Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Latin American applied research
versión impresa ISSN 0327-0793
Lat. Am. appl. res. v.37 n.2 Bahía Blanca abr. 2007
Hemodialysis access failure: viscoelastic vascular properties an intimal hyperplasia development
D. Bia1, W. Barmak5, Y. Zócalo1, C. Galli4, H. Perez-Campos6, M. Saldias6, W. Silva6, I. Alvarez6, E. I. Cabrera Fischer2,3 and R. Armentano1,2
1 Dto. de Fisiología, Facultad de Medicina, Universidad de la República, Montevideo, Uruguay, CP:11800. dbia@fmed.edu.uy
2 Universidad Favaloro, C1078AAI, Buenos Aires, Argentina fischer@favaloro.edu.ar
3 Member of the Research Career, CONICET, Buenos Aires, Argentina.
4 Universidad Tecnológica Nacional-FRBA, 1179, Buenos Aires, Argentina cngalli@gmail.com
5 University of Connecticut, CT, U.S.A.
6 INDT, MSP-Facultad de Medicina, Universidad de la República, Montevideo, Uruguay.
Abstract — Intimal hyperplasia development is largely the most important cause of vascular access failure in patients submitted to hemodialysis.
Differences in the biomechanical properties between the vascular substitutes and the native vessels have been related to the development of intimal hyperplasia. This work aim was to characterize the biomechanical behavior of arteries, veins, cryopreserved arteries and ePTFE prostheses.
Fresh and cryopreserved human arteries and veins and ePTFE conduits were in-vitro studied in order to analyze their viscoelastic properties.
Our results show that ePTFE has an elastic index, which is significantly different from that of the other conduits. This determines a high elastic mismatch that has been involved as a cause of intimal hyperplasia development.
We conclude that the biomechanical study of tubular segments to be used as vascular accesses demonstrate viscoelastic differences that could be an important determinant of access viability.
Keywords — Vascular Access. Biomechanical Study. ePTFE. Intimal Hyperplasia. Cryopreserved Arteries and Veins.
I. INTRODUCTION
The introduction of hemodialysis has determined a prolonged survival in patients with end stage renal disease. However, maintenance of permanent vascular access has limited the adequate use of chronic renal replacement therapy.
An ideal vascular access must be able to withstand a considerable flow to allow the hemodialysis procedure, cause minimal complications and have a high patency rate (Rodriguez et al., 2000). Many techniques have been employed for the vascular access therapy. Among the most used are the native (Brescia-Cimino) fistula, and those that use synthetic prosthesis (Cernadas et al., 2003). Nevertheless, up to date, none of the synthetic or biologic conduits used fulfills the ideal vascular access properties (Cinat et al., 1999).
Vascular accesses have a high incidence of complications, in particular thrombotic occlusion, which determines the vascular access failure. The development of intimal hyperplasia at the arteriovenous anastomosis, graft vein anastomosis or outflow vein have been described as a major cause of thrombosis (Tordoir et al., 2004).
The development of intimal hyperplasia has been related, among other factors, to differences in viscoelasticity between the native vessels and the vascular substitutes. This works aim was to analyze the elastic properties (E), viscous behavior (V) and pulse wave velocity (PWV) of synthetic conduits of ePTFE, and of human fresh and cryopreserved arteries and veins. Additionally, the intimal hyperplasia process, and its relationship with vascular accesses mechanical behavior are described.
II. VASCULAR ACCESS
In end stage renal disease patients, the effort must be directed to obtain an arteriovenous fistula (AVF), while the use of grafts would be left as a second choice (Rodriguez et al., 2000).
The common surgical procedure for the creation of a fistula consists in the anastomosis of the radial artery and cephalic vein in the forearm (Zarin et al., 2004). This AVF is the vascular access of choice since it determines the lowest risk of dysfunction, infection, thrombosis and the longest patency rate (Añel et al., 2003; Canaud, 2004). Additionally, problems concerning vascular steal syndrome and high output cardiac failure are least frequent in AVF performed in the forearm position. Finally, it preserves proximal vessels for future vascular accesses (Rodriguez et al., 2000; Tordoir et al., 2004).
Many patients do not have adequate veins to use for AVF. In these cases the use of an arteriovenous graft (AVG) is a valid alternative. The AVG, which consists of a prosthetic material in between the native artery and vein, should remain as a secondary option (Canaud, 2004). In these cases, the expanded polytetrafluoroethylene (ePTFE) is one of the materials used and its performance is lower than that of the AVF (Cinat et al., 1999; Rodriguez et al., 2000). However, those who prefer the AVG rather than the AVF usually point out that the AVG is easier to needle and has a faster maturation period (Young et al., 2002) and that it is easily obtained in different diameters, making it an available tool at hand for a diverse number of vessel sizes (Cinat et al., 1999). On the other hand, thrombosis is more easily treated in AVGs than in AVFs (Khosla and Ahya, 2002). Nevertheless, as it was reported, they show a lower patency rate when compared to AVF, as well as a higher incidence of infection (Ezzahiri et al., 1999; Cernadas et al., 2003).
An important factor that must be considered in end stage renal failure is the patient characteristics. For instance age, sex and diabetes have been shown to modify the access duration. For instance, the mean age of patients that need hemodialysis is increasing, which means that the probability that they have adequate endogenous vessels to use for an AVF is decreasing (Ezzahiri et al., 1999).
In summary, the need of vascular access is increasing, but the alternatives available nowadays are far from the ideal. Therefore potential new vascular accesses must be considered. In this context, cryopreserved vessels should be considered and studied for their possible use in end stage renal failure patients (Ezzahiri et al., 1999; Cernadas et al., 2003; Zarin et al., 2004; Young et al., 2002).
III. INTIMAL HYPERPLASIA
Intimal hyperplasia has been thoroughly studied during the past years; despite the fact that a consensus has not been reached regarding the mechanisms that take place for it to develop. There is evidence to support the fact that intimal hyperplasia has a complex etiology and pathobiology which necessitates further study (Sivanessan et al., 1999).
It has been observed that intimal hyperplasia in vascular accesses is the consequence of high flow rates (Kohler and Kirkman, 1999). AVFs and AVGs are currently traversed by high blood flows. As a consequence of the flow disturbances, there is a proliferation of the cells, and the extracellular matrix. These tend to move into the lumen of the graft, making an obstacle to blood passing towards the outflow vein and sometimes causing thrombi (Cernadas et al., 2003; Sivanesan et al., 1999). See Fig. 1. Since the precursor of thrombosis is stenosis, it is important to control the AVG evolution, in order to detect stenosis before thrombosis appears (Vassalotti et al., 2004; Khosla and Ahya, 2002). In fact, the use of a stent can be helpful when applying it to a stenosed native fistula or synthetic graft (Turmel-Rodrigues, 2004) while several pharmacological drugs are needed, on the other hand, to treat thrombosis (Turmel-Rodrigues, 2004).

Figure 1. The angiogram shows a stenotic venous tract (straight arrow) in a vascular access performed in the left arm of a chronically dialyzed patient. Between the humerus and the diseased vein a patent ePTFE prosthesis is visualized.
Two other main factors, related to flow, that affect the development of intimal hyperplasia are compliance mismatch between the native vessel and the graft (Haruguchi and Teraoka, 2003; Hofstra et al., 1995), and the wall shear stress, which is characterized by the chan-ces that blood elements, like platelets and leucocytes adhere to the vessel wall. In particular, inordinately high shear stress might provoke the denudation of endothelial cells that promotes the passage of smooth muscle cells into the intima (Haruguchi and Teraoka, 2003).
While veins often present high levels of intimal hyperplasia, this is not observed in arteries (Johnson et al., 2001). Since veins have a thinner wall, they might be more prone to being damaged by flow (Kohler et al., 1999). There exist other differences in the outcome of intimal hyperplasia in these two segments. For example, the vein presents a relatively poor demarcation of its elastic laminae, thereby easing the passage of smooth muscle cells from the tunica media to the intima as a consequence of an elevated degree of shear stress (Roy-Chaudhury et al., 2002). The low nitric oxide and high numbers of fibroblast growth factors receptors in the vein might also predispose this vessel to a more frequent occurrence of stenosis and thrombosis (Roy-Chaudhury et al., 2002).
According to all the aforementioned possible causes of intimal hyperplasia, it would be desirable the use of conduits with mechanical properties similar to those of the native vessels when performing a vascular access procedure.
IV. DYNAMIC STUDY OF VASCULAR ACCESSES
We have designed a circulating loop that is utilized to evaluate the mechanical properties of synthetic prosthesis (ePTFE) and fresh and cryopreserved vessels. In the circulation mock, the segments can be studied while submitted to diverse ranges of intraluminal pressure and stretching rate values. (Cabrera Fischer et al., 2002; Cabrera Fischer et al., 2005).
The in vitro system is constituted of a pneumatic pump which propels a liquid (i.e. blood) throughout the circulating loop. The pump generates the pulsatile flow to the circuit and is connected to a perfusion line that finds an organ chamber in its closed loop, where there is Tyrodes solution kept at 37ºC with a pH of 7.4 bubbled with 100% oxygen. The resistance modulator can be regulated to mimic the human circulatory system. The direction of flow is as shown in Fig. 2.

Figure 2. Circulating Loop. PP: pneumatic pump, R: Resistance modulator, DD: ultrasonic crystals measuring the diameter of the vascular segment under study. PM: pressure microtransducer. Arrows indicate flow direction.
The intraluminal pressure was measured using a Konigsberg P7 or P2.5 microtransducer (1200 Hz frequency response) while the external diameter was assessed using a pair of ultrasonic crystals (5 MHz, 2 mm diameter); as has been previously reported (Bia et al., 2005; Cabrera Fischer et al., 2002).
Segments of 5 cm ePTFE were placed and studied in the perfusion line immersed in the organ chamber as can be seen in Fig. 2. Human vessels procurement was done following the rules of the Guides of the National Organ and Tissue Bank transplant program from Uruguay. The processing of arteries and veins included the written consent and was carried out according to the ethics and safety standards for therapeutic use. Exclusion criteria was determined following the International Standards for Tissue Bank guidelines.
In situ measurements of 5 cm were performed on the adventitia tissue of the right and left femoral arteries and saphenous veins of 9 human donors in brain death condition. Vessel segments were excised and those to be used as fresh were sent to the biomechanical laboratory, while the rest were submitted to the cryopreservation protocol.
Fresh and cryopreserved defrosted venous and arterial segments were mounted in the organ chamber as can be seen in Fig. 2.
The pressure and diameter signals recorded were digitized using a specific program developed in our laboratory and stored for later analysis. This analysis consisted in the calculation of the stress-strain plot once the wall thickness was obtained and the evaluation of incremental elastic index (E), viscosity index (V). We also calculated the pulse wave velocity (PWV) so as to obtain a mechanical index used in clinics (Armentano et al., 1991; Bia et al., 2005).
V. RESULTS OF BIOMECHANICAL STUDY
Fresh femoral artery (n=9), cryopreserved femoral artery (n=9), fresh saphenous vein (n=9), cryopreserved saphenous vein (n=9) and ePTFE (n=9) were in-vitro analyzed. All segments were submitted to systemic levels of pressure (see Table 1).
Table 1: Hemodynamic Measurements

MV ± SD. FFA and CFA: fresh and cryopreserved/defrosted femoral artery. FSV and CSF: fresh and cryopreserved/defrosted saphenous vein. ePTFE: expanded polytetrafluoroethylene segments. PP and MP: pulse and mean pressure (mmHg). MD: mean diameter (mm). PR: pump rate (b.p.m). ANOVA followed by Bonferroni test. a ,b ,c and d: p<0.05 with respect to FFA, CFA, FSV and CSV, respectively. No significative differences were found.
The same methodological protocol was employed to characterize the dynamic mechanical properties of the vessels and synthetic prostheses. After positioning the segment in the organ chamber (in between the perfusion line), the segments were allowed to stabilize for a period of 15 minutes under a steady state of flow (150 ml/min) and a mean pressure of 93 mmHg, at a stretching rate of 80 beats/min (1.34 Hz).
The elastic and viscous indexes were derived from the pressure-diameter loop calculated according to previous works (Armentano et al., 1991). The PWV values (see Table 2) were obtained according to the methodology previously reported (Bia et al., 2005).
Table 2: Mechanical Parameters
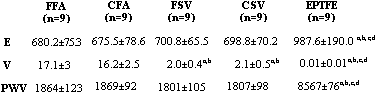
MV ± SD. FFA y CFA: fresh and cryopreserved/defrosted femoral artery. FSV and CSF: fresh and cryopreserved/defrosted saphenous vein. ePTFE: expanded polytetrafluoroethylene segments. E: elastic index (mmHg/mm). V: viscous index (mmHg.s/mm). PWV: pulse wave velocity (cm/s). ANOVA followed by Bonferroni test. a ,b ,c and d: p<0.05 with respect to FFA, CFA, FSV and CSV, respectively.
Table 1 shows that the experiments were done at isobaric and isofrequency conditions. Moreover, the mean diameter among the segments (four vessels and 1 synthetic prosthesis) was not significantly different from each another.
The elastic index (E) calculated for all vessels was similar (p > 0.05). The ePTFE elastic index was significantly higher than that of all the vascular segments (Table 2).
Regarding the viscous index (V), fresh and cryopreserved femoral arteries showed non-significantly different values. A similar trend was followed by the fresh and cryopreserved saphenous veins. However, viscous index differences between venous and arterial conduits were statistically significative. The viscous index for the ePTFE was almost zero and was significantly different from all the vessels studied (Table 2).
The pulse wave velocity (PWV), a clinical parameter used as an indicator of elasticity, was similar among the vessels, while the ePTFE showed the highest PWV, as it was expected, taking into account its high elastic index (Table 2).
VII. DISCUSSION
Biomechanical properties of veins, arteries and synthetic prostheses have been associated with intimal hyperplasia development. More specifically, it has been suggested that an increment in compliance mismatch might induce this proliferative vascular stenosis (Haruguchi and Teraoka, 2003).
Our results demonstrated that both, elastic index and PWV have similar values for the fresh and cryopreserved arteries and veins. On the contrary, the ePTFE studied segments were stiffer than the vascular segments evaluated. These findings suggest that AVG performed using this material will have a higher elastic mismatch than that obtained with the AVF. Additionally, the ePTFE showed a negligible viscosity value, while fresh and cryopreserved human vessels showed a viscoelastic behaviour.
As was shown above, ePTFE, the synthetic prosthesis mostly used in vascular access surgical procedures has very different viscoelastic properties than those observed in the biological conduits analyzed in this work. This has to do with the high levels of intimal hyperplasia observed in the literature. We conclude that cryopreserved veins and arteries show similar biomechanical behavior than that observed in fresh vessels. In this sense, cryopreserved vessels could be considered as an alternative in the construction of AVG.
ACKNOWLEDGMENT
This work was partially supported by research grants Programa Desarrollo de las Ciencias Básicas (PEDECIBA), República Oriental del Uruguay. The authors gratefully acknowledge the technical assistance of Mr. Elbio Agote.
REFERENCES
1. Añel, R.L., A.S. Yevzlin and P. Ivanovich, "Vascular access and patients outcomes in hemodialysis: questions answered in recent literature", Artificial organs, 27, 237-241 (2003). [ Links ]
2. Armentano, R.L., J. Levenson, J.G. Barra, J. Levenson, E.I. Cabrera Fischer, G.J. Breitbart, R.H. Pichel and A. Simon, "Assessment of elastin and collagen contribution to aortic elasticity in conscious dogs", Am J Physiol 260 (Heart Circ Physiol)29, 1870-1877 (1991). [ Links ]
3. Bia, D., R.L. Armentano, Y. Zócalo, W. Barmak, E. Migliaro and E. Cabrera Fischer, "In vitro model to study arterial wall dynamics through pressure-diameter relationship analysis", Latin American Applied Research, 35, 217-224 (2005). [ Links ]
4. Cabrera Fischer, E.I.,R.L. Armentano, F.M. Pessana, S. Graf, L. Romero, A.I. Christen, A. Simon and J. Levenson, "Endothelium-dependent arterial wall tone elasticity modulated by blood viscosity", Am. J. Physiol Heart Circ Physiol, 282, 389-394 (2002). [ Links ]
5. Cabrera Fischer E.I., D. Bia Santana, G.L. Cassanello, Y. Zócalo, E.V. Craword, R. Casas and R.L. Armentano, "Reduced elastic mismatch achieved by interposing vein cuff in expanded polytetrafluoroethylene femoral bypass decreases intimal hyperplasia", Artificial Organs, 29, 122-130 (2005). [ Links ]
6. Canaud, B., "Venous access for haemodialysis - facts and thoughts", Business briefing: global healthcare - advanced medical technologies (2004). [ Links ]
7. Cernadas, M.R., M. Grandjean and M.A. Tosi, "Vascular access patency and complications: a comparison of brachiobasilic AV fistulas and PTFE brachioaxillary bridge AV fistulas in hemodialysis patients", Dialysis & Transplantation, 32, 694-697 (2003). [ Links ]
8. Cinat, M.E., J. Hopkins and S.E. Wilson, "A prospective evaluation of PTFE graft patency and surveillance techniques in hemodialysis access", Sixteenth Annual Meeting of the Southern California Vascular Surgical Society, La Jolla, CA, April 24-26 (1999). [ Links ]
9. Ezzahiri, R., M.S. Lemson, P.J.E.H.M. Kitslaar, K.M.L. Leunissen and J.H.M. Tordoir, "Haemodyalisis vascular access and fistula surveillance methods in the Netherlands", Nephrol Dial Transplant, 14, 2110-2115 (1999). [ Links ]
10. Haruguchi, H. and S. Teraoka, "Intimal hyperplasia and hemodynamic factors in arterial bypass and arteriovenous grafts: a review", J Artif Organs, 6, 227-235 (2003). [ Links ]
11. Hofstra, L., D.C.J.J. Bergmans, K.M.L. Leunissen, A.P.G. Hoeks, P.J.E.H.M. Kitslaar, M.J.A.P. Daemen and J.H.M. Tordoir, "Anastomotic intimal hyperplasia in prosthetic arteriovenous fistulas for hemodialysis is associated with initial high flow velocity and not with mismatch in elastic properties", J Am Soc Nephrol, 6, 1625-1633 (1995). [ Links ]
12. Johnson, M.S., G. McLennan, S.G. Lalka, R.M. Whitfield and R.G. Dreesen, "The porcine hemodialysis access model", J Vasc Interv Radiol, 12, 969-977 (2001). [ Links ]
13. Khosla, N. and S.N. Ahya, "Improving dialysis access management", Seminars in Nephrology, 22, 507-514 (2002). [ Links ]
14. Kohler, T.R. and T.R. Kirkman, "Dialysis access failure: a sheep model of rapid stenosis", J Vasc Surg., 30, 744-751 (1999). [ Links ]
15. Rodriguez, J.A., L. Armadans, E. Ferrer, A. Olmos, S. Codina, J. Bartolome, J. Borrellas and L. Piera, "The function of permanent vascular access", Nephrol Dial Transplant, 15, 402-408 (2000). [ Links ]
16. Roy-Chaudhury, N.B.S., A. Narayana, P. Desai, M. Melhem, R. Munda, H. Duncan and S.C. Heffelfinger, "Hemodiálisis vascular access dysfunction from basic biology to clinical intervention", Advances in Renal Replacement Therapy, 9, 74-84 (2002). [ Links ]
17. Sivanesan, S., T.V. How and A. Bakran, "Sites of stenosis in AV fistulae for haemodialysis access", Nephrol Dial Transplant, 14, 118-120 (1999). [ Links ]
18. Tordoir, J.H., F.M. Van Der Sande and M.W. De Haan, "Current topics on vascular access for hemodialysis", Minerva Urol Nefrol, 56, 223-235. (2004). [ Links ]
19. Turmel-Rodrigues, L., "Stenosis and thrombosis in haemodialysis fistulae and grafts: the radiologists point of view", Nephrol Dial Transplant, 19, 306-308 (2004). [ Links ]
20. Vassalotti, J.A., A. Falk, V. Teodorescu and J. Uribarri, "The multidisciplinary approach to hemodiálisis vascular access at the Mount Sinai Hospital", The Mount Sinai Journal of Medicine, 71, 94-102 (2004). [ Links ]
21. Wilson, S.E., "New alternatives in management of the infected vascular prosthesis", Surg Infect (Larchmt), 2, 171-177 (2001). [ Links ]
22. Young, E.W., D.M. Dykstra, D.A. Goodkin, D.L. Mapes, R.A. Wolfe and P.J. Held, "Hemodialysis vascular access preferences and outcomes in the dialysis outcomes and practice pattern study (DOPPS)", Kidney International, 61, 2266-2271 (2002). [ Links ]
23. Zarin, M., I. Ahmad, D. Waheed, A. Zeb, N. Anwar, V. Aslam, T. Ayub and Z. Haider, "Arteriovenous fistula construction in chronic haemodialysis patients: comparison of end-to-side and side-to-to-side techniques", JCPSP, 14, 619-621 (2004). [ Links ]
Received: November 17, 2005.
Accepted for publication: July 17, 2006.
Recommended by Editor E. Dvorkin.














