Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Latin American applied research
versión impresa ISSN 0327-0793
Lat. Am. appl. res. v.33 n.3 Bahía Blanca jul./sept. 2003
Fortification of mushroom with calcium by vacuum impregnation
C. F. Ortíz, D. M. Salvatori and S. M. Alzamora
Dto. de Industrias, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires.
Ciudad Universitaria, 1428 Buenos Aires, Argentina.
dsalvato@di.fcen.uba.ar
Abstract ¾ Foods with added physiologically-active food components (PAC) are receiving special attention by their potential in disease prevention and health promotion. Additional intake of any nutrient might be achieved by the application of vacuum impregnation technique to fruits and vegetables. Viability of mushroom as matrix for the incorporation of calcium was then evaluated. It was found that, due to the high impregnated liquid fraction observed (17 - 40 %), the calcium incorporated in 100 g of mushroom would satisfy about 24 - 32 % of the Adequate Intake (AI). Thus, there would seem to be much potential for the introduction of high Ca2+ concentration in mushroom tissue through vacuum impregnation, combating the widespread Ca2+ deficiencies that occur in human populations. Although mechanical properties are affected by the vacuum treatment, similar or greater mushroom softening is produced during post-harvest storage or commercial thermal processing.
Keywords ¾ Mushroom. Functional Foods. Calcium.
I. INTRODUCTION
In the last years there has been a growing interest in the design of the so-called functional foods. These include foods with added physiologically-active food components (PAC) in order to facilitate or to increase the consumption of a specific vitamin or mineral that may provide an additional health benefit beyond the traditional nutrients they contain (IOM/NAS, 1994; Clare and Hasler, 1998; Hasler, 1998). In particular, calcium is a fundamental mineral for human body, since low intake is one of risk factors in the bone disease osteoporosis. Its deficiency is also endemic in Argentine. Causes are linked not to economic situation but to food habits common to the majority of population, that is, insufficient calcium consumption in the different life stages and high levels of protein, fiber, phosphate and polyphosphates containing industrialized foods which may affect the bioavailability of this nutrient.
An additional intake of this micronutrient might be achieved by impregnation soaking processes. Vacuum impregnation (VI) treatment of a porous material consists of exchanging the internal gas or liquid occluded in open pores for an external liquid phase (of controlled composition) due to pressure changes. Fruit and vegetables have a great number of pores occupied by gas or native liquid and offer the possibility of being impregnated by a determined solution (Fito and Chiralt, 2000). Thus product composition as well as its physical and chemical properties may be changed to improve the properties of the final product. The impregnated liquid fraction (X) means the pore fraction of the matrix that can be penetrated by the external solution at the mechanical equilibrium status, and is a function of the product porosity and the applied vacuum pressure.
Very few studies have been carried out by using impregnation techniques for the development of functional foods with vegetable matrices. Anino et al. (2001) compared two impregnation methods (vacuum and atmospheric) for the incorporation of Ca2+ in apple tissue. Fito et al. (2001) proposed a mathematical model to calculate the PAC concentration of the impregnation medium in order to formulate functional foods with the addition of different Ca2+ and Fe++ salts and satisfy a specific percentage of the Recommended Daily Intake (RDI). These studies suggest that these impregnation methods could be used for the incorporation of PAC in fruits and vegetables matrices.
The objective of this work was to evaluate the viability of a highly porous matrix (i.e. mushroom) for the incorporation of calcium by vacuum impregnation on the basis of matrix properties (impregnated liquid fraction, Ca2+ content, tissue microstructure and mechanical properties).
II. METHODS
A. Sample Preparation and Treatments
Fresh mushrooms (Agaricus bisporus, pH 6.8, aw @ 0.99) were cut into cylinders (1.5 cm in diameter and 1 cm in length) including three kinds of mushroom tissue (cap, stipe and cap/stipe interface). Each experiment was performed with the same lot of mushrooms in order to minimize biological variability due to age and/or cellular structure.
Two VI treatments (run 1 and run 2) were conducted at room temperature by immersion of fresh samples in agitated isotonic salt aqueous solutions (5.88 % p/p calcium salts). A mixture of calcium gluconate and calcium lactate was chosen because of its relatively high solubility at room temperature and the neutral taste it imparts to the food. Potassium sorbate (1500 ppm) was added to all the systems and the pH was adjusted to 3.5 with citric acid to inhibit and/or retard microbial growth and nonenzymatic reactions. A pulse of 15 min was applied at a vacuum pressure of 30 mmHg and then atmospheric pressure was restored and maintained for 15 min. At the end of each treatment, samples were analyzed for impregnated liquid fraction, Ca2+ content, textural properties and structure. Each specimen was identified, weighed before and after VI and its diameter and height measured.
A third treatment (run 3) was performed by using an isotonic NaCl aqueous solution to analyze the impregnation effect per se on textural characteristics.
B. Impregnated Liquid Fraction (X)
The X values were calculated from sample weight before and after each treatment. Loss of native liquid was neglected, so the calculated X value for mushrooms would represent the total external liquid penetrated into the tissue. An analysis of variance and multiple range tests were applied to establish significant differences between vacuum treatments (P £ 0.05). From the mean values of X obtained, theoretical values of the Ca2+ content finally achieved in the matrix was calculated taking into account the amount incorporated because of treatment and the amount initially present in raw material (80-90 ppm, determined by atomic absorption).
C. Determination of Ca2+ content
After being desiccated, samples were digested in a microwave digestor and calcium content was measured by atomic absorption in an atomic absorption flame photometer (Shimadzu AA-6800).
D. Texture
After each experiment, compression tests were performed in an Instron Testing Machine (model 1101), at 10 mm/min cross-head speed, 100 mm/min recording system paper speed and a load range (LR) ranging from 20-50 kgf in fresh samples and 5-10 kgf in treated samples. Each specimen was compressed perpendicular to the stem axis (75 % of original height) in order to detect rupture of the tissue. Force-strain curves (» 10 replicates) were obtained for fresh and treated mushrooms and analysis of variance and multiple range tests were applied to establish significant differences between treatments (P £ 0.05). From each curve three forces were registered: a restructuring force (Fre) corresponding to the first peak of the curve, a rupture force (Frup) corresponding to the following peak and the maximum force (Fm), when a deformation equal to 75 % of cylinder length was reached.
Modulus of deformability (Ed) defined by Eqn.(1) was also calculated :
 | (1) |
| where: |  | = stress (N/m2);F = force (N); |
| A = cross-sectional area of the specimen (m2); | ||
| Ed = modulus of deformability (N/m2); | ||
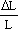 | = relative deformation; | |
| L = initial cylinder length (m) | ||
The increase in cross-sectional area due to lateral compression was neglected so that the stress values were based on the original cross sectional area of the compressed specimen.
E. Microscopy
For light microscopy (LM), specimens were taken from the inner region of cap, transitional zone and stipe. They were then fixed in 3 % (w/w) glutaraldehyde solution, in 0.1 M potassium phosphate buffer (pH=7.4) overnight at room temperature, postfixed in 1.5 % (w/w) OsO4 solution and dehydrated in a graded acetone series prior to be embedded in Spurr resin. Sections (1-2 m m thick) were stained with 1 % (w/w) toluidine blue and 1 % w/w basic fuchsin solutions and examined in a microscope (Carl Zeiss, model Axioplan).
III. CONCLUSIONS
A. Calcium Incorporation
The impregnated liquid fractions obtained (17-24 % for lot 1 and 29-40 % for lot 2) implied a high final Ca2+ content in matrices, although a great variability between lots was observed. Figure 1 shows the Ca2+ concentration of raw material (11ppm for Run 1 and 43 ppm for Run 2) and the Ca2+ concentration of VI mushrooms ranging from approximately 2400 (Run 1) to 3200 ppm (Run 2). Thus, calcium incorporated in 100 g of mushroom would satisfy about 24 - 32 % of the AI (AI = 1000 mg a day; Institute of Medicine, 1998). The figure also shows the theoretical values of Ca2+ content calculated from the experimental values of X. The differences observed would indicate the loss of native liquid during vacuum stage due to pore expansion.
B. Texture Results
The typical sigmoid shape of force/deformation curves for fresh mushrooms is shown in Fig. 2. It is possible to consider the mushroom as a spongy material (Aguilera & Stanley, 1990) which exhibits an initial increase in force (elastic region), followed by a stress plateau produced by elastic buckling and redistribution of intercellular spaces. This plateau could be attributed to a structural reorganization of the tissue after failure and liquid extraction due to compression. This behaviour was characterized by the force corresponding to the beginning of the plateau (restructuring force: Fre). After that, the force again increased until a peak (rupture force: Frup) which corresponded to the visible fracture of the material. At further deformation, the force

Fig. 1.Experimental and predicted calcium concentration (ppm) for raw and vacuum impregnated mushrooms. Run 1: Ca2+ VI; Run 2: Ca2+ VI.

Fig. 2.Pattern of force/strain curves for raw mushroom. Fre= "restructuring" force; Frup = rupture force; Fm = maximum force, after 75 % deformation.
continued increasing to higher values, ending at the predetermined deformation of the assay (maximum force: Fm).
Figure 3 shows the different patterns obtained for mushroom cylinders subjected to treatments with Ca2+ salts and NaCl. In Ca2+ impregnated tissue characteristic peaks were sharper and the forces were lower when compared with those of raw material, in addition, the constant force zone was not observed. NaCl impregnated samples did not exhibit an apparent rupture peak and softening was much more pronounced.
Figure 4 compares the Fre, Frup, and Fm values for the three lots of raw mushrooms used in the experiments. There was a great heterogeneity between the mushrooms of the lots used in the runs 1, 2 and 3. In spite of deforming in a similar way, the magnitude of developed forces (specially Fre values) was significantly different. These differences could be a consequence of the mushroom's age, stage of maturity and storage conditions. This variability in raw material would strongly affect the force values in impregnated samples. In fact, the Frup force was significantly different (P £ 0.05) when Ca2+ treatment was done with lot 1 or with lot 2 (Fig. 5a). So, each treated sample was compared

Fig.3.Pattern of force/strain curves for treated mushrooms. A) control (run 1); B) VI with Ca2+(run 1); C) control (run 2); D) VI with Ca2+ (run 2); E) control (run 3); B) VI with NaCl (run 3).
with its own control. A significant tissue softening occurred in the material after Ca2+ vacuum impregnation, denoted by the evident decrease in Frup forces of treated samples (76 % and a 93 %, respectively).
Figure 5b compares the Fm values for raw mushrooms, Ca2+ vacuum impregnated mushrooms and mushrooms treated under vacuum in the NaCl solution. These results indicate that VI process per se would have an important deteriorative effect on the textural characteristics, slightly mitigated by calcium addition.
Mean values of modulus of deformability (i.e. the relationship between stress and strain, or the resistance to the deformation) calculated at Fre and Frup points are shown in Table 1. For Na Cl treated samples only values at maximun point could be calculated. The low values obtained for impregnated samples also indicated a loss of tissue rigidity due to treatments.
Histological analysis of raw tissues (Fig. 6 A-B) showed the typical hyphal structure that composes the cap and the stem, with random crossing hyphae in the cap and longitudinal hyphae in the stem.
The longitudinal filaments in the stem were bunched together. The tissue exhibited substantial air spaces between which would allow the reorganization of the the hyphae and the observed stress plateau.
VI treatment (Fig. 6 C-F) produced citoplasmolysis and disruption of cellular membranes, loss of turgor and collapse of the cells. This tissue collapse would have contributed to volumetric shrinkage (about 30 or 50%), this fact being also observed through the greater number of cells per area in the micrographs. Hyphae cells of cap appeared less intercrossed than in control leaving enlarged intercellular spaces, which would support the tissue softening observed in instrumental analysis.

Fig. 4. Compression forces for raw mushroom of the different lots used in the experiments. Run 1: Ca2+ VI; Run 2: Ca2+ VI; Run 3: NaCl VI


Fig. 5. Compression forces for raw and impregnated mushrooms. a) Rupture forces. b) Maximum forces. Run 1: Ca2+ VI; Run 2: Ca2+ VI; Run 3: NaCl VI.
Table 1. Modulus of deformability (Ed) at restructuring point (Ed re), at rupture peak (Ed rup) and at the maximum peak (Ed m)
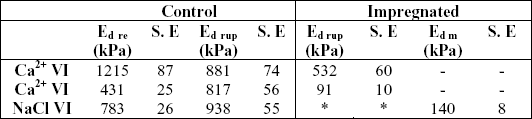
* Rupture peaks were not observed; SE = standard error
Fig. 6. LM micrographs of vacuum impregnated mushrooms. A, B: fresh (control); C-F: vacuum treated, C-D: Ca2+ impregnated; E-F: NaCl impregnated.
Acknowledgements
The authors acknowledge the financial support from Universidad de Buenos Aires, CONICET and Agencia Nacional de promoción Científica y Tecnológica de la República Argentina.
REFERENCES
1. Aguilera, J.M. and D.W. Stanley, "Microstructural Principles of Food Processing and Engineering", Elsevier, Amsterdam (1990). [ Links ]
2. Anino, S., D. Salvatori and S. M. Alzamora, "Apple matrix with calcium incorporated by impregnation processes", Ingeniería de alimentos: Nuevas fronteras en el siglo XXI. Proc. III Congreso Iberoamericano de Ingeniería de Alimentos, P. Fito, A. Mulet, A. Chiralt (eds.), Servicio de Publicaciones de la Universidad Politécnica de Valencia, Spain, 4 (2003).(En prensa) [ Links ]
3. Clare, M. and C.M. Hasler, "Functional Foods: Their Role in Disease Prevention and Health Promotion", Food Technol. 52 (11), 63-70 (1998). [ Links ]
4. Fito, P. and A. Chiralt, "Vacuum Impregnation of Plant Tissues", Minimally Processed Fruits and Vegetables. Fundamental Aspects and Applications, S.M. Alzamora, M.S. Tapia, A. López-Malo (eds.), Aspen Publishers, Inc., Maryland, 153-171 (2000). [ Links ]
5. Fito, P., A. Chiralt, N. Betoret, M. Gras, M. Cháfer, J. Martínez-Monzó, A. Andrés, D. Vidal, "Vacuum impregnation and osmotic dehydration in matrix engineering. Application in functional fresh food development", J. Food Eng. 49, 175-183 (2001). [ Links ]
6. Hasler, C.M., "A new look at an ancient concept", Chem. Industry 2, 84-89 (1998). [ Links ]
7. IOM/NAS, "Opportunities in the Nutrition and Food Sciences", Institute of Medicine/National Academy of Sciences, P.R Thomas, R. Earl (eds.), National Academic Press, Washington D.C., 109 (1994). [ Links ]
8. Institute of Medicine, "Dietary References Intake (DRI) for Calcium, Phosphorus, Magnesium, Vitamin D and Fluoride", Food and Nutrition Board, Standing Committee on the Scientific Evaluation of Dietary References Intakes, National Academy Press, Washington, D.C., 432 (1998). [ Links ]














