Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Mastozoología neotropical
Print version ISSN 0327-9383
Mastozool. neotrop. vol.19 no.1 Mendoza June 2012
ARTÍCULOS Y NOTAS
Testing simple criteria for de age estimation of six hunted mammal species in Brazilian amazon
Ana Cristina Mendes-Oliveira1, Susanne L. S. de Maria1, Renata Cecília Soares de Lima1, Alexandre S. Fernandes1, Paula C. R. de Almeida1, Luciano F. A. Montag1, and Oswaldo de Carvalho Jr.2
1 Laboratório de Ecologia e Zoologia de Vertebrados, Instituto de Ciências Biológicas, Universidade Federal do Pará, Augusto Corrêa 01, Campus Guamá, Belém, PA, Brazil. CEP 66075-110 [Correspondence: Ana Cristina Mendes-Oliveira <Cris.mastozoologia@gmail.com>].
2 Instituto de Pesquisa Ambiental da Amazônia (IPAM), Belém, PA, Brazil.
Recibido 2 noviembre 2011.
Aceptado 23 febrero 2012.
Editor asociado: A Noss
ABSTRACT: Many methods of age determination have been used to date. However, most of them require laboratory procedures which are, in general, not available for wildlife managers in the field. In this paper we attempt to standardize the age categories for six mammal species through the analysis of dental wear, dental eruption and body mass. We analyzed 632 skulls, including Mazama nemorivaga (brown brocket deer), Mazama americana (red brocket deer), Tayassu pecari (white-lipped peccary), Pecari tajacu (collared peccary), Cuniculus paca (paca) and Dasyprocta leporina (agouti). Using biological and morphological criteria we define four age categories: young, sub adult, adult and senescent adult. Scores and variables of molar cusp wear and eruption were defined for each age category within each species. We found significant differences in mean weights among age categories for D. leporina (F = 81.1; df = 3; p < 0.05), C. paca (F = 39.0; df = 3; p < 0.05), P. tajacu (F = 24.6; df = 3; p < 0.05) and M. americana (F = 30.6; df = 3; p < 0.05), suggesting that body mass can be a complementary criterion for estimating age. Despite being less quantitative than methods such as those which use deposition of cementum layers, the method described here proved to be an efficient tool for field researchers and managers in the Brazilian Amazon. Although similar methods have already been applied in the study of hunted mammals in the Peruvian and Brazilian Amazon, it is the first time such a method is described in detail and therefore it can be easily used in future works.
RESUMO: Testando critérios simples de estimativa de idade para seis espécies de mamíferos comumente caçados na amazônia brasileira. Muitos métodos para estimativa de idade de mamíferos têm sido utilizados ultimamente. Entretanto, a maioria deles requerem procedimentos laboratoriais, que geralmente, não estão disponíveis em campo. Neste artigo nós definimos categorias de idade para seis espécies de mamíferos através da análise de desgaste dentário, erupção dentária e massa corporal. Nós analisamos 632 crânios, incluindo Mazama nemorivaga (veado cinza), Mazama americana (veado vermelhor), Tayassu pecari (queixada), Pecari tajacu (cateto), Cuniculus paca (paca) and Dasyprocta leporina (cotia). Usando critérios biológicos e morfológicos nós definimos quatro categorias de idade para cada espécie: jovem, sub-adulto, adulto e adulto senescente. Foram definidos padrões de desgaste e erupção dos molares para cada categoria de idade para cada uma das espécies analisadas. Nós encontramos diferenças significativas das médias de peso entre as categorias de idade para as espécies D. leporina (F = 81.1; df = 3; p < 0.05), C. paca (F = 39.0; df = 3; p < 0.05), P. tajacu (F = 24.6; df = 3; p < 0.05) and M. americana (F = 30.6; df = 3; p < 0.05), sugerindo que a massa corporal pode ser usada como um critério complementar de estimativa de idade. Embora seja um método menos quantitativo do que aqueles citados com mais frequência na literatura, como deposição de bandas de cementum, o método descrito neste artigo mostrou ser uma ferramenta eficiente para pesquisadores e manejadores em trabalhos de campo na Amazônia brasileira. Embora métodos similares já tenham sido utilizados em estudos de caça, é a primeira vez que esta metodologia é descrita em detalhes, podendo ser replicada em outros estudos semelhantes.
Key words: Cuniculus; Dasyprocta; Mazama; Pecari;Tayassu.
Palabras chave: Cuniculus; Dasyprocta; Mazama; Pecari; Tayassu.
INTRODUCTION
Hunting is an important cultural and economic strategy for subsistence in Amazonia (Bates, 1876; Smith, 1976, 1977; Ayres and Ayres, 1979; Vickers, 1984, 1991; Bodmer et al., 1994). But even for subsistence, this activity has negative effects on the diversity of medium and large mammals reducing the populations of overexploited species (Redford and Robinson, 1987; Silva and Strahl, 1991; Bodmer et al., 1997; Leeuwenberg and Robinson, 1999; Alvard, 2000; Robinson and Bennett, 2000; Peres, 2000, 2001; Bodmer and Lozano, 2001; Sforzi and Lovari, 2000; Hurtado-Gonzalez and Bodmer, 2004; Robinson and Bennett, 2004; Sirén et al., 2004; Peres, 2007; Di Bitetti et al., 2008; Noss and Cuéllar, 2008; Fa and Brown, 2009).
From 2003 to 2009 a game management initiative was developed in the Extractive Reserve of Tapajós-Arapiuns, Santarém region, Para State, Brazil, where human communities could implement collective strategies for sustainable subsistence hunting. In that context we collected and analyzed information about diversity, biomass, sex ratio and age structure of hunted animals (Oliveira et al., 2004). The knowledge of the number of exploited animals, by sex and age, is essential to assess the population dynamics of species and therefore develop strategies for rational management (Severinghaus, 1949; Morris, 1972; Thomas and Bandy, 1975; Christensen-Dalsgaard et al., 2009).
Many methods of age determination have been used to date (Morris, 1972; Spinage, 1973; King, 1991; Fortelius and Solounias, 2000; Monakhov, 2004; Roulichová and Andera, 2007; Kaiser et al., 2009). In most of them, the age classification is based on biological characteristics that show the transition of one growth stage to another, such as deposition of cementum layers on canines (Grue and Jensen, 1979; Saez-Royuela et al., 1989; Maffei and Becerra, 2000; Nakanishi et al., 2009); sequence of tooth eruption (Ancrenaz and Delhomme, 1997) and wear (Stander, 1997; Veiberg et al., 2007; Ozaki et al., 2010); or cranial suture fusion (Landon et al., 1998).
Although the method of deposition of cementum layers presents better accuracy to age estimation in most of the investigated mammal species (Thomas and Bandy, 1975; King, 1991; Clarke et al., 1992; Choquenot and Saunders, 1993; Maffei, 2001, 2003a; Monakhov, 2004; Roulichová and Andera, 2007), it requires laboratory procedures that, in general, are not available for managers. In addition, few studies have mentioned that the deposition of cementum annuli is irregular in animals inhabiting the tropical and subtropical regions, suggesting that this method may not be reliable for age estimation in these regions (Phillips et al., 1982; Lindeque and Skinner, 1984; van Horn et al., 2003). Here we suggest that, for the Amazon study cases, the combination of tooth eruption and wear with body mass seems to be simpler, faster and more efficient for age determination, especially in the field. Although similar methods have already been applied in Peruvian and Brazilian Amazon (Townsend, 1995; Emidio-Silva, 1998; Gottdenker and Bodmer, 1998; Hurtado-Gonzalez and Bodmer, 2004; Trinca, 2004), it has not been standardized among studied cases and only the results of the ageing analyses have been published in the literature.
In this paper we analyze, define and describe the age categories of six species of medium and large mammals, based on morphological and biological parameters like tooth eruption and wear, and mean weight. The challenge here was to create simple patterns to age definition that can be used by managers in the field. The choice of species was based on Brazilian and Peruvian Amazon studies confirming the preference of hunters for peccaries, deer, agoutis and pacas (Redford and Robinson, 1987; Bodmer et al., 1997; Hurtado-Gonzales and Bodmer, 2004). These species are therefore targets of game management strategies.
MATERIALS AND METHODS
The examined skulls were collected by 51 hunters from four communities located in the Tapajós- Arapiuns Extractive Reserve in Santarém, Pará State, Brazil (55°00'53" W - 02°18'38" S), from September 2006 to August 2008. We analyzed 632 skulls of six different species of mammals, including: Mazama nemorivaga (brown brocket deer) (n = 14), M. americana (red brocket deer) (n = 61), Tayassu pecari (white-lipped peccary) (n = 23), Pecari tajacu (collared peccary) (n = 65), Cuniculus paca (paca) (n = 109) and Dasyprocta leporina (agouti) (n = 360). Due to the dental morphological similarities, the skulls of T. pecari and P. tajacu were analyzed together, as were M. nemorivaga and M. americana skulls.
The age criteria were based on upper molar eruption and wear and body mass. Incisors and canines were not considered in the dental analyses, due to their absence in the superior arch of Mazama spp., the absence of canine teeth in D. leporina and C. paca, and the common loss of these teeth in handling of some skulls. In the same way, as deciduous premolars can persist for long periods of time, unknown for most mammal species, the premolars were also not considered here.
The scores of cusp wear were adapted from "mesowear method" described by Fortelius and Solounias (2000) and Kaiser et al. (2009). The morphological age description also considered the morphology and wear of the zygomatic region in C. paca.
The body weight (kg) of all hunted individuals was compared among ages for each species individually, and the null hypothesis (H0: There is no difference of mean weight between age categories) was tested using factorial ANOVA and subsequent post hoc test with Fisher LSD. In this case, the Kolmogorov-Smirnov test was used to confirm normal distribution and Levene´s test for homogeneity of variances. All statistical analyses were performed using STATISTICA 7.0 and the significance level was set to 0.05. Considering the reduced sample of M. nemorivaga and T. pecari, these were not used in body mass analyses.
Time estimation per each age category and longevity were defined using data about the reproduction biology available in the bibliography. A mean time was calculated when more than three values were available.
RESULTS
Using biological criteria we describe four age categories: young (1), sub adult (2), adult (3) and senescent adult (4) that were biologically and morphologically distinct. The first category includes nursing animals, with the end of the lactation period considered the limit of this category. The second category includes young animals, non-nursing and non-reproductive mature individuals. The beginning of puberty was considered the limit of this second category. The third includes adult, reproductively mature animals. In this age class, reproduction events are most likely to be successful. The fourth category includes adult animals that are probably experiencing declining reproductive performance (senescence).
Table 1 presents the age classification for all examined skulls, using all criteria defined here. The upper molar analysis treats two variables: tooth eruption and tooth wear. We classified tooth eruption as "totally erupted" (te) when the tooth is totally formed, with the maximum crown exposed; "in eruption" (ie) when the tooth is still incomplete, with only part of the crown exposed; and "non erupted" (ne) when the tooth is inside the bone or breaking the alveolar bone (Table 1).
Table 1. Criteria for age classification of six taxa analyzed.
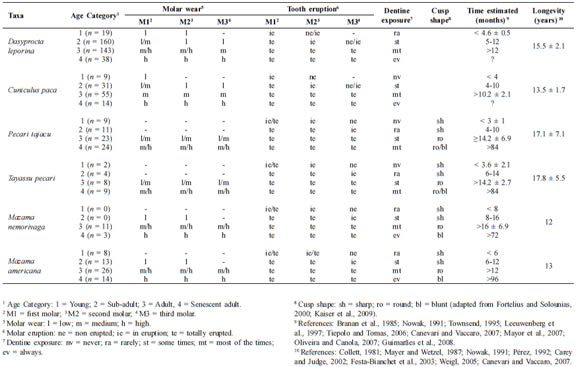
The second variable, tooth wear, was classified based on cusp wear and dentine exposure, varying among species examined (Table 1). The tooth wear was classified as high (h) (Fig. 1: C1/C2; Fig. 2: C1/C2; Fig. 3: A3/B3) when the cusps are totally worn and dentine quite exposed and dark; as medium (m) (Fig. 1: B1/ B2; Fig. 2: B1/B2; Fig. 3: A2/B2) when the cusps are worn but still visible and the dentine is exposed in some or most of the cusps; and as low (l) (Fig. 1: A1/A2; Fig. 2: A1/A2; Fig. 3: A1/B1) when the cusps are fully visible and there is no dentine exposure, with the exception of rodent species as D. leporina and C. paca, in which the dentine is already exposed from the first stage (Fig. 3). Within this variable, the cusp shapes of peccaries and deer were classified as "sharp" (Fig. 1: A1/A2; Fig. 2: A1/A2); "round" (Fig. 1: B1/B2; Fig. 2: B1/ B2) and "blunt" (Fig. 1: C1/C2; Fig. 2: C1/ C2). Here, the unworn teeth were excluded, appearing in Table 1 as a hyphen (-).

Fig. 1. Upper molar cusp shapes and dentine exposure of Mazama americana. A1 - "Sharp" shape; A2 - No dentine exposed; B1 - "Round" shape; B2 - Dentine exposed (yellow arrow), but cusps still apparent and sharp; C1 - "blunt" shape; C2 - Dentine exposed (yellow arrow) and cusps less apparent.

Fig. 2. Upper molar cusp shapes and dentine exposure of Tayassu pecari. A1 - "Sharp" shape; A2 - No dentine exposed; B1 - "Round" shape; B2 - Dentine exposed (yellow arrow) in some points; C1 - "blunt" shape; C2 - Dentine exposed (yellow arrow) and cusps less apparent.

Fig. 3. Hypselodont dentition of Dasyprocta leporine (A1, A2, A3) and Cuniculus paca (B1, B2, B3). A1/B1 - third upper molar breaking the alveolar bone, first and second upper molar with low wear; A2/ B2 - upper molar with medium wear; A3/ B3 - upper molar with high wear
In M. nemorivaga and M. americana, the high (h) level of wear presents lots of dentine exposed and cusps in "blunt" shapes (Fig. 1: C1/C2). Even with medium (m) tooth wear, the dentine can be exposed, but the cusp is still apparent, with "round" forms (Fig. 1: B1/B2). The same can happen with T. pecari and P. tajacu, but in both cases the dentine is rarely exposed in the third age category (Fig. 2: B2; Table 1) and less exposed in the fourth age category as compared to Mazama spp. for the same age categories (Fig. 2: C2; Table 1). In all four species, the low (l) level presents intact or little-worn cusps in "sharp" shapes and the dentine is never exposed (Fig. 1: A1/ A2; Fig. 2: A1/A2; Table 1).
For C. paca, the morphology of the zygomatic bone was classified into four stages, each one corresponding to one age category: the first stage including young animals with thin and smooth zygomatic bones (Fig. 4A); the second stage corresponding to sub-adult animals with smooth but not thin zygomatic bones (Fig. 4B); the third stage including adult animals with very large, but not rugged zygomatic bones (Fig. 4C); and the fourth stage corresponding to senescent adult with rugged and large zygomatic bones
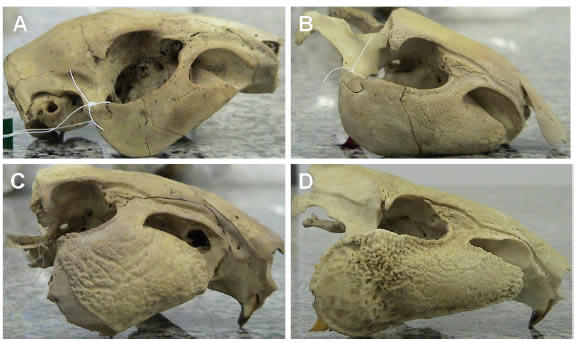
Fig. 4. The zygomatic arch of Cuniculus paca. A - young; B - sub-adult; C - adult and D - senescent adult.
In this case study, hunters preferred the adult and sub-adult age classes of Dasyprocta leporine, Cuniculus paca and Mazama americana. In contrast, for T. pecari, P. tajacu and M. nemorivaga hunters preferred the adult and senescent adult age classes (Table 1).
The factorial ANOVA analyses of body mass did not show differences between mean weight among sex for D. leporina (F = 0.87; df = 1; p < 0.05), C. paca (F = 2.34; df = 1; p < 0.05), P. tajacu (F= 3.19; df= 1; p < 0.05) and M. americana (F= 0.25; df= 1; p < 0.05). On the other hand, differences in mean weights among ages categories were statistically significant for D. leporina (F = 81.1; df = 3; p < 0.05), C. paca (F= 39.0; df= 3; p < 0.05), P. tajacu (F = 24.6; df= 3; < 0.05) and M. americana (F = 30.6; df = 3; p < 0.05) (Fig. 5).

Fig. 5. Mean weight of males and females per age category. F1: Female of age category 1 (young); M1: Male of age category 1 (young); F2: Female of age category 2 (sub adult); M2: Male of age category 2 (sub adult); F3: Female of age category 3 (adult); M3: Male of age category 3 (adult); F4: Female of age category 4 (senescent adult); M4: Male of age category 4 (senescent adult).
For D. leporina, with the exception of the third and fourth age categories (male and female), all other comparisons between mean weights among age categories were statistically different (including male and female analysis) (df = 352; p < 0.05) (Fig. 4). The same occurs with C. paca (df = 101; p < 0.05) (Fig. 5). For P. tajacu there was no difference among F1-F2, M1-M2 and M3-M4 age categories (df = 56; p < 0.05), while among F2-F3, M2-M3 and F3-F4 the mean weight was statistically different (df = 56; p < 0.05) (Fig. 4). For M. americana, with the exception of the F2-F3 comparison where there was no difference (df = 56; p < 0.05), all other comparisons were statistically different (df = 56; p < 0.05) (Fig. 5).
DISCUSSION
The major challenge of this paper was to cross the age categories, defined through biological characteristics, with morphological standards of molar wear, tooth eruption and body weight. Unfortunately, some of these biological characteristics are still unknown for most mammal species. For example, the senescence or the decline in physiological condition and cellular functioning with age, which leads to reduced survival and reproductive performance, is quite unknown in the literature (Sharp and Clutton- Brock, 2009). The lack of biological information, especially about Amazonian species, makes this classification more difficult with uncertain or fragile biological limits among age categories. Therefore, the criteria described here can be useful as a simple method of age classification by wildlife managers.
The pattern and level of cusp wear differ among groups of species, depending on the food habits and dental morphology (Herring, 1985; Popowics and Fourtelius, 1997; Mendoza et al., 2002; Kaiser et al., 2009; Ozaki et al.,2010). In most mammal species the dentine is exposed with wear, providing a proliferation of secondary dentine, of darker color than the initial dentine. In M. nemorivaga and M. americana, as in others cervids, this process seems to be more intense due to the abrasive diet (Fortelius and Solounias, 2000; Kaiser et al., 2009) and can be easily observed both in the third (3) and fourth (4) age categories (Fig. 1: B1/B2/C1/C2).
The hypselodont (open-rooted) dentition of C. paca and D. leporina promotes a different pattern of wear. The higher crown and natural lower cusps of molars make age class definition difficult (Fig. 3). Although we opted to use the tooth wear classification for these species, tooth eruption is very useful. In any case, it is easier to detect the wear observing the lack of enamel wrinkles, the teeth forming rounded structures in adult and senescent adult (Fig. 3: A3/B3). In young and sub-adult animals enamel wrinkles are easily visible (Fig. 3: A1).
The molar eruption criteria was more useful for ageing animals in the first and second age categories (Clarke et al., 1992), especially C. paca and D. leporina, whereas the molar wear criterion was more suited to ageing animals in the third and fourth age categories.
The genera Cuniculus and Dasyprocta are principally frugivores that consume not only fruit (pulp and seeds), but also leaves, fibers, flowers, and even invertebrates in smaller quantities (Henry, 1999). Dasyprocta are able to open hard endocarps with more elaborate movements than those used by pacas, sitting on their hind legs and holding the food, peeling or breaking fruit (Henry 1997). Cuniculus eat more like pigs, gnawing while supported on all four legs. They are unable to open some hard-shelled fruits, but eat the soft pericarp when the fruit has been opened by another animal (Collet, 1981). The greatest dental impact of feeding in rodents is seen in the incisors. However, behavioral differences may influence the pattern of molar and pre-molar tooth wear; most of the time, these teeth suffer more attrition on the vestibular face. In agoutis this seems to be more apparent than in Cuniculus, probably because of differences in feeding behavior.
The morphology of the zygomatic arch in the C. paca has been suggested as a good indicator of age classification, because the arch undergoes a radical change from young animals to adults. In young animals the arch is fine and smooth, whereas in adults it is rugged and rough (Nelson and Shump, 1978). Hershkovitz (1955) suggested that growth of the zygomatic arch in pacas is related to amplification of vocalization in adults. Tooth eruption also seems to be useful in age estimation for this species. Collett (1981) considered that when the third molar of a paca is fully emerged ("adult class"), the animal is about 1 year of age. Female agoutis become reproductively active at approximately 9 months of age, whereas males reach sexual maturity at 1 year (Collett, 1981).
Like ruminants, deer have pre-molars and molars with flat surfaces for grinding food. In the Amazon, however, deer in the genus Mazama are considered frugivores (Bodmer, 1989). According to Bodmer (1997), these animals tend to be frugivores in tropical forests because of an abundance of fruit resources. Thus, it is possible that the diet of animals examined in this work was less abrasive than that of deer occurring in other biomes, where they feed mainly on leaves and fibers containing large amounts of cellulose. In the Amazon forest dry season, if fruit reduction occurs, these deer can increase consumption of sprouts, leaves, and grasses. This diet pattern contributes to greater wear on the vestibular face of pre-molars and molars.
Although age classification of deer is not very accurate, Townsend (1995) and Leeuwenberg et al. (1997) considered that a life table for these animals could be constructed at age class intervals of 2 years. This means that an animal reaches adult status in 3 or 4 years. However, Branan et al. (1985) recorded a pregnant female of M. americana, in captivity, that was estimated to be 13 months old. For studies of population dynamics for game management, it is best to be conservative, as hunters generally prefer larger animals.
Herring (1985) proposed that the dental features of peccaries might be associated with feeding on moist vegetation and rooting in soft terrain. Peccaries are considered to be omnivorous, consuming fruits (including palm nuts), leaves, roots, seeds, mushrooms, adult and larval insects, frogs, snakes, lizards, and eggs of birds and turtles (Mayer and Wetzel, 1987). The manner in which these animals hold and chew food, similar to the behavior of the paca, probably causes heavy wear of pre-molar teeth, uniform on the lingual and vestibular faces.
The results suggest that for both C. paca and D. leporine body mass can be a complementary criterion to age estimation, at least at early ages. The lack of statistical differences between mean weight among the third and fourth age categories probably reflects the fast growth of these species in the first year of life (Collett, 1981; Oliveira and Canola, 2007).
Body mass is useful as a complementary method of age definition and can be used together with molar wear and tooth eruption methods. It is important to remember that individual body mass can vary according to season or nutrition. To minimize the effects of this natural seasonal variation, body mass measures should be undertaken with individuals captured during the same season. Morris (1972) and Bray et al. (2002) suggested that variables using rigid linear dimensions like foot or ear length can vary less than other volumetric variables such as body mass. But this relationship between linear measurement and age is a new issue to be investigated in future work.
Based on hunters' preferences for certain age groups, some inferences can be made regarding the survival of hunted animal populations in the medium and long term. The highest proportion of captures in the adult and sub-adult classes-as in the species D. leporina, C. paca and M. americana-can cause a decline in the number of individuals in reproductive phases. In the medium and long term this can lead to significant local population declines of these species. The preference for large animals-at least for T. pecari, P. tajacu and M. nemorivaga-suggest that hunting is not carried out at random (Caughley, 1977; Bodmer and Robinson, 2006). As mentioned by Maffei (2003b), hunters tend to choose the largest animals when these are encountered in groups as are T. pecari and P. tajacu.
The rational management of game resources is impossible without the assessment of age structure of hunted animals (Hurtado-Gonzales and Bodmer, 2004). For species with long generation times, like medium and large mammals, changes in the proportion of reproducing young and juvenile survival can provide a dramatic impact on population dynamics (Bodmer et al., 1997; Bodmer, 2004). Despite being less accurate than methods such as those which use deposition of cementum layers, the method described here can be an efficient tool for field researchers and wildlife managers in the Brazilian Amazon. Noss et al. (2003), in a study with hunters in the Bolivian Chaco, concluded that most experimental analyses of tooth wear were consistent with the hunters' subjective age classification.
A crucial problem with game management in the Amazon region has been the time to assess biological and ecological information. Considering the high growth rate of the human population and the spread of deforestation (Laurance et al., 2002; Soares-Filho et al, 2006), rational management has to be efficient in a short period of time. A positive issue is that for most of the traditional Amazon communities, high game stocks can be an important reason to maintain the forest (Oliveira et al., 2004).
ACKNOWLEDGMENTS
We first thank to CNPq/Edital Universal and Boticário Fundation for the financial support. We also thank the hunters and the communities (Pinhel, Aminã, Pascoal and Solimões) who participated in the field study; as well as the TAPAJOARA Association and IBAMA/CNPT for support within the communities.
LITERATURE CITED
1. ALVARD M. 2000. The impact of traditional subsistence hunting and trapping on prey populations: data from Wana Horticulturalist of Upland Central Sulawesi, Indonesia. Pp. 214-230, in: Hunting in Neotropical forests: review of the issues, identifying gaps, and developing strategies (E Bennett, F Arengo, eds.). Wildlife Conservation Society, New York, USA. [ Links ]
2. ANCRENAZ M and A DELHOMME. 1997. Teeth eruption as a means of age determination in captive Arabianoryx, Oryx leucoryx (Bovidae, Hippotraginae). Mammalia 61:131-138. [ Links ]
3. AYRES JM and C AYRES. 1979. Aspectos da caça no alto rio Aripuanã. Acta Amazonica 9:287-298. [ Links ]
4. BATES HW. 1876. The naturalist on the river Amazons. São Paulo University Press, Brazil. [ Links ]
5. BODMER RE. 1989. Frugivory in Amazonian Arctiodactyla: evidence for the evolution of the ruminant stomach. Journal of Zoology 219:457-467. [ Links ]
6. BODMER RE, TG FANG, L MOYA, and R GILL. 1994. Managing wildlife to conserve Amazonian Forest: population biology and economic considerations of game hunting. Biological Conservation 67:29-35. [ Links ]
7. BODMER RE. 1997. Ecologia e conservação dos veados mateiro e catingueiro na amazônia. Pp 70-77, in: Biologia e Conservação de Cervídeos Sul-Americanos: Blastocerus, Ozotocerus e Mazama (JM Barbante, ed.). FUNEP Press, Jaboticabal, São Paulo. [ Links ]
8. BODMER RE, JF EISENBERG, and KH REDFORD. 1997. Hunting and the likelihood of extinction of Amazonian mammals. Conservation Biology 11:460-466. [ Links ]
9. BODMER RE and EP LOZANO. 2001. Rural development and sustainable wildlife use in Peru. Conservation Biology 15:1163-1170. [ Links ]
10. BODMER RE. 2004. Evaluating the sustainability of harvesting in the Neotropics using the unified harvest model. Pp. 201-206, in: Hunting in Neotropical Forests: Review of the issues, identifying gaps, and developing strategies (E Bennett and F Arengo, eds.), Wildlife Conservation Society, New York, USA. [ Links ]
11. BODMER RE and JG ROBINSON. 2006. Análise da sustentabilidade de caça em florestas tropicais no Peru - estudo de caso. Pp. 579-615, in: Métodos de Estudo em Biologia da Conservação & Manejo da Vida Silvestre (L Cullen, R Rudran, and C Valadares-Padua, eds.), Universidade Federal do Paraná, Brazil. [ Links ]
12. BRANAN WV, MCM WERKHOVEN, and RL MARCHINTON. 1985. Food habitats of brocket and white-tail deer in Suriname. Journal of Wildlife Management 49:972-976. [ Links ]
13. BRAY Y, S CHAMPELY, and D SOYEZ. 2002. Age determination in leverets of European hare Lepus europaeus based on body measurements. Wildlife Biology 8:31-39. [ Links ]
14. CANEVARI M and O VACCARO. 2007. Guía de Mamíferos del Sur de América del Sur. Editorial L.O.L.A., Buenos Aires, Argentina. [ Links ]
15. CAREY JR and DS JUDGE. 2002. Longevity records: life spans of mammals, birds, amphibians, reptiles, and fish. Monographs on Population Aging 8, Odense University Press, Odense, Denmark. [ Links ]
16. CHOQUENOT D and G SAUNDERS. 1993. A comparison of three ageing techniques for feral pigs from subalpine and semi-arid habitats. Wildlife Research 20:163-171. [ Links ]
17. CHRISTENSEN-DALSGAARD SN, J AARS, M ANDERSEN, C LOCKYER and NG YOCCOZ. 2009. Accuracy and precision in estimation of age of Norwegian Arctic polar bears (Ursus maritimus): using dental cementum layers from known-age individuals. Polar Biology 10:300-309. [ Links ]
18. CLARKE CMH, RM DZIECIOLOWSKI, D BATCHELER, and CM FRAMPTON. 1992. A comparison of tooth eruption and wear and dental cementum techniques in age determination of New Zealand feral pigs. Wildlife Research 19:769-777. [ Links ]
19. COLLETT S. 1981. Population characteristics of Agouti paca (Rodentia) in Colombia. Publication of the Museum of the Michigan State University. Biological Series 5:485-602. [ Links ]
20. DI BITETTI MS, A PAVIOLO, CA FERRARI, C DE ANGELO, and Y DI BLANCO. 2008. Differential responses to hunting in two sympatric species of brocket deer (Mazama americana and M. nana). Biotropica 40:636-645. [ Links ]
21. EMIDIO-SILVA C. 1998. A caça de subsistência praticada pelos Índios Parakanã - Sudeste do Pará: características e sutentabilidade. Biology Msc. Dissertation, University of Pará UFPA/MPEG. Belém, PA, Brazil. [ Links ]
22. FA JE and D BROWN. 2009. Impacts of hunting on mammals in African tropical moist forests: a review and synthesis. Mammal Review 39:231-264. [ Links ]
23. FESTA-BIANCHET M, JM GAILLARD, and SD CôTÉ. 2003. Variable age structure and apparent density dependence in survival of adult ungulates. Journal of Animal Ecology 72:640-649. [ Links ]
24. FOURTELIUS M and N SOLOUNIAS. 2000. Functional characterization of ungulate molars using the abrasion-attrition wear gradient: a new method for reconstructing paleodiets. American Museum Novitates 3301. [ Links ]
25. GOTTDENKER N and RC BODMER. 1998. Reproduction and productivity of white-lipped and collared peccaries in the Peruvian Amazon. Journal of Zoology 245:423-430. [ Links ]
26. GRUE H and B JENSEN. 1979. Review of the formation of incremental lines in tooth cementum of terrestrial mammals. Danish Review of Game Biology 11(3):1-48. [ Links ]
27. GUIMARãES DAA, LV BASTOS, ACS FERREIRA, RS LUZ-RAMOS, OM OHASHI, and HL RIBEIRO. 2008. Características reprodutivas da paca fêmea (Agouti paca) criada em cativeiro. Acta Amazonica 38:531-538. [ Links ]
28. HERSHKOVITZ P. 1955. On the cheek pouches of the tropical American paca Agouti paca (Linnaeus, 1766). Saugetierkundliche Mitteilugen 3:67-70. [ Links ]
29. HERRING SW. 1985. Morphological correlates of masticatory patterns in peccaries and pigs. Journal of Mammalogy 66:603-617. [ Links ]
30. HURTADO-GONZALEZ JL and RE BODMER. 2004. Assessing the sustainability of brocket deer hunting in the Tamshiyacu-Tahuayo Communal Reserve, northeastern Peru. Biological Conservation 116:1-7. [ Links ]
31. KAISER TM, J BRASCH, JC CASTELL, E SCHULZ, and M CLAUSS. 2009. Tooth wear in captive wild ruminant species differs from that of free-ranging conspecifics. Mammalian Biology 74:425-437. [ Links ]
32. KING CM. 1991. A review of age determination methods for the stoat Mustela ermine. Mammal Review 1:31-49. [ Links ]
33. LANDON DB, CA WAITE, RO PETERSON, and LD MECH. 1998. Evaluation of age determination techniques for gray wolves. Journal of Wildlife Management 62:674-682. [ Links ]
34. LAURANCE WF, AKM ALBERNAZ, G SCHROTH, PM FEARNSIDE, S BERGEN, EM VENTICINQUE, and C DA COSTA, C. 2002. Predictors of deforestation in the Brazilian Amazon. Journal of Biogeography 29:737-748. [ Links ]
35. LEEUWENBERG F, S LARA-RESENDE, FHG RODRIGUES, and XA BEZERRIL. 1997. Home range, activity and habitat use of the Pampas deer (Ozotoceros bezoarticus L. 1758, Artiodactyla, Cervidae) in the Brazilian Cerrado. Mammalia 61:487-495. [ Links ]
36. LEEUWENBERG FJ and JG ROBINSON. 1999. Traditional management of hunting by a Xavante community in central Brazil: the search for sustainability. Pp. 375-394, in: Hunting for sustainability in tropical forests (JG Robinson and EL Bennett, eds). Columbia University Press, New York, USA. [ Links ]
37. LINDEQUE M and JD SKINNER. 1984. Size frequency analysis of tooth wear in spotted hyaenas Crocuta crocuta. African Journal of Zoology 19:291-294. [ Links ]
38. MAFFEI LF. and MN BECERRA. 2000. Técnica básica para determinar la edad en ungulados silvestres en base al análisis de dientes. Ecología en Bolívia 34:39-44. [ Links ]
39. MAFFEI LF. 2001. Estructura de edades de la urina (Mazama gouazoubira) en el Chaco boliviano. Mastozoología Neotropical 8:149-155. [ Links ]
40. MAFFEI LF. 2003a. The age structure of tapirs (Tapirus terrestris) in the Chaco. Tapir Conservation. Newsletter of the IUCN/SSC, Tapir Specialist Group 12:18-19. [ Links ]
41. MAFFEI LF. 2003b. Age structure of two hunted peccary species in the Bolivian Chaco Tayassu tajacu and T. pecari. Mammalia 4:575-578. [ Links ]
42. MAYER JJ and RM WETZEL. 1987. Tayassu pecari. Mammalian Species 293:1-7. [ Links ]
43. MAYOR P, DAA GUIMARãES, Y PENDU, JV SILVA, F JORI, and M LOPEZ-BEJAR. 2007. Reproductive performance of captive collared peccaries (Tayassu tajacu) in the eastern Amazon. Animal Reproduction Science 102:88-97. [ Links ]
44. MENDOZA M, CM JANIS, and P PALMQVIST. 2002. Characterizing complex craniodental patterns related to feeding behaviour in ungulates: a multivariate approach. Journal of Zoology 258:223-246. [ Links ]
45. MONAKHOV VG. 2004. Assessment of the age structure of sable samples using three methods of age determination. Russian Journal of Ecology 35: 383-388. [ Links ]
46. MORRIS P. 1972. A review of mammalian age determination methods. Mammal Review 2:69-100. [ Links ]
47. NAKANISHI N, F ICHINOSE, G HIGA, and M IZAWA. 2009. Age determination of the Iriomote cat by using cementum annuli. Journal of Zoology 279:338-348. [ Links ]
48. NELSON TW and K SHUMP JR. 1978. Cranial variation and size allometry in Agouti paca from Ecuador. Journal of Mammalogy 59:387-394. [ Links ]
49. NOSS AJ, ES CUÉLLAR, and RL CUÉLLAR. 2003. Hunter self-monitoring as a basis for biologica research: data from the bolivian Chaco. Mastozoología Neotropical 10:49-67. [ Links ]
50. NOSS AJ and RL CUÉLLAR. 2008. La sostenibilidad de la cacería de Tapirus terrestris y de Tayassu pecari en la tierra comunitaria de origen isoso: el modelo de cosecha unificado. Mastozoología Neotropical 15:241-252. [ Links ]
51. NOWAK RM. 1991. Walker´s Mammals of the World. 5th Ed., V. II, The Johns Hopkins University Press, London. [ Links ]
52. OLIVEIRA ACM, O CARVALHO JR, and R CHAVES. 2004. Gestão participativa e a atividade de caça na Reserva Extrativista do Tapajós Arapiuns, Santarém, PA. Raízes: Revista de Ciências Sociais e Economia 23:42-51. [ Links ]
53. OLIVEIRA FS and JC CANOLA. 2007. Erupção dental em pacas (Agouti paca) criadas em cativeiro. Arquivo Brasileiro de Medicina Veterinária e Zootecnia 2:390-394. [ Links ]
54. OZAKI M, G SUWA, K KAJI, T OHBA, E HOSOI, T KOIZUMI, and S TAKATSUKI. 2010. Correlations between feeding type and mandibular morphology in the sika deer. Journal of Zoology 272:244-257. [ Links ]
55. PERES CA. 2000. Effects of subsistence hunting on vertebrate community structure in Amazonian forests. Biological Conservation 14:240-253. [ Links ]
56. PERES CA. 2001. Synergistic effects of subsistence hunting and habitat fragmentation on Amazonian forest vertebrates. Conservation Biology 15:1490-1505. [ Links ]
57. PERES CA. 2007. Basin-wide effects of game harvest on vertebrate population densities in Amazonian forests: implications for animal-mediated seed dispersal. Biotropica 39:304-315. [ Links ]
58. PÉREZ EM. 1992. Agouti paca. Mammalian Species 404:1-7. [ Links ]
59. PHILLIPS CJ, B STEINBERG, and TH KUNZ. 1982. Detin, cementum, and age determination in bats: critical evaluation. Journal of Mammalogy 63:197-207. [ Links ]
60. POPOWICS TE and M FOURTELIUS. 1997. On the cutting edge: tooth blade sharpness in herbivorous and faunivorous mammals. Annales Zoologici Fennici 34:73-84. [ Links ]
61. REDFORD KH and JG ROBINSON. 1987. The game of choice: patterns of Indian and colonist hunting in the Neotropics. American Anthrpologist 89:650-657. [ Links ]
62. ROBINSON JG and E BENNETT. 2000. Carrying capacity limits to sustainable hunting in tropical forests. Pp. 13-30, in: Hunting for sustainability in tropical forest (JG Robinson and EL Bennett, eds.), Columbia University Press, New York, USA. [ Links ]
63. ROBINSON JG and E BENNETT. 2004. Having your wildlife and eating it too: an analysis of hunting sustainability across tropical ecosystems. Animal Conservation 7:397-408. [ Links ]
64. ROULICHOVÁ J and M ANDERA. 2007. Simple method of age determination in red fox, Vulpes vulpes. Folia Zoologica 56:440-444. [ Links ]
65. SÁEZ-ROYUELA C, RP GOMARIZ, and JL TELLERÍA, 1989. Age determination of European wild boar. Wildlife Society Bulletin 17:326-329. [ Links ]
66. SEVERINGHAUS CW. 1949. Tooth development and wear as criteria of age in white-tailed deer. Journal of Wildlife Management 13:195-216. [ Links ]
67. SHARP SP and TH CLUTTON-BROCK. 2009. Reproductive senescence in a cooperatively breeding mammal. Journal of Animal Ecology 79:176-183. [ Links ]
68. SIRÉN A, P HAMBACK and J MACHOA. 2004. Including spatial heterogeneity and animal dispersal when evaluating hunting: a model analysis and an empirical assessment in an Amazonian community. Conservation Biology 18:1315-1329. [ Links ]
69. SFORZI A and S LOVARI. 2000. Some effects of hunting on wild mammalian populations. Ibex Journal of Mountain Ecology 5:165-171. [ Links ]
70. SMITH NJH. 1976. Utilization of game along Brazil's transamazon highway. Acta Amazônica 6(4):455-466. [ Links ]
71. SMITH NJH. 1977. Human exploitation of terra firme fauna in Amazonian. Ciência e Cultura 30:17-23. [ Links ]
72. SOARES-FILHO BS, DC NEPSTAD, LM CURRAN, GC CERQUEIRA, RA GARCIA, C AZEVEDO-RAMOS, E VOLL, A MCDONALD, P LEFEBVRE, and P SCHLESINGER. 2006. Modeling conservation in the Amazon basin. Nature 440:520-523. [ Links ]
73. SPINAGE S J. 1973. A review of the age determination of mammals by means of teeth, with special reference to Africa. East African Wildlife Journal 11:165-187. [ Links ]
74. STANDER PE. 1997. Field age determination of leopards by tooth wear. African Journal of Ecology 35:156-161. [ Links ]
75. TIEPOLO LM and WM TOMAS. 2006. Ordem Artiodactyla. Pp. 283-303, in: Mamíferos do Brasil (NR Reis, AL Peracchi, WA Pedro, IP Lima, eds.). Editora da Universidade Estadual de Londrina, Londrina, Brasil. [ Links ]
76. THOMAS DC and PJ BANDY. 1975. Accuracy of dental-wear age estimates of black-tailed deer. Journal of Wildlife Management 39:674-678. [ Links ]
77. TOWNSEND WR. 1995. Living on the edge: Sirinó hunting and fishing in lowland Bolivia. Ph.D. Dissertation, University of Florida, USA. [ Links ]
78. TRINCA CT. 2004. Caça em assentamento rural no sul da floresta Amazônica. Msc. Dissertation of Zoology, University of Pará, UFPA/MPEG. Belém, PA, Brazil. [ Links ]
79. VAN HORN RC, TL MCELHINNY, and KE HOLEKAMP. 2003. Age estimation and dispersal in the spotted hyena (Crocuta crocuta). Journal of Mammalogy 84:1019-1030. [ Links ]
80. VEIBERG V, LE LOE, A MYSTERUD, EJ SOLBERG, R LANGVATN, and NC STENSETH. 2007. The ecology and evolution of tooth wear in red deer and moose. Oikos 116:1805-1818. [ Links ]
81. VICKERS WT. 1984. The faunal components of lowland South American hunting kills. Interciência 9:366-376. [ Links ]
82. VICKERS WT. 1991. Hunting yields and game composition over ten years in Amazon indian territory. Pp. 53-81, in: Neotropical Wildlife Use and Conservation (JG Robinson and KH Redford, eds.). The University of Chicago Press, Chicago, USA. [ Links ]
83. WEIGL R. 2005. Longevity of Mammals in Captivity: From the living collections of the world. Stuttgart, E. Schweizerbart'sche Verlagsbuchhandlung. [ Links ]














