Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Mastozoología neotropical
Print version ISSN 0327-9383
Mastozool. neotrop. vol.18 no.2 Mendoza July/Dec. 2011
ARTÍCULOS Y NOTAS
New record of a stranded sperm whale (Physeter macrocephalus) and a review of strandings along the continental Argentine coast
Mariana Degrati1,2, Néstor A. García1, M. Florencia Grandi1, M. Soledad Leonardi1, Rocío Loizaga de Castro1,2, Damián G. Vales1, Silvana L. Dans1,2, Susana N. Pedraza1,2, and Enrique A. Crespo1,2
1 Centro Nacional Patagónico (CENPAT-CONICET), Bvd. Brown 2915, 9120 Puerto Madryn, Chubut, Argentina [Correspondence: M. Degrati <degrati@cenpat.edu.ar>].
2 Universidad Nacional de la Patagonia "San Juan Bosco", Bvd. Brown 3051, 9120 Puerto Madryn, Chubut, Argentina.
ABSTRACT: We present a new record of sperm whale with notes on diet, age determination and a review of strandings along the continental Argentine coast. A complete necropsy was made in the field, and morphometric data were taken. The specimen stranded was a mature male of 64+ years old. This is the oldest specimen determined for a sperm whale in the Southwest Atlantic coast. In the stomach contents a total of 302 cephalopod lower beaks were recovered. Cestodes were present in the blubber but parasites were not found in stomach and intestine contents.
RESUMEN: Nuevo registro de un cachalote (Physeter macrocephalus) varado y una revisión de varamientos en las costas de Argentina continental. Presentamos un nuevo registro de un cachalote, con notas sobre la dieta, la determinación de edad y la presencia de parásitos, junto con una revisión de los varamientos a lo largo de toda la costa continental argentina. El animal varado fue un macho maduro de 64+ años. Este es el individuo más longevo registrado para esta especie en el Atlántico Sudoccidental. En el contenido estomacal se colectaron 302 picos inferiores de cefalópodos. Los únicos parásitos presentes fueron cestodes en la capa de grasa; no se encotraron helmintos gastrointestinales.
Key words: Age determination; Continental Argentine coast; Sperm whale; Stranding.
Palabras clave: Cachalote; Costa continental argentina; Determinación de edad; Varamientos.
The sperm whale, Physeter macrocephalus, is the largest toothed cetacean and one of the best known cetacean species. It has been captured since 19th century and as a result of this, the sperm whale is currently listed as "Vulnerable" by the IUCN (Taylor et al., 2008). This species has a cosmopolitan distribution across the oceans, ranging through the tropics to the ice barrier in both hemispheres (Whitehead, 2009). It shows a strong sexual dimorphism and a high degree of geographical segregation between sexes (Jefferson et al., 1994). Pods of females with calves stay on or near the breeding grounds throughout the year, roughly between 40º N and 40º S (Rice, 1989; Whitehead, 2009). On the other hand, adult males undertake long migrations between low-latitude breeding areas and high-latitude feeding grounds (Best, 1979). Sperm whales usually move in herds of up to 50 individuals (Jefferson et al., 1994; Bastida and Rodríguez, 2003). Their social system has been studied in detail and herds are segregated by age and sex. Males are temporarily associated with groups of females and their offspring for a short period of time. Mature but non-breeding males are segregated from their maternal herds and form solitary male's groups, which can be compose by adults and sub-adult individuals (Jefferson et al., 1994).
Despite sperm whale is a cosmopolitan species little is known about its ecology in the Southwestern Atlantic. Strandings of fresh specimens in the continental Argentine coastline (from 34º to 52.3º S) are rare as a consequence of the pelagic and deep feeding habits of the species and the extremely wide and shallow continental shelf. Here we present a new record of a stranded sperm whale with notes on age determination, diet, parasites and a review of strandings along the Argentine continental coast.
In April 2010 a dead male sperm whale (Fig. 1) was found stranded in Los Cangrejales beach (43º 22' S, 65º 02' W), Chubut Province, Argentina (Fig. 2). Fortunately, the specimen was in fresh condition in order to perform parasitological and trophic studies. A complete necropsy was performed in the field, and morphometric data (summarized in Table 1) were taken following Norris (1961). Excepting the scars in its anterior region as a consequence of inter and intra-specific fights, reported elsewhere in the literature (McCann, 1974; Best, 1979; Kato, 1984) the sperm whale had no additional lesions in its skin. Stomach and intestine contents, teeth, skin and blubber samples were taken and preserved. One mandibular tooth was selected and sectioned along the sagittal plane (bucolingual plane). One half-section was polished with sand paper (400-1000 grit) until clear and easily discernible dentinal growth layers groups (GLGs) were visible. Then GLGs were counted and age estimated considering each GLG as one-year period (Bow and Purday, 1966; Perrin and Myrick, 1980). Counts were performed by three observers independently and the final age was determined as the most repeated count (the mode) of all observers pooled (Evans et al., 2002). Parasites and the stomach content were fixed in 70% ethanol. The items were identified by means of the reference collection of the Marine Mammal Lab (LAMAMA) and published guides (Clarke, 1980; 1986).

Fig. 1. Sperm whale stranded in Los Cangrejales, Chubut, Argentina, in April 2010.
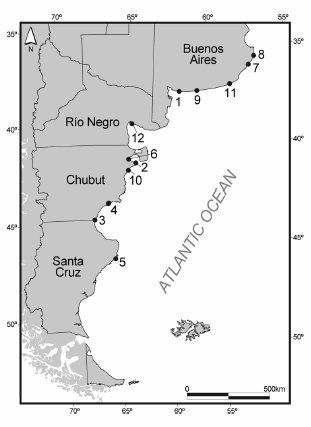
Fig. 2. Map showing the points of stranding of sperm whales in the Argentine continental coast. The numbers make reference to stranding listed in Table 2.
Table 1
Selected measurements of a sperm whale stranded in Los Cangrejales, Chubut, Argentina. All measurements are given in cm; L= left. We also registered 25 teeth in each side of the lower jaw and 6 in the upper jaw.

The specimen stranded at Los Cangrejales was a mature male of 14.7 m and 64+ years old. The polished surface of the tooth section is shown in Fig. 3a. The tooth was worn and the neonatal line could not be identified clearly. The first apical line was considered to be the first GLG corresponding to the first year of life, since this line agreed with the starting point of cementum. This specimen was probably older than 64 years, but no more than one or two GLGs over this estimation. The tooth also showed osteodentine in several parts along the occluded pulp cavity (Fig. 3a).

Fig. 3. Photograph of sperm whales teeth with the detail of: a) 64+, b) 29, c) 35 d) 45, and e) 49 GLGs (photograph a, b, c and e correspond to a lower jaw tooth and d correspond to a thin ground section of upper jaw tooth)
In the stomach contents we could only found cephalopod beaks. No soft parts of cephalopods or fish remains were found. A total of 302 cephalopod lower beaks were recovered, which was identified as Mesonychoteuthis hamiltoni (n = 99), Kondakovia longimana (n = 22), Gonatus antarcticus (n = 57), Haliphron atlanticus (n = 19), two species of Histioteuthis spp. (n = 87) and three unidentified species (n = 18). All these species have been recorded previously in the diet of the sperm whale (Pascoe et al., 1990; Crespo et al., 1990; Santos et al., 1999; 2002; Evans and Hindel, 2004a). With the exception of H. atlanticus, which is a cosmopolitan species, the rest of the cephalopods belongs to species with Antarctic or sub-Antarctic distribution (Xavier and Cherel, 2009). Therefore, their presence in the stomach contents suggests that the whale was foraging in Antarctic or Sub Antarctic waters and moving south-north prior to the stranding in northern Patagonia. The absence of any flesh remains, or fresh beaks with soft cartilaginous tissues, makes it difficult to draw any conclusions on the temporal movements of the whale within these areas.
Regarding parasites, only one species was found. Cestodes were present in the blubber. The blubber-encysted larval cestodes, Phyllobothrium delphini (Cestoda: Tetraphyllidea), is probably the most frequent parasite that appear in deep diving odontocetes and it was reported infecting sperm whale individuals by several authors (Baylis, 1932; Baer, 1961; Dailey and Brownell, 1972; González de Baccino and Praderi, 1984; Raga and Balbuena, 1987; Crespo et al., 1990). Despite the fact that both stomach and intestine contents were fully examined, parasites were not found. Nevertheless, some species of Acantocephala (Bolbosoma sp.) and Nematoda (Anisakis sp.) were documented previously for the sperm whale (Baylis, 1932; Lambertsen and Kohn, 1987; Crespo et al., 1990). Several helminths are significant from a disease standpoint and include species of four genera of nematodes (Anisakis, Stenurus, Halocercus and Crassicauda) and four genera of trematodes (Nasitrema, Campula, Pholeter and Braunina). The cestodes and the acanthocephalan apparently cause little harm to animals (Dailey, 1985), thus in this case, parasites could not be the death cause, although it is unknown.
The present work reports the maximum age determined for a male sperm whale in the Southwestern Atlantic. On the Brazilian coast (from 4º N to 33.6º S) the older male reported was 13.8 m in length and 40 years old (Ramos et al., 2001). The age structure of a male pod stranded along the Adriatic Coast, Italy, revealed that ages were between 15 and 25 years (Mazzoriol et al., 2011). In the case of females, the maximum age was also determined in 64 years, among several female groups' mass strandings in Australia (Evans and Hindell, 2004b).
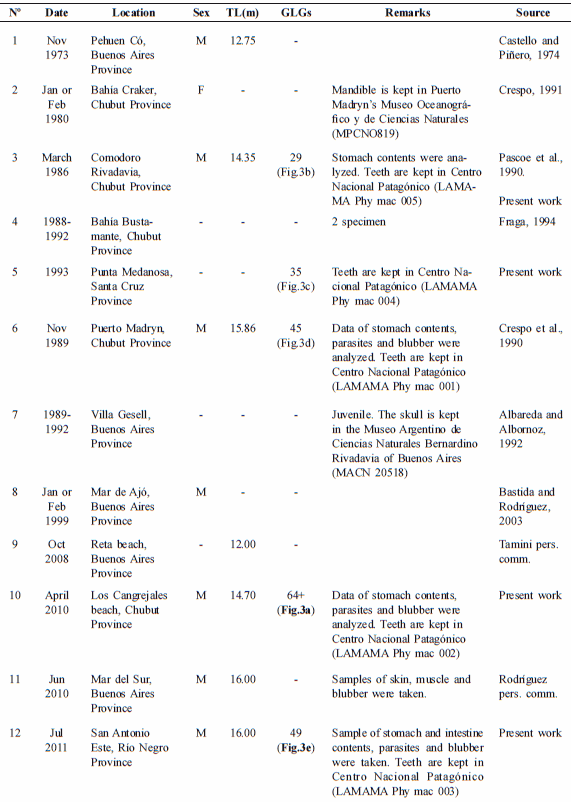
This work reports a total of thirteen strandings of sperm whale occurred in the Argentine continental coast between 1973 and the present (Table 2, Fig. 2). Most of these stranding events corresponded to solitary and mature males. Four other individuals of known age were 29, 35, 45 and 49 years old (Table 2, Figs. 3b-e). In Tierra del Fuego Island (from 52.5º to 55º S), most strandings involved single males, possibly mature and old, since many of them were larger than 14 m (Pimper et al., 2008). One-third of stranded animals on the Brazilian coast were males, while only 6% were females. Although ages are unknown for most of these males, length-to-age data indicated mature as well as immature individuals (the maximum length was 18 m for males; Ramos et al., 2001).
Table 2
Records of stranding of sperm whale, Physeter macrocephalus along the Argentine continental coast. M: male, F: female, TL: total length. GLGs: dentinal growth layers groups
In Tierra del Fuego Island, almost half of stranded animals documented from 1908 to 1983, corresponded to mass strandings. Four mass strandings were reported between 1977 and 1981, and these events involved mainly young immature or maturing males between 12 and 20 years old (Pimper et al., 2008). On the other hand, only one mass stranding involving 33 animals was recorded along the Brazilian coast from 1967 to 2000, although neither age nor length data were published (Castello and Piñero, 1974; Ramos et al., 2001). Both Brazil and Tierra del Fuego constitute areas close to the points with the highest concentration of these cetaceans, the low-latitude breeding areas and the high-latitude feeding grounds respectively, therefore it would be areas of higher occurrence of mass strandings.
In contrast with the Brazilian and Tierra del Fuego coast, no mass stranding was reported along the continental coast off Argentina. The lack of records of mass strandings and the low frequency of single strandings of sperm whales are related to environmental factors. The extremely wide and shallow Argentine continental shelf would not be the appropriate habitat for sperm whales, therefore their presence is not expected in shallow and coastal waters.
On the other hand, it is difficult to assess the cause of the single strandings off the Argentine coast. These specimens would be migrating between feeding and breeding areas through the continental slope, at approximately 200 nm far from the coastline, and then they would have entered waters of the continental shelf searching for alternative preys if food was scarce. The other option is that they could be unhealthy and disorientated and finally died. Studies on cetaceans elsewhere in the world support the general hypothesis that trends in strandings can result from climatic variation and, in particular, climatic effects on food availability (e.g. Evans et al., 2005; Walker et al., 2005; Pierce et al., 2007). Pierce et al. (2007) showed that a rise in the annual temperature anomaly may reflect changes in the distribution of the main sperm whales' squid prey and it is associated with a higher probability of strandings in the North Sea.
This note constitutes a new and valuable contribution to the knowledge of the species and along with those findings of Ramos et al. (2001) and Pimper et al. (2008) complete the long list of sperm whales stranded in the Southwestern Atlantic Ocean. As more data become accumulated a better understanding of the status and life history of the sperm whale emerges.
Acknowledgements. The authors would like to thank Centro Nacional Patagónico (National Research Council of Argentina - CONICET) for logistic and institutional support. Funds were received from Fundación BBVA BIOCON 04. Also we like to thank M. Buono, A. Varisco, N. Martínez Román, G. Svendsen, R. González, S. Montanelli and staff from Dirección de Fauna de la Provincia de Chubut for their collaboration in the field. We also thank M. Buono and an anonymous referee who provided many constructive suggestions and comments, which much improved this manuscript. At the time this manuscript was written, M.D., M.F.G., R.L.C., M.S.L. and D.G.V. were supported by a Fellowship from National Research Council of Argentina (CONICET).
LITERARURE CITED
1. ALBAREDA D and N ALBORNOZ. 1992. Varamientos y avistajes de mamíferos marinos en el área costera del Cabo San Antonio, provincia de Buenos Aires y adyacencias. V Reunión de Trabajo de Especialistas en Mamíferos Acuáticos de América del Sur. Buenos Aires. Argentina. [ Links ]
2. BASTIDA R and D RODRIGUEZ. 2003. Mamíferos marinos de Patagonia y Antártida (Vazquez Mazzini, ed.). Buenos Aires. Argentina. [ Links ]
3. BAER JC. 1961. Embranchement des acanthocéphales. Pp. 733-782, in: Traite de Zoologie: Anatomie, Systématique, Biologie IV (1) (PP Grassé, ed.). Masson et Cie Editeur. Paris [ Links ]
4. BAYLIS HA. 1932. A list of worms parasitic in cetacea. Discovery Reports, 6:393-418 [ Links ]
5. BEST PB. 1979. Social organization in sperm whales, Physeter macrocephalus. Pp. 227-289, in: Behaviour of marine animals Vol. 3 (HE Winn and BL Olla, eds.). Plenum, New York. [ Links ]
6. BOW JM and C PURDAY. 1966. A method of preparing sperm whale teeth for age determination. Nature, London 210(5034):437-8. [ Links ]
7. CASTELLO HP and ME PIÑERO. 1974. Varamientos de cachalotes, Physeter catodon, en las costas del Atlántico del Brasil y la Argentina (Cetacea, Physeteridae). Physis 86:371-374. [ Links ]
8. CLARKE MR. 1980. Cephalopoda in the diet of sperm whales of the Southern Hemisphere and their bearing on sperm whale biology. Discovery Reports 37:1e324. [ Links ]
9. CLARKE MR. 1986. A Handbook for the Identification of Cephalopod Beaks. Oxford University Press, Oxford. [ Links ]
10. CRESPO EA. 1991. Cachalote, ballena de esperma, Physeter macrocephalus. Pp. 121-124, in: Estado de conservación de los mamíferos marinos del Atlántico Sudoccidental. Informes y estudios del Programa de Mares Regionales del PNUMA (HL Capozzo and M Junín, eds.). Nairobi, Kenya, No. 138, PNUMA. [ Links ]
11. CRESPO EA, M KOEN-ALONSO, NA GARCÍA, L REYES, M COZZUOL, J CORCUERA, and M LEWIS. 1990. Varamiento de un cachalote (Physeter macrocephalus) en la costa del Golfo Nuevo, Chubut, Argentina. IV Reunión de Trabajo de Especialistas en Mamíferos Acuáticos de América del Sur. Valdivia. Chile. [ Links ]
12. DAILEY MD. 1985. Diseases of mammalia: cetacea. Pp. 805-847, in: Disease of marine animals, Vol. IV, Part 2. Introduction reptilia, aves, mammalia (O Kinne, ed.). Biologische Anstalt Helgoland, Hamburg. [ Links ]
13. DAILEY MD and RL BROWNELL. 1972. A checklist of marine mammals' parasites. Pp. 528-589, in: Mammals of the Sea, Biology and Medicine (DH Ridgwad, ed.). C.C. Thomas, Springfield, Illinois. [ Links ]
14. EVANS K, AM HINDALL, K ROBERTSON, C LOCKYER, and D RICE. 2002. Factors affecting the precision of age determination on whales (Physeter macrocephalus). Journal of Cetacean Research and Management 4:193-201. [ Links ]
15. EVANS K and MA HINDELL. 2004a The diet of sperm whales (Physeter macrocephalus) in southern Australian Waters. ICES Journal of Marine Science 61:1313-1329. [ Links ]
16. EVANS K and MA HINDELL. 2004b The age structure and growth of female sperm whales (Physeter macrocephalus) in southern Australian Waters. Journal of Zoology, London 263:237-250. [ Links ]
17. EVANS K, R THRESHER, RM WARNEKE, CJA BRADSHAW, M POOK, D THIELE, and MA HINDELL. 2005. Periodic variability in cetacean strandings: links to large-scale climate events. Biological Letters 1: 147-150. [ Links ]
18. FRAGA F. 1994. Proyecto Bustamante: Estudios sobre mamíferos marinos en islas y costas, al sur de la Provincia del Chubut. VI Reunión de Trabajo de Especialistas en Mamíferos Acuáticos de América del Sur. Florianópolis, Brasil. [ Links ]
19. GONZÁLEZ DE BACCINO R and R PRADERI. 1984. Observaciones sobre un varamiento poco usual de cachalote (Physeter macrocephalus), en la costa Atlántica de Uruguay. Actas de I Reunión de Trabajo de Especialistas en Mamíferos Acuáticos de América del Sur. Buenos Aires, Argentina. [ Links ]
20. JEFFERSON TA, S LEATHERWOOD, and MA WEBBER. 1994. FAO species identification guide. Marine Mammals of the World. [ Links ]
21. KATO H. 1984. Observation of tooth scars on the head of males sperm whale, as an indication of intra-sexual figthings. The Scientific reports of the Whales Research Institute 35:39-47. [ Links ]
22. LAMBERTSEN RH and BA KOHN. 1987. Unusual multisystenemic pathology in a sperm whale bull. Journal of Wildlife Diseases 23(3):510-514. [ Links ]
23. MCCANN C. 1974. Body scaring on cetacean-odonocetes. The Scientific Reports of the Whales Research Institute 26:145-157. [ Links ]
24. MAZZARIOL S, G DI GUARDO, A PETRELLA, L MARSILI, CM FOSSI, et al. 2011. Sometimes Sperm Whales (Physeter macrocephalus) Cannot Find Their Way Back to the High Seas: A Multidisciplinary Study on a Mass Stranding. PLoS ONE 6(5):e19417. doi:10.1371/journal.pone.0019417. [ Links ]
25. NORRIS KS. 1961. Standardized methods for measuring and recording data on the smaller cetaceans. Journal of Mammalogy 42(4):471-476. [ Links ]
26. PASCOE PL, M MICKIEWICZ, and HP CASTELLO. 1990. Cephalopod remains from the stomach of a sperm whale stranded off Patagonia. Marine Biology 104:1-4. [ Links ]
27. PIERCE GJ, MB SANTOS, C SMEENK, A SAVELIEV, and AF ZUUR. 2007. Historical trends in the incidence of strandings of sperm whales (Physeter macrocephalus) on North Sea coasts: An association with positive temperature anomalies. Fisheries Research 87:219-228. [ Links ]
28. PERRIN WF and AC MYRICK JR. 1980. Report of the workshop. Pp. 1-51, in: Age determination of toothed whales and sirenians. Report of the International Whaling Commission (Special Issue 3). La Jolla, California. [ Links ]
29. PIMPER L, N GOODALL, J GIBBSONS, A SOBRAL, C LOCKYER, and R PRADERI. 2008. A review of strandings of sperm whales from the Strait of Magellan to Cape Horn. Santiago de Chile. Report of the International Whaling Commission SC/60/O3.[Unpublished] [ Links ]
30. RAMOS RMA, S SICILIANO, M BOROBIA, NA ZERBINI, JLA PIZZORNO, ABL FRAGOSO, J LAILSON‑ BRITO JR, A FREITAS-AZEVEDO, PC SIMÕES LOPES and MC OLIVEIRA-SANTOS. 2001. A note on strandings and age of sperm whales (Physeter macrocephalus) on the Brazilian coast. Journal of Cetacean Research and Management 3(3):321-327. [ Links ]
31. RAGA JA and JA BALBUENA. 1987. Algunas características zoogeográficas de los helmintos de los cetáceos en el Mediterráneo, con especial referencia a la helmintofauna del delfín listado. Pp. 195-201, in: Mamíferos y helmintos. Retres, Barcelona. [ Links ]
32. RICE DW. 1989. Sperm whale Physeter macrocephalus Linnaeus, 1758. Pp. 177-233, in: Handbook of Marine Mammals, Volume 4, River Dolphins and the Larger Toothed Whales (SH Ridgway and RJ Harrison, eds.). Academic Press, London. [ Links ]
33. SANTOS MB, GJ PIERCE, PR BOYLE, RJ REID, HM ROSS, IAP PATTERSON, CC KINZE, S TOUGAARD, R LICK, U PIATKOWSKI, and V HERNÁNDEZ-GARCÍA. 1999. Stomach contents of sperm whales Physeter macrocephalus stranded in the North Sea 1990-1996. Marine Ecology Progress Series 183:281-294. [ Links ]
34. SANTOS MB, GJ PIERCE, M GARCIA-HARTMANN, C SMEENK, MJ ADDINK, T KUIKEN, R RJ EID, IAP PATTERSON, C LORNAN, E ROGAN, and E MENTE. 2002. Additional notes on stomach contents of sperm whales Physeter macrocephalus stranded in the NE Atlantic. Journal of the Marine Biological Association of the U.K. 82:501-507. [ Links ]
35. TAYLOR BL, R BAIRD, J BARLOW, SM DAWSON, J FORD, JG MEAD, G NOTARBARTOLO DI SCIARA, P WADE, and RL PITMAN. 2008. Physeter macrocephalus. In IUCN Red List of Threatened Species. Version 2010.4. http://www.iucnredlist.org. [ Links ]
36. WALKER RJ, EO KEITH, AE YANKOVSKY, and DK ODELL. 2005. Environmental correlates of cetacean mass stranding sites in Florida. Marine Mammal Science 21:327-335. [ Links ]
37. WHITEHEAD H. 2009. Sperm Whale (Physeter macrocephalus). Pp. 1091-1097, in: Encyclopedia of Marine Mammal. 2nd edition (WF Perrin, B Wursig, and JGM Thewissen, eds.). Academic Press. London. [ Links ]
38. XAVIER JC and Y CHEREL. 2009. Cephalopod Beak Guide For The Southern Ocean. British Antarctic Survey, Cambridge, UK. [ Links ]
Recibido 28 junio 2011.
Aceptado 8 setiembre 2011.
Editor asociado: U Pardiñas














