Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Mastozoología neotropical
versión impresa ISSN 0327-9383versión On-line ISSN 1666-0536
Mastozool. neotrop. vol.25 no.1 Mendoza jun. 2018
ARTÍCULO
Effects of sex, seed size and amount of dietary fiber on retention time of captive black howler monkeys
Raymundo T. M. dos Santos Neto, Roberto Guevara, and Maria A. Lopes
Instituto de Ciências Biológicas, Universidade Federal do Pará, Belém, Pará, Brazil [Correspondence: Raymundo Tomaz Melo dos Santos Neto <raymundotomaz@hotmail.com>]
Recibido 3 junio 2017.
Aceptado 22 noviembre 2017.
Editor asociado: F Bozinovic
ABSTRACT.
The effectiveness of a seed disperser depends, among various factors still poorly understood for many Neotropical primates, on the amount of time seeds are retained within its digestive tract. This study tested the effects of sex, seed size and dietary fiber content on seed retention time in the digestive tract of black howler monkeys (Alouatta caraya). Experiments were performed using six captive individuals (three of each sex), six seed species distributed in three size classes (small, medium, and large), and three percentages of leaves in the diet (15, 30 and 60% of total food ingested). Retention times were recorded for 1767 recovered seeds, corresponding to 55% of the total ingested. More than 98% of these seeds were undamaged, suggesting the potential role of howlers as seed dispersers. Sex, seed size and dietary fiber content alone showed no effect on retention time, but the interaction between sex and seed size showed an effect on the mean retention time, which tended to decrease for males and increase for females as the seed size increased. Thus, males and females may have complementary influences on recruitment and dispersion patterns for different plant species, contributing to community diversity.
RESUMO.
Efeito do sexo, do tamanho de sementes e da quantidade de fibra na dieta sobre o tempo de retenção em guariba-preto.
A efetividade de um dispersor de sementes depende, dentre outros fatores ainda pouco compreendidos em relação a muitos primatas Neotropicais, do tempo em que as sementes são mantidas no trato digestivo do animal. Este estudo testou o efeito do sexo, tamanho da semente e teor de fibra alimentar sobre o tempo de retenção de sementes no trato digestivo de bugios pretos (Alouatta caraya). As sessões experimentais foram realizadas com seis indivíduos cativos (três de cada sexo), seis espécies de sementes distribuídas em três classes de tamanho (pequenas, médias e grandes) e três porções de folhas na dieta (15, 30 e 60% do alimento total ingerido). Os tempos de retenção foram registrados em 1767 sementes recuperadas, correspondendo a 55% do total ingerido. Mais de 98% dessas sementes foram defecadas intactas, sugerindo o potencial dos bugios como dispersores de sementes. O sexo, o tamanho da semente e o teor de fibras alimentares não apresentaram efeito sobre o tempo de retenção, mas a interação entre sexo e tamanho da semente mostrou um efeito sobre a média do tempo de retenção, com tendência a diminuir para machos e aumentar para fêmeas à medida que o tamanho da semente aumenta. Deste modo, machos e fêmeas podem ter influências complementares nos padrões de recrutamento e dispersão para diferentes espécies de plantas, contribuindo para a diversidade da comunidade.
Key words: Alouatta caraya; Gastrointestinal passage time; Diet composition; Seed dispersal; Seed disperser effectiveness.
Palavras-chave: Alouatta caraya; Composição da dieta; Dispersão de sementes; Efetividade de dispersor de sementes; Tempo de passage gastrointestinal.
INTRODUCTION
Seed dispersal is a key element for understanding spatial and demographic distribution patterns of plant species, from the colonization of new areas and population recruitment to individual reproductive success. Endozoochory is the main method of seed dispersal by primates (Chapman & Russo 2007), in which seeds pass unharmed through the digestive tract (Tsuji et al. 2010).
Endozoochory depends on morphological, physiological and behavioral aspects of the animals: diet quality (amount of dry material ingestion), health, age, sex, stress level, and body mass, and also to seed characteristics. Extrinsic factors such as size, weight and rigidity of seeds may also be of influence (Clauss et al. 2008; Tsuji et al. 2010). All Neotropical primates are at least partially frugivorous, and therefore potential seed dispersers (Oliveira & Ferrari 2000; Chapman & Russo 2007). Consequently, several studies have investigated frugivory and seed dispersal processes from different perspectives.
The effectiveness of a seed disperser encompasses quantitative factors, such as number of visits and number of seeds dispersed per visit, and qualitative factors related to seed viability after passage through the digestive tract, rate of germination, and deposition quality, which involves animal movement and fecal deposition patterns (Schupp 1993). In turn, deposition quality depends on the amount of time that seeds stay within the animal's gut and, combined with its movement patterns, will influence the spatial-temporal dispersion of the voided seeds.
Studies with different species have obtained contrasting results regarding the effects of seed size, animal body size and type of diet on retention time. In a study with brown-eared bulbuls (Hypsipetes amaurotis), Fukui (2003) found that smaller and lighter seeds were retained the longest within individuals' digestive tracts. Conversely, Wotton et al. (2008), in a study with New Zealand pigeons (Hemiphaga novaezeelandiae novaeseelandiae), both in captivity and in the wild, observed that heavier seeds were retained for longer periods.
There are also differences in primates regarding the influence of seed characteristics on retention time. In a study with captive Japanese macaques (Macaca fuscata), Tsuji et al. (2010) verified that heavier seeds remained for longer periods within the animals' guts, whereas in a study with captive brown wooly monkeys (Lagothrix lagothricha), Stevenson & García (2003) found out that smaller seeds had longer retention times. These apparently contradictory results suggest that the influence of seed size on retention time may depend on the animals' morphological and physiological characteristics (Tsuji et al. 2010).
Other factors such as the animal's body size and type of diet may also influence seed retention time. Demment (1983) recognized that the average retention time in herbivores varies as a function of body mass ("body mass hypothesis"), while Milton (1984) found notable exceptions to this relation in non-human primates. Other authors also did not observe this correlation in various primates, from species weighing just a few grams to species with over 100 kg of body mass (Caton & Hume 1996; Clauss et al. 2008). Opposing the body mass hypothesis, some authors have argued that the mechanisms affecting food retention must reflect, fundamentally, an organism's diet (Sibly 1981). This hypothesis has been confirmed by Clauss et al. (2008) and Stevenson & García (2003).
In South America, Alouatta is the most widely distributed primate genus (Gregorin 2006), occurring in a variety of forest habitats, as well as representing one of the largest biomasses of the Amazon forest (Peres & Dolman 2000). Although the genus is well known and has been extensively studied (Liesenfeld 2003), most investigations of its role as seed dispersers are restricted to A. palliata and A. pigra in Central America (Estrada & Coates-Estrada 1984; Amato & Estrada 2010). Besides the need to encompass other species, the possible effects of body mass and diet should be considered, since the genus is the most folivorous Neotropical primate species (Miranda & Passos 2000; Aguiar et al. 2003; Ludwig et al. 2008) and presents sexual dimorphism, with males heavier than females, averaging 6.4 kg and 4.3 kg of body weight, respectively (Smith & Jungers 1997). Thus, the objective of this work is to evaluate the effects of fiber content (leaf proportion) and seed size on the retention time of captive A. caraya of both sexes.
MATERIALS AND METHODS
Study animals and location
Six adult individuals of A. caraya (three males [9.3 ± 1.3 kg] and three females [4.5 ± 0.5 kg]), raised in captivity at Centro Nacional de Primatas (CENP), State of Pará, Brazil. At CENP, animals live in social groups and are housed in enclosures with two compartments connected through an exchange door. They were fed fruits, leaves, roots and tubers. Sanitation was performed daily by washing the enclosures and feeding trays. Drinking water was also changed daily and animals were periodically inspected and dewormed by veterinarians.
Seeds
Six plant species were chosen to represent the spectrum of seed sizes consumed by wild howlers, based on a study by Amato & Estrada (2010). In their study at Parque Nacional de Palenque, Mexico, the size of seeds ingested by A. pigra and defecated intact varied in length between < 1 (Ficus spp.) and 34 mm (unidentified species). The average length of seeds used in this study was estimated from a random sample of 10 seeds from different fruits. Measurements were obtained using a caliper (Table 1).
Table 1 Average and standard deviation for length of seeds used in this study. Ten seeds of each species were measured.

Of the six species, four (passion fruit, watermelon, muskmelon and pumpkin) were already part of the diet offered at CENP. Kiwi (A. deliciosa) and linseed (L. usitatissimum) seeds were included to better represent the spectrum of seed sizes ingested by wild howlers, as inferred from the paper cited above.
Diet
To evaluate the effect of relative quantity of ingested fiber on seed retention time, three leaf percentages (15%, 30% and 60%) relative to total offered food weight were tested. These percentages were chosen based on the spectrum of leaf consumption variation (measured as a percentage of recordings) by wild howlers (Rímoli et al. 2012). Wild cabbage was used as a source of fiber as it is a food item in the normal diet of howlers at CENP. In three occasions out of the 36 experimental sessions using 60% leaf percentage, the animal did not ingest the total amount offered, but leaves still comprised more than 54% of the consumed food weight.
Experimental protocol
Individuals used in the study were separated from their respective social groups during each experimental session, observing a one-week rest period between sessions for the same individual. Each session lasted 48 hours, timed from seed ingestion. In each session, seeds from only one species were served and the experiment was repeated three times for each plant species. Thus, 18 experimental sessions were conducted per individual. Sessions were systematically distributed over the study period, and data was collected by increasing seed sizes and leaf percentages.
During each session, animal feeding followed the normal handling routine at CENP. Morning feeding (09:00) was composed solely of cabbage leaves (15%, 30% or 60% of total food weight for the day), and afternoon feeding was composed of fruits, roots and tubers. To avoid interference on study results, fruit seeds were removed before presentation.
Experimental seeds were offered inside banana pieces, the best-accepted food item by CENP animals. Quantity of seeds for each species was established through a pilot study, in order to guarantee the recovery of a representative number of seeds during an experimental session period: kiwi, linseed and passion fruit (40 seeds), muskmelon (30 seeds), pumpkin (24 seeds) and watermelon (20 seeds). Quantity of bananas used in each session varied between two to four pieces, in order to accommodate the experimental seeds.
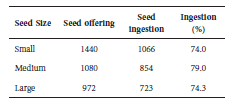
Offering of experimental seeds preceded the animals' normal afternoon feeding. Both the time when the banana pieces with experimental seeds were offered and the time when a satisfactory quantity of seeds had been ingested were recorded. These two moments established a seed-offering period, after which the experimental session began (Table 2). To quantify the real number of ingested seeds, the banana leftovers were collected after the offering period and remaining seeds were counted.
Table 2 Number of seeds offered and ingested, and percentage of ingestion, by seed size class, for the six captive individuals at CENP.
Animals were monitored through direct observations and camera recordings during the diurnal period, between 06:00 and 18:00 h. Defecation times were recorded, feces collected and seeds counted until the end of each session (48 hours).
Data analysis
Voided seeds were collectively used to calculate the mean retention time for each experimental session. Retention time (RT) was evaluated with two parameters (Fukui 2003), namely mean retention time (MRT) and standard deviation of retention time (SDRT). These calculations included only the seeds voided during the diurnal period, to which the exact moment of defecation was recorded.
The start timing of each experimental session could be defined based on two moments: when the animal ingested the first banana piece with experimental seeds (first ingestion) or when the animal ingested the last banana piece (last ingestion) during the offering period. As the RT calculated using first or last ingestion to define the start of the experimental session were highly correlated, both for the mean (r = 0.9984; p < 0.01) as well as for the standard deviation (r = 0.9999; p < 0.01), only one entry (first ingestion) was reported here.
For the analysis, seeds were grouped in three size classes (small, medium, large). After a normality test (Shapiro-Wilk test), MRT data were transformed. To test the effect of sex, seed size, diet fiber content (percentage of leaves in the diet) and the interaction between these factors on MRT and SDRT, we used repeated ANOVA measures (Zar 1999), since the same animals were used for the various treatments. Tests were performed with the R package.
RESULTS
Seed retrieval
We recovered 2644 seeds (83%) out of 3194 ingested. From those recovered, 1767 (824 for males and 943 for females) were defecated during the diurnal period (between 06:00 and 18:00 h) and a precise recording of defecation time was annotated. This figure corresponds to 55% of ingested seeds and 67% of recovered seeds (Table 3).
Table 3 General study description: number of offered, ingested and recovered seeds, by seed size class. Exact moment of defecation was recorded only for seeds voided during the diurnal period (06:00 h-18:00 h) of experimental sessions. In parenthesis, the percentage of intact seeds recovered relative to ingested seeds.
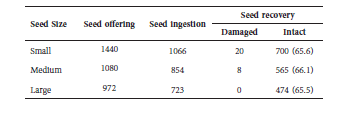
Among the 28 damaged seeds recovered, 71.4% were small seeds and none were large. This observation suggests the animals are able to perceive larger seeds still in their mouths before swallowing, while smaller seeds were inadvertently chewed and ingested with the banana pieces.
Relationship between retention time, sex, seed size and diet fiber quantity
The general means for the two RT statistics were: 1) MRT - 1304 ± 199 min (range: 48-2886 min); 2) SDRT - 462 ± 160 min (range: 130-851 min). Unlike SDRT (), which presented a normal distribution (Shapiro-Wilk test W = 0.9770; p = 0.39), MRT data were log-transformed (W = 0.9578; p = 0.05). Sex (F = 1.444; p = 0.30), seed size (F = 0.666; p = 0.54) and diet fiber content (F = 0.503; p = 0.52), did not influence RT for the two tested statistics (Fig. 1).

Fig. 1: MRT and SDRT (in minutes) for each treatment (seed size and fiber content—proportion of ingested leaves). S = small, M = medium and L = large; white and gray boxes represent females and males, respectively. Values 15, 30 and 60 represent the leaf percentage in the offered diet. MRT = Mean retention time; SDRT = Standard deviation of retention time.
Only the interaction between sex and seed size had a significant effect (F = 4.924; p = 0.04) on MRT (Table 4). Although no variable significantly influenced SDRT, there might be a tendency of SDRT reduction with increasing seed size (F = 3.439; p = 0.08) for both males and females.
Table 4 Effect of variables and their interactions on MRT and SDRT, using repeated measures of ANOVA. MRT = Mean retention time; SDRT = Standard deviation of retention time.

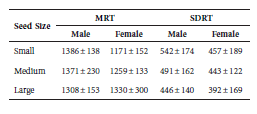
As the seed size increases, MRT decreases for males and increases for females (Table 5), suggesting that the effect of this interaction depends upon either sex or animal size.
Table 5 Values for the two statistics of retention time (minutes) by seed size class and sex category, based on 108 experimental sessions equally distributed among sex categories (males and females) and seed size classes (small, medium, large). MRT = Mean retention time; SDRT = Standard deviation of retention time.
DISCUSSION
Alouatta caraya has the potential to disperse the seeds of different plant species with lengths between 2.1 ± 0.1 and 14.0 ± 0.9 mm. The high percentage of recovered seeds (83%) in a 48- hour period, of which 98.5% were intact, reinforces this potential, considering that damaged seeds may not be viable.
The hypothesis of a difference in retention time for males and females due to their difference in body size was not confirmed. This result does not support the body mass hypothesis (Demment 1983), corroborating Milton (1984), Caton & Hume (1996), and Clauss et al. (2008) results and suggesting that howlers' body size is not the principal factor affecting seed retention time within their digestive tract.
Seed size did not affect their retention time for captive howlers, differing from the study of Lagothrix lagothricha, in which smaller seeds remained longer in the animals' digestive tract (Stevenson & Garcia 2003). Fiber content also did not affect the RT in the present study, differing from studies with L. lagothricha (Stevenson & Garcia, 2003) and 19 other primate species (reviewed by Clauss et al. 2008) that found a positive correlation between RT and fiber content.
Sex and seed size alone did not affect RT, but their interaction did significantly affect MRT. Males retained smaller seeds longer in their digestive tract, while females retained the larger ones. This difference suggests that the role of male and female as seed dispersers may vary, depending on the fruit species consumed by wild howlers. Thus, males and females may have complementary influences on recruitment and dispersion patterns for different plant species, contributing to community diversity.
Due to the difficulty of direct recording of spatial dispersion patterns (seed shadow) generated by different dispersal agents, many authors have used indirect methods to evaluate the role of animals as dispersers, based on parameters such as retention time and animal movement patterns (Stevenson & García 2003). Studies with captive animals allow the control of variables often difficult to control in natural environments (Stevenson & García 2003) and also facilitate the retrieval of voided seeds.
Retention time data obtained from these studies, in association to data about daily movements, spatial and temporal variation of wild primates diet, may be used to evaluate the effectiveness of seed dispersers and, thereafter, the spatial-temporal dynamics of plant populations and communities. However, this is still a rarely used approach, especially with Neotropical primates, relative to studies with other animal groups, such as birds (Murphy et al. 1993; Stanley & Lill 2002; Wotton et al. 2008).
Although results suggest that the captive A. caraya group is a legitimate dispenser of seeds, due to the ingestion and defecation of a high percentage of intact seeds with varying lengths of 2.1 ± 0.1 and 14.0 ± 0.9 mm, future research should evaluate the quality of this dispersion. There is a need to analyze the germination rate of defecated seeds (Bustamante et al. 1992; Soto-Gamboa & Bozinovic 2002); abundance, visitation rate, feeding behavior (feeding rate and fruit manipulation) and seed deposition site (Jordano & Schupp 2000); as well as monitoring of the fate of depositions in a natural environment (Bustamante et al. 1992; Silva et al. 2005).
CONCLUSIONS
The high rate of ingestion and defecation of intact seeds by the subjects of this study indicate the potential of A. caraya as a seed disperser. However, to determine this potential, further studies need to clarify the effectiveness of dispersion (germination rate of defected seeds and deposition site in a natural environment) for a more robust understanding of the efficiency of A. caraya as a seed disperser, the reflection on the spatial-temporal dynamics of populations and vegetation communities and the possible implications in the habitat.
ACKNOWLEDMENTS
We would like to thank CENP for their logistical support, and its staff for their help, in particular to the veterinarians Dr. Paulo Castro and Dr. José Augusto Muniz, the health monitors and research technicians Amauri Junglos and Osvaldo Marques, the biology student Jhone Mendes and all others indirectly involved in this research.
LITERATURE CITED
1. Aguiar, L. M., N. R. R. G. Ludwig, & V. J. Rocha. 2003. Dieta, área de vida, vocalizações e estimativas populacionais de Alouatta guariba em um remanescente florestal no norte do estado do Paraná. Neotropical Primates 11:78-86. [ Links ]
2. Amato, R. K., & A. Estrada. 2010. Seed dispersal patterns in two closely related howler monkey species (Alouatta palliata and A. pigra): a preliminary report of differences in fruit consumption, travelling behavior, and associated dung beetle assemblages. Neotropical Primates 17:59-66. [ Links ]
3. Bustamante R. O., J. A. Simonetti & J. Mella. E. 1992. Are foxes legitimate and efficient seed dispersers? A field test. Acta Oecologica 13:203-208. [ Links ]
4. Caton, J., & I. D. Hume. 1996. Digestive strategy of the Asian colobinae genus Trachypithecus. Primates 40:311-325. [ Links ]
5. Chapman C. A., & S. E. Russo. 2007. Primate seed dispersal. Primate in perspective. (C. J. Campbell, A. Fuentes, K. C. Mackinnon, M. Parger & S. K. Boarder, eds.). Oxford University Press, Oxford. [ Links ]
6. Clauss, M. et al. 2008. The influence of natural diet composition, food intake level, and body size on ingesta passage in primates. Comparative Biochemistry and Physiology, Part A 150:274-281. [ Links ]
7. Demment, M. W. 1983. Feeding ecology and the evolution of body size of baboons. African Journal of Ecology 21:219-233 [ Links ]
8. Estrada, A., & R. Coates-Estrada. 1984. Fruit eating and seed dispersal by howling monkeys (Aloutta palliata) in the tropical rain forest of Los Tuxtlas, Mexico. American Journal of Primatology 6:77-91. [ Links ]
9. Fukui, A. 2003. Relationship between seed retention time in bird's gut and fruit characteristics. Ornithological Science 2:41-48. [ Links ]
10. Gregorin, R. 2006. Taxonomia e variação geográfica das espécies do gênero Alouatta Lacépède (Primates, Atelidae) no Brasil. Revista Brasileira de Zoologia 1:64-144. [ Links ]
11. Jordano, P., & E. W. Schupp. 2000. Seed disperser effectiveness: the quantity component and patterns of seed rain for Prunus mahaleb. Ecological Monographs 70:591-615. [ Links ]
12. Liesenfeld, M. V. A. 2003. O destino pós-dispersão das sementes do caquizinho-do-mato (Diospyros inconstans) ingeridas pelo bugio-ruivo (Alouatta guariba clamitans) em uma floresta subtropical no sul do Brasil. Tesis de Doctorado. Universidade Federal de Campinas, São Paulo, Brasil. [ Links ]
13. Ludwig, G. et al. 2008. Comparison of the diet of Alouatta caraya (Primates: Atelidae) between a riparian island and mainland on the Upper Parana River, southern Brazil. Revista Brasileira de Zoologia 25:419-426. [ Links ]
14. Milton, K. 1984. The role of food-processing factors in primate food choice. Adaptations for foraging in nonhuman primates (P. S. Rodman & J. G. H. Cant, eds.). Columbia University Press, New York. [ Links ]
15. Miranda, J. M., & F. C. Passos. 2000. Hábito alimentar de Alouatta guariba (Humboldt) (Primates, Atelidae) em Floresta de Araucária, Paraná, Brasil. Revista Brasileira de Zoologia 21:821-826. [ Links ]
16. Murphy, S. R., N. Reid, Z. Yan & W. N. Venables. 1993. Differential passage time of mistletoe fruits through the gut of honeyeaters and flowerpeckers: effects on seedling establishment. Oecologia 93:171-176. [ Links ]
17. Oliveira, A. C. M., & S. F. Ferrari. 2000. Seed dispersal by black-handed tamarins, Saguinus midas niger (Callitrichinae, Primates): implications for the regeneration of degraded forest habitats in eastern Amazonia. Journal of Tropical Ecology 16:709-716. [ Links ]
18. Peres, C. A., & P. M. Dolman. 2000. Density compensation in neotropical primate communities: evidence from 56 hunted and non-hunted Amazonian forests of varying productivity. Oecologia 122:175-189. [ Links ]
19. Rímoli, J., R. S. Nantes, & A. E. Lázaro Júnior. 2012. Diet and activity patterns of black howler monkeys Alouatta caraya (Humboldt, 1812, Primates, Atelidae) in ecotone Cerrado-Pantanal in the left bank of Aquidauana River, Mato Grosso do Sul, Brazil. Oecologia Australis 16:933-948. [ Links ]
20. Schupp, E. W. 1993. Quantity, quality, and the effectiveness of seed dispersal by animals. Frugivory and seed dispersal: ecological and evolutionary aspects (T. H. Fleming & A. Estrada, eds.). Kluwer Academic Publishers, Dordrecht, The Netherlands. [ Links ]
21. Sibly, R. 1981. Strategies of digestion and defecation. Physiological ecology: an evolutionary approach to resource utilization (C. Townsend & P. Calow, eds.). Blackwell Scientific, Oxford. [ Links ]
22. Silva, S. I., F. Bozinovic, & F.M. Jaksic. 2005. Frugivory and seed dispersal by foxes in relation to mammalian prey abundance in a semiarid ecosystem. Journal of Austral Ecology 30:749-756. [ Links ]
23. Smith, R. J., & W. L. Jungers. 1997. Body mass in comparative primatology. Journal of Human Evolution 32:523–559.
24. Soto-Gamboa, M., & F. Bozinovic. 2002. Fruit-disperser interaction in a mistletoe-bird system: a comparison of two mechanisms of fruits processing on seed germination. Plant Ecology 159:171-174 [ Links ]
25. Stanley, M., & A. Lill. 2002. Does seed packaging influence fruit consumption and seed passage in an avian frugivore? Condor 104:136-145. [ Links ]
26. Stevenson, P. R., & C. García. 2003. Modelos para predecir las distancias de dispersión de semillas por micos churucos (Lagothrix lagothricha): aportes de estudios en campo y en cautiverio. Universitas Scientiarum 8:13-22. [ Links ]
27. Tsuji, Y., M. Morimoto, & K. Matsubayashi. 2010. Effects of the physical characteristics of seeds on gastrointestinal passage time in captive Japanese macaques. Journal of Zoology 280:171-176. [ Links ]
28. Wotton, D. M., M. N. Clout, & D. Kelly. 2008. Seed retention times in the New Zealand pigeon (Hemiphaga novaezeelandiae novaeseelandiae). New Zealand Journal of Ecology 32:1-6. [ Links ]
29. Zar, J. H. 1999. Biostatistical Analysis. Prentice Hall, New Jersey. [ Links ]














