Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Latin American applied research
versión impresa ISSN 0327-0793
Lat. Am. appl. res. v.33 n.3 Bahía Blanca jul./sept. 2003
Infinite dilution activity coefficients of solvents in fatty oil derivatives
O. Ferreira1 and G. M. Foco2
1 LSRE, Departamento de Engenharia Química, Faculdade de Engenharía
Universidade do Porto, 4200-465 Porto, Portugal
olgafer@fe.up.pt
2 PLAPIQUI (Universidad Nacional del Sur – CONICET)
Camino La Carrindanga Km 7 - 8000 Bahía Blanca - Argentina
gfoco@palpiqui.edu.ar
Abstract ¾ Inverse gas-liquid chromatography has been applied to measure infinite dilution activity coefficients (g¥ ) of different solutes in low and high molecular weight triacylglycerides (TAGs) and in mixtures of carboxylic acids with TAGs. The g¥ data obtained were used to determine binary interaction and size related parameters for the GCA-EOS group contribution with association equation of sate.
Keywords ¾ Activity Coefficients. Infinite Dilution. Carboxylic Acids. Triglycerides. Parameters Determinations. Binary Interaction.
I. INTRODUCTION
Experimental information on thermodynamic properties of fatty acids and triacylglycerides (TAGs) is required for the design and simulation of separation processes applied in the extraction and fractionation of fatty oil derivatives from vegetable and animal oils.
In previous work (Foco et al., 1996; Bermudez et al., 2000), infinite dilution activity coefficients (g¥ ) of a series of organic solutes in palmitic acid, tripalmitin and triacetin, were measured. These data were obtained by inverse gas chromatography, using the non volatile TAG or fatty acid as stationary phase and injecting the volatile solute at infinite dilution in the carrier gas stream. Those data have been used (Bottini et al, 1999) to determine molecular and group interaction parameters for the group contribution with association equation of state GCA-EOS proposed by Gros et al (1996).
The functional groups characteristic of TAGs and fatty acids are, respectively, the triglyceride TG ((CH2COO)2-CHCOO) and acid (COOH) groups. The interaction parameters between the TG and COOH groups are not available in the GCA-EOS parameter table. Also the interaction between TG and the primary (CH2OH) and secondary (CHOH) alcohol groups are missing. The purpose of this work is to measure infinite dilution activity coefficients in order to fit those interaction parameters to the experimental data.
The use of inverse gas chromatography to measure infinite dilution activity coefficients requires the solvent in the stationary phase to be a non-volatile compound, so that the amount of solvent in the stationary phase remains constant during the measurements. On the other hand, the solutes injected into the gas carrier should give sharp and neat chromatograms. Solutes of low volatility and/or highly retained in the stationary phase will give broad chromatographic peaks. In this case the values of the retention times, and consequently the calculated g¥ would be uncertain.
All TAGs are low volatile compounds and can always be used as solvents in a chromatographic stationary phase. Even the first member of the family, triacetin, has negligible vapor pressures at temperatures up to 353K. Unfortunately, carboxylic acids are highly retained in TAGs stationary phases; this makes inverse gas chromatography an unsuitable technique to measure g¥ of carboxylic acids in TAGs.
In order to obtain experimental information to fit the GCA-EOS interaction parameters between TG and COOH groups, an alternative procedure was followed in this work. Stationary phases were prepared using mixtures of TAGs with high molecular weight non volatile fatty acids and g¥ of volatile solutes in these mixtures were measured. With these values and the corresponding g¥ of the same solutes in each pure solvent (TAG and fatty acid) it was possible to quantify the interaction between TG and COOH. Mixtures of palmitic acid with two different TAGs (triacetin and tripalmitin) were prepared, and the g¥ of a series of organic solutes in these mixtures were measured.
II. EXPERIMENTAL RESULTS
The infinite dilution activity coefficients were measured by gas-liquid chromatography. The specific retention volume Vºg at 273.15 K, i.e. the normalized volume of carrier gas necessary to elute solute i out of a column with a mass Ws of solvent, is calculated from the measured retention time (ti) of solute i by:
 | (1) |
where ta is the inert gas retention time; F is the saturated gas flow measured at temperature Tf and pressure Pf ,
PsW the vapor pressure of water at Tf, Po is the column outlet pressure and J32 the James Martin correction factor for pressure gradient and gas compressibility inside the column (Conder and Young, 1979).
The infinite dilution activity coefficient of solute i, (g¥ i) is calculated from the solute retention volume by:
 | (2) |
In this equation, the gas phase non idealities are take into account by the virial equation; Bii is the second virial coefficient, vi the molar volume and PiS the saturation pressure of component i at temperature T; MS is the molecular weight of the solvent and R is the universal gas constant.
A Varian (Model Star 3400 CX) gas chromatograph with a FID detector and a Hewlett-Packard (Model 3392) integrator were used in the measurements. The columns temperatures were measured by Platinum Resistance Thermometers (Systemteknik S 1220) and the thermal stability was ± 0.1 K. The carrier gas was hydrogen, flowing at about 35 cm3/min; a soap-film meter was used to measure the gas flow rates. Column inlet and outlet were connected to a mercury manometer in order to obtain the pressure drop and atmospheric pressure was measured with a quartz transducer (Paroscientific). Pressure drops in the column ranged from 600 to 800 mbar. The solutes were injected into the carrier gas by using Hamilton 1 l syringes.
The stationary phase for each column was prepared by dissolving, in chloroform, weighed amounts of solvent and inert support (Chromosorb W, 60/80 mesh), at a ratio of 3 parts of chromosorb per part of solvent. The chloroform was then evaporated from the solution, in an inert atmosphere, using a roto-evaporator (Heidolph). Stainless steel tubing, 1/8 in. O.D. by 2 m long, were packed with each stationary phase.
In order to obtain the dead volume or reference retention time ta, methane was always injected together with each solute. The non-retention of methane in the stationary phase was checked against the constancy of methane retention time at different column temperatures. A complete description of the experimental technique is presented in a previous work (Foco et al, 1996).
Reagent grade products from Aldrich Chem. Co. were used in the experiments.
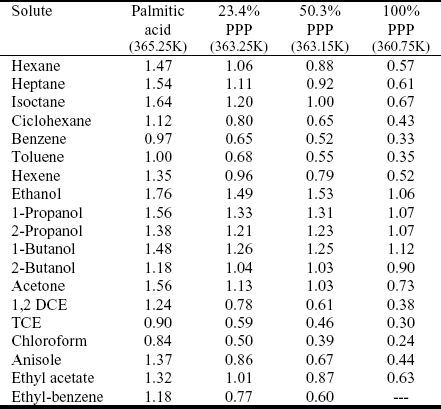
Table 1 presents, for a series of organic solutes, the g¥ measured at 353K, in two mixtures of palmitic acid (PA) with triacetin (AAA): 30% PA + 70% AAA and 50% PA + 50% AAA. Tables 2, 3 and 4 present the g¥ values obtained at 353K, 363K and 373K, in mixtures of palmitic acid (PA) with tripalmitin (PPP) for the following molar fractions: 25% PA + 75% PPP, 50% PA + 50% PPP. The tables also show the g¥ of the solutes in the pure solvents (palmitic acid, triacetin and tripalmitin).
From a propagation of error analysis, an overall uncertainty of 3 to 5% is estimated for the g¥ values reported in Tables 1 to 4. These errors were calculated on the basis of the following uncertainties in the measured variables: ± 0.13mbar for column pressure drop, ± 0.7cm3/min for the carrier gas flow, ± 0.1K for temperatures, ± 0.002g for the solvent mass in the column, ± 0.12s for the retention times.
Table 1. Experimental g ¥ in mixtures of triacetin and palmitic acid

DCE: Dichloroethane
TCE: Trichloethylene
MTBE: Methyl Terbuthyl Ether
Table 2. Experimental g ¥ in mixtures of tripalmitin and palmitic acid

Figure 1 represents the experimental g¥ of heptane in mixtures of PA + AAA and PA + PPP, as a function of the molar fraction of palmitic acid in the mixtures.

Fig. 1. Representation of g ¥ of heptane versus the molar fraction of palmitic acid: n m experimental data; ¾¾ GCA-EOS.
The limiting values of g¥ in the pure solvents (palmitic acid, triacetin and tripalmitin) are also shown. It is interesting to notice the high g¥ value of heptane in pure triacetin (MW=218), the first member of the family of triglycerides. This indicates important energy interactions between the TG and paraffinic (CH2/CH3) functional groups, which are responsible for the partial liquid miscibility found in heptane – triacetin mixtures.
Table 3. Experimental g ¥ in mixtures of tripalmitin and palmitic acid
The effect of this strong interaction vanishes in the case of tripalmitin (MW=806), due to the TAG long hydrocarbon chains. Here the combinatorial effects are predominant, and the g¥ values of heptane in tripalmitin are lower than one. The addition of the long-chain palmitic acid to triacetin increases the combinatorial effects and dilutes the TG - CH2 and COOH – CH2 interactions; these combined effects lower drastically the g¥ values of heptane. On the other hand, the addition of palmitic acid to PPP decreases the average molecular weight of the mixture and increases the energy interactions due to the presence of the COOH group in the mixture; this gives rise to higher g¥ values.
Table 4. Experimental g¥ in mixtures of tripalmitin and palmitic acid

In the narrow range of temperatures covered in this work, no temperature dependence of g¥ was observed, with the exception of the alcohols series. The g¥ of these compounds decreases with temperature, which indicates positive partial excess enthalpies in fatty acids and TAGs solutions. Figure 2 shows the g¥ of heptane, isopropanol and trichloroethylene in an equimolar solution of palmitic acid and tripalmitin, at the three temperatures measured in this work.
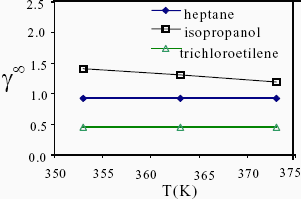
Fig. 2. Temperature dependence of g¥ for heptane, isopropanol and trichloroethylene in a mixture containing a 50%palmitic acid / 50%tripalmitin.
III. PARAMETERIZATION
There are four binary interaction parameters in the GCA-EOS model: the symmetric, temperature dependent kij (kij = kij* (1 + kij´ ln (T /600))) and the unsymmetric nonrandomness parameters a ij and a ji The g¥ data obtained in this work were used to fit the binary interaction parameters between the triglyceride group TG and the carboxylic acid COOH, primary CH2OH and secondary CHOH alcohol groups.
Table 5. Binary interaction parameters

Table 5 reports the values of those parameters. Figures 1 and 3 show the values of infinite dilution activity coefficients of heptane and alcohols in triglycerides calculated by the GCA-EOS model.

Fig. 3. g¥ of alcohols versus temperature: u m experimental data; ¾¾ GCA-EOS.
IV. CONCLUSIONS
In this work g¥ of several chemicals in mixtures of TAGs with palmitic acid were measured. The g¥ of alkanes were used to quantify the interaction between the triglyceride and the COOH functional groups. The experimental data were properly fitted by using a single temperature independent binary interaction parameter. More experimental information, for a wider range of temperatures and for mixtures with lower palmitic acid concentrations, is needed in order to test the validity of these parameters.
REFERENCES
1. Bermudez, A., Foco, G., Bottini, S. B. Infinite Dilution Activity Coefficients in Tributyl Phosphate and Triacetín, J. Chem. Eng. Data, 45(6),1105-1107 (2000). [ Links ]
2. Bottini, S.B., Fornari, T., Brignole, E.A.. Phase Equilibrium Modelling of Triglycerides with Near Critical Solvents. Fluid Phase Equilibria 158-160, 211-218 (1999). [ Links ]
3. Conder, J.; Young, C. (1979). Physicochemical Measurements by Gas Chromatography, Wiley Interscince, New York. [ Links ]
4. Foco, G.; Bermudez, A.; Bottini, S. B. Infinite Dilution Activity Coefficients in mono-, di-, tri-palmitin and palmitic acid. J. Chem. Eng. Data, 41,1071-1074 (1996). [ Links ]
5. Gros H.P., S.B. Bottini and E.A. Brignole. A group contribution equation of state for associating mixtures. Fluid Phase Equilibria, 116, 537-544 (1996). [ Links ]














