Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Latin American applied research
versión impresa ISSN 0327-0793
Lat. Am. appl. res. v.38 n.4 Bahía Blanca oct. 2008
Reduction of sulfur levels in kerosene by Pseudomonas sp strain in an airlift reactor
O. F. Sanchez1, C. J. Almeciga-Diaz1, E. Silva1, J. C. Cruz2, J. D. Valderrama2 and L. A. Caicedo2
1 Dto. de Farmacia, Universidad Nacional de Colombia, Sede Bogotá. A.A 14490, Colombia
ofsanchezm@unal.edu.co, cjalmeciga@javeriana.edu.co, esilvag@unal.edu.co
2 Dto. de Ingeniería Química, Universidad Nacional de Colombia, Sede Bogotá. Cll 45 Kra 30, Colombia
jccruzj@unal.edu.co, jdvalderramar@unal.edu.co, lacaicedom@unal.edu.co
Abstract — Combustion of organic sulfur from fossil fuels can produce acid rain that deteriorates the environment and infrastructure. Sulfur removal by microorganism has appeared as an alternative for this challenge. In this work, biodesulfurization of 50:50 water-kerosene emulsions were carried out at 100 mL scale and in a 0.01 m3 airlift reactor with resting cells of the reference strain ATCC 39327 and Pseudomonas native strains Nº 02, 05 and 06. The reactor conditions were 30°C, pH 8.0 and 0.34 m3 h-1 air flow. After 7 culture days, the mean sulfur removal for the strains Nº 06 and ATCC 39327 was 64 and 53%, respectively, with a mean calorific power loss of 4.5% for both strains. The use of the native strain Nº 06 and the designed airlift reactor is shown as an alternative for biodesulfurization process and constitute a first step for its scale-up to pilot plant.
Keywords — Biodesulfurization. Airlift Reactor. Kerosene. Pseudomonas.
I. INTRODUCTION
The main problem in fossil fuel combustion is sulfur and nitrogen conversion to oxides. These oxides make part of acid rain which deteriorates the environment and infrastructures (Monticello and Finnerty, 1985; Izumi et al., 1994; Oshiro et al., 1995; Sagardia et al., 1975). Furthermore, SO2 emission is a precursor of sulfated aerosols that are considerated as one of the main solid particulate agents that affect the human health (Sagardia et al., 1975).
Worldwide sulfur levels in fuels are between 15 ppm in North America and 5000 ppm in some countries of Africa and Middle East (United Nations Environment Programme, 2007a). In Latin-America, Brazil, Bolivia, Chile, and Argentina have levels between 500 and 2000 ppm, while Venezuela, Ecuador, Peru, Paraguay, Uruguay and Centro-America have sulfur levels up to 5000 ppm in diesel. Mexico is the only Latin country with sulfur levels less than 500 ppm (Felix, 2007; United Nations Environment Programme 2007b). Since 1995, in Colombia some decrees have been created in order to control the sulfur level in fuels. They established for the year 2008 a level up to 300 and 500 ppm in gasoline and diesel, respectively. However, actual sulfur levels are 1200 ppm in Bogotá and 4500 ppm in other places (United Nations Environment Programme 2007b).
The microbiological degradation of sulfur compounds in fossil fuels, biodesulfurization (BDS), has arisen as an alternative instead of the catalytic process of hydrodesulfurization (HDS) which leaves sulfur remnants between 300 and 500 ppm (McFarland et al., 1998). In contrast, some microorganisms with sulfur removal activity show advantages like high sulfur specificity without fuel calorific power lost. BDS processes can be carried out under moderated conditions and allow to have lower sulfur remnant levels than HDS. However, these microorganisms have a high sensibility to organic solvents (Monticello, 2000). At present there are about fifty approved patents for desulfurization processes on derived fossil fuels from petroleum and coal which use microorganisms, enzymes and vectors that keep genes of desulfurization metabolic pathways (Biodesulfurization in United States Patent and Trademark, 2007). It is estimated that the BDS process could reduced CO2 emissions and up to 80% less energy consumed per barrel compared to HDS (Le Borgne and Quintero, 2003).
In addition to elemental sulfur, sulfate, sulfite and thiosulfate, sulfur can be present in more than 200 organic compounds like sulfides, thiols, thiophenes, mercaptans, diphenilsulfides, benzothiophenes and dibenzothiophenes (Monticello and Finnerty, 1985; Fedorak and Westlake, 1983; Fedorak and Grbic-Galic, 1991; Lee et al., 1995). Dibenzothiophene (DBT) and its derivates have been the most studied compounds due their resistant to HDS process (Monticello, 2000). Three metabolic pathways for the desulfurization of DBT have been reported: (a) 4S pathway where the sulfur is selectively removed of the molecules without a significant lost in the fuel calorific power, (b) Kodoma's pathway where hydrophilic products are obtained with a lost in the fuel calorific power, and (c) a completely oxidative pathway where CO2, SO3= and H2O are produced. (Oshiro et al., 1995; Lee et al., 1995; Armstronset al., 1995; Omori et al., 1992; Konishi et al., 1986; Kodama et al., 1973; Kodama et al., 1970). The enzymes and genes involved in 4S pathway have been all elucidated and constitute the Dsz desulfurization system that involves four enzymatic steps. Cofactors participation (NADH and FMNH2) and the complexity of the metabolic pathway force to work with whole cells (Le Borgne and Quintero, 2003).
The sulfur removal from fuels and organic solvents spiked with DBT or DBT-derived compounds have been reported for some strains of Pseudomonas (Huertas et al., 1998; McFarland et al., 1998; Izumi et al., 1994; Laborde and Gibson, 1977), Corynebacterium (Omori et al., 1992), Arthrobacter (Konishi et al., 1986), Beijerinkia (Laborde and Gibson, 1977), Rhodococcus (Monticello and Finnerty, 1985; Fedorak and Westlake, 1983; Laborde and Gibson, 1977) Desulfovibrio (Armstrons et al., 1995), Rhizobiummeliloti (Frassinetti et al., 1998) and Cunninghamella elegans (Crawford and Gupta, 1990). Pseudomonas strains have shown BDS activity and resistance to organic solvents like n-decane, toluene, dimethylphtalate, etc., which represent an important characteristic for bioremedation processes. (Cruden et al., 1992; Fedorak and Grbic-Galic, 1991; Sagardia et al., 1975).
The choice of the process design for BDS depends on the economics of the production and recovery of the desired product. Recycling and operational stability of the biocatalysts are also important. Stirred tanks have been the most used reactors in BDS and there is just a few reports using airlift reactors, emulsion phase contactors and fluidized bed reactors (Le Borgne and Quintero, 2003; Monticello, 2000; McFarland et al., 1998). New processes include the use of multiple-staged airlift reactors to overcome poor reaction kinetics at low sulfur concentrations and reduce mixing costs (Monticello, 2000). Nevertheless, scale-up this process is a problem and there are not published reports that describe its engineering or operation to estimate it economically. The estimated capital costs for a BDS process is about two-thirds of a HDS process, whereas the operation cost is reduced by a 15% (Le Borgne and Quintero, 2003).
Previously, we reported the sulfur remove ability over n-hexane spiked with DBT of ATCC 39327 and 23 native strains of Pseudomonas sp. ATCC 39327 and native strains Nº 02, 05 and 06 used the 4S enzymatic pathway to remove sulfur in a 9.4, 6.0, 7.6 and 6.6%, respectively. Also, was proved that the proportion of 50:50 water:n-hexane and surfactant presence (ethanolamine oleate) had effect over the Pseudomonas sp. sulfur removal activity (Alméciga-Díaz et al., 2005; Sánchez et al., 2004).
In this work, the sulfur removal ability of ATCC 39327 and Pseudomonas sp. strains Nº 02, 05 and 06 over kerosene was initialed evaluated at 100 mL scale. Strain Nº 06 showed the highest sulfur removal levels and it was selected for BDS in an airlift reactor together with the reference strain. This system is shown as an alternative for biodesulfurization process and constitutes a first step for its scale-up to pilot plant.
II. METHODS
A. Microorganisms.
Pseudomonas sp. native strains and ATCC 39327 were stored in 10% free-fat milk and 3% meso-inositol at -20±1°C. Native strains were obtained from Corporación Autonoma Regional (CAR) wastewater ponds located in Cundinamarca, Colombia (Salazar, 1996). All microorganisms were biochemical characterized as Pseudomonas sp. (Sánchez et al., 2004).
B. Reactor Description.
Biodesulfurization assays were made in a concentric draft-tube airlift reactor (Fig. 1). Reactor vessel was 0.15 m in diameter and its overall height was 0.60 m. The draft-tube, 0.10 m in internal diameter and 0.30 m tall, was located 0.10 m above the bottom of the tank. The vessel was sparged in the concentric zone through a 0.005 m diameter sparger. The riser and downcomer area ratio was 0.80. The working volume and the overall volume of the reactor was 0.01 y 0.008 m3, respectively. A dissolved oxygen electrode (HI8043 Dissolved Oxygen Meter, Hanna Instruments) and a pH-meter (HI98170 pH-meter with temperature probe, Hanna Instruments) were placed in the downcomer zone at 0.15 and 0.25 m above the base of the tank, respectively.
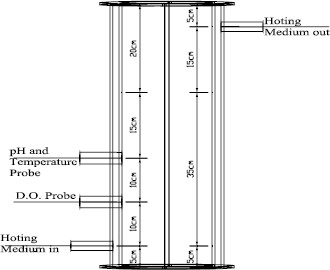
Figure 1. Airlift Reactor diagram.
Air from ~700KPa mains was supplied to the reactor through a filter, pressure regulator, floor control valve and rotameter.
C. Hydrodynamic Characterization
Mixing time and liquid circulation velocity was carried out with water, while the overall gas-liquid volumetric mass transfer coefficient (kLa) was carried out with water, kerosene and emulsion (50:50 kerosene:water and surfactant). These parameters were evaluated for three different air flows (0.142, 0.283 and 0.425 m3 s-1) and measures were made by triplicate. Data were analyzed with Statgraphics Plus v. 5.0.
The mixing time and liquid velocity was determined by the acid tracer technique (Shariati et al., 2007; Sánchez Mirón et al., 2004; Chisti, 1989). The mixing time was defined like the time needed to reach 95% of the final tracer concentration at steady-state from the instance of tracer input. Once the reactor was filled with water, HCl (35 % w/V) was added until pH 2 and later air was bubbled by 20 minutes (0.34 m3 h-1) to remove any carbonate; later with NaOH (6 M) pH was established in 5.50±0.05. The acid tracer (1 mL of HCl 35% w/V) was added in the bioreactor through a port in the downcomer section. Dimensionless acid concentration [H+] was estimated with Eq. 1. While, mean liquid circulation velocity was calculated by Eq. 2.
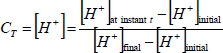 | (1) |
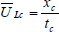 | (2) |
where xc is the circulation path length and tc is the average time for one complete circulation.
The kLa was measured with the dynamic gassing-in method (Shariati et al., 2007; Chisti and Jáuregui-Haza, 2002; Chisti, 1989). For each test the fluid was purged by bubbling nitrogen until a dissolved oxygen concentration lower than 5% of air saturation. Later the nitrogen flow was suspended, the exit of its bubbles was allowed and the air flow was established to the required condition. The increase in dissolved oxygen concentration was followed in the time until the fluid became nearly saturated with oxygen (>90%). The kLa was calculated as the slope of the linear equation:
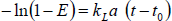 | (3) |
In Eq. 3, E is the fractional approach to equilibrium (Chisti, 1989) and can be estimated Eq. 4.
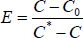 | (4) |
where C* is the saturation concentration of dissolved oxygen, C0 the initial concentration of dissolved oxygen at time t0 when a hydrodynamic steady-state has been reestablished (≤ 60 s) upon the beginning of aeration and C the dissolved oxygen concentration at any time t.
D. Pre-inoculum.
Microorganisms were initialed cultured in 0.3% CASO broth (Oxoid) at 30±1°C for 24 hours and then isolated in CVN agar (5% violet crystal solution, 2 mL L-1; CASO, 3 g L-1; agar-agar 15 g L-1; 5% nitrofurantoine in dimethylformamide, 7mL L-1) at 30±1°C for 24 hours. Finally a colony was inoculated in 200 mL of Luria-Bertani both (LB broth; Triptone 10 g L-1; yeast extract 5 g L-1; NaCl, 0.5 g L-1; and glycerin, 1 mL L-1) and incubated at 30±1°C and 200 rpm up to an OD600 25±1%, equivalent to a resting cell population of 108 CFU mL-1 (Castro, 2000).
E. Fermentation Systems
Fermentations in 100 mL scale were carried out for each native and the reference strain by triplicate in 500 mL shake flasks. Culture medium was 50:50 aqueous: kerosene with 500 ppm of ethanolamine oleate (sur-factant). One milliliter of pre-inoculum (108 CFU mL-1) was inoculated in the aqueous phase conformed by a free-sulfur minimum salt medium (FSMSM), (Frassinetti et al., 1998). Shake flasks were incubated for seven days at 30±1°C and 200 rpm (Castro, 2000).
Fermentations in the airlift reactor were carried out in a 50:50 aqueous:kerosene ratio in presence of 3500 ppm of ethanolamine oleate. A 200 mL pre-inoculum of ATCC 39327 or strain Nº 6 was inoculated in 7.8 L of culture medium (FSMSM:kerosene). Fermentations were incubated for 7 days at 30°C and were carried out twice. A 50 mL sample was taken each 24 h for assays, and pH and dissolved oxygen were measured in the reactor. Air flow was 0.34 m3 h-1.
All he inntoculus were carried out with resting cells (Le Borgne and Quintero, 2003).
F. Assays
Phases were separated by centrifugation at 3000 g for 30 minutes. Viability and sulfate quantification, by turbidimetry assay with barium chloride at 420 nm, were carried out in the aqueous phase. Qualitative analysis of kerosene was carried out by HPLC (Waters 600) in serial acetonitrile extractions of kerosene phase. Chromato-graphic system used a NovaPak C-18 column (30 cm x 3.9 mm x 5 µm) and a mobile phase with lineal elution gradient of water-acetonitrile (47:53) at a flow rate of 1 mL/min. Peaks were registered at 254 nm and scattered through 210 and 400 by a diodes array detector (DAD - Waters 996) coupled to the HPLC. Spectrum of each peak was compared to the standard solution spectrums of DBT, DBT-sulfone and 2-hydroxybiphenyl (Crawford and Gupta, 1990, Alméciga-Díaz et al., 2005).
Kerosene calorific power was determinated in an adiabatic calorimetric pump (ICONTEC, 2003). Total sulfur was carried out by barium chloride in the condensed residue of the adiabatic calorimetric pump. Data were analyzed statistically using the software Statgraphics plus v. 5.0.
G. Viability
From the aqueous phase 100 µL were diluted with 900 µL of sterile water, serial dilutions were carried out up to 10-8 dilution and CFU count was measured after 12 hours incubation in CVN agar at 37ºC.
III. RESULTS AND DISCUSSION
A. Sulfur Removal from Kerosene in 100 mL scale
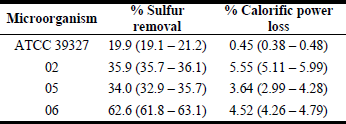
Initial kerosene calorific power and sulfur content was 49.7 KJ g-1 and 319 ppm, respectively. ATCC 39327 and strains Nº 02, 05 and 06 had an average sulfur removal of 19.9, 35.9, 34.0 and 62.6% and an average calorific power loss of 0.45, 5.55, 3.64 and 4.52%, respectively (Table 1). Kerosene chromatograms showed that neither DBT nor any of its metabolites were detected before and after culture, but after 7-culture days was noticed a decrease in the area of some non-identified peaks between 7.5 and 10.5 min.
Table 1. Average and range of sulfur removal and calorific power loss for kerosene in 100 mL scale.
For all strains, microorganism viability during the culture was around 108 CFU mL-1. This result is likewise with previous reports using the same strains in n-hexane spiked with DBT (Alméciga-Díaz et al., 2005; Sánchez et al., 2005). Since the microorganisms kept their viability along the culture, they could become in a sulfur removal alternative for desulfurization studies in two-phase fermentation systems with high molecular weight hydrocarbons. Although, viability and sulfur removal results over kerosene have not been reported, similar results were reported for a Psudomonas strain in p-xylene 50% (Cruden et al., 1992). However, P. putida and P. mendocina showed high sensitive to n-heptane concentrations greater than 10% (Huertas et al., 1998).
Strains 02 and 06 produced the greatest calorific power lost, 5.5 and 4.5, respectively (Table 1). This could be associated with the use of fuel like a carbon source or the generation of free-sulfur hydrophilic compounds (Monticello, 2000). Although, there are no reports that allows saying whether this is an important calorific power lost or not, these values are lower that obtained in DBT chemical oxidation followed by solvent separation of the corresponding sulfones that led to a 10% decrease in the fuel energetic value (Le Borgne and Quintero, 2003).
B. Reactor Characterization.
For different air flows the designed reactor showed a circulation liquid velocity range between 0.060 and 0.095 m s-1 with a maximum standard deviation of 8%. The mixing time range was between 38 and 27 s with a maximum standard deviation of 4%. These results are likewise to the reported for airlift reactors (Chisty, 1989).
For the different superficial air rates kerosene showed the highest kLa (Table 2). Although, there are no reported data for systems like these (water:kerosene emulsion and kerosene), these results are similar to reported by Subczynski and Hyde (1984) who found higher oxygen concentrations in organic compounds like sec-butyl and paraffin oil than in water. Also, Clarke et al. (2006) showed a kLa enhanced up to 4-folds in a 20% alkane (n-C13) – water system at 30°C, 1.5 vvm and an agitation rate in the range of 400 and 1000 r.p.m.
Table 2. Average kLa* measures in airlift reactor.
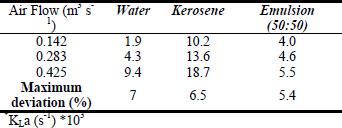
Oxygen supply to the microorganisms grown in the fermentors depends on the kLa. The kLa values were determined for three different air flows (Table 2). Although the highest value of kLa obtain was not very great we could hardly use better conditions for air feeding, especially for aeration rates. For the selected aeration rates is observed that the highest kLa is obtain with the highest air flow (0.425 m3 s-1). This is an important fact, now that besides oxygen is important for Pseudomonas growth, it is also important for the enzymatic reaction since in the 4S metabolic pathway there are three monooxygenases involved (Gray et al., 2003).
C. Sulfur Removal from Kerosene in Airlift Reactor.
Strain Nº 06 was selected for DBS assays in airlift reactor due to its high sulfur removal levels in 100 mL scale. Power capacity loss and percentage of sulfur removed were determinated after seven days culture in the airlift.
Previously, we showed that the presence of ethanolamine oleate increase near 4-fold the ATCC 39327 biodesulfurization activity respect to culture medium without surfactant (Sánchez et al., 2004). Due to the ionic characteristics of ethanolamine oleate, culture medium pH modification from 7.6 to 8.0 allowed reducing its amount from 5000 ppm to 3500 pm, getting the same phase separation time. This pH modification did not have any major effect over cell viability. For both strains, the airlift reactor culture conditions were the same (air flow 0.34 m3 h-1 and 30°C for 7 days).
After 7-culture days, sulfur removal for ATCC 39327 and strain Nº. 06 were 64 and 53 %, respectively (Fig. 2). It is observed a high biodesulfurization rate in the first 24h but no major changes are displayed in the last 48 h. Additional studies with ATCC 39327 have not displayed an increment in BDS activity after 12 days (data not shown). Similar results were obtained by Lee et al. (1995), who using a Gram-positive isolated bacterium did not enhance the sulfur removal after 8 days of culture.
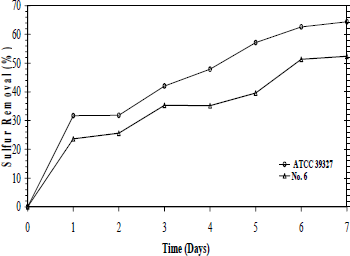
Figure 2. Sulfur removal level from Kerosene in airlift reactor by ATCC 39327 and native strain Nº 6.
The ATCC 39327 sulfur removal was 2.6-folds higher in the reactor than in the 100 mL scale. Since this is a mixed culture, it is possible that the airlift operation conditions (not presented at 100 mL scale) like aeration, gas renovation and flow pattern could improve the growth and metabolism of other microorganisms rather than Pseudomonas with BDS activity. On the other hand, sulfur removal with the native strain Nº 06 at reactor scale was reduced in about a 15%. This could be associated with less accurate oxygen and nutrients transport at the selected operation conditions in the reactor than in 100 mL culture. This was an expected finding, due to the normal decrease in the mass transfer during the scale up by heterogeneities in the mixing pattern (Hewitt and Nienow, 2007). Although, further evaluation of operational conditions with strain Nº 06 are necessary to improve the sulfur removal levels; these results show the BDS potential of strain Nº 6 in an airlift reactor.
Average calorific power loss with both strains was about 20 KJ g-1 (about a 4,5%) (Fig 3). As it was mentioned, this reduction in the calorific power is lower than previous BDS reports (Le Borgne and Quintero, 2003). The calorific power loss in the airlift reactor cultures with strain Nº 06 were similar to the obtained results in 100 mL culture, while ATCC 39327 showed a higher calorific power loss in the airlift reactor compare to 100 mL scale. This agrees with their biodesulfurization activity. Previously, we described that ATCC 39327 and strains Nº 02, 05 and 06, use the 4S pathway for sulfur removal from DBT (Alméciga-Díaz et al, 2005). In this pathway sulfur is removed like sulfate, while carbon backbone remains like 2-hydroxybiphenyl (McFarland et al., 1998; Monticello, 2000). However, chromatographic analysis of kerosene before and after fermentation did not show DBT or its metabolites, pointing to the presence of a different pathway on these Pseudomonas strains to carry out the BDS process, where sulfate should be one of the end products Additionally, the calorific power loss could be caused by the generation of water-soluble end products like in the Kodoma's pathway, where sulfur is removed like 3-hydroxy-2-formyl-benzotiophene, or the use of a completely oxidative pathways to CO2 and SO42+ (McFarland et al., 1998). In the same way, the higher calorific power loss observed with ATCC 39327 in the airlift reactor could be associated to the presence of other strains than Pseudomonas with different metabolic pathways. This hypothesis is withstood by the higher BDS activity observed for ATCC 39327 in the airlift reactor. However, further analyses are necessary to determine if the calorific power loss has a deleterious effect over the kerosene
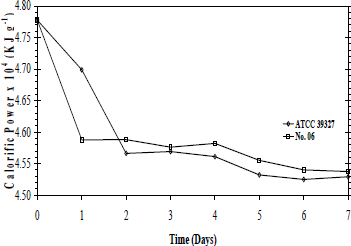
Figure 3. Kerosene calorific power in airlift reactor by ATCC 39327 and native strain Nº 06.
Sulfur removal percentages of this work were similar to obtained using Continuos Stirred Tank Reactor designed by Energy Biosystems Corporation which permitted a sulfur removal level between 50 to 70% over diesel (McFarland et al., 1998), and better than obtained with a similar system designed by Brookhaven National Laboratory which permitted a sulfur removal level between 25 to 35% (McFarland et al., 1998). Other bioreactors, like Fluidized Bed Reactors or Emulsion Phase Contactor (where biocatalyst is spread out in the fuel using a spray that make a fine emulsion or where biocatalyst is immobilized and fuel pass through it), are still in development and there is not data of their sulfur removal capacity (McFarland et al., 1998; Monticello 2000).
IV. CONCLUSIONS
Native strain Nº 06 is showed like a BDS alternative of fossil fuels due to its high sulfur removal percentage (53%), low calorific power loss (4,5%) and viability preservation. Scale-up process allowed establishing that hydrocarbon presence has a positive effect over KLa. Although there was not an increment on the sulfur removal level from 100 mL scale to airlift reactor scale, this paper show the potential use of native strain Nº 06 and airlift reactor for fossil fuels BDS.
REFERENCES
1. Alméciga-Díaz, C.J., O.F. Sánchez and E. Silva, "Evaluación de la actividad de sulfurizadora de aislados nativos de Pseudomonas spp. en presencia de hidrocarburo", Rev. Col. Biotec., 7, 15-24 (2005). [ Links ]
2. Armstrons, S., B. Sankey and G. Voordouw, "Conversion of debenzothiophene to byphenyl by sulfate-reducing bacteria isolated from oil field production facilities", Biotechnol. Lett., 17, 1133-1136 (1995). [ Links ]
3. Biodesulfurization in United States Patent and Trademark http://www.uspto.gov/. (2007). [ Links ]
4. Castro, S., "Estudio de la biodesulfurización del DBT (dibenzotiofeno) en un sistema emulsionado por acción de una cepa de Pseudomonas sp.", Tesis. Departamento de Ingeniería Química. Universidad Nacional de Colombia, Bogotá. (2000). [ Links ]
5. Clarke, K.G., P.C. Williams, M.S. Smit and S.T.L. Harrison, "Enhancement and repression of the volumetric oxygen transfer coefficient through hydrocarbon addition and its influence on oxygen transfer rate in stirred tank bioreactors", Biochem. Eng. J., 28, 247-232 (2006). [ Links ]
6. Chisti, Y., Airlift bioreactor, Elsevier, New York. (1989). [ Links ]
7. Chisti, Y. and U.J. Jáuregui-Haza, "Oxygen transfer and mixing in mechanically agitated airlift bioreactor", Biochem. Eng. J., 10, 143-153 (2002). [ Links ]
8. Crawford, D. and R. Gupta, "Oxidation of dibenzothiophene by Cunninghamella elegans", Curr. Microbiol., 21, 229-231 (1990). [ Links ]
9. Cruden, D., J. Wolfram, R. Rogers and D. Gibson, "Physiological properties of a Pseudomonas strain which grows with p-xylene in a two-phase (organic-aqueous) medium", Appl. Environ. Microbiol., 58, 2723-2729 (1992). [ Links ]
10. Fedorak, P. and W. Westlake, "Microbial degradation of organic compounds in Prudhoe Bay crude oil", Can. J. of Microbiol., 29, 291-296 (1983). [ Links ]
11. Fedorak, P. and D.Grbic-Galic, "Aerobic cometabolism of benzothiophene and 3-methylbenzothiophene", Appl. Environ. Microbiol., 57, 932-940 (1991). [ Links ]
12. Felix, J., "Fundamentos para la disminución de azufre en los combustibles de ALC: Enfoque sistémico. Desafíos de Refinación. Conferencias obre Azufre en Combustibles en Sur América", http://arpel.org (2007). [ Links ]
13. Frassinetti, S., L. Setti, A. Corti, P. Farrinelli, P. Montevecchi and G. Vallini, "Biodegradation of dibenzothiophene by a nodulating isolate of Rhizobiummeliloti", Can. J. of Microbiol., 44, 289-297 (1998). [ Links ]
14. Gray, K.A., G.T. Mrachkoyz and C.H. Squiresy, "Biodesulfurization of fossil fuels", Curr. Opin. Microbiol. 6, 229-235 (2003). [ Links ]
15. Hewitt, C. and A. Nienow, "The scale-up of microbial batch and fed-batch fermentation processes", J. Biotechnol., 131, S134-S135 (2007). [ Links ]
16. Huertas, M., E. Duque, S. Marqués and J. Ramos, "Survival in soil of different toluene-degrading Pseudomonas strains after solvent shock", Appl. Environ. Microbiol., 64, 38-42 (1998). [ Links ]
17. ICONTEC, Determinación del poder calorífico bruto de carbón y coque, NTC 2128, (2003). [ Links ]
18. Izumi, Y., T. Ohshiro, H.Ogino, Y. Hine and M. Shimao, "Selective Desulfurization of Dibenzothiophene by Rhodococcuserythropolis D-1", Appl. Environ. Microbiol., 60, 223-226 (1994) [ Links ]
19. Kodama, K., S. Nakatani, K. Umehara, K. Shimizu, Y. Minoda and K. Yamada, "Induction of dibenzothiophene oxidation by Pseudomonajainii", Agr. Biol. Chem., 34, 1320-1324 (1970). [ Links ]
20. Kodama, K., K. Umehara, K. Shimizu, S. Nakatani, Y. Minoda and K. Yamada, "Identification of microbial products from dibenzothiophene and its proposed oxidation pathway", Agr. Biol. Chem., 37, 45-50 (1973). [ Links ]
21. Konishi, J., Y. Ishii, T. Onaka, K. Okamura and M.Suzuki, "Thermophilic carbon-sulfur-bond-targed biodesulfurization", Appl. Environ. Microbiol., 63, 3164-3169 (1986). [ Links ]
22. Laborde, A. and D. Gibson, "Metabolism of dibenzothiophene by Beijerinckiaspecies", Appl. Environ. Microbiol., 34, 783-790 (1977). [ Links ]
23. Le Borgne, S. and R. Quintero, "Biotechnological processes for the refining of petroleum", Fuel Process. Tech., 81, 155-169 (2003). [ Links ]
24. Lee, M., J. Senius and M. Grossman, "Sulfur-Specific Microbial Desulfurization of Sterically Hindered Analogs of Dibenzothiophene", Appl. Environ. Microbiol., 61, 4362-4366 (1995). [ Links ]
25. McFarland, B., D. Boron, W.Deber, J. Meyer, A. Jonson and R. Atlas, "Biocatalytic sulfur removal from fuels: applicability for producing low sulfur gasoline", Crit. Rev. Microbiol, 24, 99-147 (1998). [ Links ]
26. Monticello, D., "Biodesulfurization and upgrading of petroleum distillates", Curr. Opin. Biotechnol., 11, 540-546 (2000). [ Links ]
27. Monticello, D. and W. Finnerty, "Microbial Desulfurization of Fossil Fuels", Ann. Rev. Microbiol., 39, 371-389 (1985). [ Links ]
28. Omori, T., L. Monna, Y. Saiki and T. Kodama, "Desulfurization of dibenzothiophene by Corynebacterium sp. strain SY1", Appl. Environ. Microbiol., 58, 911-915 (1992). [ Links ]
29. Oshiro, T., T. Hirata and Y. Izumi, "Microbial desulfurization of dibenzothiophene in the presence of hydrocarbon", Appl. Microbiol. Biotechnol., 44, 249-252 (1995). [ Links ]
30. Sagardia, F., J. Rigua, A. Martinez, F. Fuentes, C. Lopez and W. Flores, "Degradation of benzothiophene and related compounds by soil Pseudomonas in a oil-aqueusenvironment", Appl. Environ. Microbiol., 29, 722-725 (1975). [ Links ]
31. Salazar, T., "Aislamiento de Pseudomonas nativas degradadoras de dibenzotiofeno", Tesis. Bacteriología. Universidad Colegio Mayor de Cundinamarca (1996). [ Links ]
32. Sánchez, O.F., C.J. Alméciga and E. Silva, "Efecto de la presencia de oleato de etanolaminasobre la actividaddesulfurizadora de Pseudomonas sp. ATCC 39327", Rev. Col. Cienc. Quím. Farm. 33, 13-19 (2004). [ Links ]
33. Sánchez, O.F., C.J. Alméciga-Díaz and E. Silva, "Desulfurization activity evaluation of Pseudomonas sppnative strains over n-hexane DBT spiked and kerosene", ENPROMER, 2nd Mercosur Congress on Chemical Engineering, Rio de Janeiro, Brasil, (2005). [ Links ]
34. Sánchez Mirón, A., M.C. Cerón García, F. García Camacho, E. Molina Grima and Y. Chisti, "Mixing in bubble column and airlift reactors", Trans. I. Chem E. Part A., 82, 1367-1374 (2004). [ Links ]
35. Shariati, F.P., B. Bonakdarpour and M.R. Mehrnia, "Hydrodynamics and oxygen transfer behaviour of water in diesel microemulsions in a draft tube airlift birreactor", Chem. Eng. Proc., 46, 334-342 (2007). [ Links ]
36. Subczynski, W.K. and J.S. Hyde, "Difusión of oxygen in water and hydrocarbons using a electron spin resonante spin - label technique", Biophys. J. 45, 743-748 (1984). [ Links ]
37. United Nations Environment Programme, "Diesel Fuel Sulphur Levels: Global Status", Febrary, http://www.unep.org (2007a). [ Links ]
38. United Nations Environment Programme, "Latin America and the Caribbean sulphur levels in diesel fuel", February, http://www.unep.org (2007b). [ Links ]
Received: July 5, 2007.
Accepted: December 28, 2007.
Recommended by Subject Editor: Orlando Alfano.














