Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Latin American applied research
versión impresa ISSN 0327-0793
Lat. Am. appl. res. v.37 n.3 Bahía Blanca jul. 2007
Electrocatalytic oxidation of ethanol on platinum nanoparticles modified polyaniline electrode
D. A. Fungaro, A. Oliveira-Neto and M. Linardi
Institute for Energy and Nuclear Research, Av. Prof. Lineu Prestes, 2242, CEP 05508-000, São Paulo, SP, Brazil dfungaro@ipen.br
Abstract — The electrocatalytic properties of platinum nanoparticles incorporated into polyaniline films was investigated for oxidation of ethanol in sulfuric acid solutions. The electropolymerization of aniline and the electrochemical depositions of plati-num particles into the polyaniline film were achieved by cyclic voltammetry. The effect of the formation conditions of platinum-modified polyaniline films (as cycle numbers, sweep rates) and alcohol concentra-tion were evaluated. The platinum-modified polyani-line films formed by the optimization conditions can be used as a convenient conducting substrate for the ethanol electrooxidation.
Keywords — Polyaniline Films. Ethanol. Platinum Particles.
I. INTRODUCTION
Conducting polymers are a very important materials which are currently being investigated with regard their application in energy storage, microelectronics, electro-chromic displays, electrocatalysis, chemical sensors, etc. (Inzelt et al., 2000; Fungaro, 2001, 2004). These polymers, especially polyaniline, are usually used as matrix to incorporate noble metal catalysts in the application for electrooxidation of small molecules such as hydrogen, methanol and formic acid, etc. (Kost et al., 1988; Gholamian and Contractor, 1990; Hable and Wrighton, 1991). The reason for incorporating metallic particles into porous matrixes is to increase the specific area of these materials and thus improve catalytic efficiency. Another reason is the higher tolerance of the platinum particles to poisoning effect due to adsorption of CO species, in comparison with the serious problem of poisoning effect on bulk platinum electrodes. On the other hand, it is of great importance to investigate the catalytic activity of low loading platinum catalysts and to determine the lowest metal loading necessary to reach the practical performances in terms of current density and power density because of relatively high cost of catalytic material.
Platinum particles are generally electrodeposited in-to the polyaniline films at a constant potential (Croissant et al., 1998; Ficicioglu and Kadrigan, 1997; Mikhaylova et al., 2001; Laborde et al., 1994; Kitani et al.
Ethanol, among other small organic molecules, offers an attractive alternative as a liquid fuel with respect to methanol because is less toxic and it is the major renewable biofuel from the fermentation of biomass. However, few studies have been reported so far regarding ethanol oxidation on polyaniline film electrode (Hable and Wrighton, 1993). In view of its application pros-pects, fundamental insight for the electrooxidation of ethanol is important.
The purpose of the present work was to study the electrooxidation of ethanol as function of formation condi-tions of platinum-modified polyaniline films (cycle numbers, sweep rates) and alcohol concentration.
II. EXPERIMENTAL
All the solutions were prepared with ultrapure water Millipore Milli-Q. Aniline (Aldrich) was purified by distillation with zinc dust and stored under nitrogen gas. All other reagents were of analytical grade and were used as-received: ethanol (Merck), H2SO4 (Merck) e H2PtCl6. 6H2O (Acros Organics). The experiments were performed at ambient temperature (25 C) and under a nitrogen atmosphere.
Electrochemical experiments were carried out using an Autolab 30 (EcoChemie - Netherlands) voltammetric analyzer. The cell assembly contained a 3 mm diameter glassy carbon electrode as working electrode, a plati-num wire as auxiliary electrode and a Ag/AgCl (3 mol L-1 KCl) as reference electrode.
The polyaniline films were formed by cyclic voltam-metry at 50 mV s-1 between - 0.16 and + 0.94 V in solu-tion of 0.1 mol L-1 of aniline + 0.5 mol L-1 H2SO4. The film thickness, varied by changing the number of poten-tial cycles, was estimated using the cathodic charge value of the last cycle during film formation (Genies and Tsintavis, 1986). The dispersion of platinum into the polyaniline films was performed by cyclic voltam-metry at 50 mV s-1 between -0.063 and +0.84 V in solution of 3 mmol L-1 of H2PtCl6 + 0.5 mol L-1 H2SO4, except for specific indication of sweep rates used. Prior to the reduction the electrode was soaked in the hexa-chloroplatinic acid solution for 15 min. The amount of platinum deposited into the polyaniline film can be estimated from measurements of the charge involved during the reduction of Pt4+ to Pt0 (Laborde et al., 1994).
Electrocatalytic oxidation of ethanol on polyaniline films modified with platinum particles was examined in 0.5 mol L-1 CH3CH2OH + 0.5 mol L-1 H2SO4 by cyclic voltammetry at 5 mV s-1 between - 0.063 and 0.74 V.
III. RESULTS AND DISCUSSION
A. Thickness of polyaniline films
Polyaniline is a very interesting material because is ho-mogeneous, strongly adherent to the support, and chemically stable in acid medium. Electropolymeriza-tion offers the possibility of controlling the thickness and homogeneity of the conducting polymer film on the electrode surface.
The effect of polyaniline films on the electrodeposition of platinum particles and the subsequent electrocatalytic activity for ethanol oxidation was studied by changing the cycle sweep numbers to 13, 15 and 17. The thickness of the polyaniline films electropolymerized with the above three cycle numbers corresponds to 0.05, 0.08 e 0.1 μm, respectively. Figure 1 shows the effect of thick-ness of polyaniline films modified with same amount of platinum particles (25 cycles) on the oxidation of ethanol.

Fig 1. Effect of thickness of polyaniline films on the oxidation of 0.5 mol L-1 CH3CH2OH+0.5 mol L-1 H2SO4 at 5 mV s-1.
The current of ethanol oxidation with the thicker polyaniline films (0.1 μm) falls remarkably compared to that with two thinner polyaniline films due the more difficulty for the distribution of platinum particles in three dimensions. The superimposition of the curves during the positive and negative sweeps indicates that strongly chemisorbed posing species are not formed extensively (Laborde et al., 1994). The poisoning effect was less significant with the polyaniline film of intermediate thickness (0.08 μm), which was used as the matrix to electrodeposit platinum particles in the following experiments.
B. Effect of cycle numbers of platinum electrodeposition
The electroactivity for ethanol oxidation depends on the amount of deposited platinum. The platinum-modified polyaniline electrodes were prepared with different platinum loadings. The platinum particles were electro-deposited into polyaniline films (0.08 μm) by cyclic voltammetry with same potential limits and sweep rate, but the cycle numbers increase from 15, 25 to 50. As expected, the platinum loadings increased successively with increasing cycle numbers of platinum electrode-position. The platinum loadings calculated by the method used in literature (Laborde et al., 1994) were 8.1, 9.3 e 16.4 μg cm2, corresponding to the cycle num-bers 15, 25 and 50 used in the electrodeposition of platinum particles, respectively. Figure 2 shows the effect of cycle numbers of platinum electrodeposition on the oxidation of ethanol.
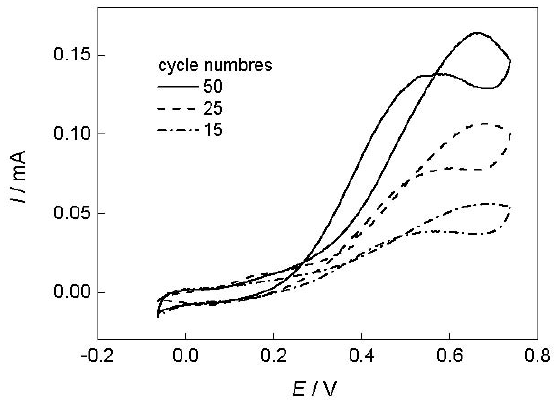
Fig 2. Effect of cycle numbers of platinum electrode-position on the oxidation of 0.5 mol L-1 CH3CH2OH + 0.5 mol L-1 H2SO4 at 5 mV s-1.
The anodic peak currents, which appear in the positive going sweeps, increased with the amount of deposited platinum. The platinum nanoparticles electrodeposited up to the 50 cycles were sufficiently dispersed in the overall structure of the polyaniline film. Figure 3 represents the variation of the maximum current density as a function of the amount of deposited platinum. The current density increase regularly up to 9.5 μg cm2, then reaches a plateau. The decrease observed after 16.4 μg cm2 is probably due to the formation of platinum clusters, leading to a smaller dispersion (Laborde et al., 1994).

Fig 3. Maximum current density as a function of the amount of deposited platinum on the oxidation of 0.5 mol L-1 CH3CH2OH + 0.5 mol L-1 H2SO4 at 5 mV s-1.
C. Effect of sweep rates of platinum electrodeposition
The effect of sweep rate of platinum electrodeposition on the electrooxidation of ethanol was studied. The platinum particles are electrodeposited into polyaniline films (0.08 μm) by cyclic voltammetry with the same potential limits (-0.063 to + 0.84 V), and same cycle numbers (25 times), while the sweep rates increase from 10 to 100 mV s-1. The current peak of ethanol oxidation increases with the decrease of the sweep rate from 100 to 10 mV s-1 (Fig. 4).

Fig 4. Effect of sweep rates of platinum electrodeposi-tion on the oxidation of 0.5 mol L-1 CH3CH2OH + 0.5 mol L-1 H2SO4 at 5 mV s-1.
The current for ethanol oxidation reaches the highest value (about 0.101mA) at the lowest sweep rate (10 mV s-1), owing to highest platinum loading (24.5μg cm-2). However, since the sweep rate is slower, it will spend much longer time (75 min) compared to other two rates of 80 and 50 mVs-1 (9 and 15 min, respectively) to accomplish the electrodeposition of platinum particles and may lead to a certain extent of degradation of polyaniline matrix. The platinum particles electrodeposited with sweep rate of 100 mV s-1 have no electrocatalytic actives on the oxidation of ethanol. Niu et al. (2003) found that the electrocatalytic activities of platinum particles electrodeposited into polyaniline films with sweep rate of 100 mV s-1 on methanol oxidation was not remarkable and decreased quickly during the con-secutive potential cycle. From the point of view of both electrocatalytic activities of platinum particles and op-timization of polymer matrix properties, therefore, an intermediate sweep rate of 50 mV s-1 is selected to de-posit platinum particles within the polymer matrix.
D. Effect of the concentration of ethanol
The investigation of the effect of varying the ethanol concentration on oxidation electrocatalytic at polyani-line/Pt electrode was examined and was shown in Fig. 5.

Fig 5. Effect of alcohol concentration on the oxidation of CH3CH2OH + 0.5 mol L-1 H2SO4 at 5 mV s-1.
The concentration of ethanol was varied from 0.10 to 1.0 mol L-1 and maximum current peak of oxidation was obtained for 0.50 mol L-1. The main products observed during ethanol electrochemical oxidation on platinum electrodes are acetaldehyde, acetic acid and carbon dioxide, with predominance for acetic acid and acetaldehyde (Souza et al., 1997; Ianniello et al., 1999; Fujiwara et al., 1999; Iwasita, 2002; Iwasita and Pastor, 1994). The formation of CO2 goes through strongly chemisorbed intermediates, CO and hydrogenated absorbates, whereas acetaldehyde, etc, are formed through a weakly adsorbed ethanol intermediate (Iwasita and Pastor, 1994). It has been reported that the production of the partially oxidized acetaldehyde increases as the ethanol concentration increases (Bergamaski, 2003). The activity of electrode is limited because polyaniline can be degraded by the acidic solutions of acetaldehyde.
IV. CONCLUSIONS
Polyaniline films can be used as a convenient conduct-ing substrate for the dispersion of platinum nanoparticles for electrooxidation of ethanol. It was found that the oxidation of ethanol depends on the nature of both polymer matrix and platinum particles. The optimum catalytic activity for a polyaniline film of 0.08 μm thickness was obtained for the following conditions: amount of deposited platinum = 9.5 μg cm-2, sweep rate = 50 mV s-1 and ethanol concentration = 0.50 mol L-1. Experiments of current-potential curves using platinum-modified polyaniline films in direct ethanol fuel cell are currently in progress.
ACKNOWLEDGEMENTS
The financial support of Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) are gratefully acknowledged.
REFERENCES
1. Bergamaski, K., J.F. Gomes, B.E. Goi and F.C. Nart, "Effect of alcohol concentration and electrode composition on the ethanol electrochemical oxidation", Ecl. Quim., 28, 87-92 (2003). [ Links ]
2. Croissant, M.J., W.T. Napporn, J.M. Léger and C. Lamy, "Electrocatalitic oxidation of hydrogen at platinum-modified polyaniline electrodes", Electrochim. Acta, 43, 2447-2457 (1998). [ Links ]
3. Ficicioglu, F. and F. Kardigan, "Electrooxidation of metanol on platinum doped polyaniline electrodes: deposition potential and temperature effect", J. Electroanal. Chem., 430, 179-182 (1997). [ Links ]
4. Fujiwara, N., K.A. Friedrich and U. Stimming, "Ethanol oxidation on PtRu electrodes studied by differential electrochemical mass spectrometry", J. Electroanal. Chem., 472, 120-125 (1999). [ Links ]
5. Fungaro, D.A., "Sulfonated polyaniline coated mercury film electrodes for voltammetric analysis of metals in water", Sensors, 1, 206-214 (2001). [ Links ]
6. Fungaro, D.A., "Determinação voltamétrica de Cd e Zn usando eletrodo modificado com polianilina sulfonada", Ecl. Quim., 2, 63-69 (2004). [ Links ]
7. Genies, E.M. and C. Tsintavis, "Electrochemical behaviour, chronocoulometric and kinetic study of the redox mechanism of polyaniline deposits", J. Electroanal. Chem., 200, 127-145 (1986). [ Links ]
8. Gholamian, M. and A.Q. Contractor, "Oxidation of formic acid at platinum microparticles dispersed in a polyaniline matrix: influence of long-range order and metal-polymer interaction", J. Electroanal. Chem., 289, 69-83 (1990). [ Links ]
9. Hable, C.T. and M.S. Wrighton, "Electrocatalytic oxidation of methanol by assemblies of platinum-Tin catalyst particle in a conducting polyaniline matrix", Langmuir, 7, 1305-1309 (1991). [ Links ]
10. Hable, C.T. and M.S. Wrighton, "Electrocatlytic oxida-tion of methanol and ethanol: A comparison of platinum-Tin and platinum-ruthenium catalyst particles in a conducting polyaniline matrix", Langmuir, 9, 3284-3290 (1993). [ Links ]
11. Ianniello, R., V.M. Schmidt, J.L. Rodriguez and E. Pastor, "Electrochemical surface reactions of intermediates formed in the oxidative ethanol adsorption on porous Pt and PtRu", J. Electroanal. Chem., 471, 167-179 (1999). [ Links ]
12. Inzelt, G., M. Pireni, J.W. Schultze and M.A. Vorotynstev, "Electron and proton conducting polymers: recent development and prospects", Electrochim. Acta, 45, 2403- 2421(2000). [ Links ]
13. Iwasita, T. and E. Pastor, "A DEMS and FTIR spectroscopy investigation of adsorbed ethanol on polycrystalline platinum", Electrochim. Acta, 39, 531-537 (1994). [ Links ]
14. Iwasita, T., "Fuel cells: spectroscopic studies in the electrocatalysis of alcohol oxidation", J. Braz. Chem. Soc., 13, 401-409 (2002). [ Links ]
15. Kelaidopoulou, A., E. Abelidou, A. Papoutsis, E.K. Polychroniadis and G. Kokkinidis, "Electrooxidation of ethylene glycol on Pt-based catalysts dispersed polyaniline." J. Appl. Electrochem.28, 1101-1106 (1998). [ Links ]
16. Kelaidopoulou, A., A. Papoutsis, G. Kokkinidis, W.T. Napporn, J.M. Léger and C. and Lamy, "Electrooxidation of beta-D(+) glicose on bare and upd modified particles dispersed in polyaniline", J. Appl. Electrochem., 29, 101-107 (1999). [ Links ]
17. Kitani, A., T. Akashi, K. Sugimoto and S. Ito, "Electrooxidation of methanol on platinum modified polyaniline electrode", Synth. Met., 121, 1301-1302 (2001). [ Links ]
18. Kost, K.M., D.E. Bartak, B. Kazaee and T. Kuwana, "Electrodeposition of platinum microparticles into polyaniline films with electrocatalytic applications", Anal. Chem., 60, 2379-2384 (1988). [ Links ]
19. Laborde, H., J.M. Léger and C. Lamy, "Electrocatalytic oxidation of methanol and C1 molecules on highly dispersed electrodes Part 1: Platinum in polyaniline", J. Appl. Electrochem., 24, 219-226 (1994). [ Links ]
20. Mikhaylova, A.A., E.B. Molodkina, O.A. Khazova and V.S. Bagottzky, "Electrocatalytic and adsorption properties of platinum microparticles electrodeposited into polyaniline films", J. Electroanal. Chem., 509, 119-127 (2001). [ Links ]
21. Napporn, W.T., H. Laborde, J.M. Léger and C. Lamy, "Electrooxidation C1 molecules at Pt-based catalyst highly dispersed into a polymer matrix: effect of the method of preparation", J. Electroanal. Chem., 404, 153-159 (1996). [ Links ]
22. Niu, L., Q. Li, W. Fenghua, X. Chen and H. Wang, "Formation optimization of platinum-modified polyaniline films for the electrocatalytic oxidation of methanol", Synth. Met., 139, 271-276 (2003). [ Links ]
23. Souza, J.P.I., F.J.B. Rabelo, I.R. Moraes and F.C. Nart, "Performance of a co-electrodeposited Pt-Ru electrode for the electro-oxidation of ethanol studied by in situ FTIR spectroscopy", J. Electroanal. Chem., 420, 17-20. (1997). [ Links ]
Received: December 6, 2005.
Accepted: December 7, 2006.
Recommended by Subject Editor Ana Lea Cukierman.














