Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Mastozoología neotropical
versión impresa ISSN 0327-9383versión On-line ISSN 1666-0536
Mastozool. neotrop. vol.25 no.2 Mendoza dic. 2018
ARTÍCULO
Description of digging behavior in armadillos Dasypus novemcinctus (Xenarthra: Dasypodidae)
Giannina P. Clerici1, Patrícia S. Rosa2 and Fabiana R. Costa3
1 Departamento de Ciências Biológicas, Universidade Federal de São Paulo, São Paulo, SP, Brazil [Correspondence: <gipclerici@gmail.com>.
2 Instituto Lauro de Souza Lima, Bauru, SP, Brazil.
3 Centro de Ciências Naturais e Humanas, Universidade Federal do ABC, São Paulo, SP, Brazil.
ABSTRACT
Although digging is an essential behavior for foraging and burrow construction in the fossorial armadillo Dasypus novemcinctus, this behavior has never been clearly described. Here, we provided the first detailed description of D. novemcinctus digging activity. We observed the behaviors of eight D. novemcinctus at the Lauro de Souza Lima Institute in Bauru. Subjects were individually recorded while digging in an outdoor, former vivarium belonging to the institute. Videos were scored frame-by-frame to yield nine distinct behavioral acts. These were organized into an ethogram and a kinematic flow diagram indicating the most common behavioral transitions. From 248 sequences, we observed that digging generally began with forelimb movement, while hind-limbs were used to remove accumulated soil on the ventral side; the tail provided support against the soil. Before digging, all subjects also half-plunged their heads into the ground, thus breaking up soil. The observed behaviors corroborate the classification of D. novemcinctus as "scratch-diggers” and also clarifies species-specific aspects of digging behavior.
RESUMO
Descrição do comportamento de escavação em tatus-galinha Dasypus novemcinctus (Xenarthra: Dasypodidae).
Embora cavar seja um comportamento essencial para o forrageamento e a construção de tocas para Dasypus novemcinctus, este comportamento nunca foi claramente descrito. O presente estudo fornece a primeira descrição detalhada para a atividade fossorial de D. novemcinctus. Foram observados os comportamentos de oito indivíduos de D. novemcinctus no Instituto Lauro de Souza Lima em Bauru. Estes espécimes foram filmados individualmente enquanto cavavam dentro de uma área externa previamente delimitada deste instituto. Os filmes foram analisados quadro a quadro e um total de nove atos comportamentais foram reconhecidos e detalhados para a produção de um etograma, bem como um diagrama comportamental cinemático de fluxo produzido para indicar as transições mais comuns de um ato para outro. A partir das 248 sequencias analisadas foi possível observar que o movimento dos membros anteriores foi na maior parte do tempo requisitado para iniciar esta atividade, e os posteriores foram usados para remover o solo que se acumulava sob o ventre dos indivíduos enquanto a cauda fornecia sustentação ao corpo a partir do seu apoio contra o solo. O ato de inserir parte da cabeça no solo a fim de torna-lo mais inconsolidado antes da atividade de escavação ter início foi aqui reportado como inédito nesta atividade exercida pelos tatus-galinha. As sequencias aqui observadas corroboram, portanto, a prévia classificação pelo menos para D. novemcinctus como "scratch-digger” e também fornece informação relevante sobre este comportamento específico para a espécie em questão.
Key words: Armadillos; Cingulata; Digging behavior; Ethology; Xenarthra.
Palavras-chave: Cingulata; Comportamento de escavação; Etologia; Tatus; Xenarthra.
Recibido 11 marzo 2017.
Aceptado 26 abril 2017.
Editor asociado: R. Zenuto
INTRODUCTION
Digging is a major part of armadillo natural history, but the behavior remains under-studied (Superina 2013). Virtually all species of armadillos are fossorial (Vizcaíno et al. 1999) and considered "scratch-diggers” (Hildebrand 1985), the most common category among mammals. Armadillos are classified into three groups based on the Index of Fossorial Ability (IFA), or the ratio of olecranon to ulna length (Vizcaíno et al. 1999): cursorial, non-digging forms; diggers that do not depend on digging for foraging success; and extensive diggers (subsequently modified by Milne et al. 2009; see Abba et al. 2005 and Vizcaíno et al. 2012). Dasypus novemcinctus Linnaeus, 1758 falls into the second category of moderately good diggers (Vizcaíno et al. 1999; Milne et al. 2009), with the likelihood that selection could have shaped limb morphology (Loughry & McDonough 2013). Approximately 70-90% of the species’ active time is spent foraging for insects via digging (Ancona & Loughry 2009).
Burrows are an important diagnostic feature for species identification (Carter & Encarnação 1983). The burrows of D. novemcinctus can even reflect age differences because their dimensions are dependent on animal size (juveniles are smaller) (McDonough et al. 2000). Burrows function most obviously as refuges (McDonough et al. 2000; Layne 2003), but can also act as food storage during drought or cold (Taber 1945). Although a solitary species, D.novemcinctus burrow density is very high in Brazil. This density could reflect considerable precautions against predators, or the amount of time the species has been present in the area, given that the structures are permanent and will remain even after vacated (McDonough et al. 2000).
No study specifically examining D.novemcinctus digging exists and the behavior has only been briefly reported in general natural history research (Taber 1945; Loughry & McDonough 2013). Nine-banded armadillos are known to loosen soil with their noses and forelimbs before pushing it back with hind-limbs (Taber 1945; Moeller 1990; McDonough & Loughry 2008; Loughry & McDonough 2013). This general pattern also holds for other species (Ancona & Loughry 2009; Duarte et al. 2015). However, the highly fossorial pygmy armadillo (Chlamyphorous truncatus Harlan, 1825) appears to possess a unique digging style among mammals (Rood 1970). Apart from these reports, we know little about armadillo digging behavior. Our study thus aims to fill this gap by providing the first detailed description of digging in D. novemcinctus.
MATERIALS AND METHODS
Subjects were eight D. novemcinctus (five males and three females) from the savannah reserve of Lauro de Souza Lima Institute (ILSL) at Bauru County in São Paulo State, Brazil. Animals were trapped between February and May 2015 using rectangular, single-entrance, and wire-mesh cages with metal doors. They were then kept in the institute’s vivarium for the experiment in June 2015. After capture, weight and total body length were determined. Subjects did not require sedation for these procedures.
After the experiment a mixture of moistened dog food and eggs was provided ad libitumfor the subjects. All procedures were in accordance with the Research Ethics Committee of the Federal University of São Paulo (UNIFESP), under protocol number 1701280316.
Subjects were placed individually in a glass terrarium (83.5 × 29 × 59.5 cm) situated inside the vivarium, a larger outdoor area (3.77 × 4.10 m) near the savannah reserve. Formerly used to raise armadillos for behavioral observations, the vivarium was filled with Dark Red Latosol (sandy phase) from the reserve (Cavassan et al. 1984), a highly weathered mineral soil mainly comprising sand, clay, and silt (Camargo et al. 1987). Within the vivarium, small rocks, pebbles, and roots were dispersed throughout the soil, but no plant cover was available and the bottom was completely cemented to prevent animals from digging out. The terrarium was built in the left corner and had wooden sides (29 × 59.5cm) and a glass window (83.5 × 59.5 cm) in front to allow video recording of subjects while digging. A camera (SONY® Cyber-shot DSC-H50 model, 9.1 megapixels) was placed 50 cm away from the window, guaranteeing full coverage of the area, and placed on a flat platform (27 × 17 × 12.5 cm) perpendicular to the ground (Fig. 1).
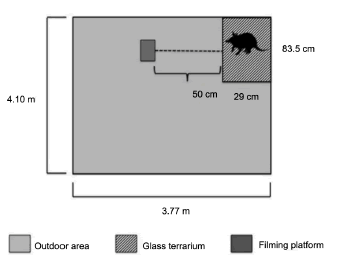
Fig. 1. Schematic of the glass terrarium built inside the former outdoor vivarium where subjects were observed.
Focal sampling was performed on each individual (Lehner 1979) for 15 min during morning (starting at 06:00 h) and afternoon (starting at 16:00 h). No artificial light was used, and the afternoon recordings were finished as dusk began. During the sampling period, an observer simultaneously reported all digging-related activity by writing it down, which was later confirmed with video scoring. When continuous digging occurred, observation time was doubled to 30 min. When a subject became stressed, it was removed and replaced with another armadillo until it was calm enough to be re-introduced into the area. Stress was identified as complete immobility, with eyes closed for over 5 min, or repetitive attempts to escape from the enclosure. Animals were placed inside the terrarium based on an established sequence that guaranteed a 4-h resting period for all animals between morning and afternoon recordings. Total recorded observation time was 20 h.
Video footage was analyzed frame-by-frame in Launch Tracker®; all digging-related behaviors were described in detail and an illustration produced. Behaviors were then noted in the order of occurrence per individual. Subsequently, data from all matrices were combined to determine the percentage of transitions (from one behavioral act to another) that occurred. This pattern of individual actions was outlined as a behavioral kinematic flow diagram (Brockmann 1994). Additionally, sex differences were determined with an unpaired t-test for size and weight, as well as a Mann-Whitney U test for digging duration. Significance was set at P<0.05.
RESULTS
We identified nine digging behaviors (kinematic-diagram codes in parentheses) (illustrations in Fig. 2):
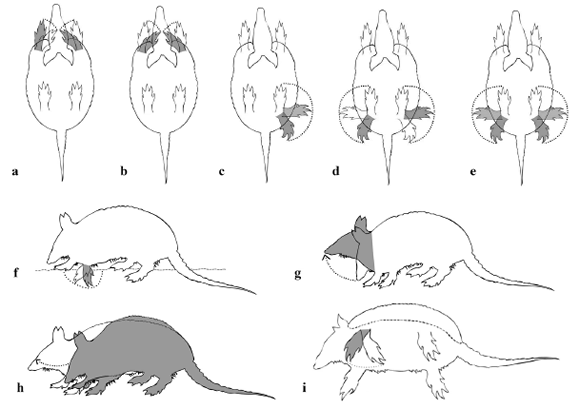
Fig. 2. Illustrations of digging-related behaviors. (a) Digging alternately with two limbs (DATL). (b) Digging simultaneously with two limbs (DSTL). (c) Pushing away the earth with one limb (PEOL). (d) Pushing away earth with two limbs alternating (PETLA). (e) Pushing away earth with two limbs simultaneously (PETLS). (f) Digging with one limb (DOL); (g) Pushing earth with head (PEH). (h) Pushing earth by stretching body (PESB). (i) Digging with body resting laterally on ground (DBRG). Drawings by André Baltieri.
– Digging with one limb (DOL): subject uses one forelimb to dig while other limbs remain on the ground. This brief scratching movement appears to be a preliminary behavior to digging.
– Digging simultaneously with two limbs (DSTL): both forelimbs are engaged together in the same two movements (protract and extend) to displace soil.
– Digging alternately with two limbs (DATL): both forelimbs dig with alternating movements (one stretches while the other retracts).
– Digging with body resting laterally on ground (DBRG): subject uses both forelimbs to dig while one side of the body rests on the ground.
– Pushing away earth with two limbs simultaneously (PETLS): both hind-limbs move soil piled up below abdomen after forelimbs have displaced sediment from the front. Forelimbs support weight during hind-limb motion, which occurs whenever there is contact between sediment and abdomen. This behavior appears to be related to clearance of space to allow more sediment to be pushed backwards.
– Pushing away earth with one limb (PEOL): one hind-limb pushes sediment while other limbs remain on the ground.
– Pushing earth with head (PEH): nose is half on the ground to push soil away for a short distance.
– Pushing earth by stretching body (PESB): the body arches and stretches, thus projecting the head forward and consequently pushes some earth that lies over it.
– Pushing away earth with two limbs alternating (PETLA): soil piled below abdomen is moved away with one forelimb at a time.
We also observed eight non-digging behaviors (previously reported by Duarte et al. 2015). These were: 1) perceiving odors (walking and sniffing simultaneously), 2) bipedal position (standing on hind legs and sniffing; identical to "bipedal sniffs” described by McDonough & Loughry 1995), 3) being alert (standing still with ears pointing forward while sniffing; similar to "quadrupedal sniffs” from McDonough & Loughry 1995), 4) jumping (rising from the ground or other surface and landing either in the same or a different location; see also Carlisi & Cooper 1975), 5) "eating from substrate” (seeking food and feeding with forelimbs, producing numerous "food probes”; Abba et al. 2005), 6) depositing droppings (defecating or urinating on substrate), 7) bobbing (removing earth with strong up-and-down motions, especially of the head), and 8) scratching (moving claws over one portion of body rapidly).
We observed 248 behavioral sequences. Most began with forelimb movement, specifically with alternating digging (DATL; 115 sequences). This act was generally followed by simultaneous digging with two limbs (DSTL; 113 sequences) or one limb (DOL; 28 sequences). When the first digging behavior was DATL, the most probable transition was to DSTL (52%), whereas DOL was more frequently followed by DATL (55%). We also observed high rates of digging while the subject was lying on its side (DBRG), followed by a repeat of DBRG and by one-limbed pushing of soil (PEOL) (both 50%). Starting with DSTL most frequently ended with DSTL (38%). Another common transition began with head-pushes of the soil (PEH), followed by forelimb (DATL,44%) or hind-limb (PETLA and PETLS) digging, then ending either with forelimbs (DSTL,50%) or hind-limbs again (PETLS, 44%). No sequences began with body stretches (PESB). We used these transition percentages (Table1) to generate a behavioral kinematic flow diagram (Fig. 3).
Table 1
Observed transition frequencies (%) between behavioral acts.
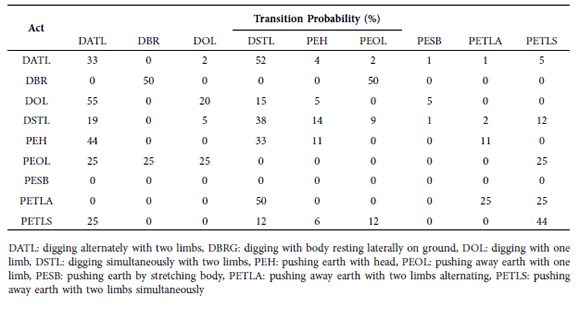
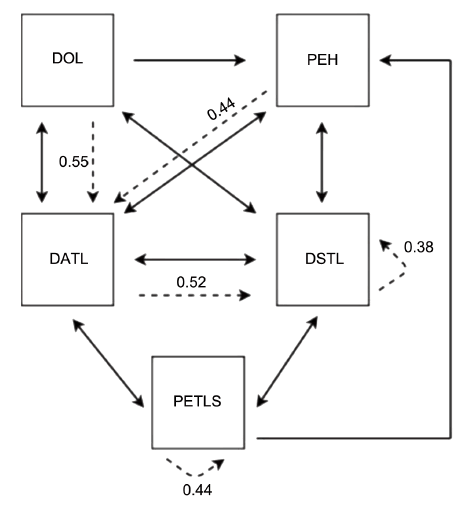
Fig. 3. Behavioral kinematic flow diagram of D. novemcinctus. Full bidirectional arrows indicate equal probability for both acts. Full unidirectional arrows indicate that the indicated sequence was recorded only once. Dashed unidirectional arrows indicate that the subsequent act has the highest probability of occurring. The highest values reported (dashed arrows) are used for plotting. DATL: digging alternately with two limbs, DOL: digging with one limb, DSTL: digging simultaneously with two limbs, PEH: pushing earth with head, PETLS: pushing away earth with two limbs.
The diagram revealed a general digging pattern. First, animals performed preliminary movements (DOL and PEH). They then continued with movements of both forelimbs (DATL and DSTL). Finally, they pushed away earth with both hind-limbs (PETLS) before returning to forelimb digging. We did not find sex differences in body length (P=0.3722; for n=5: mean ñ SD=73.6 ñ 2.619 and for n=3: mean ñ SD=67 ñ 5.686), weight (P=0.5603; for n=5: mean ñ SD=3740 ñ 136.4 and for n=3: mean ñ SD=3533 ñ 284.8), or digging duration (P=0.1429; for n=5: median with range=19 and for n=3: median with range=226) (Fig. 4).

Fig. 4. Body weight (a), body length (b), and total digging duration among D. novemcinctus males (n=5) and females (n=3)
DISCUSSION
Most digging sequences began with forelimb movement (216 of 248), specifically DATL (115 sequences). This alternating motion with both forelimbs is then succeeded by simultaneous digging (DSTL, 52%) or continued alternation (DATL; 33%). Thus, armadillos primarily use forelimbs to initiate digging, as well as remove and displace soil that piles up under the abdomen. To free the forelimbs for movement, we observed armadillos arching their body while using hind-limbs and tail for support. This act has been previously described (Taber 1945).
Behaviors that were less frequently observed included digging with one forelimb (DOL; 28 sequences) and while lying sideways on the ground (DBRG; 13 sequences). Simultaneous digging with both forelimbs more frequently followed the former (Table 1), suggesting a natural progression from superficially scratching to active digging. This sequence is likely related to foraging and not to burrow construction, as we never observed it leading to a burrow. Instead, burrow-related digging involves support from one forelimb, a hind-limb, and the lateral side of the body (including tail). We observed subjects pushing soil away with one limb (PEOL) when they attempted to dig near and under the glass window, suggesting that the window may limit free movement of both limbs. This behavior probably corresponds to situations when armadillos attempt to dig under trunks, roots, or rocks, where many burrows are found.
Hind-limb-initiated digging typically began with simultaneous pushing of soil (PETLS) and rarely with alternating movement (PETLA) or the use of a single limb (PEOL). Digging with the hind-limbs seems to only occur when soil has accumulated beneath the animal after forelimb digging (Taber 1945). Alternating the hind-limbs in a fast, back-pedaling motion pushes soil out from under the abdomen. Using a single limb achieves the same purpose but with less soil removed. In contrast, large amounts of accumulated soil can be efficiently removed through simultaneous pushing by both hind-limbs, combined with stretching and strong support from the forelimbs. This effectiveness explains why the latter behavior occurs more often than PETLA or PEOL.
Nine-banded armadillos also use their noses to loosen soil (PEH) (Taber 1945; Chapmans & Feldhamer 1982; McDonough & Loughry 2008). Indeed, some animals plunge their heads half into the ground to such an extent that they can be categorized as "head-lift diggers,” similar to moles and some rodents (Hildebrand 1985). In this study, we found that the head was only sporadically used as a spade to throw soil sideways or backwards while the animal stretches (PESB) or remains still (PEH). Study animals always followed a single head movement with subsequent forelimb digging, either simultaneously (DSTL; 33%) or alternately (DATL; 44%). This sequence was particularly common when armadillos had just begun to dig. Therefore, using the head may be a preliminary act to loosen soil for easier forelimb digging.
The only digging behavior that did not start a sequence was when the armadillo stretched to push soil (PESB). This act actually combines head and hind-limb pushing and, if observed, always followed forelimb digging (DATL, DOL, and DSTL). By arching the body and stretching, the animal is able to push soil away toward the front and back. Although infrequent, this behavior was observed in more than one animal and has never been previously described in relation to D. novemcinctus digging.
Armadillos bear long claws on their forelimb toes. Well-developed, fast-growing claws are common in fossorial mammals, allowing them to loosen soil (Hildebrand 1985). For example, Tolypeutes Illiger, 1811 was reclassified as burrow-digging (Attias et al. 2016), a change from the genus’ original status as non-diggers that re-used the burrows of other species (Nowak 1999; Vizcaíno & Milne 2002). Although less fossorial than other species, Tolypeutes was observed to dig and possess well-developed, spatulate claws (Attias et al. 2016) common to many terrestrial mammals with fossorial adaptations (Hopkins 2005), including other armadillos such as D. novemcinctus.
The tail supports the body when mammals dig. Besides this role, tails have also been documented to arrange and smooth removed soil during burrowing (Hickman 1985). Other researchers have suggested that the tail increases sensitivity to the ground in species that pushes soil back when digging (Sharpe 1943). In Ctenomys Blainville, 1826, the tail may orient animals when moving backward inside tunnels (Giannoni et al. 1996), a function already previously described for Geomys Rafinesque, 1817 (Hickman 1985). Here, we found that for nine-banded armadillos, the tail acted as support, forming a tripod with the hind-limbs when subjects stood upright (DATL, DSTL, and DOL), or else upholding one side (DBRG) while digging. When entering a burrow, nine-banded armadillos may also use their tails similarly to Ctenomys. However, more experiments are necessary to test this hypothesis, as subjects were not allowed to dig burrows deeper than half of their body length.
No sex differences were detected in body weight, body length, or digging duration, but given the small sample size in this study, we stress the importance of corroboration by future studies. Nevertheless, our preliminary data contributes to a better understanding of armadillo natural history, especially as little attention has been paid to this issue. We also provided an empirical basis for further studies of this kind.
Although digging behavior has been generally outlined in armadillos (Taber 1945), this study is the first to produce an ethogram detailing the specific behavior of using hind-limbs to kick away soil that had accumulated from forelimb digging. To the best of our knowledge, we are also the first to investigate sequential patterns in D. novemcinctus digging behavior. Our observations provide novel and detailed descriptions supporting the classification of armadillos as "scratch-diggers” (Hildebrand 1985). Armadillos share this digging pattern with other fossorial animals, such as rodents (e.g., Ctenomys; Giannoni et al. 1996) and ground squirrels (Drabek 1970). This digging style is the most common in mammals and indicates rapid, parasagittal, and alternating forelimb movement. Importantly, however, we also observed a few instances of "head-lift digging,” a less common style among mammals (Hildebrand 1985). Although use of the head is clearly secondary to use of fore- and hind-limbs, our report demonstrates that the behavioral repertoire of armadillos is more diverse than previously believed.
CONCLUSIONS
Here, we showed that armadillo digging typically begins with forelimb movement. Armadillos used their hind-limbs to remove accumulated soil beneath their abdomens after a period of forelimb digging. During this process, the tail provided support for the body against the ground. Prior to digging, armadillos occasionally half-plunged their heads and thus loosened sediment for digging. This behavior has never been described previously for any armadillo species. Our observations corroborate the classification of D. novemcinctus as "scratch-diggers.” Moreover, our detailed description of this important behavior improves existing knowledge on D. novemcinctus natural history. Consequently, this study makes a major contribution to general armadillo ethology, which remains poorly understood.
ACKNOWLEDGEMENTS
The authors thank the employees of the Lauro de Souza Lima Institute animal house for assistance with maintenance and handling during the experiments. We also thank André Baltieri for behavioral illustrations.
LITERATURE CITED
1. Abba, A. M., D. E. Udrizar Sauthier, & S. F. Vizcaíno. 2005. Distribution and use of the burrows and tunnels of Chaetophractus villosus (Mammalia, Xenarthra) in the Eastern Argentinean pampas. Acta Theriologica 50:115-124. [ Links ]
2. Ancona, K. A., & W. J. Loughry. 2009. Time budgets of wild nine-banded armadillos. Southeastern Naturalist 8(4):587-598. [ Links ]
3. Attias, N., F. R. Miranda, L. M. M. Sena, W. M. Tomas, & G. M. Mourão. 2016. Yes, they can! Three-banded armadillos Tolypeutes sp. (Cingulata: Dasypodidae) dig their own burrows. Zoologia (Curitiba) 33(4):1-8. [ Links ]
4. Brockmann, H. J. 1994. Measuring behaviour: Ethograms, kinematic diagrams, and time budgets. <http://college.holycross.edu/faculty/kprestwi/behavior/e&be_notes/E&BE_ethograms.pdf> [ Links ].
5. Camargo, M. N., E. Klant, & J. H. Kaufmam. 1987. Classificação de solos usada em levantamentos pedológicos no Brasil. Boletim Informativo da Sociedade Brasileira de Ciência do Solo, Campinas 12(1):11-33. [ Links ]
6. Carlisi, J., & G. Cooper. 1975. The nine-banded armadillo. Harvard Magazine 78(3):30-31. [ Links ]
7. Carter, T. S., & C. Encarnação. 1983. Characteristics and use of burrows by four species of armadillos in Brazil. Journal of Mammalogy 64:103-108. [ Links ]
8. Cavassan, O., O. Cesar, & F. R. Martins. 1984. Fitossociologia da vegetação arbórea da reserva estadual de Bauru, Estado de São Paulo. Revista Brasileira de Botânica 7(1):91-106. [ Links ]
9. Chapman, J., & G. Feldhamer. 1982. Wild Mammals of North America: Biology, Management, and Economics. John Hopkins University Press, New York. [ Links ]
10. Drabek, C. M. 1970. Ethoecology of the round-tailed ground squirrel, Spermophilus tereticaudus. Ph.D. thesis. University of Arizona, Tucson, USA. [ Links ]
11. Duarte, A. C., M. Superina, & F. Trujillo. 2015. Etograma para tres especies de armadillos (Dasypus sabanicola, D. novemcinctus y Cabassous unicinctus) mantenidas en cautiverio em Villavicencio, Colombia. Edentata 16:1-10. [ Links ]
12. Giannoni, S. M., C. E. Borghi, & V. G. Roig. 1996. The burrowing behavior of Ctenomys eremophilus (Rodentia, Ctenomyidae) in relation with substrate hardness. Mastozoologıa Neotropical 3:161-170. [ Links ]
13. Hickman, G. 1985. Surface-mound formation by the Tuco-tuco, Ctenomys fulvus (Rodentia: Ctenomydae), with comments on earth-pushing in the other fossorial mammals. Journal of Zoology 205:385-390. [ Links ]
14. Hildebrand, M. 1985. Digging of quadrupeds.Functional Vertebrate Morphology (M. Hildebrand, D. Bramble, K. Liem & D. Wake, eds.). Harvard University Press, Cambridge. [ Links ]
15. Hopkins, S. S. B. 2005. The evolution of fossoriality and the adaptive role of horns in the Mylagaulidae (Mammalia: Rodentia). Proceedings B of Biological Sciences 272 (1573):1705-1713. [ Links ]
16. Layne, J. N. 2003. Armadillo, Dasypus novemcinctus. Wild Mammals of North America: Biology, Management, and Conservation (G. A. Feldhamer, B. C. Thompson & J. A. Chapman, eds.). Johns Hopkins University Press, Baltimore and London. [ Links ]
17. Lehner, P. N. 1979. Handbook of Ethological Methods. New York, Garland. [ Links ]
18. Loughry, W. J., & C. M. Mcdonough. 2013. The Nine-banded Armadillo: A Natural History. University of Oklahoma Press, Norman. [ Links ]
19. Mcdonough, M. C., & W. J Loughry. 1995. Influences on vigilance in nine-banded armadillos. Ethology 100:50-60. [ Links ]
20. Mcdonough, M. C., & W. J Loughry. 2008. Behavior ecology of armadillos. The Biology of the Xenarthra (S. F. Vizcaíno & W. J. Loughry, eds.). University Press of Florida, Gainesville. [ Links ]
21. Mcdonough, M. C., M. A. Delaney, P. Q. Le, M.S. Blackmore, & W. J. Loughry. 2000. Burrow characteristics and habitat associations of armadillos in Brazil and the United States of America. Revista de Biología Tropical 48(1):109-120. [ Links ]
22. Milne, N., S. F. Vizcaíno, & J. C. Fernicola. 2009. A 3D geometric morphometric analysis of digging ability in the extant and fossil cingulate humerus. Journal of Zoology 278:48-56. [ Links ]
23. Moeller, W. 1990. Modern Xenarthrans. Encyclopedia of Mammals (S. Parker, ed.). McGraw-Hill, Inc., New York. [ Links ]
24. Nowak, R. 1999. Walker´s Mammals of the World. John Hopkins University Press, Baltimore. [ Links ]
25. Rood, J. P. 1970. Notes on the behavior of the pigmy armadillo. Journal of Mammalogy 51:179. [ Links ]
26. Sharpe, W. M. 1943. A pocket gopher (Geomys lutescens) travels backward. Journal of Mammalogy 24:99. [ Links ]
27. Superina, M. 2013. What do we know about armadillos? An analysis of four centuries of knowledge about a group of South American mammals, with emphasis on their conservation. Mammal Review 44:69-80. [ Links ]
28. Taber, W. F. 1945. Contribution of the life history and ecology of the nine-banded armadillo. Journal of Mammalogy 26(3):211-226. [ Links ]
29. Vizcaíno, S. F., & N. Milne. 2002. Structure and function in armadillo limbs (Mammalia, Xenarthra: Dasypodidae). Journal of Zoology 257(1):117-127. [ Links ]
30. Vizcaíno, S. F., J. Fernicola, & M. S. Bargo. 2012. Paleobiology of Santacrucian glyptodonts and armadillos (Xenarthra, Cingulata). Early Miocene Paleobiology in Patagonia: High-Latitude Paleocommunities of the Santa Cruz Formation (S. F. Vizcaíno, R. Kay & M. S. Bargo, eds.). Cambridge University Press, Cambridge, MA. [ Links ]
31. Vizcaíno, S. F., R. A. Fariña, & G. V. Mazzetta. 1999. Ulnar dimensions and fossoriality in armadillos. Acta Theriologica 44(3):309-320. [ Links ]














