Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Latin American applied research
versión impresa ISSN 0327-0793
Lat. Am. appl. res. vol.39 no.2 Bahía Blanca abr./jun. 2009
ARTICLES
Synthesis and structural characterization of L-(-)-malic acid pillared layered double hydroxides
F.P. Jiao†‡, X.Q. Chen†, Z.D. Fu†,Y. H. Hu*‡ and Y. H. Wang‡
† College of minerals processing and bioengineering, * Author to whom the correspondence should be addressed
‡ College of Chemistry and Chemical Engineering, Central South University, Changsha 410083, china
jiaofp@163.com
Abstract - A new organic-inorganic hybrid material was obtained by the intercalation of L-(-)-malic acids into Magnesium Aluminum layered double hydroxides (LDHs). The intercalation was performed by reprecipitation and coprecipitation method, respectively, from which high crystallized supramolecular chrial materials were obtained and L-(-)-malic acid was orientated perpendicularly between layers without carbonate ions. The structure of supramolecular chrial materials was characterized using XRD, FT-IR and DSC-TG, EA, ICP, and the supramoleclar structural model of the compound was given.
Keywords - Layered Double Hydroxides; L-(-)-Malic Acid; Intercalation; Reprecipitation And Coprecipitation; Structural Characterization.
I. INTRODUCTION
Hydrotalcite-like compounds, also known as anionic clays or layered double hydroxides (LDHs), have been investigated mainly because of their potential industrial uses such as catalysts, electrodes, ion exchangers, optical hosts and antacids (Cavani et al.,1991; Qiu and Villemure 1997). Among layered compounds, LDHs and organo-LDHs represent one of the most technologically promising materials because of their relative ease of preparation (Boclair et al., 2001). The LDHs also can be described as layered compounds of brucite-like structure (Mg(OH)2), with positively charged layers due to a partial substitution of divalent M2+ metals by trivalent M3+ metals. Between the layers there are counter anions to balance the electrical charge and coordinating water molecules (Nakayama et al., 2004; Khan et al., 2001). Its general formula: [M2+1-xM3+x(OH)2]x+(An-x/n).mH2O, where M2+ and M3+ can be any divalent and trivalent metal ions (with ionic radius similar to Mg2+), x is the metal ratio M3+/(M2++M3+) and An- is the interlamellar charge-compensating anion. Certain bulky organic anions may be used as pillaring agents via anion exchange. LDHs are used for preparing pillared layer materials, most frequently organo-LDHs (Parida and Das, 2000; Ambrogi et al., 2001). It is especially so, if the pillars are chiral, as these quasi two-dimensional channel systems are not only well defined structures with huge pore openings, but the optically active pillars create a chiral environment. Materials like these may be easily found using in chiral field (Aisawa et al., 2001; Jiao et al., 2008).
The paper reports the intercalation of L-(-)-malic acid (MA), served as a model of chiral drug, into LDHs through two different methods, reprecipitation and coprecipitation. The interaction between the host substance LDHs and the guest molecules MA was investigated. And then, the supramolecular structural model of the compound is suggested.
II. EXPERIMENT
A. Synthesis of LDHs
All reactants used in this work have high purity degree and all water used is distilled. This paper adopted the Reichle method (Dos Reis et al., 2004) to synthesize LDHs. Solution A was prepared by dissolving quantitative MgSO4·7H2O and Al2(SO4)3·18H2O in distilled water maintaining a [Mg2+]/[Al3+] molar ratio of 2. Solution B was prepared by dissolving quantitative NaOH and Na2CO3 (with a slight excess) in distilled water. LDHs was prepared by adding solution A to solution B in 3-4h with vigorous stirring while maintaining the pH > 9. The material obtained was submitted to hydrothermal treatment at 70 °C for 20 h. The slurry was centrifuged and washed several times with warm deionized water until neutral pH was reached and finally dried at 70 °C for 18 h.
B. Reprecipitation method (Ren et al., 2003)
L-(-)-malic acid pillared LDHs (MA-LDHs) was prepared by adding superfluous MA (6.5 g) to a suspension of the above LDHs in 100 ml distilled water. The suspension slowly dissolved with effervescence and a clear solution was obtained. This solution was added dropwise to an alkaline solution (NaOH (2.0g) dissolved in 100 ml decarbonated water) maintaining the pH above 9, followed by refluxing for 6 h at 140 °C. The resulting solid was recovered by filtration, washed, and dried at 75 °C for 14 h, giving the MA-LDHs that is named as sample 1.
C. Co-precipitation method (Aisawa et al., 2001)
7.70 g (0.03 mo1) of Mg(NO3)2·6H20 and 5.75 g (0.015 mo1) of Al(NO3)3·9H20 were both dissolved in 20 mL of decarbonated water (solution C). 5.94 g (0.485 mo1) of sodium hydroxide and 3.91 g MA were both dissolved in 70 mL of deionized water (solution D). Solution C was added dropwise under vigorous stirring and nitrogen atmosphere to a 250 ml three-neck round-bottomed flask. During the procedure, the pH value was maintained constant by continuous addition of NaOH solution (PH>8). The slurry was vacuum dried in an oven at 75 °C for 24 h. The intercalation product thus obtained is named as sample 2.
D. Characterization
Powder X-ray diffractometry (XRD) measurements were performed on a Rigaku Rint 6000 powder X-ray diffractometer, using Cu\Ka radiation at 30 mA, 40 kV. Fourier transform infrared (FTIR) spectra were obtained using a NICOLET AVATAR 360 FTIR spectrophotometer by the standard KBr disk method. DSC-TG measurement was conducted on Netzsch STA-449C thermal analyzer under air with a heating rate of 10 °C min-1 up to 1000 °C. EA and ICP were performed by Elementar vario EL III analyzer and SHIMADZU ICPS 7510 Emission Spectrometer respectively.
III. RESULTS AND DISCUSSION
A. Control of intercalated layer's condition
The molar ratio of Mg/Al in intercalation product obtained is 2, the XRD pattern for the LDHs, consisted of both sharp and symmetrical peak and some high angle asymmetrical peak. This provides evidence of characteristic reflections corresponding to a well-crystallized layered phase (Fig.1 a).

Fig. 1. Powder XRD patterns for Mg-A1-LDHs. (a) L-(-)-malic acid-LDHs, (b)-reprecipitation, (c)-coprecipitation
The MA-LDHs synthesized by coprecipitation and reprecipitation method are mainly affected by the temperature of reactive system, pH, molar ratio of MA/LDHs, reactive time. The solvent must be decarbonated water lest the carbon dioxide intercalate into the MA-LDHs and influence the crystal structure.
B. Crystal structure
The powder XRD patterns of the materials are typical of layered material, showing the basal peak and higher order reflections at low angle. The patterns of LDHs, sample 1 and sample 2 are shown in Fig.1. a, b, and c. The diffraction peaks (003), (006), (009) and (110) are easily recognized. The diffraction Peak (003) of LDHs shifts to lower angle after MA being intercalated into LDHs in sample 1 and 2, and their interlayer spaces are calculated as 1.25 nm and 1.21 nm (Table 1), respectively. These values indicate that an expansion of the interlayer spacing, compared with the LDHs (d003: 0.76 nm), may be caused by the MA intercalation into LDHs by both reprecipitation and coprecipitation methods. Given the thickness of the brucite-like layer of LDHs of 0.48 nm (Boclair et al., 2001), the gallery height are 0.77 nm and 0.73 nm, respectively.
Table 1. XRD date of diffraction peaks of LDHs and L-(-)-malic acid -LDHs

These values match up to the chain length of MA, which demonstrates a monolayer arrangement for the intercalated MA oriented perpendicular to the LDHs layers and the carboxyl of MA joint with LDHs layers. The intercalation of MA anions are clearly seen in each case by virtue of the expanded interlayer space. Comparing with LDHs, the MA-LDHs have an inirratic spiculate peak which is proper of sandwich. All of these demonstrate that MA intercalated into LDHs by reprecipitation and coprecipitation methods successfully.
From Fig. 1, it is also clearly observed that the reflection line of diffraction peak (ambient 60.8° (110)) was not moved, which indicates the intercalation of the MA has not changed the structure of layer but only has changed the interlayer space.
C. FTIR spectra
The LDHs and MA-LDHs materials are further confirmed by examination of the FTIR spectra shown in Fig. 2. a, b and c. The IR spectra of LDHs shows very broad bands at ambient 3448 cm-1, relating to the ? (OH) stretching vibration of hydroxyl groups of the host layers and to the interlayer and physically absorded water molecules. The absorption peak of the spectra corresponding to symmetric vibration of CO32- at 1363 cm-1 and the peak of the spectra at 788 cm-1 is attributed to bending vibration of CO32-. The peaks of the spectra corresponding to the lattice vibration modes are attributed to M-O (840 and 650 cm-1) and O-M-O (430 cm-1) vibrations. The characteristic peaks of CO32- in the curve b and c disappeared, and two new bands at 1575 cm-1 and 1400 cm-1 are due to anti-symmetric vibration and symmetric vibration of ? (coo-) mode, respectively. All of these demonstrate that MA is intercalated into the LDHs (Kloprogge and Frost, 1999).

Fig. 2. FTIR spectra for Mg-A1.LDHs (a) and L-(-)-malic acid-LDHs. (b-reprecipitation ,c-coprecipitation)
D. Supramolecular structure
The supramolecular structure of MA-LDHs depicted in Fig. 3. The height of gallery in sample 1 and 2 are 0.77 nm and 0.73 nm once the thickness of LDHs layers (0.48 nm) (Boclair et al., 2001)has been measured. It demonstrates that MA is intercalated into the LDHs. The supramolecular force is attributed to two main interactions, hydrogen bond and covalent bond. The covalent bond existed inside of layers is strong while the interaction existed the interlayers is weak. The host substance and guest molecules form inorganic-organic chiral material through the interactions of electrovalent bond, hydrogenolysis, Van der Waals Force.
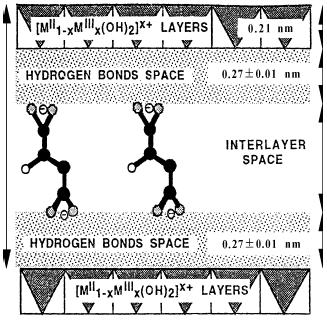
Fig. 3 Sketch of L-(-)-Malic acid-LDHs
E. DSC-TG analysis
The thermal behavior of the LDHs and MA-LDHs was examined by DSC-TG. The LDHs (Fig. 4.) losses 22.82% of its weight in the first step, and about 12.17% in the second step. The corresponding DSC curve shows the first decalescent peak at 110 °C arising from loss of surface adsorbed water. And the second decalescent peak is observed around 234 °C resulting from dehydroxylation of the brucite-like layer and decomposition of intercalated carbonate anions. After the intercalation of MA into LDHs host, the thermal decomposition characteristics of the resulting product are significantly different from the precursor. The DSC-TG curves of MA-LDHs is shown in Fig. 5. Comparing the DSC curves of LDHs with MA-LDHs, a significant change can be seen on the thermal behavior of the materials after intercalation. Liberation of absorbed water from the galleries of the intercalate result in an endothermic peak at 234 °C in the DSC curve, approximately 120 °C higher than the corresponding peak for the LDHs indicating that the water is more strongly bound in the interlayer galleries after intercalation of the MA. The TG curve shows two weight losses, the first one, 15.93% of weight, is attributed to the elimination of surface absorbed and interlayer water, another weight loss due to the dehydroxylation and combustion of MA is observed in the temperature of 454 °C. The account of water incorporated in the MA-LDHs according to the TG data is approximately 15.93%, which compares well with a calculated value for Mg0.66Al0.34(OH)2 (C4H4O5)0.23. 0.94H2O of 16.0%. In the case of the MA-LDHs, the combustion of the intercalated MA is shown in the main exothermic peaks in the temperature region 454 °C, which are higher than the combustion temperature region(250-330°C) of MA. It indicates MA is intercalated into the LDHs. The thermal stability of intercalated LDHs increased when the MA is intercalated into LDHs layers.

Fig. 4 .DSC-TG curves for Mg-Al-LDHs

Fig. 5. DSC-TG curves for MA- LDHs
Based on IR spectra and DSC-TG curves, it is possible to indicate the existence of an interattraction induced by supramolecular interaction between the host LDHs layers and the guest MA molecules.
F. Element chemical analysis
The results from elemental chemical analysis (metals) are included in Table 2.
Table 2 Chemical compositions (wt%) and structural formulae of samples

IV. CONCLUSIONS
In this work, a new organic-inorganic hybrid materials have been synthesized by the intercalation of MA into LDHs by coprecipitation and reprecipitation method. From the results of characterization, it indicates that the reprecipitation method is better than coprecipitation method. The MA-LDHs have a crystallized supramolecular, inoganic-organic chiral structure and well thermal stability. Their interlayer spaces are expanded to 1.25 nm and 1.21 nm, respectively. These data in this paper suggest a monolayer arrangement for the intercalated MA oriented perpendicular to the LDHs layers. The thermal stability of intercalated LDHs increased when the MA is intercalated into LDHs layers.
ACKNOWLEDGEMENTS
We wish to acknowledge the supports of Nation Natural Science Foundation of China (project No. 20776162) and by the Postdoctoral Science Foundation of Central South University on this work gratefully.
REFERENCES
1. Aisawa, S., S. Takahashi, W. Ogasawara, Y. Umetsu and E. Narita, "Direct Intercalation of Amino Acids into Layered Double Hydroxides by Coprecipitation," J. Solid State Chem., 162, 52-62 (2001). [ Links ]
2. Ambrogi, V., G. Fardella, G. Grandolini, L. Perioli and M.C. Tiralti, "Intercalation compounds of hydrotalcite -like anionic clays with anti-inflammatory agents - I. Intercalation and in vitro release of ibuprofen," International J. Pharm., 220, 23-32 (2001). [ Links ]
3. Boclair, J.W., P.S. Braterman, B.D., Brister, Z.M., Wang and F. Yarberry, "Physical and Chemical Interactions between Mg:Al Layered Double Hydroxide and Hexacyanoferrate," J. Solid State Chem., 161, 249-258 (2001). [ Links ]
4. Cavani, F., F. Trifiro and A. Vaccari, "Hydrotalcite-type anionic clays: Preparation, properties and applications," Catal. Today, 11, 173-301 (1991). [ Links ]
5. Jiao, F.P., X.Q. Chen, L. Yang and Y.H. Hu, "Enantioselective extraction mandelic acid enantiomers using ester alcohol L-tartarate as chiral selector," Lat. Am. Appl. Res., 38, 249-252 (2008). [ Links ]
6. Khan, A.I., L.X. Lei, A.J. Norquist and D. O'Hare, "Intercalation and controlled release of pharmaceutically active compounds from a layered double hydroxide," Chem. Commun., 22, 2342-2343 (2001). [ Links ]
7. Kloprogge, J.T., and R.L. Frost, "Fourier Transform Infrared and Raman Spectroscopic Study of the Local Structure of Mg-, Ni-, and Co-Hydrotalcites," J. Solid State Chem., 146, 506-515 (1999). [ Links ]
8. dos Reis, M.J., F. Silvério, J. Tronto and J.B. Valim, "Effects of pH, temperature, and ionic strength on adsorption of sodium dodecylbenzenesulfonate into Mg-Al-CO3 layered double hydroxides," J. Phys. Chem. Solids., 65, 487-492 (2004). [ Links ]
9. Nakayama, H., N. Wada and M. Tsuhako, "Intercalation of amino acids and peptides into Mg-Al layered double hydroxide by reconstruction method," International J. Pharm., 269, 469-478 (2004). [ Links ]
10. Qiu, J.B., and G.J. Villemure, "Anionic clay modified electrodes: electron transfer mediated by electroactive nickel, cobalt or manganese sites in layered double hydroxide films," Electroanal Chem., 428, 165-172 (1997). [ Links ]
11. Parida, K., and J. Das, "Mg-Al hydrotalcites: preparation, characterisation and ketonisation of acetic acid," J. Mole. Catal. A: Chemical, 151, 185-192 (2000). [ Links ]
12. Ren, L.L., J. He, D.G. Evans and X. Duan, "Intercalation and Assembly Glutamic Acid Pillared of Supramolecular Materials of Layered Double Hydroxides," Chem. J. Chinese Universities, 24, 169-173 (2003). [ Links ]
Received: April 3, 2008.
Accepted: August 1, 2008.
Recommended by Subject Editor: Ricardo Gómez.














