Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Mastozoología neotropical
versión impresa ISSN 0327-9383versión On-line ISSN 1666-0536
Mastozool. neotrop. v.12 n.2 Mendoza jul./dic. 2005
Main food categories in diets of sigmodontine rodents in the Monte (Argentina)
Stella M. Giannoni 1, 2, Carlos E. Borghi 1, 2, Mariana Dacar 1, and Claudia M. Campos 1
1Grupo de Investigaciones de la Biodiversidad , Instituto Argentino de Investigaciones de las Zonas Áridas (CRICYT - CONICET), CC 507, 5500 Mendoza, Argentina. giannoni@lab.cricyt.edu.ar; stella.giannoni@gmail.com. 2 Instituto y Museo de Ciencias Naturales y Departamento de Biología, Facultad de Ciencias Exactas, Físicas y Naturales, Universidad Nacional de San Juan, Argentina.
ABSTRACT: The present study aims at improving the characterization of the dietary status of four sigmodontine rodents (Graomys griseoflavus, Akodon molinae, Calomys musculinus and Eligmodontia typus) by quantifying the major food categories (leaves, stems, seeds, Prosopis flexuosa pods, and arthopods) with a modified microhistological technique. The use of this new technique enhances the likelihood of accurately identifying plant parts, such as stems, seeds and fruits that are usually underestimated. Results reveal that hard tissues, like seeds, are a major resource in the diets while other hard tissues, such as stems and P. flexuosa pods, appear for the first time in the diet of all four species of sigmodontine rodents. Rodent species display considerable trophic flexibility that may be particularly advantageous in arid environments where resource availability is unpredictable and heterogeneous.
Key words. Diet. Food categories. Microhistological technique. Monte desert. Sigmodontine rodents.
INTRODUCTION
The terms "granivore", "insectivore" and "herbivore" refer to animals whose diet contains over 50% of seeds, insects (or arthropods in a wider sense), and green plant parts (leaves, stems) respectively, whereas "omnivore" refers to those species in whose diets no particular category prevails (Kerley and Whitford, 1994).
Desert small mammals adopt one of two alternative dietary strategies, either granivory or herbivory/omnivory (Kerley, 1992). In North American deserts granivorous rodents are dominant, according to the information on diets obtained mostly for species of the family Heteromyidae (Reichman, 1975; Mares and Rosenzweig, 1978; Abramsky, 1983). No evidence of strict granivory by small mammals has been found in Australian, South African or South American deserts, where the most frequent dietary strategies are omnivory, insectivory and herbivory (e.g. Glanz, 1977; Morton, 1979; Meserve, 1981a,b; Morton, 1985; Kerley, 1989, 1992; Murray and Dickman, 1994a, b; Murray et al., 1999; Campos et al., 2001).
The diets of these groups of rodents have been examined using the microhistological technique (Baumgartner and Martin, 1939; Dusi, 1949; Butet, 1987) for analyzing fecal and stomach contents. Nonetheless, some food items as hard tissues, for example stems, fruits, seeds, and roots, may be underestimated in the microhistological analysis (Campos, 1997; Campos et al., 2001). Recently, Dacar and Giannoni (2001) modified this technique in order to increase the likelihood of accurately identifying underestimated plant parts such as stems, seeds, and fruits.
The small mammals that occur in the central-western Monte desert (Argentina) were categorized as omnivores by Campos et al. (2001): Akodon molinae (with a strong tendency to insectivory), Calomys musculinus (with a strong tendency to granivory), Eligmodontia typus and Graomys griseoflavus (with a strong tendency to folivory). Campos et al. (2001) characterized the specific composition of diets by using the microhistological technique, this characterization was as accurate as this technique allowed. So, in this study we attempted to improve diet characterization for the same rodent species and the same habitat as those studied by Campos et al. (2001) by using the microhistological technique modified by Dacar and Giannoni (2001), because it allows identification of hard tissues.
MATERIAL AND METHODS
Study site
The study was conducted in the Biosphere Reserve of Ñacuñán, located in the central-western part of the Mendoza plain (34º 02' S, 67º 58' W), 200 km southeast of Mendoza city, Argentina (Ojeda et al., 1998). The Reserve is in the Monte phytogeographical province (Morello, 1958), and comprises approximately 13 000 ha of xerophytic vegetation. The climate is semiarid with a long-term average annual rainfall of 322 ± 103 mm (ISD, range 192-533; N = 17 yrs), concentrated in the summer months (November to March; mean= 235.22 ± 89.5 mm). Mean daily temperatures are lower than 10ºC in winter and above 20ºC in summer (Ojeda et al., 1998).
The vegetation consists of an open woodland and shrubland steppe (Roig, 1971), and the area has been protected from grazing and human disturbance since 1970. The open mesquite woodland is the most widespread plant community, dominated by Prosopis flexuosa , accompanied by Geoffroea decorticans, a shrub layer of Larrea divaricata , L. cuneifolia, Atriplex lampa, Lycium chilense , and a species-rich herbaceous layer (e.g., Pappophorum caespitosum, Digitaria californica, Setaria leucopila; Roig, 1971).
Mammal species
We studied four sigmodontine rodents (less than 100 g ) that coexist in the Reserve of Ñacuñán: Graomys griseoflavus, Akodon molinae, Calomys musculinus, and Eligmodontia typus. In total, 69 samples of feces and intestinal contents were analyzed: 31 G . griseoflavus, 21 A . molinae, 6 C . musculinus, and 11 E. typus. The material was taken from animals trapped during the dry season (1996-1997; 2001-2002) when, because of food shortage, animals would be forced to eat a wide variety of items. Fecal samples were obtained from animals captured with Sherman traps, whereas samples of intestinal and stomach contents were taken from animals captured with Victor snap-traps.
Diet composition was determined by a microhistological analysis. We used the technique developed by Dacar and Giannoni (2001) for hard tissues such as stems and seeds. This procedure uses a macerating solution of 17.5% of NaHCO 3 to soften and clarify hard tissues. Fifty microscope fields were systematically examined on each slide under a microscope at 400x. Histological features of epidermis of leaves, seeds, fruits, and stems, and arthropod body parts were used to identify food items on slides prepared from stomach and intestinal contents, and feces. Presence of each food item was recorded, and its percent frequency of occurrence was determined on all microscope fields (Holechek and Gross, 1982).
Food items were grouped into the following categories: leaves, seeds, Prosopis flexuosa pods, stems, and arthropods. A multivariate analysis of variance (MANOVA) was used to test for differences among rodent species in the consumption of 5 food categories. The Newman-Keuls test was used to assess differences in the consumption of each food category by all four rodent species (Zar, 1999). The frequency of occurrence of each dietary category was expressed as mean, standard deviation and coefficient of variation (CV). A nonparametric analysis of variance (Kruskall-Wallis) and a Tukey test were used to test for differences among rodent species in the consumption of food categories, per plant species, only considering the most common plant species found in the diets.
RESULTS
Significant differences in the consumption of seeds, stems, leaves, Prosopis flexuosa pods and arthropods were observed among mammal species (Fig. 1). Calomys musculinus consumed the highest proportion of seeds: nearly 60% of its diet was composed mainly of seeds of Lycium spp., Larrea spp., and grasses. The highest proportions of arthropods were found in the diets of Akodon molinae and C. musculinus, although the latter species showed no significant differences compared to Eligmodontia typus. The highest proportion of leaves was found in the diet of Graomys griseoflavus, the most consumed plant species being P. flexuosa. All rodents consumed stems and P. flexuosa pods, showing no differences among them (Fig. 1 and Table 1) .

Fig. 1. Categories consumed by sigmodontine rodents of the Monte desert in the dry season. Letters and numbers indicate differences between means (MANOVA and Newman-Keuls test, P < 0.001).

In analyzing the proportions of the different food categories present in the diet of each sigmodontine species, C. musculinus showed the highest variation in the consumption of the different categories as indicated by the coefficient of variation, which agreed with the high percentages of seed found in its diet, and with the little variation in seed consumption among the individuals studied. On the other hand, E. typus exhibited the least variation in the proportions of the categories consumed, since this species showed an even use of all food categories. In the diet of E. typus, P. flexuosa pods, arthropods and stems were the most highly variable food categories (Table 1). In the case of G. griseoflavus, leaves represented more than 50% of its diet, and was the least variable category; arthropods, instead, were the least consumed but most highly variable category. In the diet of A. molinae, arthropods and seeds were the most abundant and less variable categories (Table 1).
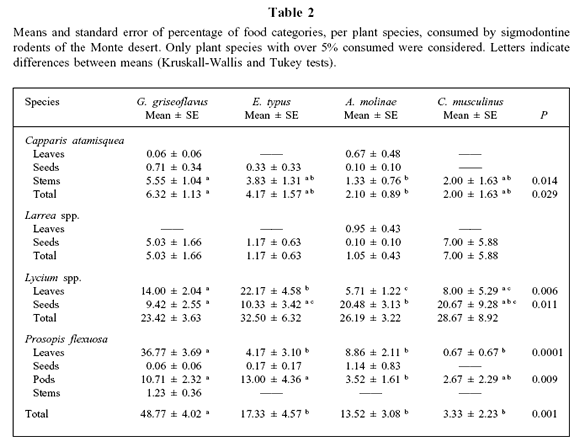
The most consumed plant species (with a frequency higher than 5% in the diet) were Capparis atamisquea, Larrea spp., Lycium spp., and P. flexuosa. All rodent species consumed stems of C. atamisquea, Larrea seeds, leaves and seeds of Lycium spp., and leaves and pods of P. flexuosa (Table 2).
DISCUSSION
Granivory, rather than omnivory, has been frequently considered the most advantageous dietary strategy for desert-dwelling rodents (Price and Brown, 1983; Mares 1993). However, several recent studies on diets and food preferences in African, Australian, and South American deserts have shown that rodents are more usually omnivorous than granivorous (Morton, 1979; Meserve, 1981 a , b; Morton, 1985; Kerley, 1989, 1992; Murray and Dickman, 1994 b; Campos et al., 2001).
Some anecdotal and casual observations have suggested that G. griseoflavus is a folivorous species (Mares, 1973; Rosi, 1983). In Chilean shrublands E. typus has been reported to include a large proportion of insects in its diet (Mann, 1945; Hershkovitz, 1972). In the cropfields of central Argentina , the diet of C. musculinus is composed primarily of seeds (Maldonado Curti, 1989; Dellafiore and Polop, 1994; Ellis et al., 1998). The trophic status of the four most abundant sigmodontine rodents of the Monte desert is poorly known as yet. Campos (1997) and Campos et al. (2001) made a detailed dietary study using a microhistological analysis that may have underestimated some food items such as stems, fruits, seeds, and roots. According to these authors, small rodents of the Monte desert are omnivorous, with a tendency towards granivory (C. musculinus), insectivory (A. molinae), and folivory (G. griseoflavus).
Results of the present study, using a new technique developed for increasing accuracy in the identification of plant parts, support preliminary indications on the dietary status as stated by Campos et al. (2001), however some interesting differences were found. Campos et al. (2001) found a higher use of leaves by all four rodent species compared to the present study. Although some hard items, such as stems, were found for the first time, others, such as pods and seeds, increased the percentage of occurrence in diets of Monte desert rodents.
Higher proportions of seed found in the diets reveal a higher use of this resource. This is consistent with results of ongoing studies on food hoarding (Giannoni et al., 2001), seed handling ability, and ability of sigmodontine rodents to find buried seeds (P. Taraborelli, 2003, in litt.). Prosopis pods are a major source of nutrients for Dipodomys merriami (Reynolds and Glendening, 1949) and also for small mammals such as G. griseoflavus and E. typus. These results support field experiments that indicate that P. flexuosa pods are intensively removed by small rodents of the Monte (C. Campos and C. Borghi, pers. obs.). Moreover, considering that P. flexuosa produces fruit during the wet season, consumption of pods and seeds during the dry season strongly suggests that rodents feed on stored pods, especially because P. flexuosa seeds were not found in the soil seed-bank during the dry season (Marone et al., 1998). Stems were also consumed by all sigmodontines, particularly by G. griseoflavus , the most herbivorous species. For other herbivores of the Monte desert (Microcavia australis), bark consumption was recorded during the dry season due to low availability of green vegetation (Borruel et al., 1998; Tognelli et al., 1999), and this could also be related to the high water and carbohidrate contents of bark (Frank, 1988).
In this study, results reflect rodent diets in periods of harsh conditions, such as drought, that coincide with periods of maximum relative abundance of rodents (autumn). So the trophic status of rodents indicates what they are capable of eating for surviving the season of most critical food shortage, rather than their potentially preferred food items in seasons of food abundance. According to definitions of the terms "granivore", "insectivore", "herbivore" and "omnivore" (Kerley and Whitford, 1994), the present study supports the suggestion that C. musculinus is a granivore, G. griseoflavus an herbivore, and that E. typus and A. molinae are omnivores, although the latter exhibits a strong tendency towards insectivory.
Like in other deserts, species display considerable dietary flexibility, as indicated by the high standard deviations found in all five food categories. This flexibility may be particularly advantageous in arid environments where resource availability is unpredictable and heterogeneous (Reichman, 1975; Meserve, 1981b; Glanz, 1982; Meserve et al., 1988; Stafford Smith and Morton, 1990). These results show a new and more complex scenario in the relationship of South American rodents with desert plants, increasing the importance of plant parts other than leaves, i.e. fruits, stems and seeds, in these interactions.
ACKNOWLEDGEMENTS
This research was partly funded by CONICET Grant PIP Nº 4684, PICT Nº 03281, and PIP Nº 2884. We thank G. Diaz and A. Orofino (Grupo de Investigaciones de la Biodiversidad, IADIZA-CRICYT-CONICET) for providing us with some samples of feces and intestinal contents. Nelly Horak assisted us in preparing the English version.
LITERATURE CITED
ABRAMSKY Z. 1983. Experiments on seed predation by rodents and ants in the Israeli desert. Oecologia 57:328-332. [ Links ]
BAUMGARTNER LL and AC MARTIN. 1939. Plant histology as an aid in squirrel food-habitat studies. Journal of Wildlife Management 3:266-268. [ Links ]
BORRUEL N, CM CAMPOS, SM GIANNONI, and CE BORGHI. 1998. Effect of herbivorous rodents (cavies and tuco-tucos) on a shrub community in the Monte desert. Journal of Arid Environments 39:33-37. [ Links ]
BUTET A. 1987. L'analyse microscopique des feces: une technique non perturbante d'etude des regimes alimentaires des mammiferes phytophages. Arvicola IV:33-38. [ Links ]
CAMPOS CM. 1997. Utilización de recursos alimentarios por mamíferos medianos y pequeños del desierto del Monte. Tesis Doctoral Inédita, Universidad Nacional de Córdoba, Argentina. [ Links ]
CAMPOS CM, RA OJEDA, S MONGE, and M DACAR. 2001. Utilization of food resources by small and medium-sized mammals in the Monte Desert biome, Argentina. Austral Ecology 26:142-149. [ Links ]
DACAR M and SM GIANNONI. 2001. Technical note: A simple method for preparing reference slides of seed. Journal of Range Management 54:191-193. [ Links ]
DELLAFIORE CM and JJ POLOP. 1994. Feeding habits of Calomys musculinus in the cropfield and its borders. Mastozoología Neotropical 1:45-50. [ Links ]
DUSI J L. 1949. Method for the determination of food habits by plant microtechniques and histology and their application on cottontail rabbit food habits. Journal of Wildlife Management 13:295-298. [ Links ]
ELLIS BA, JN MILLS, GE GLASS, KT MCKEE Jr., DA ENRIA, and E CHILDS. 1998. Dietary habits of the common rodents in a agroecosystem in Argentina. Journal of Mammalogy 79:1203-1220. [ Links ]
FRANK CL. 1988. Diet selection by a heteromyid rodent: role of net metabolic water production. Ecology 69:1943-1951. [ Links ]
GIANNONI SM, M DACAR, P TARABORELLI, and CE BORGHI. 2001. Seed hoarding by rodents of the Monte desert, Argentina. Austral Ecology 26:259-263. [ Links ]
GLANZ WE . 1977. Comparative ecology of small mammal communities in California and Chile. PhD thesis, University of California, Berkeley . [ Links ]
GLANZ WE. 1982. Adaptive zones of Neotropical mammals: a comparison of some temperate and tropical patterns. Pp. 95-110, in: Mammalian Biology in South America (MA Mares and HH Genoways, eds.). Special Publications, Series N° 6, Pymatuning Laboratory of Ecology, Linnesville, PA. [ Links ]
HERSHKOVITZ P. 1972. The Recent Mammals of the Neotropical Region: A Zoogeographic and Ecological Review. Pp. 311-431, in: Evolution, Mammals, and Southern Continents (AF Keast, C Erk, and BP Glass, eds.). New York Press, New York . [ Links ]
HOLECHEK JL and BD GROSS. 1982. Evaluation of different calculation procedures for microhistological analysis. Journal of Range Management 35:721-730. [ Links ]
KERLEY GIH. 1989. Diet of small mammals from the Karoo, South Africa. South African Journal of Wildlife Research 19:67-72. [ Links ]
KERLEY GIH. 1992. Trophic status of small mammals in the semi-arid Karoo, South Africa . Journal of Zoology 226:563-572. [ Links ]
KERLEY GIH and WG WHITFORD. 1994. Desert-dwelling small mammals as granivores: intercontinental variations. Austral Journal of Zoology 42:543-555. [ Links ]
MALDONADO CURTI S. 1989. Impacto del pastoreo sobre micromamíferos: un estudio comparativo en ambientes semiáridos. Tesis de Maestría, Facultad de Ciencias, Universidad de Chile. [ Links ]
MANN G. 1945. Mamíferos de Tarapaca: Observaciones realizadas durante una expedición al alto norte de Chile. Biológica 2:23-141. [ Links ]
MARES MA. 1973. Climates, mammalian communities and desert rodent adaptations: An investigation into evolutionary convergence. Ph.D. Thesis, University of Texas at Austin . [ Links ]
MARES MA. 1993. Desert rodents, seed consumption, and convergence. BioScience 43:162-379. [ Links ]
MARES MA and ML ROSENZWEIG. 1978. Granivory in North and South American deserts: rodents, birds, and ants. Ecology 59:235-241. [ Links ]
MARONE L, BE ROSSI, and ME HORNO. 1998. Timing and spatial patterning of seed dispersal and redistribution in a South American warm desert. Plant Ecology 137:143-150. [ Links ]
MESERVE PL. 1981a. Resource partitioning in a Chilean semi-arid small mammal community. Journal of Animal Ecology 50:745-57. [ Links ]
MESERVE PL. 1 981b. Trophic relationships among small mammals in a Chilean semiarid thorn scrub community. Journal of Mammalogy 62:304-14. [ Links ]
MESERVE PL, BK LANG, and BD PATTERSON. 1988. Trophic relationships of small mammals in a Chilean temperate rainforest. Journal of Mammalogy 69:721-30. [ Links ]
MORELLO J. 1958. La provincia fitogeográfica del Monte. Opera Lilloana 2:1-155. [ Links ]
MORTON SR. 1979. Diversity of desert-dwelling mammals; a comparison of Australia and North America. Journal of Mammalogy 60:253-64. [ Links ]
MORTON SR. 1985. Granivory in arid regions: comparison of Australia with North and South America. Ecology 66:1859-66. [ Links ]
MURRAY BR and CR DICKMAN. 1994 a . Granivory and microhabitat use in Australian desert rodents: are seeds important? Oecologia 99:216-225. [ Links ]
MURRAY BR and CR DICKMAN. 1994b. Food preferences and seed selection in two species of Australian desert rodents. Wildlife Research 21:647-655. [ Links ]
MURRAY BR, CR DICKMAN, CH WATTS, and SR MORTON. 1999. The dietary ecology of Australian desert rodents. Wildlife Research 26:421-437. [ Links ]
OJEDA RA, CM CAMPOS, JM GONNET, CE BORGHI, and V ROIG. 1998. The MaB Reserve of Ñacuñán , Argentina: its role in understanding the Monte Desert biome. Journal of Arid Environments 39:299-313. [ Links ]
PRICE MV and J BROWN. 1983. Patterns of morphology and resources use in North American desert rodent communities. Great Basin Naturalist Memoirs 7:117-134. [ Links ]
REICHMAN OJ. 1975. Relation of desert rodent diets to available resources. Journal of Mammalogy 56:731-751. [ Links ]
REYNOLDS HG and GE GLENDENING. 1949. Merriam Kangaroo Rat a Factor in Mesquite Propagation on Southern Arizona Range Lands. Journal of Range Management 2:193-197. [ Links ]
ROIG, FA. 1971. La Reserva Forestal de Ñacuñan. Deserta, 1:21-239. [ Links ]
ROSI MI. 1983. Notas sobre la ecología, distribución y sistemática de Graomys griseoflavus griseoflavus (Watherhouse, 1837) (Rodentia, Cricetidae) en la provincia de Mendoza. Historia Natural 3(17):161-167. [ Links ]
STAFFORD-SMITH, DM, and SR MORTON. 1990. A framework for the ecology of arid Australia. Journal of Arid Environments 18(3):255-278 [ Links ]
TOGNELLI MF, CE BORGHI, and CM CAMPOS. 1999. Effect of gnawing by Microcavia australis (Rodentia, Caviidae) on Geoffroea decorticans (Leguminosae) plants. Journal of Arid Environments 41:79-85. [ Links ]
ZAR JH. 1999. Biostatistical Analysis. 4th Edition. 661 pp. Prentice-Hall International, Inc. [ Links ]
Recibido 7 mayo 2004.
Aceptación final 19 mayo 2005.














