Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Latin American applied research
Print version ISSN 0327-0793
Lat. Am. appl. res. vol.41 no.2 Bahía Blanca Apr. 2011
ARTICLES
Formulation of water in paraffin emulsions
C. J. Morales, U. Riebel, N.M. Guzmán§ and M. Guerra*
EIQ-UCV, Caracas, Venezuela. Actually: Lehrstuhl Mechanische Verfahrenstechnik, Brandenburgische Technische Universität Cottbus, PF 101344, D-03013, Cottbus, Germany. moralesc@gmx.net
Lehrstuhl Mechanische Verfahrenstechnik, Brandenburgische Technische Universität Cottbus, PF 101344, D-03013, Cottbus, German.
§ Escuela de Ingeniería Química. Universidad Central de Venezuela. P.O. Box 50656 - Sabana Grande, Caracas 1051-A, Venezuela.
* Departamento de Ingeniería de Proyectos MCL Control, S.A., Av. Don Diego Cisneros, Centro Empresarial Los Ruices, Piso 3, Of. 308. Caracas 1071, Venezuela.
Abstract - In this work, the effect of surfactant content, water content, HLB value, alcohol content, salinity, emulsion volume and mixing properties on water in paraffin emulsion stability was studied. Emulsion stability was determined by the extent of water and oil resolved after 30 days. After finishing the variables scans, the most appropriate formulation conditions were established, and a formulation protocol was defined. Emulsion density and apparent viscosity were measured, and viscosity was modeled by the Power Law.
Keywords - Stability; Span 80; Arkopal N40; Viscosity.
I. INTRODUCTION
In Venezuela there are more than 11,000 lagoons of waste oil emulsions. Due to environmental regulations, these materials require processing and/or disposal, but their characteristics and properties are quite variable. The problem has been extensively handled by petroleum engineers with little success and the existence of a single approach for the treatment and separation is far from possible.
The extraordinary stability of these waste oil emulsions is due to a combination of high viscosity, low density difference, and presence of solids and natural emulsifiers in part formed by the oxidation of crude oil components during the ageing process.
The scope of this work was to develop highly stable synthetic emulsions with reproducible properties, serving as a model to study several techniques for emulsion destabilization that could be also used in crude oil emulsions. Although comparisons between synthetic and crude oil emulsions is not the objective of this work, ranges of water content, density and viscosity of synthetic emulsions are in agreement with the available data of the crude oil emulsion samples.
The effect of different variables was studied in one-dimension scans and as result, an emulsion providing emulsions with maximum stability is obtained.
II. THEORETICAL BACKGROUND
Emulsions are thermodynamically unstable systems; however, the kinetic mechanisms involved in emulsion breaking can be so slow that the corresponding emulsion may be considered as metastable as far as the application is concerned (Salager, 2000).
In a broad sense, producing an emulsified system involves several choices and activities considered as variables. Each of them has an independent effect on formulation; for this reason, one-dimensional scans are used to study their effects on the emulsion properties.
Emulsions are often non-Newtonian fluids with complex rheological behavior; most of them follow the Power Law model (Fox and McDonald, 2003).
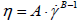 | , (1) |
η is the so-called apparent viscosity that would indicate the resistance to flow under some specific conditions; 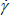 is the shear rate; A is called Consistency Index and B Power Law Index. The viscosity increases within the internal phase content. Many studies have reported empirical relationships to describe this behavior (Becher, 1977; Becher, 1982; Arai and Shinoda, 1967; Kunieda and Shinoda, 1985; Bourrel et al., 1980; Antón et al., 1997), but none of them are valid in the general case, because many other effects, as the non-Newtonian behavior, have to be considered as well (Shinoda et al., 1990; Graciaa et al., 1984).
is the shear rate; A is called Consistency Index and B Power Law Index. The viscosity increases within the internal phase content. Many studies have reported empirical relationships to describe this behavior (Becher, 1977; Becher, 1982; Arai and Shinoda, 1967; Kunieda and Shinoda, 1985; Bourrel et al., 1980; Antón et al., 1997), but none of them are valid in the general case, because many other effects, as the non-Newtonian behavior, have to be considered as well (Shinoda et al., 1990; Graciaa et al., 1984).
Griffin (1949) introduced the Hydrophilic-Lipophilic Balance or HLB concept, which is a surfactant characteristic parameter used as a measure of the relative affinity of the surfactant for the water and oil phases. The HLB concept has been extensively used (Davies, 1957; Lin, 1980; Becher and Griffin, 1970; Becher, 1982; Becher, 1996). For cost and efficiency reasons, a mixture of several surfactants is generally used as emulsifier.
III. MATERIALS AND METHODS
Surfactant suppliers maintain that to obtain a W/O emulsion, the HLB value should be between 4 and 6, approximately. Therefore, a mixture of Sorbitan Monooleate (Span 80®) with a HLB=4.3 and Nonylphenol Polyglycol Ether 4 EO (Arkopal N 040®) with a HLB=8.7 (both highly soluble in oil), were used. Paraffin and surfactants were mixed. Then, distilled water was added slowly (using a burette) during 5 minutes, while it was mixed with a Braun stirrer model MR-5550, at 13400 rpm. After adding all the water, the components were mixed for 5 more minutes. 100 mL of emulsion (water plus paraffin) were prepared in each run in a 600 mL volumetric flask (see Fig. 1-a and 1-b).
(a) 
(b) 
(c) 
Figure 1. Appearance of water-in-oil emulsion stabilized by Span 80® and Arkopal N 040®. Immediately after the preparation (a: 100 mL, b: 500 mL) and 30 days later (c: 50 mL sample in test tube). fw = 0.75, HLB = 5.5, Cs = 2% v/v, vm = 13400 rpm, tm = 5 min
Samples of 50 mL were transferred into test tubes, and monitored for 30 days. Stability of emulsions against sedimentation and coalescence was assessed altogether by the total separated volume of water plus paraffin (Vs = Vw + Vp) divided by the total sample volume (V = Vw + Vp + Ve) (see Fig. 1-c). The emulsions with acceptable stability were defined as those in which the total separation was lower than 10% in 30 days.
The emulsion type was inferred by observing what happened when a drop of each emulsion was added to a volume of either pure paraffin or pure water.
After finishing the experimental tests, the formulation to produce the most stable emulsion for each water content was defined. The emulsions were prepared in triplicate to validate the reproducibility of the proposed formulation protocol. Emulsions density and apparent viscosity were experimentally determined. The density was calculated from the ratio between the emulsion weight and volume and the apparent viscosity was measured with a standard Brookfield LVF dial reading viscometer with a #3 spindle. For the viscosity measurement, it was necessary to prepare batches of 500 mL emulsion. Apparent viscosity was fitted by a function of the power law form. Parameters A and B were obtained from a plot of apparent viscosity versus angular speed. Even though the values of A and B will be different from the rigorously derived from the standard power law expression for viscosity, they will still track the same trend. The correlations obtained for A and B are function of the water content.
A. Experimental plan
One-variable scan was used to study the effect of the main variables on the emulsion properties. The variables and ranges were: surfactant content (0.5 - 10% v/v), water content (25 - 75% v/v), HLB value (4.5 - 6.5), alcohol content (Octanol 0 - 4% v/v), salinity (0 - 16 g/dL water), mixing velocity (6100 - 13400 rpm), mixing time (5 -20 min), mixing type (continuous or intermittent), and batch emulsion volume (50 - 500 mL). Finally, emulsions with Sodium Dodecyl Sulphate were also formulated to allow comparison between a nonionic and an ionic surfactant. The range of variation of each parameter was established taking into account theoretical considerations, previous experience in formulation and available capacities and resources.
All tests were carried out at room temperature (25±1 °C). Surfactant and co-surfactant content are given in relation to the water plus paraffin volume. Salinity is given as NaCl in water. Mixing time is defined after all water addition. Surfactant mixtures HLB values were calculated from the Span 80® and Arkopal N040® HLB values assuming ideal mixture behavior.
IV. RESULTS AND DISCUSSION
A. Variables Scan
First, the effect of surfactant content on emulsion stability was evaluated. Figure 2 shows the results of these tests. Surfactant content influence on emulsion stability is not really significant because all formulated emulsions were stable according to the defined criterion. The higher stabilities were obtained for surfactant contents above of 1.5 % v/v. Sedimentation was demonstrated in all cases by the presence of a clarified paraffin layer in the top of the test tubes. Coalescence was identified by the presence of separated water at the bottom, and was observed only for surfactant contents of 4.0 and 8.0 %v/v.
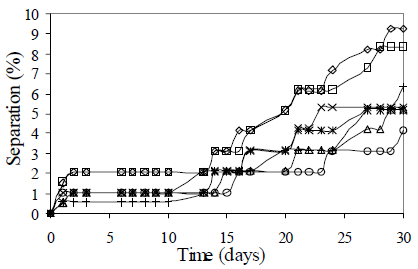
Figure 2. Surfactant Content Effect on Emulsion Stability. fw = 0.50, HLB = 5.5 with a mixture of Span 80® and Arkopal N 040®, vm = 13400 rpm, tm = 5 min., T = 25°C. Cs = 0.5-10% v/v: ( ) 0.5%, (
) 0.5%, ( ) 1.0%, (
) 1.0%, ( ) 1.5%, 2.0 (
) 1.5%, 2.0 ( ), (+) 4.0%, (×) 8.0%, (
), (+) 4.0%, (×) 8.0%, ( ) 10.0%.
) 10.0%.
Although the highest stability was obtained for surfactant content of 10% v/v, the most appropriate formulation value should also take into account the costs; therefore, a surfactant content of 2% v/v was selected for the rest of the formulations. In practical applications, a surfactant concentration between 0.2-3% is used, which corresponds with our selection.
Stability of the emulsions can be explained based on surfactant concentration which is, in all cases, above the mixture critical micelle concentration and the surfactant solubility. The CMC of pure Span 80 at room temperature, for example, is 0.026% w/v (Foyeke and Burgess, 1998).
The second variable studied was the surfactant HLB. A range of HLB from 4.5 to 6.5 was selected. Outside this range W/O emulsion stability is very low or the formulation results in a O/W emulsion. HLB describes the surfactant affinity to the water and oil phases, and its influence on emulsion stability depends strongly on the volumetric proportion of the phases. Three scans of HLB were made for emulsions with water contents of fw = 0.25, 0.50 and 0.75, respectively.
Figures 3 and 4 show HLB effect on emulsion stability. In these tests, not all the emulsions were stable enough. In some cases a separation up to 60% was obtained within 30 days after preparation.

Figure 3. HLB Effect on Emulsion Stability. fw = 0.25 (above) and fw= 0.50 (below), Cs=2% v/v, vm=13400 rpm, tm=5 min., T = 25°C. HLB = 4.5-6.5 with a mixture of Span 80® and Arkopal N 040®: ( ) 4.5, (×) 5.0, (
) 4.5, (×) 5.0, ( ) 5.5, (
) 5.5, ( ) 6.0, (
) 6.0, ( ) 6.5
) 6.5

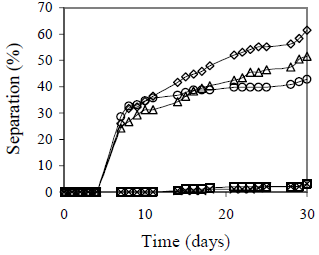
Figure 4. HLB Effect on Emulsion Stability. Picture after 30 days. fw=0.75, Cs= 2% v/v, vm = 13400 rpm, tm = 5 min., T = 25°C. HLB = 4.5-6.5 with a mixture of Span 80® and Arkopal N 040®: ( ) 4.5, (×) 5.0, (
) 4.5, (×) 5.0, ( ) 5.5, (
) 5.5, ( ) 6.0, (
) 6.0, ( ) 6.5
) 6.5
HLB effect on stability was higher for emulsions with fw = 0.25 and 0.75, but in the first case separation is only due to sedimentation, which is explained by the weak interaction between the droplets. Coalescence was observed only for fw = 0.75. Droplet approach before coalescence was not observed due to the high internal phase content; all drops were in contact and emulsion stability decreases with coalescence. The HLB values that yield best stability are 5.0 and 5.5 for fw = 0.25; 5.5 and 6.0 for fw = 0.5, and 4.5 and 5.0 for fw = 0.75.
With the HLB scan, at least one emulsion with high stability was obtained for low, medium and high water content, but the study of new variables could lead to an improved formulation and would help to understand the variables which exert higher influence on stability. HLB scans were repeated with 2% Octanol dissolved in the paraffin before adding the water. Octanol does not act as good co-surfactant, and in all cases the stability is reduced when alcohol is used, observing both coalescence and sedimentation.
Figure 5 summarizes alcohol content effect on emulsion stability for HLB = 5.5 and fw = 0.25, 0.50 and 0.75. The higher the water content and/or the Octanol content, the lower the emulsion stability with coalescence. Reduction of emulsion stability with sedimentation can be explained by a change in the required hydrophilic-lipophilic balance in presence of Octanol. The higher the fw the higher the octanol dissolved in the oil (for the same alcohol content on the total emulsion), that according to the Windsor concept (Salager, 2000; Rosen, 2004) modifies the interactions between the surfactant molecules and the oil phase, which is interpreted as a variation in the required HLB value.
(a) fw = 0.25

(b) fw = 0.50
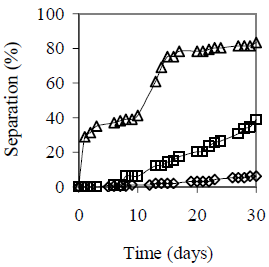
(c) fw = 0.75
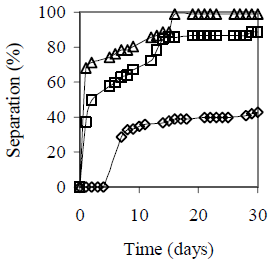

Figure 5. Octanol Content Effect on Emulsion Stability. HLB 5.5 with a mixture of Span 80® and Arkopal N 040®, Cs = 2% v/v, vm = 13400 rpm, tm = 5 min., T = 25°C. Co = 0 - 4% v/v: ( ) 0%, (
) 0%, ( ) 2%, (
) 2%, ( ) 4%. Picture (bottom) after 30 days
) 4%. Picture (bottom) after 30 days
As well, a water salinity scan was included in this work. The emulsions with the lowest separation after one month were those with a salt content of 4 and 8 g/dL. For 12 and 16 g/dL of salt a second emulsion phase, somewhat translucent, was formed. In these cases, the pure paraffin layer and the translucent emulsion were measured together.
Even though in some cases salinity increases the stability against sedimentation, stability does not differ significantly from the one obtained without salt. Salinity also increases the density difference; therefore, use of salt in the formulation is not justified, so it is not considered in the final protocol.
Figure 6 summarizes the effect of mixing conditions on emulsion stability. Figure 6-a shows the mixing velocity effect is shown. The selected velocities include all the stirrer operational range. The emulsions that presented the lower separation were those obtained with 9500 and 6100 rpm. Separation decreases from 6% at 13400 rpm to 4% at 9500 rpm. Reduction in mixing velocity is favorable not only in terms of stability, but also in operational cost. Two other stirrers with velocities up to 2000 and 4000 rpm, respectively, were tested before selecting the Braun stirrer model MR-5550, but in both cases emulsion stability was not satisfactory. Figure 6-b shows the mixing time effect on emulsion stability. The best results were obtained for 5 and 10 minutes of agitation, but for 10 minutes both coalescence and sedimentation were observed.
(a) 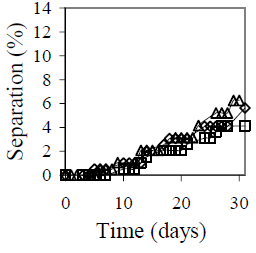
(b) 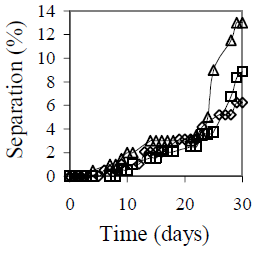
(c) 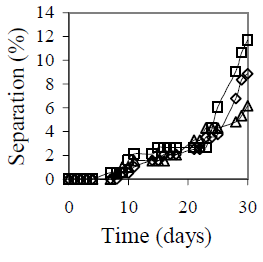
Figure 6. Mixing Conditions Effect on Emulsion Stability. fw = 0.50, HLB = 5.5 with a mixture of Span 80® and Arkopal N 040®, Cs = 2% v/v, T = 25°C. (a): Effect of mixing velocity with continuous mixing for 5 min: ( ) 6100 rpm, (
) 6100 rpm, ( ) 9500 rpm, (
) 9500 rpm, ( ) 13400 rpm. (b): Effect of mixing time with continuous mixing at 13400 rpm: (
) 13400 rpm. (b): Effect of mixing time with continuous mixing at 13400 rpm: ( ) 5 min, (
) 5 min, ( ) 10 min, (
) 10 min, ( ) 20 min. (c):1 Effect of mixing type: (
) 20 min. (c):1 Effect of mixing type: ( ) continuous for 10 min. at 13400 rpm, (
) continuous for 10 min. at 13400 rpm, ( ) Intermittent I, (
) Intermittent I, ( ) Intermittent II.
) Intermittent II.
Figure 6-c shows the mixing type effect on emulsion stability. During the first 25 days, all three emulsions showed approximately the same stability; however, after 30 days, a lower separation was obtained with the intermittent mixing at variable velocity.
Comparing the three plots in Fig. 6, it is clear that a mixing velocity reduction from 13400 rpm to 9100 rpm improves the previous result, but a longer mixing time is not recommended. Depending on mixing conditions, emulsion temperature varied from 31 °C at 6100 rpm and 5 minutes, to 58 °C at 13100 rpm and 20 minutes. Even though, the changes in stability appear much later. The results on stirrer speed, mixing time and intermittent mixing indicate that low mixing temperature could favor long time stability.
Figure 7 shows the results of batch volume scans. An emulsion stability reduction was observed when a 50 mL batch emulsion was prepared. This emulsion volume does not ensure an appropriate emulsification, because the stirrer geometry and the beaker used do not provide enough liquid height. In this case coalescence was also observed.

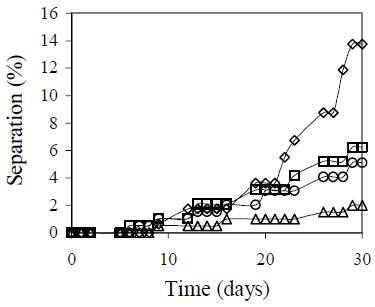
Figure 7. Batch Volume Effect on Emulsion Stability. Picture after 30 days. fw = 0.50, HLB = 5.5 with a mixture of Span 80® and Arkopal N 040®, Cs = 2% v/v, vm = 13400 rpm, tm = 5 min., T = 25°C. Batch Volume: ( ) 50 mL, (
) 50 mL, ( ) 100 mL, (
) 100 mL, ( ) 250 mL, (
) 250 mL, ( ) 500 mL
) 500 mL
On the other hand, the 250 mL and the 500 mL batch volumes represent an improvement on emulsion stability compared to the results with 100 mL. For 250 mL batch volume, the separation after 30 days is only 2%. Again, an indirect influence of mixing temperature might explain this behavior.
The ionic surfactant sodium dodecyl sulphate was also tested. A salinity scan in the range from 0 to 16 g NaCl/dL water was made, for fw =0.25 and 0.50. In both cases emulsions were unstable. For fw =0.25 the phase separation was complete within few hours, whereas for fw = 0.50 the emulsions were highly foamy and O/W type.
A. Emulsification Protocol
After the scans were made, the emulsification protocol was fixed as follows: 250 mL emulsions were prepared at 9500 rpm during 5 minutes. Reproducibility and total phase separation lower than 10% was ensured. To validate the proposed protocol each emulsion was preparedin triplicate and the stability was evaluated during a month. Average standard deviations in the separation were ±0.28, ±0.25 and ±0.14% for the samples of fw = 0.25, 0.50 and 0.75, respectively. Figure 8 shows the validation results.

Figure 8. Emulsification Protocol Validation. W/O emulsions stabilized by a Span 80® and Arkopal N 040® mixture, Cs = 2% v/v, vm = 9100 rpm, tm = 5 min., T = 25°C. fw: ( ) 0.25, (
) 0.25, ( ) 0.50, (
) 0.50, ( ) 0.75
) 0.75
B. Emulsion properties
All the emulsions formulated with Span 80® and Arkopal N 040® were W/O type. This behavior is in agreement with the theoretical background, since the HLB values used were between 4.5 and 6.5. In this case, the surfactant is highly soluble in the oil phase and this affinity favors the formation of W/O emulsion. The average surface diameter of the droplets was obtained by microscopy (Morales et al., 2008) and varies between 3 and 6 microns, but it could not be determined in all cases.
Emulsion density was measured in all tests. Density values varied from 840 to 930 kg/m3 (measured at 38 °C), basically depending on the water content. The influence of surfactant content, HLB, Octanol content and water salinity on density was not considerable, and mixing conditions did not affect density.
Figure 9 shows water content effect on emulsion viscosity. Viscosity depends on the angular speed, indicating that the emulsions are pseudoplastic fluids: the higher the angular speed, the lower the apparent viscosity. The experimental points demonstrate that increasing the water content produce a more pseudoplastic emulsion. For a constant angular speed, apparent viscosity depends strongly on the water content; for a angular speed of 0.63 s-1, for example, apparent viscosity increases from 200 to 30000 mPa.s, when water content changes from 25 to 75% v/v. Although the viscometer operational range is up to 60s-1, this study includes only measurements up to 6 s-1, according to the available data for crude oil emulsions for future comparisons.
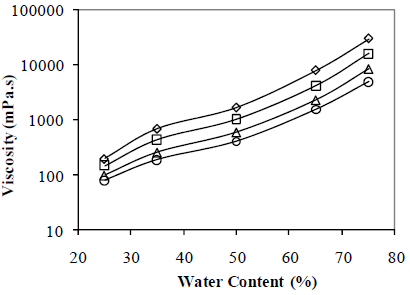
Figure 9. Water Content Effect on Emulsion Apparent Viscosity. W/O Emulsions Stabilized by a mixture of Span 80® and Arkopal N 040® .HLB = 5.5, Cs = 2% v/v, vm = 13400 rpm, tm = 5 min, T = 25°C. Angular speed w:( ) 0.63 s-1, (
) 0.63 s-1, ( ) 1.26 s-1, (
) 1.26 s-1, ( ) 3.14 s-1, (
) 3.14 s-1, ( ) 6.28 s-1
) 6.28 s-1
At low water content, drops are too far apart to interact among each other and the only interaction beyond the homogeneous fluid case is that of each drop with its surrounding fluid. When the number of drops increases, drop-drop interactions become predominant, and the resulting frictional effects drive the viscosity increase.
Apparent viscosity was fitted by a correlation with the power law form. Equivalent consistency index A[mPa.sB] and Power Law Index B[dimensionless] were calculated for each emulsion at different water content values (see Table 1).
Table 1. Correlation Constants for water-in-oil emulsion stabilized by Span 80® and Arkopal N 040®
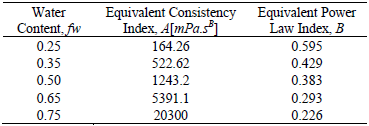
Our results agree with the general experience that demonstrates that the emulsion viscosity increases with the internal phase content. Although many empirical relations have been proposed, their precision cannot be determined since these equations do not take into account the pseudoplastic behavior of the emulsion which can be very significant for high fw values (Salager, 1999). For this reason, a polynomial adjustment of A [mPa.sB] and B [dimensionless], based on the internal phase content, is proposed,
A = 1691505.33fw4 - 2781930.33fw3 + 1673741.30fw2 - 430342.25fw + 40001.21 (2)
B= 35.691fw4 - 78.541fw3 + 62.487fw2 - 21.719fw + 2.2082 (3)
Thus, the apparent viscosity of fresh emulsions can be determined with a model that simultaneously includes the internal phase content effect and the emulsion pseudoplastic behavior as follows,
η = A · ωB-1 (4)
Figure 10 shows angular speed effect on apparent viscosity. Experimental and modeled values are presented. Modeled values were obtained using Eq. 4; equivalent Consistency Index A[mPa.sB] and Power Law Index B were calculated from Eqs. 2 and 3, respectively.
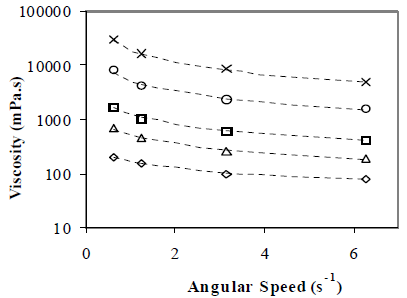
Figure 10. Effect of Angular speed on Apparent Viscosity. W/O Emulsions Stabilized by a mixture of Span 80® and Arkopal N 040® .HLB = 5.5, Cs = 2% v/v, vm = 13400 rpm, tm = 5 min, T = 25°C. Symbols correspond to the experimental values and lines correspond to the modeled ones, fw: ( ) 0.25, (
) 0.25, ( ) 0.35, (
) 0.35, ( ) 0.50, (
) 0.50, ( ) 0.65, (×) 0.75
) 0.65, (×) 0.75
Octanol content and water salinity effect on apparent viscosity was also measured. Results are shown in Figs. 11 and 12, respectively. The higher the alcohol content, the lower the apparent viscosity; the opposite effect is obtained for the water salinity, where the salinity effect is much stronger. The Octanol effect on emulsion viscosity can be explained by a reduction in the external phase viscosity.
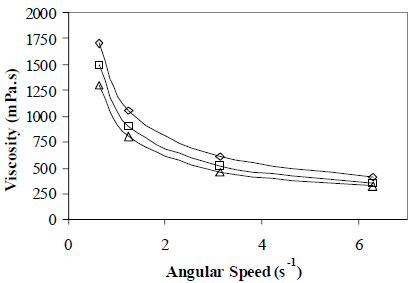
Figure 11. Octanol Content Effect on Emulsion Apparent Viscosity. W/O Emulsions Stabilized by a mixture of Span 80® and Arkopal N 040®. HLB = 5.5, Cs = 2% v/v, fw = 0.50, vm = 13400 rpm, tm = 5 min., T = 25°C, Co: ( ) 0%, (
) 0%, ( ) 2%, (
) 2%, ( ) 4%
) 4%
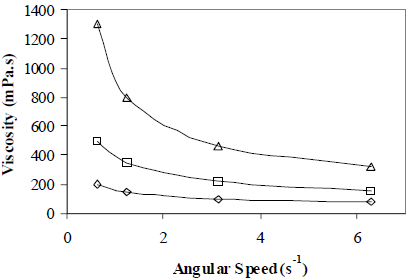
Figure 12. Water Salinity Effect on Emulsion Apparent Viscosity. W/O Emulsions Stabilized by a mixture of Span 80® and Arkopal N 040®. HLB = 5.5, Cs = 2% v/v, fw = 0.25, vm = 13400 rpm, tm = 5 min., T = 25°C, NaCl: ( ) 0 g/dL, (
) 0 g/dL, ( ) 8 g/dL, (
) 8 g/dL, ( ) 16 g/dL
) 16 g/dL
In Fig. 11, for a angular speed of 0.63 s-1 for example, viscosity diminishes by 200 mPa.s when octanol content increases from 2 to 4%, but when salinity increases from 8 to 16 g/dL (see Fig. 12) viscosity increases by 800 mPa.s. Although in this experiment the droplet size could not be measured regularly for all salt concentrations, a decrease in the average surface diameter xs from 5 microns to no detectable values was demonstrated. The reduction in droplet size and a probable narrow size distribution explain the increase in viscosity.
V. CONCLUSIONS
Span 80 and Arkopal N040 mixtures stabilize water in paraffin emulsions in a wide range of water content. A validated protocol shows maximal standard deviations of ± 0.28% in emulsion stability reproducibility and, in all cases, water plus paraffin separation was lower than 6% v/v.
The HLB required to produce a most stable emulsion depends on the emulsion water content; therefore, a HLB scan is recommended when nonionic surfactants are used. Mixing velocity and emulsion batch scans can optimize the emulsion stability.
Surfactant content and water salinity do not influence significantly on stability of water in paraffin emulsions stabilized by Span 80 and Arkopal N040 mixtures. In contrast, octanol presence harms the stability of such emulsions.
Water in paraffin emulsions stabilized by Span 80 and Arkopal N040 mixtures show a pseudoplastic rheological behavior at both low and high internal phase content. A model based on Power Law, which takes into account the water content, reproduces very well the experimental viscosity values. The range of validity of the model should be extended by including a wide range of angular speed. Salinity increases remarkably the emulsion viscosity.
Basic research should be continued with the formulation of highly stable synthetic emulsions, including complex emulsions closer to crude oil emulsions with asphaltenes and solids.
For a future destabilization study, centrifugation, temperature and electrostatic field are recommended. Experiments will be modelled including both, synthetic and crude oil emulsions.
NOMENCLATURE
A consistency index (mPa.sB)
B power law index (-)
Co octanol concentration (%v/v)
Cs surfactant concentration (%v/v)
fw water fraction (-)
HLB average hydrophilic lipophilic balance (-)
T temperature (°C)
tm mixing time (min)
vm mixing velocity (rpm)
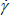 shear rate (s-1)
shear rate (s-1)
η apparent viscosity (mPa.s)
ω angular velocity (s-1)
1 Intermittent I: 5 min at 13400 rpm + 5 min pause + 5 min at 13400 rpm, Intermittent II: 5 min at 8000 rpm + 5 min pause + 5 min at 13400 rpm
ACKNOWLEDGEMENTS
The authors wish to thank CDCH (Humanistic and Scientific Development Center): Projects PI-08-00-6129-2005 and PI-08-00-6127-2005, and DAAD (German Academic Exchange Service) for the economic support of this research.
REFERENCES
1. Antón, R.E., N. Garcés and A. Yajure, "A correlation for three-phase behavior of cation1C surfactant-oil-water systems," J. Dispersion Sci. Technol., 18, 539-555 (1997). [ Links ]
2. Arai, H. and K. Shinoda, "The effect of mixing of oils and of nonionic surfactants on the phase inversion temperatures of emulsions," J. Colloid Interface Sci., 25, 396-400 (1967). [ Links ]
3. Becher, P., Emulsions: Theory and Practice. R. Krieger, Huntington NY (1977). [ Links ]
4. Becher, P. International Symposium on Surfactants in Solution, Lund, Sweden (1982). [ Links ]
5. Becher, P., Encyclopedia of Emulsion Technology, M. Dekker, New York (1996). [ Links ]
6. Becher, P. and W.C. Griffin, HLB: A bibliography 1949-1969, Atlas Chemical Industries (1970). [ Links ]
7. Bourrel, M., J.L. Salager, R.S. Schechter and W.H. Wade, "A correlation for phase behaviour of non-ionic surfactants," J. Colloid Interface Sci. 75, 451-461 (1980). [ Links ]
8. Davies, J.T., Proceedings 2nd Intern. Congress Surface Activity, 1, 426, Butterworth, London (1957) [ Links ]
9. Fox, R. and A. McDonald, Introduction to Fluid Mechanics, John Wiley & Sons Inc, New York (2003). [ Links ]
10. Foyeke, O. and J. Burgess, "Influence of interfacial properties of lipophilic surfactants on water-in-oil emulsion stability," Journal of Colloid and Interface Science. 197, 142-150 (1998). [ Links ]
11. Graciaa, A., J. Lachaise, M. Bourrel, I. Osborne-Lee, I. R.S. Schechter and W.H. Wade, "Partitioning of nonionic and anionic surfactant mixtures between oil/microemulsion/water phases," SPE Reservoir Eng., 2, 305-314 (1984). [ Links ]
12. Griffin, W.C., "Classification of surface active agents by HLB," J. Soc. Cosm. Chem., 1, 311-326 (1949). [ Links ]
13. Kunieda, H. and Shinoda, K., "Evaluation of the hydrophile-lipophile balance (HLB) of nonionic surfactants. I. Multisurfactant systems," Journal of Colloid and Interface Science, 107, 107-121 (1985). [ Links ]
14. Lin, I, "Critical micelle concentration, hidrophil-lipophil balance, effective chain length and hydrophobicity index of ionic surfactants containing two long-chain alkyl groups," Tenside Detergents, 17, 119-123 (1980). [ Links ]
15. Morales, C., U. Riebel, A. Padrón, P. Zúñiga and J.A. Sorrentino, "Use of video enhance microscopy for characterization of solid-liquid-liquid mixtures," Particle and Particle System Characterization, 25, 136 - 141 (2008). [ Links ]
16. Rosen, M., Surfactants and Interfacial Phenomena, John Wiley & Sons, inc., New Jersey, (2004). [ Links ]
17. Salager, J.L., Formulación, composición y fabricación de emulsiones para obtener las propiedades deseadas. Estado del Arte. Parte B. Propiedades de las emulsiones y su medición, Laboratorio FIRP, Mérida (1999). [ Links ]
18. Salager, J.L., "Formulation concepts for the emulsion maker," Pharmaceutical Emulsions and Suspensions (Eds.: F. Nielloud, G. Marti-Mestres), Marcel Dekker, Inc., New York - Basel, 19-125 (2000). [ Links ]
19. Shinoda, K., M. Fukuda and A. Carlsson, "Characteristic solution properties of mono-, di- and triglyceryl alkyl ethers: lipophobicity of hydrophilic groups," Langmuir, 6, 334-337 (1990) [ Links ]
Received: April 14, 2009.
Accepted: March 25, 2010.
Recommended by Subject Editor Orlando Alfano.














