu.ar (L.R. Cavaglieri).
0325-7541/© 2017 Asociación Argentina de Microbiología. Published by Elsevier Espana, S.L.U. This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
ABSTRACT
Aflatoxin B1 is a carcinogenic and mutagenic mycotoxin produced mainly by Aspergillus flavus and Aspergillus parasiticus. It is the predominant mycotoxin found in raw materials used for the manufacture of broiler feeds. The aim of the present study was to develop a new and optimized method for the detection and quantification of aflatoxin B1 (AFB1) residues in broiler liver using solid phase extraction (SPE) clean-up and liquid chromatography-electrospray ionization/tandem mass spectrometry (LC-ESI-MS/MS) detection. The method was validated for linearity, accuracy, precision, limit of detection (LOD) and limit of quantification (LOQ). The validation parameters indicated satisfactory linearity (r2 >0.99), accuracy and precision (4.57% intra-day RSD; 14.65% inter-day RSD) a very high recovery (99 ±13%) and high sensitivity achieved for AFB1 in animal samples (LOD = 0.017 and LOQ= 0.050 ng/g). The method was effective for the detection and quantification of AFB1 residues in broiler liver and could also be potentially used for detecting AFB1 in other edible animal tissues after natural or experimental AFB1 exposure with high sensitivity and precision.
KEYWORDS
Aflatoxin B1; Liver; Broilers; LC-MS/MS; Method development
RESUMEN
La aflatoxina B1 (AFB1) es una micotoxina carcinogénica y mutagénica producida principalmente por Aspergillus flavus y Aspergillus parasiticus. Es la principal toxina que contamina las materias primas utilizadas para la elaboración de alimentos balanceados destinados a la alimentación de pollos parrilleros. El objetivo de este trabajo fue desarrollar un método nuevo y optimizado para detectar y cuantificar bajos niveles de AFB1 en hígado de pollo, usando limpieza por extracción en fase sólida (SPE) y cromatografía líquida acoplada a detección por espectrometría de masa en tándem con ionización por electrospray (LC-ESI-MS/MS). Se validaron la linealidad, la exactitud, la precisión, el límite de detección (LOD) y el límite de cuantificación (LOQ). El método resultó tener linealidad (r2>0,99), exactitud y precisión muy satisfactorias (4,57% RSD intradía; 14,65% RSD interdía), un alto porcentaje de recupero (99 ± 13%) y la sensibilidad más alta lograda para la detección de AFB1 en muestras de origen animal (LOQ=0.050 ng/g y LOD = 0.017). El método fue muy efectivo para detectar y cuantificar bajos niveles de AFB1 en hígados de pollos parrilleros. Este método podría potencialmente utilizarse para la detección de esta toxina en otros tejidos y subproductos de origen animal luego de su exposición a AFB1 con una mayor sensibilidad y precisión.
PALABRAS CLAVE
Aflatoxina B1; Higado; Pollos parrilleros; LC-MS/MS; Desarrollo de método
Introduction
Mycotoxins are toxic secondary metabolites produced by fungi. They are unavoidable contaminants in foods and feeds, exerting harmful effects upon animal and human health39. The most important mycotoxins in naturally contaminated foods and feeds are aflatoxins (AFs), ochratoxins, zearalenone, T-2 toxin, deoxynivalenol and fumonisins7,32. Aflatoxin contaminated feeds are a severe threat to both livestock productivity and human health and cause significant worldwide economic losses every year3. Aflatoxins produced by toxigenic strains of Aspergillus flavus and Aspergillus parasiticus are the most hazardous naturally occurring toxins for both human and animals. Among the four major AFs - B1, B 2, G1 and G2 - aflatoxin B1 (AFB1 is the most toxic and carcinogenic. The manifestation of chronic or acute toxicosis as well as carcinogenicity depends on the dose, duration of exposure, and rate of metabolism to less toxic metabolites3. Aflatoxin B1 was classified as a Group 1 carcinogen (carcinogenic to humans) by the International Agency for Research on Cancer19. The toxicity of AFB1 in birds has been widely investigated, being the liver target organ for the toxin. Biochemical, hematological, immunological, and pathological effects of AFB1 have also been well-described38,23,25,27. The liver is also the main organ where AFB1 is stored, metabolized and/or conjugated to nucleic acids and proteins. Aflatoxin B1 is activated by cytochrome P450 enzymes (CYP), including CYP1A2, CYP3A4 and CYP2A6 and converted to epoxides (AFB1-8,9-exo-epoxide and AFB1-8,9-endo-epoxide), aflatoxin M1 (AFM1), aflatoxin P1 (AFP1), aflatoxin Q1 (AFQ1), or its reduced form aflatoxicol (AFL)38. Recent studies have shown that CYP2A6 and, to a lesser extent, CYP1A1 are the enzymes responsible for bio-activation of AFB1 into epoxide forms in chicken and quail liver8. Residues of AFB1 and some of its metabolites have been detected in eggs, liver, gizzard and kidney, as a consequence of the direct intake of contaminated feed28,2,29,26.
The poultry industry has evolved over the past 30 years to be a major source of animal protein in the human diet. In Argentina and other Latin American countries, the poultry industry is one of the most important animal production systems and has acquired great economic significance, mainly in the meat production sector21. Aflatoxin contamination in broiler feeds represents a potential risk for broilers' health and cause economic losses associated to acute and chronic aflatoxicosis. The presence of AFB1 residues in edible broiler tissues leads to the toxin carryover in the food chain. Since AFB1 may remain unchanged in the liver even when exposure levels are relatively low, the development of simple and precise methods for the quantification of mycotoxins in different animal products and edible tissues is mandatory. The methods for AFs detection in the liver described to date use less sensitive techniques or are multi-toxin methods that are not optimized for AFB1. The aim of the present study was to develop a new and optimized method for the detection and quantification of AFB1 residues in broiler liver using SPE clean-up and LC-ESI-MS/MS detection.
Materials and methods
Chemicals and materials
Aflatoxin B1 standard was purchased from Romer Labs® (Tulln, Austria). HPLC-grade organic solvents such as acetonitrile and methanol were purchased from Sintorgan (Buenos Aires, Argentina). HPLC-grade water was obtained from a Labconco WaterPro Mobile purification system (model 90901-01). Clean-up OASIS®, HLB, 6 cc (200mg) SPE cartridges were purchased from Waters Corporation (Milford, MA, USA). Pro-analysis grade solvents such as dichloromethane, methanol, acetonitrile and hexane were purchased from Cicarelli (Santa Fe, Argentina).
Samples
Aflatoxin-contaminated liver samples (n = 20) were collected from broilers (Gallus gallus domesticus, purchased from Cobb) fed a 50ng/g AFB1 contaminated corn-soybean meal standard basal diet, in a previous in vivo experiment23 performed in accordance with international sanitary and ethical guidelines (the protocol was approved by the Committee of Ethics and Biosecurity of the Universidad Nacional de Río Cuarto). In that study, one-day-old male chicks (Cobb) were obtained from a commercial hatchery and placed under continuous fluorescent lighting with feed and water available ad libitum until they were 18day old. On day 18, a total of 160 birds (4 replicates/treatment, with 5 chicks/replicate) were individually weighed and randomly selected. Birds were fed the dietary treatments for 28 days (day 18-46). During the experimental period, broilers received the diet corresponding to each treatment. A grower diet was given from day 1 to 35, and a finisher diet was provided from day 36 until the end of the experiment. A standard corn-soybean meal diet (basal diet) meeting NRC (1994) requirements was fed from day 1 until the end of the experiment. The basal diet contained 2 µg of AFB1 /kg of diet, as determined by the method described by Trucksess et al.37 Broilers were monitored daily for signs of morbidity and mortality. At the end of the assay, productivity parameters (i.e., body weight gain, feed consumption and feed conversion ratio), biochemical parameters, macroscopic and microscopic changes in the liver of broilers were evaluated. Non-contaminated liver samples (n = 5) used for the recovery assay were obtained from broilers fed an AFB1 -free diet.
Spiking and recovery assay
Five blank liver samples (50g each) were spiked with 50µl of a 1 mg/ml AFB1 solution to obtain a concentration of 1 ng AFB1 per g of tissue. These samples were processed and analyzed in the same way as naturally contaminated samples and the recovery percentage was calculated.
Extraction
Aflatoxin B1 from liver tissue was extracted according to the method described by Tavčar-Kalcher et al.35 with some modifications. Five ml of citric acid (20%) and 10g of diatomaceous earth were thoroughly mixed with 50 g of ground liver tissue. The toxin was extracted with 100 ml dichloromethane by shaking for 30 min at room temperature. The organic phase was filtered through Whatman N°4 filter paper (Whatman International Ltd., Maidstone, UK). Water was eliminated by adding 5 g anhydrous sodium sulfate and the extract was filtered for a second time. Twenty ml of the filtrate were evaporated to dryness at 60°C. The residue was resuspended in 20 ml acetonitrile:H2O (75:25, v/v) and extracted with 10 ml hexane for fat separation. The mixture was thoroughly mixed, centrifuged and then let to settle until two phases were formed. An aliquot of 10 ml of the aqueous phase (bottom) was evaporated to dryness in a rotary evaporator. For clean-up, the dried extract was resuspended in 10 ml methanol:H2O (80:20, v/v), diluted by adding 90 ml distilled water and passed through an OASIS®, HLB, 6cc (200mg) SPE cartridge (Waters Corporation, Milford, MA, USA) previously conditioned with 5 ml methanol and 5 ml water according to the methodology described by Sørensen and Elbaek30. Solid phase extractions were performed in a Vac Elut 20 position Manifold SPE extractor (Agilent Technologies Inc., Santa Clara, CA, USA). The toxin was eluted with 7ml methanol, evaporated to dryness and stored at -20°C until analysis. Before LC-MS/MS analysis, the extracts were resuspended in 500µl methanol:H2O (20:80, v/v).
LC-MS/MS instrumental conditions
Detection and quantification were performed in a Waters Alliance 2695 HPLC system (Waters Corporation, Milford, MA, USA) equipped with a Waters Alliance 2685 pump, a Waters Alliance 2695 autosampler and a Waters 2996 Photo Diode Array (PDA) detector, interphased to a Quattro Ultima Platinum Tandem quadrupole Micro Mass Spectrometer with electrospray ionization (ESI) source. Chromatographic separation was performed at 25°C in an X-BridgeTM C18, 150 x 2.1mm i.d., 3.5 µm particle size column equipped with a 2.1 x 10 mm i.d. security guard cartridge (all from Waters Corporation, Milford, MA, USA). The mobile phase of the chromatographic procedure was a gradient of aqueous
0.1% formic acid (solvent A) and 0.1% formic acid-methanol (solvent B). At the initial time, the eluent was 20% solvent B. This composition was kept over 3 min, after which a linear gradient to 100% of solvent B was performed in 12 min, and then the eluent was kept at 100% of solvent B to clean the column. The initial conditions were stabilized over 10min before the next injection.
The flow rate was 0.2ml/min and the injection volume was 50 µl. ESI-MS/MS was performed in multiple reaction monitoring (MRM) mode in positive polarity. The most abundant trace in the MRM mode was used for quantitative analysis. Nitrogen gas was used for both nebulization and desolvation heated to 150 and 200°C, respectively. The capillary voltage was 3.00kV. The nitrogen flow was adjusted to 100 and 725 l/h for cone and desolvation gases, respectively. Dwell time was set at 200 ms for all transitions. Cell exit potential was 50 V and cone voltage was 35 V. Data acquisition and processing were performed using Mass Lynx V.4.1 (Waters Corporation, Milford, MA, USA) software. The MRM transitions and experimental conditions are shown in Table 1. Aflatoxin B1 retention time and the MRM-transitions of the precursor ion [M+H]+ (m/z 313) to two product ions (m/z 241 and 285) were met to identify AFB1 in liver tissue extracts. A calibration curve was created by injecting 50 µl of different AFB1 standard solutions concentration (1-10ng/ml).
Table 1 Transitions and instrument parameter settings used for data collection in positive ion mode

Validation
The method was validated for linearity, accuracy, precision, LOD and LOQ. Linearity of the method was tested by injecting three replicates (50 pl) of four levels of AFB1 standard solutions (1-10ng/ml). The accuracy of the method was determined by a recovery assay as described above and the average content of AFB1 obtained was used to calculate the recovery percentage. Precision was determined by intra-day and inter-day repeatability experiments making three injections of each spiked liver extract per day during four days. The extracts used for inter-day injections were the same as those used in the first day, and were properly kept at -20°C in the darkness to avoid degradation of the toxin. The mean AFB1 concentrations, intra-day and inter-day relative standard deviation (RSD) values were calculated. The limit of detection (LOD) and limit of quantification (LOQ) for AFB1 were calculated based on signal-to-noise (S/N) ratios of 3:1 and 10:1, respectively, by were experimentally obtained injecting standard dilutions with the corresponding S/N ratio.
Results
The average concentration of AFB1 found in liver of broilers fed the 50ng/g AFB1 diet was 0.4 ±0.1 ng/g. Figure 1 shows the LC/ESI(+)-MS/MS MRM chromatograms evidencing the presence of AFB1 (RT 11.5min) in liver of broilers fed AFB1. The precursor ion of AFB1 obtained from ESI was [M+H]+ m/z 313. When fragmented in the collision cell, the most sensitive transition ions were m/z 241 and 285.
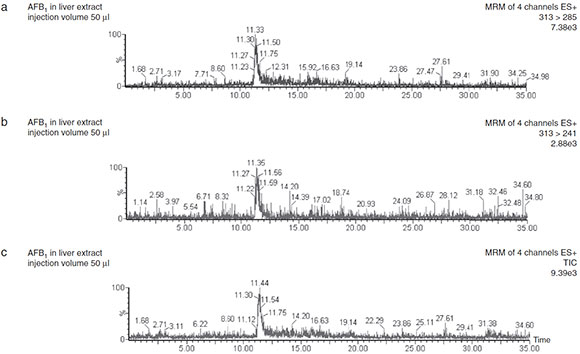
Figure 1 LC-ESI-MS/MS chromatograms obtained in positive ion mode showing the total ion current and selective AFB1 transitions in liver extracts obtained from broilers fed 50 pg/kg AFB1 diet.
The minimal amounts of AFB1 detectable and quantifiable with this method were 0.025 and 0.083 ng, respectively. The LOD and LOQ for this method were 0.017 and 0.05ng AFB1/g of sample, respectively (Fig. 2). Linear correlation (r2 >0.993) was obtained between peak area and concentration of AFB1 standard solutions ranging from 0.05 to 0.50 ng. The average AFB1 recovery was 99 ±13% (Table 2). Intra-day RSD was 4.57% and inter-day RSD was 14.65%.
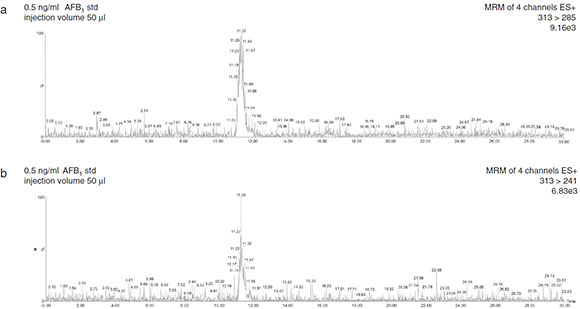
Figure 2 Total ion current and selective reaction monitoring transitions of AFB1 in solvent-only standard.
Table 2 Linearity, limit of detection (LOQ), limit of quantification (LOQ) and recovery percentage for aflatoxin B1 in LC-MS/MS

Discussion
In the present study, a sensitive, optimized and precise method for the analysis of AFB1 residues in broiler chicken liver using organic solvent extraction, SPE clean-up and LC-MS/MS detection and quantification was developed. Other methods for the detection of AFB1 in tissues and animal products developed since the 1980s involved less sensitive detection techniques such as ELISA, TLC or HPLC1,36,17,31,16,6,13,9. The use of LC-MS/MS improves detection and accurate quantification of AFB1 residues in tissue of animals fed mycotoxin-contaminated feeds. Many multi-mycotoxin LC -MS methods have been developed to detect fungal toxins and metabolites in various types of samples, mainly foods and feeds9,33,34,4,20,18. The advantages of LC-MS techniques rely in the improved detection limits, the confirmation provided by mass spectral fragmentation and the ability to filter out by mass any impurities interfering in spectrophotometric detectors. This study evaluates AFB1 residues in broiler liver, but the method could also be applied to muscle and other kinds of meat for human consumption. Chen et al.5 developed a similar method using SPE clean-up and LC-ESI-MS/MS for the detection of AFs and ochratoxin A in various animal-derived foods (swine, bovine and sheep muscle, liver, kidney and fat, chicken muscle and liver, fish muscle and skin, hen eggs and dairy milk) obtaining LODs that varied from 0.07 ng/g to 0.59 ng/g and recoveries that ranged from 68.3% to 105.7% with RSDs of less than 17.6%. Sørensen and Elbaek30 applied another method using SPE clean-up and LC-ESI-MS/MS to quantify 18 mycotoxins and metabolites in bovine milk obtaining a LOD of 20ng/l for aflatoxin M1. Zhao et al.40 developed the latest method for the quantification of 30 micotoxins in swine and poultry feeds and derived foods (meat, edible tissues and milk), obtaining LODs and LOQs of 0.1 and 0.5 ng/g AFB1, in pork meat and of 0.02 and 0.01 ng/g in swine feed and dairy milk, respectively. All these methods have the advantage of detecting multiple mycotoxins and derived metabolites in a single run. However, the conditions of method described in the present study were optimized for extraction and detection of AFB1, the most hazardous of mycotoxins due to its carcinogenic and genotoxic effects. Therefore, the LOD and LOQ obtained for meat products were improved compared to the multi-toxin methods described by other authors. The importance of AFB1 in broiler feed and tissues is higher than that of other toxins since AFB1 constitutes a major public health concern, especially in Latin American countries where there is no regulation for the toxin in matrices such as animal meat or eggs.
When applying the method, AFB1 residues were found in the liver of broilers fed a 50ng/g AFB1 diet showing clear LC/ESI(+)-MS/MS MRM chromatograms showing a well defined peak at 11.5 min. The precursor ion of AFB1 obtained from ESI was the adduct [M+H]+, m/z 313. When fragmented in the collision cell, the most sensitive transition ions were m/z 241 and 285. Multiple reaction monitoring transitions exhibited high signal intensity in positive ESI mode. The second product ion was monitored for confirmation of the compound identity. Four identification points (retention time, molecular mass of the precursor ion and two characteristic product ions) are included in the method as required for the confirmation of substances listed in group B of Annex I of Directive 96/23/EC (European Commission)11. Quantification was performed using the most abundant signal by comparing the areas under integrated peaks of the 313 >285 reaction monitoring transition in samples with those obtained with AFB1 standards.
The method was evaluated for linearity, accuracy, precision, LOD and LOQ. A high repeatability in the retention time (11.5 min) was obtained for standards as well as for sample extracts. Lower LOD and LOQ values were achieved (0.017 and 0.05 ng/g, respectively) improving sensitivity compared to other existing methods. A linear correlation (r2 >0.993) was observed between peak area and concentration of AFB1 standard injections (50 µl) in the range of 0.05-5.00 ng of AFB1 injected to the column (1-10ng/ml solutions), confirming the linearity of the method. The high mean recovery value obtained (99 ±13%) was in the range between 50% and 120%, required by commission regulation No. 401/2006/EC (European Commission)10, yielding very satisfactory accuracy. Intra-day and inter-day RSD values were 4.57% and 14.65%, respectively, indicating the high repeatability and precision of the developed method.
Many countries have established AFB1 tolerance levels in food in order to reduce toxin exposure. Feeding animals with mycotoxin-contaminated grains that are not suitable for human consumption (diluted with clean grain or feed to reduce mycotoxin levels) is a common practice in developing countries where contaminated material cannot be thrown away. The European Union has established maximum limits ranging from 5 to 20 ng/g for AFB1 in animal feeds, depending on the type of product and animal species (European Commission)12. An action level of 100 ng/g has been established by the Food and Drug Administration for aflatoxins in corn and peanut based feeds intended to mature poultry (FDA)15. In Latin America, Brazil has established a limit of 50 ng/g for aflatoxins B1, G1, B2 and G2, while MERCOSUR, the Latin American common trade market, accepts 20 ng/g of aflatoxins B1, G1, B2 and G2 (FAO)14. In Argentina, the existing regulation concerning AFs in animal products for human consumption only refers AFM1 in milk establishing tolerance levels of 0.5 µg/kg (fluid) and 5 µg/kg (powdered) (FAO)14. Hence, it is important to control AF residues in meat and other animal products even when there is still no regulation established for them since AFs, when consumed in low levels for long periods of time, have been associated to chronic aflatoxicosis and liver cancer22.
Conclusion
In conclusion, the developed method proved to be effective for the detection and quantification of AFB1 residues in broiler liver with great sensitivity. The validation parameters showed satisfactory linearity, accuracy and precision and very high recovery. Therefore, the method could be used for detecting AFB1 residues in broiler liver and eventually other edible animal tissues after natural or experimental AFB1 exposure.
Funding
This work was funded by Secretaría de Ciencia y Técnica de la Universidad Nacional de Río Cuarto (SECyT) and Consejo Nacional de Investigaciones Científicas y Técnicas, Argentina (CONICET).
Conflict of interest
Alejandra P. Magnoli, María L. González Pereyra, María P. Monge, Lilia R. Cavaglieri and Stella M. Chiacchiera declare they have no conflict of interest.
Acknowledgements
The authors are grateful to Secretaría de Ciencia y Técnica de la Universidad Nacional de Río Cuarto (SECyT) and Consejo Nacional de Investigaciones Científicas y Técnicas, Argentina (CONICET), which supported this study through grants. The authors also thank Dr. Ana M. Dalcero who supervised many of their research projects, including this one, and has recently passed away.
1. Beltrán E, Ibáñez M, Sancho JV, Cortés MA, Yusa V, Hernández A. UHPLC-MS/MS highly sensitive determination of aflatoxins, the aflatoxin metabolite M1 and ochratoxin A in baby food and milk. Food Chem. 2011;126:737-44. [ Links ]
2. Bintvihok A, Thiengnin S, Doi K, Kumagai S. Residues of aflatoxin in the liver, muscle and eggs of domestic fowls. J Vet Med Sci. 2002;64:1037-9. [ Links ]
3. CAST. Mycotoxins: risks in plant, animal, and human systems. In: Richard JL, Payne GA, (eds.). Task Force Report No. 139. Ames, IA, USA: Council of Agricultural Science and Technology; 2003. [ Links ]
4. Cavaliere C, Foglia P, Guarino C, Nazzari M, Samperi R, Lagana A. A sensitive confirmatory method for aflatoxins in maize based on liquid chromatography/electrospray ionization tandem mass spectrometry. Rapid Commun Mass Spectrom. 2007;21:550-6. [ Links ]
5. Chen D, Ca X, Tac Y,Wu Q, Pan Y, Huang L, Wang X, Wang Y, Peng D, Liu Z, Yuan Z. Development of a sensitive and robust liquid chromatography coupled with tandem mass spectrometry and a pressurized liquid extraction for the determination of aflatoxins and ochratoxin A in animal-derived foods. J Chromatogr A. 2012;1253:110-9. [ Links ]
6. Corcuera LA, Ibánez-Vea M, Vettorazzi A, González-Penas E, Cerain AL. Validation of a UHPLC-FLD analytical method for the simultaneous quantification of aflatoxin B1 and ochratoxin A in rat plasma, liver and kidney. J Chromatogr B: Anal Technol Biomed Life Sci. 2011;879:2733-40. [ Links ]
7. Devegowda GM, Raju VLN, Swamy HVLN. Mycotoxins: novel solutions for their counteraction. Feedstuffs. 1998;70:12-6. [ Links ]
8. Diaz G, Murcia HW, Cepeda SM. Cytochrome P450 enzymes involved in the metabolism of aflatoxin B1 in chickens and quail. Poult Sci. 2010;89:2461-9. [ Links ]
9. Díaz-ZaragozaM, Carvajal-MorenoM,Méndez-Ramírez I, Chilpa-Galván NC, Avila-González E, Flores-Ortiz CM. Aflatoxins, hydroxylated metabolites and aflatoxicol from breast muscle of laying hens. Poult Sci. 2014;93:3152-62. [ Links ]
10. European Commission (EC) Commission Regulation 401/2006/EC Laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs, Brussels. [ Links ]
11. European Commission (EC) Commission Decision 2002/657/EC implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. Brussels: Commission Regulation 472/2002/EC; 2002. [ Links ]
12. European Commission Directive 2002/32/ec of the European Parliament and of the Council of 7 may 2002 on undesirable substances in animal feed. [ Links ]
13. Fan Y,Zhao L, Ma Q, Li X, Shi H, Zhou T, Zhang J, Ji C. Effects of Bacillus subtilis ANSB060 on growth performance, meat quality and aflatoxin residues in broilers fed moldy peanut meal naturally contaminated with aflatoxins. Food Chem Toxicol. 2013;59:748-53. [ Links ]
14. Food and Agriculture Organization of the United Nations (FAO). Worldwide regulations for mycotoxins in food and feed. Food Nutrition paper 81; 2003. Rome, Italy. Available at: http://www.fao.org [accessed on: 09.08.15].
15. Food and Drug Administration (FDA) CPG (1994) Sec. 683.100 Action Levels for Aflatoxins in Animal Feeds. [ Links ]
16. Gathumbi JK, Usleber E, Ngatia TA, Kangethe EK, Martlbauer E. Application of immunoaffinity chromatography and enzyme immunoassay in rapid detection of aflatoxin B1 in chicken liver tissues. Poult Sci. 2003;82:585 -90. [ Links ]
17. Gregory JF, Manley D. High performance liquid chromatographic determination of aflatoxins in animal tissues and products. J Assoc Off Anal Chem. 1981;64:144-51 http://www.fda.gov/ICECI/ComplianceManuals/CompliancePolicyGuidance-Manual/ucm074703.htm [ Links ]
18. Huang B, Han Z, Cai Z, Wu Y,Ren Y. Simultaneous determination of aflatoxins B1, B2, G1, G2, M1 and M2 in peanuts and their derivative products by ultra-high-performance liquid chromatography -tande mass spectrometry. Anal Chim Acta. 2010;662:62-8. [ Links ]
19. International Agency for Research on Cancer (IARC). Some traditional herbal medicines, some mycotoxins, naphthalene and styrene. Summary of data reported and evaluation. IARCMonogr Eval Carcinog Risks Hum. 2002;82:590. [ Links ]
20. Kokkonen MK, Jestoi MN. A multi-compound LC-MS/MS method for the screening of mycotoxins in grains. Food Anal Methods. 2009;2:128-40. [ Links ]
21. Lamelas K, Mair G, Beczkowski G. Evolución del sector Avícola año 2009, perspectiva 2010. Bol Avíc. 2016;58:1-26. Ministerio de Agricultura, Ganadería, Pesca y Alimentación. http://www.minagri.gob.ar [accessed on November] [ Links ].
22. Liu Y, Wu F. Global burden of aflatoxin-induced hepatocellular carcinoma: a risk assessment. Environ Health Perspect. 2010;118:818-24. [ Links ]
23. Magnoli AP, Monge MP, Miazzo RD, Cavaglieri LR, Magnoli CE, Merkis CI, Cristofolini AL, Dalcero AM, Chiacchiera SM. Effect of low levels of aflatoxin B1 on performance, biochemical parameters and aflatoxin B1 in broiler liver tissues in the presence of monensin and sodium bentonite. Poult Sci. 2011;90: 48 -58. [ Links ]
24. Magnoli AP, Texeira MR, Rosa CA, Miazzo RD, Cavaglieri LR, Magnoli CE, Dalcero AM, Chiacchiera SM. Sodium bentonite and monensin under chronic aflatoxicosis in broiler chickens. Poult Sci. 2011;90:352-7. [ Links ]
25. Magnoli AP, Monge MP, Nazar FN, Magnoli CE, Cavaglieri LR, Bagnis G, Dalcero AM, Marin RH. Combined effects of aflatoxin B1 and corticosterone treatment on selected performance indices and liver histopathology in Japanese quail. Poult Sci. 2012;91:354-61. [ Links ]
26. Mutlu AG, Kursun O, Kasimoglu A, Muzaffer D. Determination of aflatoxin M1 levels and antibiotic residues in the traditional Turkish desserts and ice creams consumed in Burdur city center. J Anim Vet Adv. 2010;9:2035-7. [ Links ]
27. Nazar FN, Magnoli AP, Dalcero AM, Marin RH. Effect of feed contamination with aflatoxin B1 and administration of exogenous corticosterone on Japanese quail biochemical and immunological parameters. Poult Sci. 2012;91:47-54. [ Links ]
28. Oliveira CA, Kobashigawa E, Reis TA, Mestieri L, Albuquerque R, Correa B. Aflatoxin B1 residues in eggs of laying hens fed a diet containing different amounts of mycotoxin. Food Addit Contam. 2000;17:459-62. [ Links ]
29. Rizzi L, Simioli M, Roncada P, Zaghini A. Aflatoxin B1 and clinop-tilolite in feed for laying hens: effects on egg quality, mycotoxin residues in livers and hepatic mixed function oxidase activities. J Food Prot. 2003;66:860-5. [ Links ]
30. Sørensen LK, Elbsk TH. Determination of mycotoxins in bovine milk by liquid chromatography tandem mass spectrometry. J Chromatogr B. 2005;820:183-96.
31. Stubblefield RD, Shotwell OL. Determination of aflatoxins in animal tissues. J Assoc Off Anal Chem. 1981;64:964-8. [ Links ]
32. Sultana N, Hanif NQ. Mycotoxin contamination in cattle feed and feed ingredients. Pak Vet J. 2009;29:211-3. [ Links ]
33. Sulyok M, Berthiller F, Krska R, Schuhmacher R. Development and validatio of a liquid chromatography/tandem mass spectrometric method for the determination of 39 myco-toxins in wheat and maize. Rapid Commun Mass Spectrom. 2006;20:2649-59. [ Links ]
34. Sulyok M, Krska R, Schuhmacher R. A liquid chromatography/tandem mass spectrometric multi-mycotoxin method for the quantification of 87 analytes and its application to semiquantitative screening of moldy food samples. Anal Bioanal Chem. 2007;389:1505-23. [ Links ]
35. Tavčar-Kalcher G, Vrtac K, Pestevsek U, Vengust A. Validation of the procedure for the determination of aflatoxin B1 in animal liver using immunoaffinity columns and liquid chromatography with post-column derivatizatio and fluorescence detection. Food Control. 2007;18:333-7.
36. Taylor ST, King JW, Greer JI, Richard JL. Supercritical fluid extraction of aflatoxin M1 from beef liver. J Food Prot. 1997;60:698-700. [ Links ]
37. Trucksess MW, Stack ME, Nesheim S, Albert RH, Romer TR. Multifunctional column coupled with liquid chromatography for determination of aflatoxins B1, B2, G1, G2 in corn, almonds, Brazil nuts, peanuts and pistachio nuts: collaborative study. J. AOAC Int. 1994;6:1512-21. [ Links ]
38. Yunus AW, Razzazi-Fazeli E, Bohm J. Aflatoxin B1 in affecting broiler's performance, immunity, and gastrointestinal tract: a review of history and contemporary issues. Toxins. 2011;3:566-90. [ Links ]
39. Zahoor-ul-Hassan M, Khan Z, Khan A, Javed I. Pathological responses of white leghorn breeder hens kept on ochratoxin A contaminated feed. Pak Vet J. 2010;30:118-23. [ Links ]
40. Zhao Z, Liu N, Yang L, Deng Y, Wang J, Song S, Lin S, Wu A, Zhou Z, Hou J. Multi-mycotoxin analysis of animal feed and animal-derived food using LC-MS/MS system with timed and highly selective reaction monitoring. Anal Bioanal Chem. 2015, http://dx.doi.org/10.1007/s00216-015-8898-5. [ Links ]














