Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Latin American applied research
versión impresa ISSN 0327-0793
Lat. Am. appl. res. v.36 n.4 Bahía Blanca oct./dic. 2006
Effect of sodium butyrate and zinc sulphate supplementation on recombinant human IFN-β production by mammalian cell culture
R. Zuqueli, C. Prieto, M. Etcheverrigaray and R. Kratje
Laboratorio de Cultivos Celulares, Facultad de Bioquímica y Ciencias Biológicas, Universidad Nacional del Litoral, Ciudad Universitaria -C.C. 242- (S3000ZAA) Santa Fe, Pcia. Santa Fe, Argentina
rkratje@fbcb.unl.edu.ar
Abstract — Recombinant human interferon-β1 (rhIFN-β1) glycoprotein is used as a therapeutic agent for a variety of diseases, such as multiple sclerosis and hepatitis.
In the present work, different strategies were applied to produce rhIFN-β1a in mammalian cell cultures. Transfected population of CHO-K1, CHO dhfr-, BHK and HEK cells were compared for their ability to produce rhIFN-β1a, and clones of the most promising cell line (CHO-K1) were isolated by the limit dilution method.
Likewise, different culture conditions were assayed by changing the amounts of fetal calf serum, sodium butyrate and/or ZnSO4, to improve cell productivity. The presence of each additive increased the rhIFN-β1a yield ranging from 2 to 8 times, depending on the tested cell clone, but when these components were simultaneously added to the medium, the rhIFN-β1a concentration in the supernatants was even greater.
Keywords — rhIFN-β1a Production. CHO Cells. Zinc Sulphate. Sodium Butyrate.
I. INTRODUCTION
Interferons (IFNs) comprise a conserved family of antiviral proteins secreted by specific mammalian cells upon virus infection. Binding of IFNs to specific cell surface receptors triggers an intracellular signal cascade resulting in the transcriptional activation of a large number of cellular genes (de Veer et al., 2001; Bekisz et al., 2004). While some of these proteins modulate viral replication, others have additional effects such as modulation of the immune system and direct anti-tumor effect. According to this, IFN-based therapy has been demonstrated to be beneficial in the treatment of human malignancies such as chronic myelogenous leukemia, non-Hodgking's lymphoma, infectious diseases such as hepatitis B and C and immune disorders such as multiple sclerosis (Gutterman, 1994; Revel, 2003; Lutton et al., 2004).
Recombinant IFNs for therapeutic use are currently being produced in bacterial and mammalian cells. Recombinant human IFN-β1a (rhIFN-β1a) is a commercially available glycosylated form of IFN-β expressed in Chinese Hamster Ovary (CHO) cells, while rhIFN-β1b is a non-glycosylated variant expressed in E. coli. The rhIFN-β1b shows an in vitro biological activity similar to that of rhIFN-β1a, but it has an extremely lower half life in vivo and the generation of neutralizing antibodies, during the treatment with both molecules, have suggested the convenience of the treatment with the glycosylated form (Runkel et al., 1998; Brickelmaier et al., 1999; Kivisakk et al., 2000).
Difficulties to produce large amounts of rhIFN-β1a in mammalian cell cultures may be due in part to destabilizing sequences present in the IFN-β mRNA coding region and the 3' untranslated region (Raj and Pitha, 1993) and/or by growth arrest or apoptosis induction mediated by the recombinant protein itself.
Sodium butyrate (NaBu) has been widely used in recombinant CHO cell cultures for high-level expression of recombinant protein such as antibodies (Kim and Lee, 2000), erythropoietin (Chung et al., 2001), recombinant B-domain-deleted factor VIII (Chun et al., 2003), thrombopoietin (Sung et al., 2004) and tissue plasminogen activator (Hendrick et al., 2001). One of the most evident changes brought about by butyrate is the acetylation of histones via inhibition of the histone deacetylase enzyme. The acetylation of core histones is expected to exert a major influence on the accessibility of chromatin to regulatory molecules (D'Anna et al., 1980; Lee et al., 1993), but it can also inhibit cell growth and induce cellular apoptosis (Mimura et al., 2001; Kim and Lee, 2000). Zinc has been proposed to block apoptosis in human cells under certain conditions (Perry et al., 1997) and to confer increasing mRNA stability, either by altering its secondary structure or by promoting the binding of stabilizing proteins to the mRNA (Harford and Klausner, 1990; Cao, 2004). Likewise, zinc inhibits, directly or indirectly, one or more ribonucleases responsible for the degradation of labile mRNAs (Hartmann et al., 2001).
In this work, we studied the effect of the addition of different amounts of FCS (fetal calf serum), butyrate, zinc or a combination of them on the productivity of recombinant cells engeneered to obtain high expression levels of rhIFN-β1a.
II. METHODS
A. Cell Culture
The Chinese Hamster Ovary (CHO-K1), Syrian Hamster Kidney (BHK-21) and Human Embryonic Kidney (HEK-293) cell lines were grown in culture medium containing 5 % FCS (Bioser, Argentina) as described previously (Kratje et al., 1994).
CHO cells deficient in the dihydrofolate reductase gene (CHO dhfr-) were grown in the medium described above supplemented with 10 % FCS or in selective medium: Iscove Modified Dulbecco's Medium, IMDM (Gibco, USA) supplemented with 10 % FCS extensively dialyzed against phosphate-buffered saline (PBS).
WISH cells were grown in Minimum Essential Medium, MEM (Gibco, USA) supplemented with 10 % FCS.
B. Plasmid Construction
The human IFN-β1 coding sequence was amplified using the polymerase chain reaction (PCR) with the 5' forward primer 5'-ATGACCAACAAGTGTCTCCTCCAA-3' and the 3' reverse primer 5'-TCAGTTTCGGAGGTAACCTGTAAG-3'. The PCR fragment was gel-purified (GFXTM PCR DNA and Gel Band, GE Healthcare, USA) and inserted into the plasmid pGEM-T easy (Promega, USA). An EcoRI fragment, containing the human IFN-β coding sequence, was removed from the pGEM-T-IFN-β1 plasmid and inserted into the EcoRI site of the pCIneo (Promega, USA) and p91023 (Wong et al., 1985) plasmids. A restriction site mapping analysis was performed to confirm the orientation of the insertion and the region checked out by dideoxy DNA sequencing.
C. Transfections
Transfections of cells were performed using 10 μg/ml of LipofectAMINE 2000 (Invitrogen, UK) and 6 μg/ml of plasmidic DNA, using p91023 plasmid for CHO dhfr- and pCIneo plasmid for CHO-K1, BHK-21 and HEK-293 cell lines. In brief, cells were grown up to 80% confluence in 12-well plates before transfection. Then, the culture medium was replaced by serum-free medium and the transfection mixture was added. After 4 h this medium was replaced by a complete medium. 24 h post transfection, the cells were selected with Geneticin (400 μg/ml) (Gibco, USA) or selective medium. This medium was replaced by fresh medium every 3-4 days until death of the control cells. When transformant cells were in confluence, the medium was assayed for rhIFN-β1a production by immunoblot. After this, producing cell lines were cloned by the limit dilution method (Freshney, 2000).
D. Butyrate Treatment
Sterile 0.5 M n-butyrate (sodium salt) (Sigma-Aldrich, USA) in PBS was prepared and stored as a stock solution at -20oC.
To examine the effect of FCS and NaBu on the production of rhIFN-β1a, recombinant CHO cells were seeded into each well of a 24-well plate containing growth medium. When cells were in confluence, the medium was replaced by another one containing combinations of 0, 0.1, 0.5, 1 and 2 % FCS and 0, 1, 2, 3 and 5 mM NaBu. The media were replaced every 2 days during 8 days.
E. Zinc Treatment
Sterile 2 M ZnSO4 (Sigma-Aldrich, USA) in PBS was prepared and stored as a stock solution at 4oC.
To examine the effect of Zn2+ on the production of rhIFN-β1a, recombinant CHO cells were seeded into 24-well plates containing growth medium. When cells were in confluence the medium was replaced by fresh medium containing 0.5 % FCS and 0, 25, 50, 100, 150 or 200 μM ZnSO4. The media were replaced every day and the treatment was carried out during 3 days.
F. Butyrate and Zinc Treatment
To study the additive effect of butyrate and Zn2+ on the production of rhIFN-β1a, recombinant CHO cells were seeded into each well of a 24-well plate containing growth medium. When cells reached confluence the medium was replaced by another one containing 0.5 % FCS and 2 mM NaBu for clone 1D5, 0.1 % FCS and 1 mM NaBu for clone 1D7 and 0.1 % FCS and 5 mM NaBu for clone 1G5, plus 0, 50, 100 or 150 μM ZnSO4. The media were replaced every day and the treatment was carried out for 3 days.
G. Pulse and Rest Treatment
To examine the effect of FCS, butyrate and Zn2+ on the production of rhIFN-β1a by the clone 1D5 and to try to prolong the production time, recombinant CHO cells were seeded into each well of a 24-well plate containing growth medium. When cells were in confluence, the medium was replaced by medium supplemented with FCS, NaBu and ZnSO4 as shown in Table 1 and the incubation proceeded for 24 h (pulse). Then, the media were replaced by another medium supplemented with 1 % FCS and cells were incubated for 24 h (rest). Every day cells were treated with 0.25 % tripsine / 0.04 % EDTA (Gibco, USA) and counted by the trypan blue dye exclusion method (Griffiths, 1986). The treatment was carried out for 8 days.
Table 1. Different media used in the pulse treatment.

H. Immunoblot Assay
The total concentration of rhIFN-β1a was quantified using a rapid screening method of immunoblot. For this, 40 μl of different dilutions of the protein standard (rhIFN-β1b, Betaferon, Schering Plough) were used. Culture supernatants were blotted onto a nitrocellulose membrane (GE Healthcare, USA). This membrane was incubated in tris buffered saline (TBS) containing 1 % bovine serum albumin (BSA) for 1 h. After washing with TBS, the incubation proceeded with TBS-BSA 0.1 % containing the mouse anti-rhIFN-β1b MAb 2C12 previously obtained in our laboratory. This MAb 2C12 has also the capacity to recognize the rhIFN-β1a molecule. After this, the membrane was incubated with peroxidase-conjugated anti-mouse immunoglobulin (DAKO A/S, Denmark) and revealed by incubation with 10 ml of a solution containing 0.25 % α-chloronaphtol in methanol, with the addition of 40 μl of 0.5 % H2O2 (30 V) in TBS.
I. Western Blot Analysis
Different forms of rhIFN-β1a were characterized by a Western blot assay. Sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) was performed in an 18 % polyacrylamide gel (Laemmli, 1970). 40 μl samples and 500 ng/ml of protein standard (rhIFN-β1b, Betaferon, Schering Plough) were applied to the gel. Proteins were electrophoretically transferred to a nitrocellulose membrane and probed with the mouse anti-rhIFN-β1b MAb 2C12. Immunoreactive bands were visualized using a peroxidase-conjugated anti-mouse immunoglobulin kit (Perkin Elmer Life Science, USA).
J. Antiviral Activity
The antiviral activity of rhIFN-β1a was determined using a cytopathic effect bioassay that measures the ability of the protein to protect WISH cells from cytotoxicity due to infection with the vesicular stomatitis virus (VSV). For this, 100 μl of serial dilutions of the protein standard (rhIFN-β1b, Betaferon, Schering Plough) and of the test samples were placed into the wells of microtiter plates. Afterwords, 4x104 WISH cells suspended in 100 μl of growth medium were added to each well and the plates incubated for 2 h at 37oC. Afterwards, 50 μl of VSV virus were added to each well and the plates were incubated for another 24 h. Supernatants were removed and 50 μl of 0.75 % crystal violet in 40 % methanol were added. Plates were incubated for 10 minutes, the colorant was washed and 250 μl of 20 % acetic acid were added. Color was read at 540 nm. Signal intensity for the samples was reported as the mean of the absorbance measured in duplicate wells.
III. RESULTS AND DISCUSSION
A. Analysis of rhIFN-β1a Expressing Cells
An aliquot of different recombinant cell lines supernatants were assayed by Western blot to select the mammalian host cells that produce rhIFN-β1a properly glycosylated. All recombinant cell lines produced glycosylated and non-glycosylated forms of rhIFN-β1a but in different proportions (Fig. 1). CHO-K1 and HEK-293 cells mainly produced the glycosylated form (lane 2 and 4, respectively), BHK-21 principally produced the non-glycosylated form (lane 3) and CHO dhfr- cells produced undetectable quantities of the recombinant protein (lane 1).

Fig. 1. rhIFN-β1 production by different recombinant cell clones. Supernatants of cell lines CHO dhfr-, CHO.K1, BHK-21, HEK-293 (line 1 to 4, respectively), and 20 ng of the non-glycosylated protein used as standard (line 5) were analyzed by Western blot.
According to these results, CHO-K1 cells were chosen to continue with the study. A rhIFN-β1a producing CHO-K1 cell line was propagated and cloned by the limit dilution method and three positive clones were selected according to their higher expression level. These clones were named 1D5, 1D7 and 1G5 and their productivities were 1.9, 2.5 and 2.9 mIU/cell/d, respectively.
B. Effect of the Addition of NaBu and FCS on the Production of rhIFN-β1a
In order to analyze the effect of NaBu and FCS on the rhIFN-β1a expression, different medium conditions were assayed. The amount of rhIFN-β1a in the supernatants was estimated by immunoblot. As shown in Fig. 2, the rhIFN-β1a production raised when NaBu was present in the culture medium and this effect was marked during the first and second medium exchange. This gain achieved with the NaBu addition decreased during subsequent medium exchanges, probably due to the deleterious effects of NaBu such as inhibition of cell growth and induction of cellular apoptosis, described by Kim and Lee (2000) and Wang et al. (2004), respectively. Besides, an increase in cellular detachment was observed. This effect may be attributed to changes in the cell surface and the cytoskeleton which occur during apoptosis (Mimura et al., 2001). In addition, it is favoured by the depletion of FCS, as it had been observed in previous experiments in our laboratory, where cells detached from the bottom of the plates as the FCS concentration decreased.
Fig. 2. Effect of FCS and NaBu on the production of rhIFN-β1a for clones 1D5, 1D7 and 1G5. After reaching confluence, cells were incubated in medium containing combinations of 0, 0.1, 0.5, 1 and 2 % FCS and absence ( ) or presence of 1 mM (
) or presence of 1 mM ( ), 2 mM (
), 2 mM ( ), 3 mM (
), 3 mM ( ) and 5 mM (
) and 5 mM ( ) NaBu. The medium of each well was replaced every 2 days during a period of 8 days, performing 4 medium exchanges.
) NaBu. The medium of each well was replaced every 2 days during a period of 8 days, performing 4 medium exchanges.
We found that the productivity of each clone was affected in a different manner depending on the concentration of FCS and NaBu. For the first medium exchange, the rhIFN-β1a expression increased almost 6 times for clone 1D5 in medium supplemented with 0.5 % FCS and 2, 3 and 5 mM NaBu or 1 % FCS and 5 mM NaBu; 6 times for clone 1D7 in medium supplemented with 0.1 % FCS and 1, 2, 3 and 5 mM NaBu or 0.5 % FCS and 5 mM NaBu or 1 % FCS and 5 mM NaBu; and 5 times in the case of clone 1G5 in medium supplemented with 0.1 % FCS and 5 mM NaBu. Conditions that increased the productivity with the lower concentration of NaBu (to avoid apoptosis) and FCS (to reduce costs and facilitate the downstream processing) were chosen as optimal conditions for each clone.
C. Effect of Zn2+ on the Production of rhIFN-β1a
Constitutive expression of human IFN-β1a gene was also analyzed in the presence of Zn2+. Different concentrations of ZnSO4 were added to the culture and the supernatants were assayed by immunoblot 24 hours later. As shown in Fig. 3, the expression of rhIFN-β1a was higher with 150 μM ZnSO4, but concentrations higher than 50 μM were deleterious for cell viability (results not shown). Similar observations have been found by Taylor and Blackshear (1995) with TK-L cells exposed to concentrations higher than 100 μM ZnSO4. In those cases they found that the accumulation of the tristetraprolin and c-fos mRNA was not increased and cell death was even greater. In cultures containing 100 or 150 μM ZnSO4, the protein production raised during the first day and then declined.
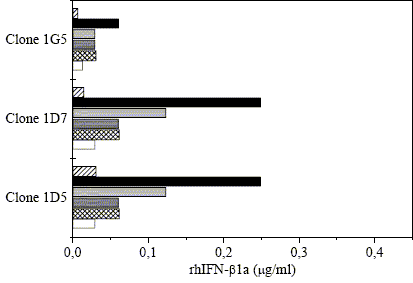
Fig. 3. Effect of ZnSO4 on the production of rhIFN-β1a during the first medium replacement estimated by immunoblot when medium supplemented with 0.5 % FCS and absence ( ) or presence of 25 μM (
) or presence of 25 μM ( ), 50 μM (
), 50 μM ( ), 100 μM (
), 100 μM ( ), 150 μM (
), 150 μM ( ) or 200 μM (
) or 200 μM ( ) ZnSO4 was used.
) ZnSO4 was used.
Clones 1D5 and 1D7 displayed an increment in the production of 4 and 8 times when the Zn2+ concentration was about 100 and 150 μM, respectively, and 2 times when concentrations were between 25 and 50 μM. The clone 1G5 showed a similar pattern but the production of the recombinant protein was much lower.
D. Effect of the Combination of NaBu and Zn2+ on the Production of rhIFN-β1a
To examine the effect of the combination of NaBu and Zn2+ on the production of rhIFN-β1a, clones 1D5, 1D7 and 1G5 were cultured with optimal amounts of NaBu plus 0, 50, 100 or 150 μM ZnSO4 and the rhIFN-β1a concentrations in the supernatants were measured by immunoblot. Fig. 4 shows that the production of the different assayed clones generally increased with time in the presence of concentration of Zn2+ higher than 50 μM concentration. This enhancing effect of Zn2+ was more evident for clones 1D5 and 1D7. With optimal amounts of NaBu and FCS plus 50 or 100 μM ZnSO4, the increase in the recombinant protein production was about 2.5 times for the clone 1D7 and about 3 times for the clone 1D5. The clone 1G5 showed a similar pattern but the production of the recombinant protein again was much lower.

Fig. 4. Effect of NaBu and ZnSO4 on the production of rhIFN-β1a estimated by immunoblot, when medium supplemented with 0.5 % FCS and 2 mM NaBu for clone 1D5, 0.1 % FCS and 1 mM NaBu for clone 1D7 and 0.1 % FCS and 5 mM NaBu for clone 1G5, in the absence ( ) or presence of 50 μM (
) or presence of 50 μM ( ), 100 μM (
), 100 μM ( ) or 150 μM (
) or 150 μM ( ) ZnSO4 was used.
) ZnSO4 was used.
The production levels of biological active rhIFN-β1a in optimal medium containing 50 or 100 μM ZnSO4 for clones 1D5 and 1D7 were analized by antiviral activity (Table 2). The 1D5 clone showed the highest production in the presence of 50 μM of ZnSO4.
Table 2. Antiviral activity of different clones in optimal medium containing 50 or 100 μM ZnSO4.

Comparing the results from Fig. 4 and Table 2, it is clear that the levels of rhIFN-β1a production reached by clones 1D5 and 1D7 in the presence of 50 or 100 μM ZnSO4 are different. This difference may be attributed to the characteristics of the methods applied. In fact, while the immunoblot assay measures the total amount of recombinant protein whether it is active or not, the antiviral activity bioassay detects only the protein that is biologically active. Consequently, clone 1D5 appears as a better producer of the biological active molecule, and for this reason this clone was chosen for further studies.
E. Pulse and Rest Treatment
In order to prolong the production time of clone 1D5, a pulse and rest treatment was applied during 8 days.
Fig. 5 shows that the productivity rose every day when ZnSO4, NaBu or a mix of NaBu and ZnSO4 were added to the culture, displaying an increment of 2.5, 5.5 and 6 times, respectively. Furthermore, we found that the cellular monolayer remained attached and that the rhIFN-β1a production was held in similar levels during the pulses with each one of the different media, in contrast to the previous results without the rest period.

Fig. 5. Effect of NaBu, ZnSO4 and ZnSO4 plus NaBu on the production of rhIFN-β1a during the different pulses. Results obtained by the antiviral activity assay are expressed as the ratio of the specific productivity reached by clone 1D5 cultivated in medium B, C or D in comparison to the specific productivity reached in medium A (absence of NaBu and ZnSO4). The values obtained for medium A in each pulse were 10.6, 11.4, 15.6 and 12.3 mIU/cell/d, respectively. The results corresponding to the different rests are not shown.
To examine whether the culture conditions affected the glycosylation of rhIFN-β1a, supernatants were compared with each other after each pulse by Western blot. Fig. 6 shows that the relative amount of each isoform, glycosylated and non-glycosylated rhIFN-β1a, was the same in all cases. These results suggest that the quality of the protein was mantained in spite of the different supplements added to the culture.

Fig. 6. Analysis of rhIFN-β1a production. Supernatants of the clone 1D5 during the 1st pulse with medium A, B, C or D (line 1 to 4, respectively) and 20 ng of the non-glycosylated protein used as standard (line 5) were visualized by Western blot.
On the other hand, it can be seen from the gel that the rhIFN-β1a production increases with the addition of ZnSO4, NaBu or a mix of both.
IV. CONCLUSIONS
The results presented here showed that the addition of butyrate, Zn2+ or both simultaneously to the culture medium of recombinant cell clones producing rhIFN-β1a is of practical interest. We found that the production could be increased almost 5 times for the CHO-K1 clone 1G5 in medium supplemented with 0.1 % FCS and 5 mM NaBu and 6 times for clones 1D5 and 1D7 in media supplemented with 0.5 % FCS and 2 mM NaBu and 0.1 % FCS and 1 mM NaBu, respectively. Therefore, optimal conditions should be studied for each clone. On the other hand, the presence of Zn2+ in the culture medium, which according to the literature acts through stabilization of the mRNA (Taylor and Blackshear, 1995; Worthington et al., 2002), also increased the production of the recombinant protein. There was a 4-fold increase in the production of the recombinant protein for the CHO-K1 clone 1G5 and a 8-fold one for clones 1D7 or 1D5 using medium supplemented with 0.5 % FCS and 150 μM ZnSO4. With both additives, the increase in the rhIFN-β1a concentration was remarkable during the first day of culture; but then declined. Further improvements in the production levels were achieved by a combination of NaBu and Zn2+, reaching higher levels of production than the one obtained with each one of the additives alone. On the other hand, it was demonstrated that the high levels of production could be maintained for at least 8 days by pulsing the culture with NaBu and Zn2+, without any alteration in the quality of the product.
In summary, we have established stable cell clones and conditions for the production of high levels of glycosylated rhIFN-β1a of high specific activity. Therefore, the production of the recombinant protein in our laboratory, in order to perform preclinical and clinical studies, is warranted.
REFERENCES
1. Bekisz, J., Schmeisser, H., Hernandez, J., Goldman, N. and Zoon, K., "Human Interferons Alpha, Beta and Omega",Growth Factors, 22(4), 243-251 (2004). [ Links ]
2. Brickelmaier, M., Hochman, P., Baciu, R., Chao, B. and Whitty, A., "ELISA methods for the analysis of antiboby responses induced in multiple sclerosis patients treatred with recombinant interferon-β", J. Immunol. Methods, 227(1-2), 121-135 (1999). [ Links ]
3. Cao, H., "Expression, Purification, and Biochemical Characterization of the Antiinflammatory Tristetraprolin: A Zinc-Dependent mRNA Binding Protein Affected by Posttranslational Modifications", Biochemistry, 43(43), 13724-13738 (2004). [ Links ]
4. Chun, B.-H., Park, S.-Y., Chung, N. and Wang, W.-G., "Enhanced production of recombinant B-domain deleted factor VIII from Chinese hamster ovary cells by propionic and butyric acids", Biotechnol. Lett., 25(4), 315-319 (2003). [ Links ]
5. Chung, B. S., Jeong, Y. T., Chang, K. H., Kim, J. S. and Kim, J. H., "Effect of sodium butyrate on glycosylation of recombinant erythropoietin", J. Microbiol. Biotechnol., 11(6), 1087-1092 (2001). [ Links ]
6. D'Anna, J., Tobey, R., and Gurley, L. R., "Concentration-Dependent Effects of Sodium Butyrate in Chinese Hamster Cells: Cell-Cycle Progression, Inner-Histone Acetylation, Histone H1 Dephosphorylation, and Induction of an H1-like Protein", Biochemistry, 19, 2656-2671 (1980). [ Links ]
7. de Veer, M. J., Holko, M., Frevel, M., Walker, E., Der, S., Paranjape, J. M., Silverman, R. H. and Williams, B. R., "Functional classification of interferon-stimulated genes identified using microarrays", J. Leukocyte Biol., 69(6), 912-20 (2001). [ Links ]
8. Freshney, R. I., Cloning and selection. In: Culture of Animal Cells. A Manual of Basic Technique. Fourth edition, Wiley-Liss, Canada, 195-215 (2000). [ Links ]
9. Griffiths, B., Scaling-up of animal cell cultures. In Animal Cell Culture, Freshney, R. (ed.) IRL Press, Washington, USA, 33-69 (1986). [ Links ]
10. Gutterman, J. U., "Cytokine therapeutics: lessons from interferon alpha", Proc. Natl. Acad. Sci. USA, 91(4), 1198-1205 (1994). [ Links ]
11. Harford, J. B. and Klausner, R. D., "Coordinate post-transcriptional regulation of ferritin and transferrin receptor expression: the role of regulated RNA-protein interaction", Enzyme, 44(1-4), 28-41 (1990). [ Links ]
12. Hartmann, R., Walko, G. and Justesen, J., "Inhibition of 2'-5' oligoadenylate synthetase by divalent metal ions", FEBS Lett., 507(1), 54-58 (2001). [ Links ]
13. Hendrick, V., Winnepenninck, P., Abdelkafi, C., Vandeputte, O., Cherlet, M., Marique, T., Renemann, G., Loa, A., Kretzmer, G., Werenne, J., "Increased productivity of recombinant tissular plasminogen activator (t-PA) by butyrate and shift of temperature: a cell cycle phases analysis", Cytotechnology, 36(1-3), 71-83 (2001). [ Links ]
14. Kim, N. S. and Lee, G. M., "Overexpression of bcl-2 inhibits sodium butyrate-induced apoptosis in Chinese hamster ovary cells resulting in enhanced humanized antibody production", Biotechnol. Bioeng., 71, 184-193 (2000). [ Links ]
15. Kivisakk, P., Alm, G. V., Fredikson, S. and Link, H., "Neutralizing and binding anti-interferon-beta (IFN-beta) antibodies. A comparison between IFN-beta-1a and IFN-beta-1b treatment in multiple sclerosis", Eur. J. Neurol., 7(1), 27-34 (2000). [ Links ]
16. Kratje, R. B., Reimann, A., Hammer, J. and Wagner, R., "Cultivation of recombinant baby hamster kidney cells in a fluidezd bed biorreactor system whit porous borosilicate glass", Biotechnol. Prog., 10(4), 410-420 (1994). [ Links ]
17. Laemmli, U. K., "Cleavage of structural proteins during the assembly of the head of bacteriophage T4", Nature, 227, 680-685 (1970). [ Links ]
18. Lee, D. Y., Hayes, J. J., Pruss, D. and Wolffe, A. P., "A positive role for histone acetylation in transcription factor access to nucleosomal DNA", Cell, 72(1), 73-84 (1993). [ Links ]
19. Lutton, J. D., Winston, R. and Rodman, T. C., "Multiple Sclerosis: Etiological Mechanisms and Future Directions", Exp. Biol. Med., 229(1), 12-20 (2004). [ Links ]
20. Mimura, Y., Lund, J., Church, S., Dong, S., Li, J., Goodall, M. and Jefferis, R., "Butyrate increases production of human chimeric IgG in CHO-K1 cells whilst maintaining function and glycoform profile", J. Immunol. Methods, 247, 205-216 (2001). [ Links ]
21. Perry, D. K., Smyth, M. J., Stennicke, H. R., Salvesen, G. S., Duriez, P., Poirier, G. G. and Hannun, Y. A., "Zinc Is a Potent Inhibitor of the Apoptotic Protease, Caspase-3. A novel target for Zinc in the inhibition of apoptosis", J. Biol. Chem., 272(30), 18530-18533 (1997). [ Links ]
22. Raj, N. and Pitha, P., "65-KDa protein binds to de-stabilizing sequences in the IFN-β mRNA coding and 3'UTR", FASEB J., 7(8), 702-710 (1993). [ Links ]
23. Revel, M., "Interferon-β in the treatment of relapsing-remitting multiple sclerosis", Pharmacol. Ther., 100(1), 49-62 (2003). [ Links ]
24. Runkel, L., Meier, W., Pepinsky, R. B., Karpusas, M., Whitty, A., Kimball K., Brickelmaier, M., Muldowney, C., Jones, W. and Goelz, S. E., "Structural and functional differences between glycosylated and non-glycosylated forms of human interferon-beta (IFN-beta)", Pharm. Res., 15(4), 641-649 (1998). [ Links ]
25. Sung, Y. H., Song, Y. J., Seung, W. L., Chung, J. Y. and Lee, G. M., "Effect of sodium butyrate on the production, heterogeneity and biological activity of human thrombopoietin by recombinant Chinese hamster ovary cells", J. Biotechnol, 112(3), 323-335 (2004). [ Links ]
26. Taylor, G. A. and Blackshear, P. J., "Zinc Inhibits Turnover of Labile mRNAs in Intact Cells", J. Cell. Physiol., 162(3), 378-387 (1995). [ Links ]
27. Wang, X. H., Xu, J., Zhang, Y., Li, L., Feng, Q., Mi, L. and Chen, Z. N., "Inducible expression of Bcl-XL inhibits sodium butyrate-induced apoptosis in hybridoma, resulting in enhanced antibody production", Cell. Biol. Int., 28(3), 185-191 (2004). [ Links ]
28. Wong, G. G., Witek, J. S., Temple, P. A., Wilkens, K. M., Leary, A. C., Luxenberg, D. O., Jones, S. S., Brown, E. L., Kay, R. M., Orr, E. C., Shoemaker, C., Golde, D. W., Kaufman, R. J., Hewik, R. M., Wang, E. A. and Clark, S. C., "Human GM-CSF: Molecular Cloning of Complementary DNA and Purification of the Natural and Recombinant Proteins", Science, 228, 810-815 (1985). [ Links ]
29. Worthington, M., Pelo, J., Sachedina, M., Applegate, J., Arseneau, K., Pizarro, T., "RNA Binding Properties of the AU-rich Element-binding Recombinant Nup475 / TIS11 / Tristetraprolin Protein", J. Biol. Chem., 277, 48558-48564 (2002). [ Links ]
Received: December 14, 2005.
Accepted for publication: June 20, 2006.
Recommended by Editor A. Bandoni.














