Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO  uBio
uBio
Compartilhar
Biocell
versão impressa ISSN 0327-9545
Biocell v.26 n.3 Mendoza ago./dez. 2002
Oogenesis in the swamp eel Synbranchus marmoratus (Bloch, 1795) (Teleostei; synbranchidae). Ovarian anatomy, stages of oocyte development and micropyle structure
Mario Aldo Ravaglia, María Cristina Maggese
Laboratorio de Embriología Animal. Departamento de Biología, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires, Buenos Aires – ARGENTINA.
Address correspondence to: Dr. Mario Aldo Ravaglia. Laboratorio de Embriología Animal. Dpto. Biodiversidad y Biología Experimental, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires (UBA), Pabellón II, Ciudad Universitaria. (C1428EHA) Buenos Aires, ARGENTINA. Fax: (+54- 11) 4576 3384. E-mail: mario@bg.fcen.uba.ar
Keywords: Synbranchidae. Ovarian anatomy. Oogenesis. Micropyle.
Abstract:Synbranchus marmoratus (Synbranchidae), commonly known as the swamp eel, is a protogynous diandric teleost fish widely distributed throughout South America. The purpose of this work was to study the ovarian anatomy and to describe oocyte developmental stages in the swamp eel, Synbranchus marmoratus. S. marmoratus has a unique sacular ovary. It is covered by a conspicuous muscular wall, probably involved in an egg-releasing system acting as a peristaltic-like mechanism. The internal ovarian anatomy shows a U-shaped ovarian lamella delimiting a dorsal ovarian lumen. The microscopic study shows evidence of the existence of a germinal epithelium in the inner surface of the lamella, which contains germinal cells, pre-follicular cells and epithelial cells. The complete oogenesis process is divided into four stages: oogonia, primary growth, cortical alveoli and vitellogenesis. Besides, the ovulated oocytes, and atretic structures were described. The structure of the micropyle was studied by scanning electron microscopy (MEB). Near the animal pole the vitelline envelope forms crests that fuse together becoming furrow-like structures with a slightly spiraled direction that converge into the micropyle pit where is located the micropylar canal. Al-though the sex reversal process of Synbranchids has been subject of many studies, this is the first complete description of the ovarian anatomy and oogenesis.
Introduction
Marine and freshwater aquatic environments support more than 20,000 teleost species showing a broad diversity of sexual patterns and reproductive strategies. Although the wide range of gonadal morphologies reflects the complexity of teleost reproduction, basic features (i.e. the structure of germ cells and different somatic cell elements constituting the gonadal tissue) are similar (Nagahama, 1983). Most of the research on ovarian structure and morphology has been developed in a relative small number of species, mainly those with commercial value (Tyler and Sumpter, 1996).
The oogenesis of teleosts has been studied extensively. The description of the various stage series during oocyte development is based on distinct morphological, histological, physiological and/or biochemical cell characteristics (i.e. oocyte size and shape, quantity and distribution of different cytoplasmatic and nuclear inclusions) (Rinchard et al., 1998; Selman and Wallace, 1986; Begovac and Wallace, 1988; Wallace and Selman, 1990; Casadevall et al., 1993; Tyler and Sumpter, 1996). These successive stages are helpful in understanding cellular events during oogenesis and serve as a basis for experimental research and further comparison among species.
Synbranchus marmoratus, commonly known as the "swamp eel", is a teleost fish that belongs to the order Synbranchiformes (Rosen and Greenwood, 1976). It can be found in mud caves of rivers, ponds, swamps and marshy areas (Graham, 1981). They have alternative aerial respiration and are characterized by the lack of paired fins, a reduced caudal fin and the absence of squamation (Lüling, 1980).
S. marmoratus is a protogynous fish (Lo Nostro and Guerrero, 1996) with two different kind of males (diandria). "Primary males" develop directly to males from eggs; "secondary males" develop from functional females by sex reversal (Sadovy and Shapiro, 1987; Reinboth, 1983; Lo Nostro and Guerrero, 1996). In this order, the process of sex reversal has been subject of many studies (Chan et al., 1975; Chan, 1977; Chan and Yeung, 1983; Tao et al., 1993; Yeung et al., 1993; Ravaglia et al., 1997) but its biochemical and physiological bases are still not well understood. The purpose of this work was to study the ovarian anatomy and to describe oocyte developmental stages in the swamp eel, Synbranchus marmoratus. The knowledge of the complete oogenesis process is important to establish the sexual status throughout the female life, the first intersex step during sex reversal, and the life history of this interesting teleost species.
Materials and Methods
A total of forty two adult females of S. marmoratus (39 ± 8 cm length) were collected monthly during 1997 in marshy areas near Santo Tomé City (Santa Fe, Argentina), carried to the laboratory in wet canvas bags, and maintained in fresh water aquaria under natural photoperiod conditions. Within 48 hours of collection, fishes were anesthetized with benzocaine (1g/l), measured, weighed and killed by decapitation. Fresh ovaries were dissected under a stereoscopic microscope for anatomical description and then fixed in Bouin´s fluid overnight and embedded in paraffin. Sections from 7 to 20 mm thick (depending on the degree of oocyte development), were stained with haematoxilin-eosin for histomorphological studies, or with different histochemical techniques (PAS, PAS-Alcian blue, Klüver-Barrera, Giemsa L, Cajal silver technic) to identify mucoproteins and nervous components (Pearse, 1985). The PAS-metanil yellow method was used to stain basement membranes in tissue sections from regressed ovaries previously embedded in glycol-methacrylate (Quintiero-Hunter et al., 1991). Slides were examined under light microscopy.
Cell diameters were measured using a calibrated eyepiece micrometer, and results are expressed as the diameter range recorded for each stage of oocyte development.
For scanning electron microscopy, ovulated eggs from the ovarian cavity were fixed in 4% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.2) for 4 h, rinsed in cacodylate/sacarose buffer and gradually dehydrated from ethanol to acetone. The samples were then critical point dried, coated with gold/palladium alloy and examined under a JEOL JSM-25S II.
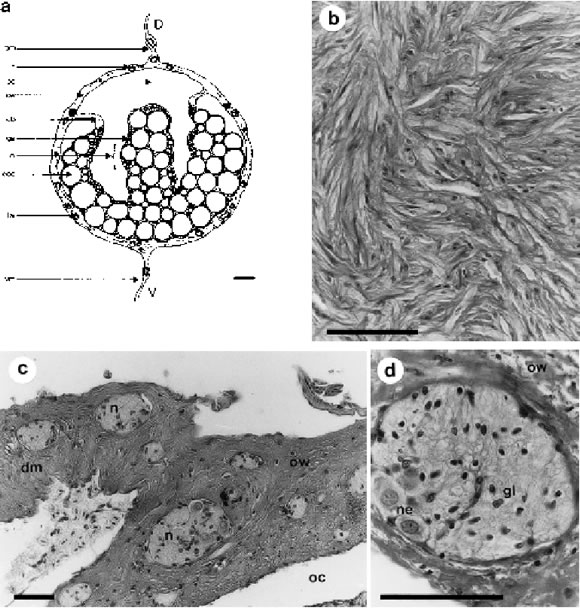
FIGURE 1. Ovarian anatomy. a: schematic drawing of S. marmoratus ovary in transverse section, showing its morphology and anatomy. b: ovarian wall showing the distribution pattern of the smooth muscular fibers. c: dorsal portion of the ovarian wall. Transverse sections of many amyelinic nerves can be seen. d: detail of transverse section of a nerve. bv: blood vessel, cb: connective bridge, dm: dorsal mesentherium, ge: germinal epithelium, gl: glial cells, n: nerve, ne: neuron, oc: ovarian cavity, ow: ovarian wall, ol: ovarian lamella, ooc: oocyte, vm: ventral mesentherium. Bars: a) 1 mm; b, c, d) 20 mm. Staining technique: b, c, d) Hematoxilyn-eosin.
Results
Ovarian anatomy
The reproductive system of the female swamp eel consists of a single tubular ovary, located above the digestive system and attached to the dorsal wall of the body cavity and to the gut by mesenteries. The ovary occupies almost the entire body cavity and is covered by a conspicuous muscular wall.
There is a "U-shaped" ovigerous lamella within the ovarian wall. This lamella is attached to the muscular wall by two lateral connective tissue bridges. Between the lamella and the ovarian wall there is a cavity, the ovarian lumen. The collapse of this cavity at final stages of vitellogenesis is due to the pressure exerted by fully developed oocytes inside the lamella. The whole gonad is homogeneous in anatomy and oocyte distribution. The germinal zone with oogonia and early oocytes rests on the inner surface of the ovarian lamella. Developing follicles are distributed along the outer surface of the ovarian lamella (Fig. 1a). The ovarian wall mainly contains smooth muscular fibers and dense connective tissue. Although the muscular fibers are oriented in many different ways, they seem to be arranged in at least three distinct layers (Fig. 1b). The numerous nervous cords found inside the ovarian wall are longitudinally oriented. The different histochemistry techniques used (Cajal silver technic and Klüver-Barrera), confirm them as amyelinic nerves forming a net-like structure (Fig. 1c, d).
The ovary narrows at its caudal end and joins the uriniferous duct to form a short urogenital duct which opens into the urogenital papilla. A small number of mature females showed their ovarian cavity full of recently ovulated eggs.
During the sampling period we found females with ovaries in different stages of development and only a few ovaries that had completely ovulated, with the ovarian cavity totally occupied with mature oocytes, ready to be spawned. However, we found no ovaries with the ovarian cavity partially occupied with mature oocytes.Stages of oocyte development
To build a discrete scale of oocyte development, we followed physiological and cytological criteria applied previously by other authors (Wallace, 1985; Cussac and Maggese, 1986; Begovac and Wallace, 1988; Casadevall et al., 1993; Tyler and Sumpter, 1996).
OOGONIA (OOG)
Oogonia are distributed homogeneously along the inner surface of the germinal epithelium which covers the ovarian cavity, and are located between epithelial and/or pre-follicular cells. They are large, generally ovoid to fairly round, with an oval-shaped nucleus, loose chromatin and usually a single nucleolus (Fig. 2a). The mean cytoplasmatic diameter varies between 18 and 36 mm, and the mean nuclear diameter between 14 and 17mm.
Germinal cells, epithelial cells and pre-follicular cells constitute the germinal epithelium and are separated from the underlying stroma by a basement membrane. This membrane is recognized as a PAS positive structure in glycol methacrylate sections of regressed ovaries stained with PAS-metanyl yellow-hematoxilin (Fig. 2a). With light microscopy, germinal cells seem to be in contact with the ovarian lumen.
There are other cells, alone or forming nests, with oogonia-like aspect (spherical shape and large nuclei). However, they can be identified as oocytes that have not begun primary growth by the presence of synap-tonemal complexes in the nucleus (Fig. 2b) and because they are clearly separated from the germinal epithelium by the basement membrane (Fig. 2c).
OOCYTE 1 (OOC 1): Primary growth or perinucleolar stage
The primary growth stage initiates when after oogonia enters in meiosis. At this stage, the ovarian follicle, considered the functional unit of the ovary, is formed. It consists of the oocyte surrounded by a follicular and a thecal cell layer.
Oocytes 1 (ooc 1) are rounded cells with abundant cytoplasm (Figs. 2d and e). Mean cellular diameter varies between 30-150 mm for the cytoplasm and 20-58 mm for the nucleus (early and late stages respectively). Aggregates of amorphous material, or "nuage", can be typically found near the nucleus. The cytoplasm is basophilic.
Follicular cells form a continuous layer around the oocyte. They have flat nuclei and with very thin cytoplasm very difficult to see at light microscopy level. At final stages of ooc 1, homogeneous and PAS positive material can be distinguished between the oocyte and the follicular layer, and will develope into the vitelline envelope.
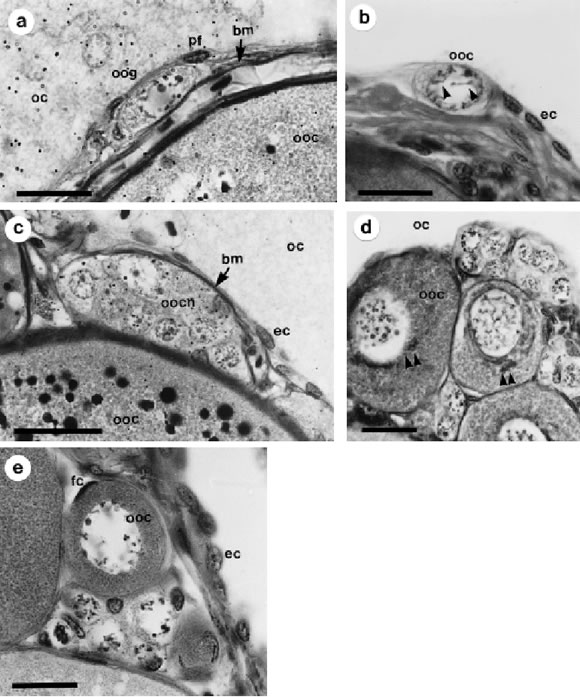
FIGURE 2. Oogonia (OOG) and Primary growth oocytes (OOC 1). a: oogonia located in the germinal epithelium. b: early oocyte with synaptonemal complexes in the nucleus (arrow heads). c: recently developed oocyte nest located just under the germinal epithelium. d: primary growth oocytes showing nuage in the perinucleolar cytoplasm (arrow heads). e: basophilic oocyte that have undergone primary growth, a surrounding follicular cell can be seen. Below it there are many other oocytes in previous phases. Double arrowhead: nuage, bm: basement membrane, ec: epithelial cell, oc: ovarian cavity, ooc: oocyte, oocn: oocyte nest, oog: oogonia, pf: prefollicular cell, fc: follicular cell. Bars: 20 mm. Staining technique: a, c) PAS-metanil yellow, b, d, e) Hematoxilyn-eosin.
OOCYTE 2 (OOC 2): Cortical alveoli stage
At this stage, the oocyte cytoplasm has a basophilic aspect. Cytoplasmatic and nuclear diameters vary among 140-360 mm and 40-100 mm, respectively The nucleo-cytoplasmatic ratio decreases progressively. Nucleoli are placed close to the nuclear envelope, which in this stage is characterized by its irregular shape (Fig. 3b). Nucleoli will remain at this position during the whole process of oocyte development. This stage is characterized by the appearance of three principal components: 1) cortical alveoli; 2) vitelline envelope and 3) the Balbiani body.
Cortical alveoli (the yolk vesicles of early literature) appear at the beginning in the peripheral zone of the ooplasm. They are membrane-limited rounded vesicles of variable size with PAS positive content. As oocyte increase in size, cortical alveoli increase in number and begin to fill the oocyte cytoplasm (Fig. 3c). At final stages, the cortical alveoli are displaced and packed in a peripheral location, just beneath the plasm membrane (Figs. 3d-f).
The vitelline envelope can clearly be distinguished almost at the same time of cortical alveoli. It appears as a thin PAS positive band with a homogeneous structure, and a mean diameter of 2 mm (Fig. 3d).
The Balbiani body (also known as "yolk nucleus"), is a conspicuous body that can be distinguished near the nucleus. This highly basophilic structure is 8 to 24 mm in diameter (Fig. 3a).
During this stage also begins the formation of cytoplasmatic lipid droplets. They accumulate at the beginning in a perinuclear position and they continue to amass possibly throughout the rest of oocyte growth. At light microscopy level it is difficult to distinguish between lipid droplets and cortical alveoli because they appear as empty vacuoles with the morphological staining methods used (Fig. 3e).
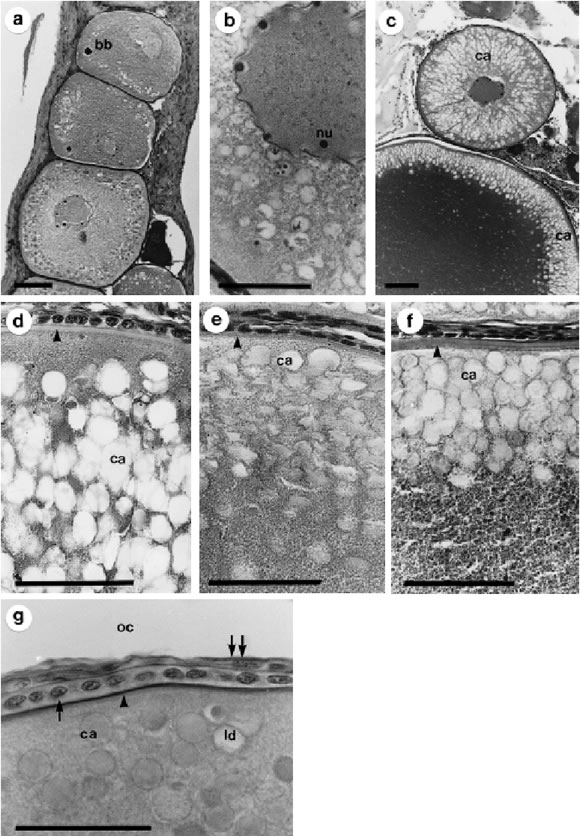
FIGURE 3. Cortical alveoli stage (OOC 2). a: general aspect of oocytes showing the Balbiani body and cortical alveoli in the peripheral cytoplasm. b: detail of the nuclear membrane showing the irregular shape that characterize this stage. c: oocytes with cortical alveoli distributed in different parts of the cytoplasm according to their degree of development. d-f: photographic sequence showing the displacement of the cortical alveoli and its packaging in the peripheral cytoplasm. g: detail of an oocyte cytoplasm showing lipid droplets and cortical alveoli. Arrow: follicular cells, double arrow: thecal cells, arrowhead: vitelline envelope, bb: balbiani body, ca: cortical alveoli, ld: lipid droplet, nu: nucleoli, oc: ovarian cavity. Bars: 50 mm. Staining technique: a-g) Hematoxilyn-eosin.
OOCYTE 3 (OOC 3): Vitellogenesis
Vitellogenesis is the principal process responsible for the significant increase in volume of the oocytes, up the 90% of the final egg size.
Oocytes in early vitellogenesis are characterized under light microscopy by the presence of little yolk globules situated under the cortical alveoli in the peripheral cytoplasm (Fig. 4a). As oocyte growth continues, these yolk globules increase in number, become distributed throughout the oocyte cytoplasm and frequently fuse in a centripetal way. The central accumulation of yolk spheres displaces the cortical alveoli to the peripheral zone (Figs. 4a, b).
At final stages of vitellogenesis the ooplasm is completely occupied with different size yolk platelets. The inner are bigger than the outer ones near the oocyte membrane (70-80 mm and 30-40 mm, respectively).
The vitelline envelope gets thicker, up to 11mm, and shows the typical radiated structure. These striations belong to the channels left in the envelope structure by the microvilli which connect both the follicular cells and the oocyte (Fig. 4e).
The follicular and thecal cells are still arranged in a single cell layer.
At this stage, the nucleus is progressively displaced towards the animal pole and some structures, probably condensed chromosomes, can be distinguished in the central part of the germinal vesicle (Fig. 4f). However, the nucleus is very difficult to find due to the huge amount of yolk spheres throughout the ooplasm.
During this part of development, oocytes reach diameters of up to 4 mm (and the germinal vesicle 180 mm). This means that during the whole process of development oocytes increases their size more than 100 times.
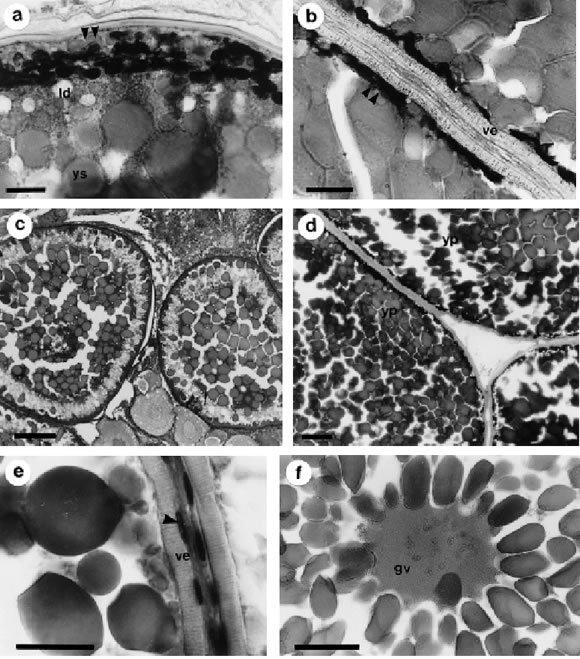
FIGURE 4. Vitellogenesis (OOC 3). a: trichromic staining of an early vitellogenic oocyte showing fluid yolk spheres, cortical alveoli and lipid droplets. b: trichromic staining of a mid vitellogenic oocyte showing the displacement of cortical alveoli to the periphery due to the huge accumulation of fluid yolk spheres. c, d: general aspect of mid and late vitellogenic oocytes, respectively, showing the accumulation of yolk as spheres (c) and crystalline platelets (d). e: high magnification of a vitellogenic follicle. The vitelline envelope is well developed with its typical radiated structure. The follicular layer remain single and flat. f: germinal vesicle of a vitellogenic oocyte showing some structures that would be condensed chromosomes. arrow: chromosomes, arrowhead: follicular cell, double arrowhead: cortical alveoli, gv: germinal vesicle, ld: lipid droplets, ve: vitelline envelope, yp: yolk platelets, ys: yolk spheres. Bars: a-b) 20mm; c-d-f) 100 mm; e) 30 mm. Staining technique: a, b, d) PAS-Alcian blue. c, e, f) Hematoxilyn-eosin.
OOCYTE 4 (OOC 4): Ovulated oocytes
In Synbranchus marmoratus oocytes are ovulated to the ovarian cavity leaving the thecal and follicular layers in the ovarian lamella. Ovulated oocytes are spherical in shape and the colored pigments contained within the lipid droplets give them the characteristic yellow-orange coloration. They are about 4 mm in diameter.
Ovulated eggs are covered by the vitelline envelope whose structure is similar to that observed in late stages of ooc 3, and no jelly-coat is observed. The cytoplasm is fully occupied with yolk platelets. The micropyle pit can be seen on the animal pole of ovulated eggs under the stereoscopic microscope. Histological differences between late stages of ooc 3 and ovulated oocytes are not conspicuous at light microscopy level.
Micropyle system
Ovulated oocytes collected from the ovarian cavity were observed with scanning electron microscopy (SEM). They showed a compact vitelline envelope surrounding the egg, with a single micropyle as the only entrance into the animal pole (Fig. 5a). The vitelline envelope in the vegetative pole has a polyhedrical surface that resembles "floor tiles" (Fig. 5b). Over the equatorial region, these "tiles" change their shape and elongate to form isolated crests. Near the animal pole these crests fuse together and become furrow-like structures with a slightly spiraled direction (Fig. 5c). These furrows converge directly into the micropyle pit. In the center of this pit is located the micropylar canal (Fig. 5d).
Atretic and ovulated follicles
Remnants of both atretic and ovulated follicles are present in the ovaries of the swamp eel (Fig. 6).
The onset of resorption is f irst detected by changes in the follicular cell layer. These cells, that are initially flat or cubic in shape, become cylindrical and vacuolized and are apparently phagocytic (Fig. 6d). Almost completely resorption of the oocyte cytoplasm, occurs in this way and the presumably phagocytic cells are finally grouped in epithelioid masses. Eosinophilic granulocytes can be identified (Giemsa-L test) (Fig. 6e) in the atretic structures and they may be also involved in the regression process.
The atretic structures show, under UV light, an in-tense autofluorescence within the range of 400-440 nm. The yolk platelets in viable oocytes have a soft green autofluorescence. Oocytes that have undergone regression processes show an intense orange fluorescence that vanishes while degradation goes on.
At the end of the atretic process, the follicular "ph-agocytic" cells, the degenerative granulocytes, the follicle remnants and pigmented debris, remain together and form a compact heterogeneous structure. This structure condenses with time and forms the brown bodies or "melano-macrophage centers" characteristic of fish (Ravaglia and Maggese, 1995) (Fig. 6f). The follicular wall of ovulated follicles remains in the ovary and undergoes a similar regression process, except that resorption of yolk material does not occur.
Discussion
Natural sex reversal occurs in Synbranchus marmoratus (Lo Nostro and Guerrero, 1996). In spite of their wide distribution and abundance, and the very interesting fact of sex reversal, the information on their life cycle and the regulation of the sexual processes is scarce. Only a few old papers on a related species (Monopterus albus) have described some reproductive characteristics (Chang and Phillips, 1967 a, b; Tang et al., 1974; Tao et al., 1993). Recently, new interesting reports have been published about S. marmoratus spermatogenesis, induction of sex reversal and brain distribution of sex hormones (Lo Nostro and Guerrero, 1996; Ravaglia and Maggese, 1995; Ravaglia et al. 1997, Vissio et al., 1996). However, for a better understanding of the basis of any reproductive study, is important to know the normal ovarian histology and oocyte development process. This is the first complete study about oogenesis in this species.
The ovarian anatomy of S. marmoratus corresponds to the cyst-ovarian type (Dodd, 1977; Nagahama, 1983) such as was also described in Monopterus albus (Chan and Phillips, 1967a) and other teleosts.
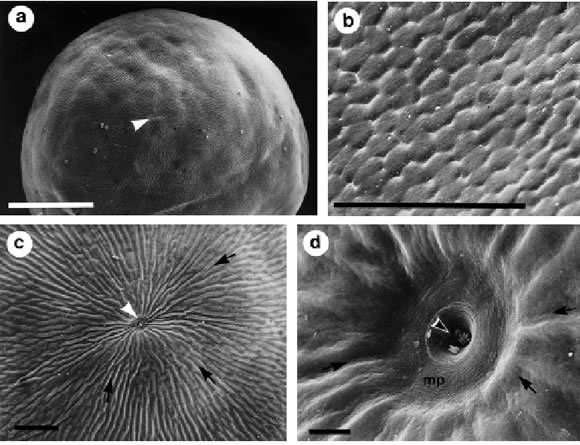
FIGURE 5. Ovulated oocytes (OOC 4). a: SEM general aspect of an ovulated oocyte observed from the animal pole. b: vegetative pole surface of an ovulated oocyte with SEM. The vitelline envelope resemble ÒtilesÓ. c: SEM detail of the oocyte animal pole showing the furrow-like structures that converge in the micro-pyle. d: SEM detail of the micropyle. The micropylar canal opens in the center of the micropyle pit. Arrow: vitelline envelope furrow, black/white arrowhead: micropyle canal, white arrowhead: micropyle, mp: micro-pyle pit. Bars: a) 1 mm; b-c) 100 mm; d) 10 mm.
The ovary has a sacular type structure and mature eggs are ovulated into the ovarian cavity. The gonad length takes almost 2/3 of the body cavity and 1/2 of the total body length. The finding of a few females with the ovarian lumen completely occupied with ovulated eggs allowed us to confirm the existence of a complete ovulation of all vitellogenic oocytes. This fact plus the absence of partially-spawned ovaries in all the reproductive fish sampled suggest the existence of only one complete spawning in each reproductive season. The histological studies and the ovarian dynamics (Ravaglia, 2000) confirm that S. marmoratus can also be considered as a "synchronous by group" spawner, according to Wallace and Selman (1981). The ovary contains a main population of large, well-developed oocytes, that would be synchronically ovulated, and a second and very little population of previtellogenic oocytes, that will be recruited in the following reproductive season. It was also confirmed that the only place for egg-storage is the ovarian lumen.
The large ovarian length plus the synchronous ovulation found, suggest the existence of a particular egg-releasing system. It might exists a kind of peristaltic-like contraction system for moving down the eggs toward the urogenital papilla, that would act contracting the ovarian wall from the anterior portion to the posterior one, moving the eggs in consecutive waves.
The terminology used to describe different stages of oogenesis varies according to authors and species. The roman numerals or the alphabetical classification are very different and sometimes misleading in many species. In the last years the classification that seems to be the most acceptable is that which briefly describes some remarkable characteristic of development (Selman and Wallace, 1986, 1989; Wallace and Selman, 1981; Selman et al., 1986, 1988; Begovac and Wallace, 1988; Tyler and Sumpter, 1996). This is the one we have used to describe S. marmoratus oogenesis.
The observations made on the ovarian anatomy and oogenesis in S. marmoratus suggest the existence of a germinal epithelium. Although this concept is not new for mammals, its application to fish gonadal anatomy is relative new. Recently Grier (2000) has redefined the idea for Centropomus undecimalis in a complete ultra-structural work.
In the swamp eel, the germinal epithelium borders the ovarian lumen and it is supported by a basement membrane that separates it from the lamellar stroma. The germinal epithelium consists of oogonia and oocytes that advance as far as the arrested diplotene of the first meiotic prophase and somatic cells such as epithelial and pre-follicular cells. A similar structure was found in the pipefish Syngnathus scovelli (Begovac and Wallace, 1987, 1988). Once oogonia enter to the first meiotic prophase they should be considered oocytes. Pre-follicular cells that surround the oocyte and a diplotene oocyte pinches off from the surface and form the ovarian follicle. In the ovarian stroma some somatic cells will be re-distributed around the follicular ones and form the theca. At this point the ovarian follicle is formed. Although our light microscopy observations support the existence of a germinal epithelium, it is very difficult to observe the differences between oogonia and recently formed oocytes. According to Grier (2000) the criteria used to differentiate both types of germinal cells are based on nuclear morphology. In general oogonia have ovoid nuclei, diffuse chromatin and a single nucleolus. On the other hand oocytes have round nuclei, also one nucleolus and conjugated chromosomes at pachytene that form synaptonemal complexes. When oocytes enter diplotene the primary growth begins and the cytoplasm becomes basophilic. This is an important character to consider in oogenesis staging, because these cells were always thought to be oogonia.
Although in S. marmoratus the nuage could be identified only in growing ooc 1 at the light microscopy level, it is also typical of oog although it can be only identified under the electron microscope in them.
Vitellogenesis is one of the most interesting processes during oogenesis. As in most oviparous animals, yolk is one of the most conspicuous elements in eggs. In teleost fishes, many related structures have been described in literature (yolk vesicles, lipid droplets, cortical alveoli, etc.). In the swamp eel, lipid droplets are the first vitellogenic structures that appear within the egg cytoplasm. Cortical alveoli appear almost simultaneously with lipid droplets. It has been well established that one of the important functions of cortical alveoli is to avoid the polyspermy by hardening the vitelline envelope after fertilization (Nagahama, 1983). Scanning electron microscope, immuofluorescense and affinity-chromatography (Selman et al., 1988) have shown that yolk vesicles and cortical alveoli are the same structure and these authors suggest to name them cortical alveoli rather than yolk vesicles, because they do not serve as a source of nutrients to the embryo.
The atretic figures are very common in fish gonads throughout the entire reproductive life cycle and their origin is variable. Van den Hurk and Peute (1979) have described very well all the atretic processes in the rainbow trout. They divided the process in four stages named a, b, g and d. The atretic process in S. marmoratus agree with the stages described in rainbow trout. The d stage of rainbow trout corresponds to the melano macrophage centers of S. marmoratus (or CMM). A complete description of these structures can be found in Ravaglia and Maggese (1995).
The complex surface of the vitelline envelope of ovulated eggs is not a common feature of fish eggs. Similar structures have been described in only a few species. Riehl and Kokoscha (1993) found the most evident and interesting case in the eggs of Luciocephalus sp. They show in mature eggs the existence of an incredible chorion architecture having lots of ridges and furrows in the animal hemisphere. These furrows have an spiraled arrangement and converge into the micro-pyle pit. Amanze and Iyengar (1990) described in the eggs of the cyprinid Barbus conchonius a micropyle region that consists of almost ten grooves and ridges, which are directed into the micropyle canal. In the catfish Sturisoma aureum, Riehl and Spatzner (1991) found eggs whose vitelline envelope have furrows running from the vegetal to the animal pole. In Luciocephalus sp. The number of furrows is similar to those of S. marmoratus, while in the other two studied species, the number of ridges is considerably lower. In Barbus conchonius, Amanze and Iyengar (1990) were able to demonstrate using time-lapse video microscopy and image analysis of sperm movements that the grooves guide sperm into the micropyle. They also calculated that this guidance role would increase sperm penetration and/or fertilization up to 99.7%. Although the ridges and furrows in the swamp eel S. marmoratus are not as developed as the Luciocephalus sp. ones, we suggest the possible existence of a similar sperm guidance role to the one found in Barbus conchonius.
We could not recognize the micropyle structure under light microscopical observations. Although the number of histological sections of mature oocytes observed was important, there was no evidence of the micropylar cell. This may be due to two reasons: the oocytes at final stages of growth are quite large, so the chances of finding the sections with the micropyle cell are low. On the other hand it is very difficult to get good and complete histological sections of mature oocytes because the fixed yolk platelets have a hard crystalline structure that make the paraffin-slicing process really difficult.
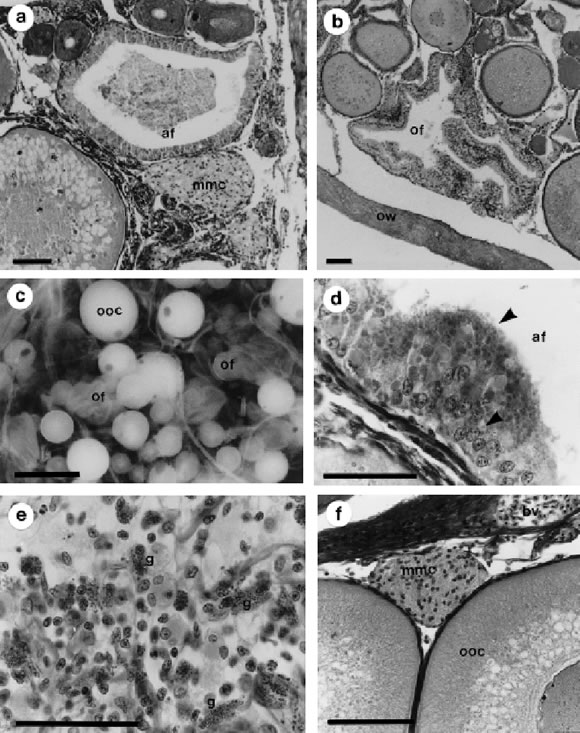
FIGURE 6. Atretic structures. a: atretic follicle in a previtellogenic ovary. b: ovulated follicle in a post spawned ovary. c: macroscopic view of a post spawning ovary showing the ovulated follicles like collapsed sacs. d: detail of phagocytic follicular cells from an initial atretic follicle. e: advanced atretic follicle. Eosinofilic granulocytes can be seen between the phagocytic follicular cells and the residual structures. f: melano-macrophage center in the ovarian lamella. af: atretic follicle, arrowhead: phagocytic follicular cells, bv: blood vessel, g: granulocytes, mmc: melano-macrophage center, of: ovulated follicles, ooc: pre-vitellogenic oocyte, ow: ovarian wall. Bars: a-b-d-e-f) 50 mm; c) 2 mm. Staining technique: a, b, d, e, f) Hematoxilyn-eosin.
Acknowledgments
The authors want to thank Dr. Harry Grier (Florida Marine Research Inst.) for his valuable advise and help with the germinal epithelium observations and Dr. Dante Paz (Universidad de Buenos Aires), for his critical review. This work was supported by grants from the University of Buenos Aires (TW41) and CONICET (PIP 4558/96 and 0539/98).
References
AMANZE D, IYENGAR A (1990). The micropyle: a sperm guidance system in teleost fertilization. Development 109: 495-500. [ Links ]
BEGOVAC P, WALLACE R (1987). Ovary of the pipefish, Syngnathus scovelli. J Morphol 193: 117-133. [ Links ]
BEGOVAC P, WALLACE R (1988). Stages of oocyte development in the pipef ish Syngnathus scovelly. J Morphol 197: 353-369. [ Links ]
CASADEVALL M, BONET S, MATALLANAS J (1993). Description of different stages in Ophiodon barbatum (Pisces, Ophiidae). Environ Biol Fishes 36: 127-133. [ Links ]
CHAN S T, PHILLIPS JG (1967a). The structure of the gonads during natural sex reversal in Monopterus albus (Pisces, Teleostei). J Zool Lond 151: 129-141. [ Links ]
CHAN ST, PHILLIPS JG (1967b). Seasonal changes in the distribution of gonadal lipids and spermatogenetic tissue in the male phase of Monopterus albus (Pisces: Teleostei). J Zool Lond 152: 31-41. [ Links ]
CHAN ST, WAI-SUM O, B SW (1975). The gonadal and adenohypophysial functions of natural sex reversal. In: Intersexuality in Animal Kingdom (Reihboth R. ed.) Berlin: Springer Verlag, pp.201-221. [ Links ]
CHAN ST (1977). Spontaneous sex reversal in fishes. In: "Handbook of sexology" (J. Money and H. Musaph, eds.) Elsevier/ North Holland Biomedical Press. Amsterdam, pp 91-105. [ Links ]
CHAN ST, YEUNG N (1983). Sex control and sex reversal in fishes under natural conditions. In: Fish Physiology. Hoar W., Randall D. and Donaldson E., eds. vol IX - part B. Academic Press. New York, pp 171-222. [ Links ]
CUSSAC VE, MAGGESE MC (1986). Oogenesis in Rhamdia sapo (Pisces: Pimelodidae). Stages of the oocyte, egg's envelopes and effects of the human chorionic gonadotropin. Rev Bras Biol 46(1): 139-147. [ Links ]
DODD JM (1977). The structure of the ovary of nonmammalian vertebrates. In: "The ovary". 2nd ed., vol. 1. Zuckerman, S. and B. Weir, eds. New York, Academic Press, pp. 219-263. [ Links ]
GRAHAM JT (1981). Burrowing and amphibious life of of the swamp eel Synbranchus marmoratus. Am Zool 21: 995. [ Links ]
GRIER HG (2000). The ovarian germinal epithelium and folliculogenesis in the common snook Centropomus undecimalis (Teleostei: Centropomidae). J Morphol 243: 265-281. [ Links ]
LO NOSTRO F, GUERRERO G (1996). Presence of primary and secondary males in a population of Synbranchus marmoratus, Bloch 1795, a protogynous fish (Teleostei - Synbranchiformes). J Fish Biol 49: 788-800. [ Links ]
LÜLING KH (1980). Biotop, begleitfauna und amphibische lebensweise von Synbranchus marmoratus (Pisces Synbranchidae) in seitengewässern des mittleren Paraná. Bonner Zool Beitr, yr. 1/2, nº 31, pp 111-143. [ Links ]
NAGAHAMA Y (1983). The functional morphology of teleost gonads. In: Fish Physiology (Hoar W., Randall D. and Donaldson E. eds.) vol IX - part A. Academic Press. New York, pp. 223-275. [ Links ]
PEARSE AG (1985) Histochemistry. Theoretical and applied. Vol. 2. Analytical Technology. Fourth Edition. Churchill Livingstone, New York, pp. 874-928. [ Links ]
QUINTIERO-HUNTER I, GRIER H, MUSCATO M (1991). Enhancement of histological detail using metanil yellow as counterstain in periodic acid Schiff´s hematoxylin staining of glycol methacrylate tissue sections. Biotechnique and Histochemistry 66: 169-172. [ Links ]
RAVAGLIA MA, MAGGESE MC (1995). Melano-macrophages Centres in the gonads of the swamp eel Synbranchus marmoratus Bloch, (Pisces, Synbranchidae). Histological and histochemical characterization. J Fish Dis 18: 117-125. [ Links ]
RAVAGLIA MA, LO NOSTRO F, MAGGESE MC, GUERRERO GA, SOMOZA GM (1997). Characterization of molecular variants of GnRH, induction of spermiation and sex reversal using salmon GnRH-A and domperidone in the protogynous diandric fish, Synbranchus marmoratus Bloch (Telesotei, Synbranchidae). Fish Physiology and Biochemistry 16: 425-436. [ Links ]
RAVAGLIA MA (2000). Reproductive biology of the swamp eel Synbranchus marmoratus Bloch 1795 (Teleostei: Synbranchidae). Oo-genesis and hormonal induction of final maturation and ovulation. Ph. D. Thesis. Biology Department, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires. Buenos Aires, 176 pp. (In Spanish). [ Links ]
REINBOTH R (1983). The peculiarities of gonad transformation in teleosts. Different 23(suppl.): 82-86. [ Links ]
RIEHL R, SPATZNER RA (1991). Breeding, egg structure and larval morphology of the catf ish Sturisoma aureum (Steindachner) (Teleostei, Lorecariidae). J Aquariculture and Aquatic Science 6: 1-6. [ Links ]
RIEHL R, KOKOSCHA M (1993). A unique surface pattern and micropylar apparatus in the eggs of Luciocephalus sp. (Perciformes, Luciocephalidae). J Fish Biol 43: 617-620. [ Links ]
RINCHARD J, PONCIN P, KESTERMONT P (1998). Croissance ovocytaire et regulation steroidienne chez les poissons a pontes unique et multiples: une revue. Annls Limnol 34(2): 211-225. [ Links ]
ROSEN DE, GREENWOOD PH (1976). A fourth Neotropical species of Synbranchid eel and the phylogeny and systematics of Synbranchiform fishes. Bull Am Mus Nat His vol. 157(1) 70 pp. [ Links ]
SADOVY I, SHAPIRO D (1987). Criteria for the diagnosis of hermaphroditism in fishes. Copeia 1: 136-156. [ Links ]
SELMAN K, WALLACE R (1986). Gametogenesis in Fundulus heteroclitus. Amer Zool 26: 173-192. [ Links ]
SELMAN K, WALLACE R (1989). Cellular aspects in oocyte growth in teleosts. Zool Sci 6: 211-231. [ Links ]
SELMAN K, WALLACE RA, BARR V (1986). Oogenesis in Fundulus heteroclitus. IV. Yolk vesicle formation. J Exp Zoology 239: 277-288. [ Links ]
SELMAN K, WALLACE RA, BARR V (1988). Oogenesis in Fundulus heteroclitus. V. The relationship of yolk vesicles and Cortical alveoli. J Exp Zoology 246: 42-56. [ Links ]
TANG F, CHANG STH, LOFTS B (1974). Effects of mammalian luteinizing hormone on the natural sex reversal of the ricefield eel, Monopterus albus (Zuiew). Gen Comp Endocrinol 24: 242-248. [ Links ]
TAO Y, LIN H, VAN DER KRAAK G, PETER R (1993). Hormonal induction of precocious sex reversal in the ricef ield eel, Monopterus albus. Aquaculture 118: 131-140. [ Links ]
TYLER CR, SUMPTER JP (1996). Oocyte growth and development in teleosts. Rev Fish Biol Fisheries 6: 287-318. [ Links ]
VAN den HURK R, PEUTE J (1979). Cyclic changes in the ovary of the rainbow trout, Salmo gairdneri, with special reference to sites of steroidogenesis. Cell Tissue Res 199: 289-306. [ Links ]
VISSIO PG, PAZ DA, MAGGESE MC (1996). The adenohypophysis of the swamp eel, Synbranchus marmoratus, an immunocytochemi-cal analysis. Biocell 20(2): 155-161. [ Links ]
WALLACE RA (1985). Vitellogenesis and oocyte growth in nonmammalian vertebrates. In: Developmental Biology. Vol 1, (L. W. Browder Eds.). Plenum Press, New York, pp. 127-177. [ Links ]
WALLACE RA, SELMAN K (1981). Cellular and dynamics aspects of oocyte growth in teleosts. Am Zool 21: 325-343. [ Links ]
WALLACE RA, SELMAN K (1990). Ultrastructural aspects of oogenesis and oocyte growth in fish and amphibians. J Electron Microsc Tech 16: 175-201. [ Links ]
YAMAMOTO TS, KOBAYASHI W (1992). Closure of the micropyle during embryonic development of some pelagic fish eggs. J Fish Biol 40: 225-241. [ Links ]
YEUNG WSB, CHEN H, CHAN STH (1993). Effects of LH and LHRH-Analog on gonadal development and in vitro steroidogenesis in the protogynous Monopterus albus. Gen Comp Endocrinol 89: 323-332. [ Links ]
Received on March 19, 2002.
Accepted on August 26, 2002.














