Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Latin American applied research
versión impresa ISSN 0327-0793
Lat. Am. appl. res. vol.41 no.1 Bahía Blanca ene. 2011
ARTICLES
Kinetics of the esterification of maleic anhydride with castor oil.
P.C. Mazo, D. Estenoz, L.A. Ríos
Grupo Procesos Fisicoquímicos Aplicados. Departamento de Ingeniería Química, Universidad de Antioquia. Calle 67 Nº 53-108 Medellín, Tel 2195539
pcmazo@matematicas.udea.edu.co
Instituto de Desarrollo Tecnológico para la Industria Química (Intec). Santa Fe Argentina
destenoz@ceride.gov.ar
Abstract - Kinetics reaction between castor oil and maleic anhydride without condensation and catalyst was studied. On the other hand, the products concentration, kinetics and thermodynamics parameters were determined using mathematical modeling and were validated with experimental data. The reaction was carried out in a semi-batch reactor at constant temperature. The reaction was conducted through the determining of the acid number (ASTM D4662-03), and showed first order kinetics with respect to the anhydride. For the modified oil, the hydroxyl value (OHv) was quantified through titration following (ASTM D4274-05). The distribution of molecular weights was determined using gel permeation chromatography (GPC), and functional group analysis was determined using infrared spectroscopy (IR).
Keywords - Castor Oil; Anhydride Maleic; Esterification; Kinetics; Mathematical Model.
I. INTRODUCTION
Castor oil is a viscous pale yellow non-volatile and non-drying oil with a bland taste and is sometimes used as a purgative. India is the world largest exporter of castor oil; other major producers are China and Brazil. The chemistry of castor oil is centered on its high content of ricinoleic acid and the three points of functionality existing in the molecule. These are: (1) the carboxyl group which can provide a wide range of transesterifications, hydrolysis, saponification, reduction; (2) the single point of unsaturation which can be altered by hydrogenation or epoxidation or vulcanization or addition reactions; and (3) the hydroxyl group which can be esterified or acetylated or alkoxylated, may be removed by dehydration to increase the unsaturation of the compound to give a semi-drying oil, can be split at that point by high-temperature pyrolysis and by caustic fusion to yield useful products of shorter chain length. The ricinoleic acid comprises over 89% of the fatty acid of the oil (Ogunniyi, 2006).
Although castor oil is not edible, it is more versatile than other vegetable oils as it is widely used as a starting material for many industrial chemical products because of its unique structure. It is one of those vegetable oils that have found usage in many chemical industries.
It is a raw material for paints, coatings, inks, lubricants and a wide variety of other products.
Because of its hydroxyl functionality, the oil is suitable for use in isocyanate reactions to make polyurethane elastomers (Quipeng et al., 1990), polyurethane millable (Yeganeh and Mehdizadeh, 2004), adhesives and coatings (Trevino and Trumbo, 2002; Somani et al., 2003), interpenetrating polymer network from castor oil-based polyurethane (Xie and Guo, 2002) and some semi-rigid polyurethane foams that have potential uses in thermal insulation were produced when castor oil/polyether mixture was reacted with toluene diisocyanate (Ogunniyi et al., 1996).
Maleinization reaction has been used to make chemical modification of vegetable oils through Diels-Alder (conjugated dienes), esterification (alcohols) and "ene" reactions (compounds with allylic hydrogens) with maleic anhidride.
Main modifications that have been made to olefinic compounds with the aim to increase its hydrofilicity are "ene" reactions and many adducts can be prepared through Diels-Alder reactions of unsaturated fatty acids with maleic andydride. Those products and their derivatives are employed as dry oils, water-soluble paints and surfactants to metals, some applications of these reactions with vegetable oils are described by Bickford et al. (1942), Candy et al. (2005), Plimmer (1949); Teeter et al. (1948), Tomodaa et al. (1998) and Warth et al., (1997); which are carried out at higher temperatures to 250 Celsius degrees, where is possible decomposition of the oil and undesirable by-products. Wang et al. (2008) obtains biodegradable foams from maleated castor oil (MACO), obtained by esterification of hydroxyls using maleic anhydride in a temperature range from 80 to 120 Celsius degrees. The mechanism of addition of nucleophiles to reactive carbonyl compounds is a subject of continuing interest (Andrés and de Rossi, 2005; Skrzypek et al., 1998).
In this work we have proposed study the kinetics of this reaction without condensation, determine some thermodynamics and kinetics parameters through mathematical modeling and validated the model obtained with experimental data.
II. METHODSMaterials
Castor oil, grade USP, with a OHv value equal to 159.51 mg of KOH/g of sample, maleic anhydride, commercial grade.
Synthesis of modified castor oil.
Maleic anhydride (MA) and castor oil (1:1 by mole, respectively) were added in a 250 mL three-necked round-bottom flask equipped with a stirrer, a thermometer, and an inlet of dry nitrogen (see Fig. 1). The reaction proceeded with continuous stirring at various temperatures (80, 87, 100 °C, respectively) for a period of time (0.5-9 h).
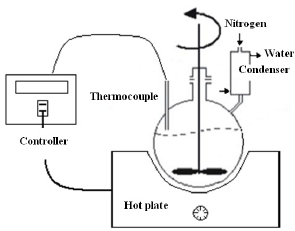
Figure 1. Schematic representation of the reactor (250 mL) used for esterification reaction.
The measurement and control the temperature with thermocouple J is necessary. Hot plate was used during heating process. The samples were washed with water in order to removed excess anhydride and/or maleic acid.
Characterization methods.
The kinetic experiments were performed at temperatures of 80, 87 and 100 Celsius degrees. The reaction was monitored through the determination of the acid number (ASTM D4662-03), which occurring as a kinetic of first order respect to anhydride. For the modified oil, the amount of hydroxyls (OHv) is quantified through titration (ASTM D4274-05). Molecular weights of castor oil and MACO were measured by using a Walter 208LC gel permeation chromatograph (GPC) at room temperature with THF as the solvent.
Fourier transform infrared (FTIR) spectra were collected by a IR Prestige Shimadzu, FTIR spectrometer within the wavenumber range of 400-4000 cm-1 at a 4 cm-1 resolution. All the samples were prepared by coating the reaction product on the surface of a KBr tablet.
III. RESULTS AND ANALYSIS
Esterification of maleic anhydride with castor oil proceeds in three stages (Fig. 2), where: P (a,b,c): specie to analyze, a: number of hydroxyl groups, b: number of acid groups, c: number of ester bridges, A: maleic anhydride.

Figure 2. Esterification reactions castor oil and maleic anhydride
When there is no condensation, the number of ester bridges between chains is zero, and this is confirmed through gel permeation chromatography analysis, GPC made to MACO samples at 6 hours of reactions, where the molecular weight no show the dimers or trimers formation, the first peak of the chromatogram correspond to the solvent, that in this case was tetrahydrofuran (Fig. 3).

Figure 3. GPC chromatograms, corresponding to the temperatures 80, 87 and 100°C.
For determine if the results obtained in the kinetic modeling represents properly the experimental data, we decide carried out the mathematical simulation of the esterification reactions and predict the evolution of the reactants and products concentration during the time of the reaction.
Self-autocatalysis system and the simulation were carried out in irreversibility conditions because without condensation there is no equilibrium. The model was resolved using Matlab 7.5. Mathematical equations of the kinetic model for this system are:
 | (1) |
 | (2) |
 | (3) |
 | (4) |
Global equation
 | (5) |
A. Kinetics of the esterification of castor oil with maleic anhidride.
To monitor the esterification reaction course, acid number was used, from which the esterification yielding can be estimated. Figure 4 indicates that the acid number drastically decreases at the beginning of the reaction and then levels off due to the reduction of the component's concentration. It is clear that the higher reaction temperature favors the reactions as a result of higher reaction speed. The majority of the reaction is nearly completed within 7 h at 80°C, while 2 h is sufficient for the reaction at 100°C.

Figure 4. Change of acid number with reaction time during MACO synthesis at different temperatures.
A kinetic study of the decreasing of the acidity grade was made and with this value was calculated the conversion percent, as shown:
 | (6) |
where η is the conversion percent, Avo initial acidity value, Avf final acidity value. The final acidity value is only one-half of initial acidity value, and for this reason it must be multiply by 2 into Eq. 6.
Comparing the variation of the concentration of the maleic anhydride, during the experiment time and comparing the results of the simulation with the experimental data (See Fig. 5), we can see that the tendency is an exponential decreasing, that is about to the results obtained for the temperature of 87 Celsius degrees. In the model, the effect of temperature is not considered, which is significant for the progress of the reaction because when this increase, there is decreasing more rapidly than maleic anhydride consumed, but can occur Diels-Alder dehydration colateral and undesirable inter- and intra-molecular cyclization reactions.

Figure 5. Variation of the concentration of the maleic anhidride during the course of the esterification reaction.
In Fig. 6, the variation of the concentration of the active species during the course of the reaction is showed. Besides, P(2,1,0) is the specie of interest, and is produced in greater quantity. On the other hand, we observe an increase in the concentration through time, and anhydride is consumed almost entirely.

Figure 6. Variation of the concentration of the reactive species during the esterification reaction.
The reaction follows a first-order kinetics respect to the decreasing of the acid groups from anhydride, which is showed by regression model presents a r2 value high. (See Table 1).
Table 1. Kinetics parameters for the esterification reaction between castor oil with maleic anhydride without catalyst.

On the other hand, we have selected the classical transition state theory (Benson, 1969; Glasstone et al., 1941) to calculate the kinetic parameters. The rate constant, k(T), was computed using this theory assuming that the transmission coefficient is equal to unity, as expressed by the following relation:
 | (7) |
where kB, h, and R are the Boltzman constant, the Planck constant, and the universal gas constant, respectively. ΔG≠(T) is the standard-state free energy of activation, at the absolute temperature T.
 | (8) |
 | (9) |
 | (10) |
When the temperature is increased, there is an increase in the rate reaction, and in all three cases, these values are very high (See Table 1).
Table 2 show the results obtained to the activation energies, Ea, and the pre-exponential factor A, have been calculated using the Arrhenius Eq. 8.
Table 2. Results obtained for the calculations to kinetic data.

The change in the sign of the enthalpy for the transition state (ΔH≠) represents an endothermic and late transition state, where occur important changes in the reactants and TS polarity. The entropy change for the transition state (ΔS≠) has positive sign indicating a decreasing of the order in the transition state because of an increase in the molecular collisions. Also, there is a change of sign in the Gibbs free energy indicating a spontaneous process.
The hydroxyl values obtained for maleinizated castor oil are showed in Table 4, for 3 hours of reaction.
Table 3. Thermodynamic parameters for esterification process.

Tabla 4. Hydroxyl values (OHv) for MACO samples, after 3 hours of reaction.

According to hydroxyl values OHv, the samples are tabulated as described in Table 4. In this Table, only integral part is considered.
During three hours of reaction, FTIR spectra of castor oil and MACO were compared as described in Fig. 7. For castor oil, the peak at 3450 cm-1 belongs to -OH absorption, while that at 1644 cm-1 can be assigned to -C=C- double bond. When castor oil reacts with maleic anhydride, the double bond peak is enhanced. No peaks corresponding to cyclic anhydride at 1779 and 1849 cm-1 can be perceived in the resultant MACO, indicating that almost all of maleic anhydride has been consumed to react with castor oil.

Figure 7. IR spectra for the maleinizated castor oil samples.
IV. CONCLUSION
Maleinization reaction is an esterification reaction obtained from maleic anhydride and the secondary hydroxyl group of castor oil. In our autocatalytic system, this reaction follows a first-order kinetics with respect to the anhydride concentration. At temperatures below 100°C are not observed dimers or trimers formed by condensation. The mathematical model predicts the concentration of active species during the reaction and the decrease in anhydride.
REFERENCES
1. Andrés, G.O and R.H. de Rossi, "Kinetics and Mechanistic Study of the Reaction of Cyclic Anhydrides with Substituted Phenols. Structure-Reactivity Relationships," J. Org. Chem, 70, 1445-1449 (2005). [ Links ]
2. Benson, S.W., The Foundations of Chemical Kinetics, McGraw-Hill, New York (1969). [ Links ]
3. Bickford, W.G., P. Krauczunas and D.H. Wheeler, "The Reaction of Nonconjugated Unsatured Fatty Esters with Maleic Anhydride," Oil Soap, 19, 23-27 (1942) [ Links ]
4. Candy, L., C. Vaca-Garcia and E. Borredon, "Synthesis and Characterization of Oleic Succinic Anhydrides: Structure-Property Relations," JAOCS, 82, 271-277 (2005). [ Links ]
5. Glasstone, K.J., K.J. Laidler and H. Eyring, The Theory of rate Processes. McGraw-Hill, New York, (1941). [ Links ]
6. Ogunniyi, D.S., W.R.O. Fakayejo and A. Ola, "Preparation and properties of polyurethanes from toluene diisoyanate and mixtures of castor oil and polyol," Iranian Polym. J, 5, 56-59 (1996). [ Links ]
7. Ogunniyi, D.S., "Review Paper Castor oil: A vital industrial raw material," Bioresource Technology, 97, 1086-1091 (2006). [ Links ]
8. Plimmer, H., "The Reaction of Maleic Anhydride with Nonconjugated Unsaturated Fatty Materials," J. Oil Colour Chem. Assoc, 32, 99-112 (1949). [ Links ]
9. Quipeng, G., F. Shixia and Z. Qingyu, "Polyurethanes from 2,4-toluene diisocyanate and a mixture of castor oil and hydroxyether of bisphenol A," Eur. Polym. J, 26, 1177-1180 (1990). [ Links ]
10. Skrzypek, J., J.Z. Sadlowski, M. Lachowska and P. Nowak, "Kinetics of the esterification of phthalic anhydride with 2-ethylhexanol Part IV: Non-catalytic process. Final kinetic comparison of the catalysts," Chemical Engineering and Processing, 37, 163-167 (1998). [ Links ]
11. Somani, K.P., S.S. Kansara, N.K. Patel and A.K. Rakshit, "Castor Oil based polyurethane adhesives for wood-to-wood bonding," Int. J. Adhesion Adhesives, 23, 269-275 (2003). [ Links ]
12. Teeter, H.M., M.J. Geerts and J.C. Cowan, "Polymerization of Drying Oils. III. Some Observations on Reaction of Maleic Anhydride with Methyl Oleate and Methyl Linoleate," J. Am. Oil Chem. Soc. 25,158-162 (1948). [ Links ]
13. Tomodaa, H., Y. Sugimotoa, Y. Tania and S. Watanabeb, "Characteristic Properties of Cutting Fluid Additives Derived from the Reaction Products of Hydroxyl Fatty Acids with Some Acid Anhydrides," Journal of Surfactants and Detergents, 1, 533-537 (1998). [ Links ]
14. Trevino, A.S. and D.L. Trumbo, "Acetoacetylated castor oil in coatings applications," Prog. Org. Coat. 44, 49-54 (2002). [ Links ]
15. Wang, H.J., M.Z. Rong, M.Q. Zhang, J. Hu, H.W. Chen and T. Czigány, "Biodegradable Foam Plastics Based on Castor Oil," Biomacromolecules, 9, 615-623 (2008). [ Links ]
16. Warth, H., R. Mulhaupt, B. Hoffmann and S. Lawson, "Polyester Networks Based upon Epoxidized and Maleinated Natural Oils," Angew. Makromol. Chem, 249, 79-92 (1997). [ Links ]
17. Xie, H.Q and J.S. Guo, "Room temperature synthesis and mechanical properties of two kinds of elastomeric interpenetrating polymer networks based on castor oil," Eur. Poly. J., 38, 2271-2277 (2002). [ Links ]
18. Yeganeh, H. and M.R. Mehdizadeh, "Synthesis and properties of isocyanate curable millable polyurethane elastomers based on castor oil as a renewable resource polyol," Eur. Poly. J. 40, 1233-1238 (2004). [ Links ]
Received: December 15, 2008.
Accepted: December 29, 2009.
Recommended by Subject Editor Ana Lea Cukierman.














