Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Biocell
Print version ISSN 0327-9545
Biocell vol.29 no.3 Mendoza Aug./Dec. 2005
Ubisch bodies and pollen ontogeny in Oxalis articulata Savigny
Sonia Rosenfeldt and Beatriz G. Galati
Departamento de Biodiversidad y Biología Experimental, Facultad de Ciencias Exactas y Naturales. Universidad de Buenos Aires. Buenos Aires, Argentina.
Address correspondence to: Lic. Sonia Rosenfeldt. Departamento de Biodiversidad y Biología Experimental, Facultad de Ciencias Exactas y Naturales, UBA. 4º piso, Pabellón II, Ciudad Universitaria. C1428EHA Buenos Aires. ARGENTINA. E-mail: soniar@bg.fcen.uba.ar / galati@bg.fcen.uba.ar
ABSTRACT: The correlation between the ontogeny of Ubisch bodies and pollen development in Oxalis articulata was studied with Transmission Electron Microscopy (TEM). The ultrastructural changes observed during the different stages of development in the tapetal cells are related to Ubisch bodies, sporopollenin and pollen-kitt formation. The pro-orbicules have the appearance of lipid globuli and their formation is related to the endoplasmic reticulum of rough type (ERr). The lipid globules or pro-orbicules disappear in the mature Ubisch bodies, and the places that they occupied remain free of contents or with pollen-kitt.
Keyword: Pollen. Ubisch bodies. Ontogeny. Utrastructure. Oxalis
Introduction
The genus Oxalis L. comprises about 800 cosmopolitan species. Previous studies on microsporogenesis and microgametogenesis in this family are meagre (Johri et al., 1992). The majority of them have been carried out in the genus Averrhoa and very few ones in the genus Oxalis (Herr, 1972; Guth and Weller 1986).
Ubisch (1927) observed granular bodies along the inner walls of tapetal cells in Oxalis and she pointed out the staining properties similarities of these bodies to pollen exine.
These granular bodies were described by Ubisch (op. cit.) as very showy small plates due their size and form. In spite of this fact, ultrastructural studies of these bodies have been performed in only one species of this genus until this moment (Carniel, 1967).
The aim of this paper is to describe the ultrastructure and development of Ubisch bodies and pollen grains in Oxalis articulata Savigny in order to advance the understanding of the orbicules morphology and function.
Material and Methods
The material of Oxalis articulata Savigny was collected in FCEyN, Ciudad Universitaria, Nuñez, Buenos Aires, Argentina.
Material was fixed in FAA (formalin, alcohol, acetic- acid) for light microscopy studies. Sections were made by means of standard microtome techniques. The slides were stained in a safranin-fast green combination, cresyl violet or toluidine blue and mounted in synthetic resin (D'Ambrogio, 1986). The material was observed with a Wild M20 microscope.
Material fixed in FAA was transferred to 100° acetone and then air dried for scanning electron microscopy (SEM) studies. The sputtering treatment was made with gold-palladium for 3 min.
For transmission electron microscopy (TEM) studies, the material was pre-fixed in 2.5% glutaraldehyde in phosphate buffer (pH 7.2) for 2 h and post-fixed in OsO4 at 2ºC in the same buffer for 3 h. Then the material was dehydrated in ascending ethanol series and embedded in Spurr's resin. Fine sections were made on a Sorvall ultramicrotome, stained with uranyl acetate and lead citrate (O'Brien and Mc Cully, 1981), observed and photographed in a JEOL 100c TEM.
Results
We identified five stages of Ubisch bodies and pollen development in Oxalis articulata.
Stage 1: Microspore Mother Cells (MMC)
The anther is tetrasporangiate and its wall consists of: epidermis (ep), endothecium (en), one or two middle layers (ml) and a secretory type tapetum (t) (Figs. 1, 3).

FIGURES 1-4. MMC stage in O. articulata. Figs. 1-2. Light micrographs. Fig. 1. Transverse section of a developing anther showing the anther wall layers and the MMC. Scale bar: 10 μm. Fig. 2. MMC with callosic wall (c) and cytoplasmic connections (arrow). Scale bar: 5 μm. Figs. 3-4. Transmission electron micrographs. Fig. 3. Anther wall layers (ep: epidermis, en: endothecium, ml: middle layer, t: tapetum) and MMC. Scale bar: 2 μm. Fig. 4. Detail of tapetal cell cytoplasm with abundant endoplasmic reticulum of rough type (ERr). Scale bar: 500 nm.
The MMC are uninucleate and with few vacuolated cytoplasm (Fig. 1). They have abundant endoplasmatic reticulum of rough type (ERr), mitochondria, proplastids, and dictyosomes (Fig. 3).
The tapetal cells cytoplasm has many free ribosomes, ERr and proplastids (Fig. 3).
When callose walls envelop the MMC, conspicuous cytoplasmic connections are observed (Fig. 2).
The cytoplasm of the tapetal cells is very dense, with a similar ultrastructure to that of the MMC (Fig. 4).
Stage 2: Tetrad formation
Cytokinesis is simultaneous resulting in tetrahedral microspore tetrads (Fig. 5). The middle lamella of the MMC is disintegrated but the primary wall is still present (Fig. 6). When the tetrad is formed, an incipient primexine is seen between the callosic wall and the plasmalemma (Fig. 6).

FIGURES 5-7. Transmission electron micrographs of stage 2 (Tetrad formation). Fig. 5. Microspores tetrad. Scale bar: 2 μm. Fig. 6. Microspore cytoplasm detail; callose (c), lipidic globule (lg). Scale bar: 500 nm. Fig. 7. Tapetal cell detail with cytoplasmic connections (arrow). Endoplasmic Reticulum of rough type (ERr), plastid (p). Scale bar: 500 nm.
The microspore cytoplasm shows high activity and has many mitochondria. Plastids and large osmiophilic globules are scattered in the cytoplasm (Fig. 6).
In this stage, the tapetal cells are binucleate and more vacuolated. Their cytoplasm shows an ERr increase. The endoplasmic reticulum cisternae are arranged in dense and uniformly thick rows, parallel to the cell wall (Fig. 7). There are also many proplastids, mitochondria and small lipidic globules (Fig. 7).
The tapetal cell wall begins to dissolve and presents a lax structure. Many cytoplasmic connections go through the radial walls of these cells (Fig. 7).
Stage 3: Free young microspores
After the dissolution of the callose wall, the sporopollenin wall begins to form. Three different electron- dense layers form the pollen grain wall: ectexine with interrupted tectum, columellae and foot layer; endexine and intine still in formation (Figs. 8, 11).
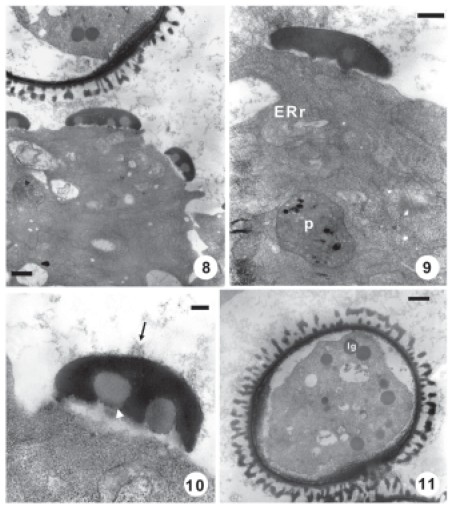
FIGURES 8-11. Transmission electron micrographs of stage 3 (Free young microspores). Fig. 8. Portion of a microspore and tapetal cell with Ubisch bodies. Scale bar: 1 μm. Fig. 9. Ubisch body and tapetal cell cytoplasm details. Endoplasmic reticulum of rough type (ERr), plastid (p). Scale bar: 500 nm. Fig. 10. Ubisch body detail; lipidic globule (white arrowhead ). Note the fibrillar electron dense material (black arrow). Scale bar: 200 nm. Fig. 11. Young microspore. Scale bar: 1μm.
The microspores have a conspicuous nucleus and the structure of their cytoplasm is similar to the previous stage (Fig. 11).
The tapetal cells show a dense cytoplasm with abundant ERr, numerous free ribosomes and well development plastids. The ERr lost the parallel configuration and its cisternae are extensive (Fig. 9).
Moderate electron-dense globules or pro-orbicules appear outside the plasmalemma at the inner tangential face of the tapetal cells (Figs. 8, 9). These globules are observed on depressions or crypts of the tapetal cells plasmalemma.
A fibrillar electron-dense material is observed inside the anther locule (Figs. 8-10). This material is deposited on the developing exine and on several globules or prorbicules (Fig. 10). One to six globules are assembled for sporopollenin deposition and they form the young Ubisch bodies (Figs. 8-10).
Stage 4: Free microspores
The microspores have a conspicuous nucleus and their cytoplasm is limited to a parietal position due to the presence of a large vacuole (Fig. 12).

FIGURES 12-18. Stage 4 (Free microspores, figs.12,14,16) and stage 5 (Mature pollen grain, figs.13,15,17,18) in O. articulata. Figs. 12,13,18. Ligth micrographs. Figs. 14,15,16. Transmission electron micrographs. Fig. 12. Free microspores. Scale bar: 10 μm. Fig. 13. Mature pollen grains and Ubisch bodies on the inner face of endothecial cells (arrow). Scale bar: 10μm. Fig. 14. Microspore wall, Ubisch bodies and tapetal cells (tc). Scale bar: 1μm. Fig. 15. Pollen wall and cytoplasm of the vegetative cell. Scale bar: 1μm. Fig. 16. Ubisch body detail with tapetal cell rests (tc). Scale bar: 500nm. Fig. 17. Mature Ubisch bodies showing the places that occupied the lipidic globules (arrow). Scale bar: 1 μm. Fig. 18. Ubsich bodies in paradermal view. Scale bar: 7,5 μm.
Many mitochondria and large lipidic globules are observed in the microspores cytoplasm (Fig. 14).
Tapetal cells start autolysis, and only rests of these cells can be observed (Figs. 12, 14, 16).
Ubisch bodies and exine have the same high electron- density (Fig. 14). The globules present in the Ubisch bodies at the previous stage are no longer observed (Figs. 14, 16).
Lipidic substances are seen among the exine columellae (Fig. 14).
Stage 5: Mature pollen grain
Pollen grains are two-celled when shed. The vegetative cell encloses the generative cell (Fig. 20).
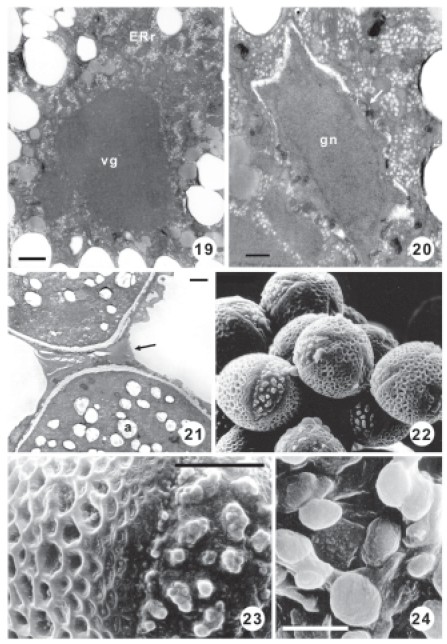
FIGURES 19-24. Stage 5 (Mature pollen grain) in O. articulata. Figs. 19-21. Transmission electron micrographs. Fig. 19. Detail of vegetative cell cytoplasm. Endoplasmic reticulum of rough type (ERr), vegetative nucleus (vn). Scale bar: 1 μm. Fig. 20. Detail of generative cell; generative nucleus (gn). Membranous inclusions in the electron- translucent wall (arrow). Scale bar: 500nm. Fig. 21. Detail of two pollen grains with abundant pollenkitt (arrow). Note the amyloplasts (a) in the vegetative cell cytoplasm. Scale bar: 2 μm. Figs. 22-24. Scanning electron micrographs in Oxalis articulata. Fig. 22. Pollen grain. Scale bar: 10 μm. Fig. 23. Details of colpus and exine. Scale bar: 10 μm. Fig. 24. Detail of Ubisch bodies. Scale bar: 10 μm.
The generative cell acquires a fusiform aspect with a lobulate outline (Fig. 20).
This cell has an electron-translucent wall with some plasmodesmata and membranous inclusions (Fig. 20). The generative cell cytoplasm is highly reduced when compared to the large nucleus (Fig. 20).
The vegetative cell cytoplasm is very dense, filled with numerous small vesicles, mitochondria, lipidic globules, amyloplasts and ERr with extended cisternae (Figs. 15, 19). The vegetative cell nucleus is lobed, for this reason, small portions of it are seen in the fine sections (Fig. 19).
The pollen grain wall and the Ubisch bodies are fully developed (Figs. 13, 15, 17). The ectexine and the Ubisch bodies sporopollenin is compressed and shows less electron-density that in the previous stage (Figs. 15, 17).
At this stage, abundant pollenkitt is present among pollen wall columellae and connects different grains in the anther locule (Figs. 15, 21).
The places occupied by the globules during Ubisch bodies formation are reduced in size and are observed as small perforations in fine sections. These places are free of contents or occupied by pollen-kitt (Figs. 16, 17).
These Ubisch bodies are seen as multiperforated plates in paradermal view under light microscopy (Fig. 18) and as plates with dome shape in lateral view and smooth surface under scanning electron microspoy (Fig. 24).
Pollen grain morphology
The pollen grain is tricolpate, anguloaperturate, prolate- spheroidal to subprolate in equatorial view, columellate, reticulate, with granular lumina (Figs. 22, 23).
The ectexine is reduced to small granular fragments in the colpus (Fig. 23).
Discussion
This is the first report about Ubisch bodies ontogeny in relation with pollen development in the genus Oxalis.
The microsporogenesis and microgametogenesis in O. articulata are similar to previous descriptions in other species of the genus (Johri et al. 1992).
In this species the pollen grains are two-celled when shed. According to Pargney (1978), O. corniculata pollen grains are three celled at the shedding stage.
The ultraestructural changes observed during the different stages of development in the tapetal cells are related to Ubisch Bodies, sporopollenin and pollen-kitt formation.
Tapetal development in Oxalis articulata is similar to other Dicotyledons secretory tapetum descriptions, however, tapetal cell walls loss occur very early, during the tetrad stage. At this stage, there is a great amount of ERr in the tapetal cell cytoplasm, and at the following stage, the cisternae of ERr are very extensive. This fact possibly marks the ERr involvement in the Ubisch bodies synthesis. Similar origin was reported in Passiflora spp. (Amela Garcia et al., 2002), Jacaranda mimosifolia (Galati and Strittmatter, 1999), Ceiba insignis (Galati and Rosenfeldt,1998), Prosopis julifora (Vijayaraghavan and Chaudhry, 1993), Catharanthus roseus (EL-Ghazaly and Nilsson, 1991), Anemarrhena asphodeloides (Chen et al., 1988), Lilium henryi (Herich and Lux, 1985), Lavandula dentata (Suarez-Cervera and Seoane-Camba, 1986) and Allium cepa (Risueño et al., 1969).
At the free young microspores stage, numerous globuli appear outside the plasmalemma at the inner tangencial face of the tapetal cells. Due to their electron- density, these globuli seem to be constituted by lipidic substances and may be consider as pro-orbicules. Similar observations and interpretations were made by Clement and Audran (1993 a,b) and Heslop-Harrison and Dickinson (1969) in Lilium; Christensen et al. (1972) in Sorgum bicolor and Steer (1977) in Avena.
Pro-orbicules are observed on depressions or crypts of the tapetal cells plasmalemma. Rowley and Walles (1987) and Steer (1977) found similar crypts and thought that they constituted a site of lipid deposition during pro-orbicule formation.
A fibrillar substance with the same electron density as sporopollenin accumulates on the globuli after these bodies are outside the plasmalemma of the tapetal cells. For this reason, the accretion of sporopollenin on them appears extracellular. These observations are in concordance with the opinion of Amela Garcia et al. (2002), Galati and Strittmatter (1999), Galati and Rosenfeldt (1998), Clement and Audran (1993 a,b), Christensen et al. (1972), and Echlin (1971).
According to Carniel (1967), the Ubisch bodies of O. pubescens are developed before tapetal cell wall total desintegration. On the contrary, in O. corniculata the tapetal cells wall begins to dissolve at the tetrade stage, and these walls are totally degraded when the sporopollenin accumulates on the globuli at the young microspore stage.
The lipid globules or pro-orbicules disappear in the mature Ubisch bodies, and the places that they occupied remain free of contains or with pollen-kitt.
These observations were also reported for O. pubescence (Carniel, 1967).
In the young pollen grain, the vegetative cell goes through a growth period, characterized by the synthesis of new cytoplasm and an increase in the number of organelles, as it has been reported for the majority of the ultrastructurally studied species (Raghavan, 1997). Many small vesicles are scattered in the cytoplasm of this cell. According to Fisher et al. (1968) these vesicles derive from dictyosomes and according to Noguchi (1990), vesicles of this type could fuse to form vacuoles in the emerging pollen tube. Amela García et al. (2002) thought that the small vesicles could constitute a membrane reservoir for the initial fast growth of the pollen tube, assuring its anchoring on the stigma.
The generative cell of Oxalis articulata presents some evaginations that protrude into the cytoplasm of the vegetative cell. Tiwari (1994) found that the wall processes arise from all faces of the generative cell and protrude into the cytoplasm of vegetative cell. According to Theunis et al. (1985) the ends of Rhododendron laetum generative cell narrow into long evaginations.
We can speculate that O.articulata generative cell morphology could support the transport of metabolites from the vegetative cell.
The pollen morphology coincides with the general description for the genus (Erdtman, 1952).
According to Wang and Chen (2001) O. acetocella pollen grains are tetracolpate and rarely tri or penta colpate. However, we have not observed pollen grains 4-or 5-colpate in O articulata, only 3-colpate.
Acknowledgements
We thank Lic. Lara Strittmatter for reviewing the English. This work was supported by Grant Nº 2514 from CONICET, Argentine.
References
1. Amela Garcia MT, Galati BG, Anton AM (2002). Microsporogenesis, microgametogenesis and pollen morphology of Passiflora spp. (Passifloraceae). Bot J Linn Soc 139: 383-394. [ Links ]
2. Carniel K (1967). Licht- und electromenmikroskopische Untersuchung der UbischKörperentwicklung in der gattung Oxalis. Österr Bot Z 114:490-501. [ Links ]
3. Christensen JE, Horner HT Jr, Lersten NR (1972). Pollen wall and tapetal orbicular wall development in Sorghum bicolor (Gramineae). Amer J Bot 59: 43-58. [ Links ]
4. Clément C, Audran JC (1993a). Cytochemical and ultrastructural evolution of orbicules in Lilium. Pl Syst Evol (Suppl.) 7: 63- 74. [ Links ]
5. Clément C, Audran JC (1993b). Orbicule wall surface characteristics in Lilium (Liliaceae). An ultrastructural and cytochemical approach. Grana 32: 348-353. [ Links ]
6. Chen ZK, Wang FH, Zhou F (1988). On the origin, development and ultrastructure of the orbicules and pollenkit in the tapetum of Anemarrhena asphodeloides (Liliaceae). Grana 27: 273-282. [ Links ]
7. D'Ambrogio A (1986). Manual de Técnicas en Histología Vegetal. Ed. Hemisferio Sur S.A. Buenos Aires. Argentina. pp. 1- 83. [ Links ]
8. Echlin P (1971). The role of the tapetum during microsporogenesis of angiosperms. In: J. Heslop-Harrison (ed.), Pollen development and physiology. Butterworths, London. pp. 41-61. [ Links ]
9. EL-Ghazaly G, Nilsson S (1991). Development of tapetum and orbicules of Catharanthus roseus (Apocynaceae). In: S. Blackmore & S.H.Barnes (eds.), Pollen and spores. Systematics Association Special Vol. 44. Clarendon Press, Oxford. Pp. 317-329. [ Links ]
10. Erdtman G (1952). Pollen morphology and plant taxonomy: Waltham Massachusset, USA. The Chronica Botanica. [ Links ]
11. Fisher DB, Jensen WA, Ashton ME (1968). Histochemical studies of pollen: storage pockets in the endoplasmic reticulum (ER). Histochemie 13: 169-182. [ Links ]
12. Galati BG, Rosenfeldt S (1998). The pollen development in Ceiba insignis (Kunth) Gibbs & Semir ex Chorisia speciosa St. Hil. (Bombacaceae). Phytomorphology 48(2): 121-129. [ Links ]
13. Galati BG, Strittmatter LI (1999). Correlation between pollen development and Ubisch bodies ontogeny in Jacaranda mimosifolia (Bignoniaceae). Beitr Biol Pflanz 71: 249-260. [ Links ]
14. Guth CJ, Weller SG (1986). Pollination, fertilization and ovule abortion in Oxalis magnifica. Am J Bot 73: 246-257. [ Links ]
15. Herich R, Lux A (1985). Lytic activity of Übisch bodies (orbicles). Cytologia 50: 563-569. [ Links ]
16. Herr JM Jr (1972). An extended investigation on the megagametophyte in Oxalis corniculata L. In: Murty YS; Johori BM; Mohan Ram HY;Varghese TM. Advances in plant morphology. Sarita Prkasham. Meerut (India). pp 92-101. [ Links ]
17. Heslop- Harrison J, Dickinson HG (1969). Time Relationships of Sporopollenin Synthesis Associated with Tapetum and Microspores in Lilium. Planta 84: 199-214. [ Links ]
18. Johri BM, Ambegaokar KB, Srivastava PS (1992). Comparative embryology of angiosperms. Vol. 1. Springer-Verlag. Berlín Heidelberg. [ Links ]
19. Noguchi T (1990). Consumption of lipid granules and formation of vacuoles in the pollen tube of Tradescantia reflexa. Protoplasma 156: 19-28. [ Links ]
20. O'Brien TP, McCully ME (1981). The study of plant structure. Principles and selected methods. Termarcarphi Pty. Ltd., Melbourne. Australia. [ Links ]
21. Pargney JC (1978). Étude ultrastructurale de la gamétogenèse mâle dans une espèce à floraison cléistogame: Oxalis corniculata, suivie de quelques considérations générales sur la cléistogamie. Can J Bot 56: 1262-1269. [ Links ]
22. Raghavan V (1997). Molecular Embryology of Flowering Plants. Cambridge. University Press. pp: 690. [ Links ]
23. Risueño MC, Giménez-Martín G, López-Sáez JF, García MIR (1969). Origin and development of sporopollenin bodies. Protoplasma 67: 361-374. [ Links ]
24. Rowley JR, Walles B (1987). Origin and structure of Ubisch bodies in Pinus Sylvestris. Acta Societatis Botanicorum Poloniae 56(2): 215-227. [ Links ]
25. Suárez-Cervera M, Seoane-Camba JA (1986). Ontogénese des grains de pollen de Lavandula dentata L. et évolution des cellules tapétales. Pollen et Spores 28: 5-28. [ Links ]
26. Steer MW (1977). Differentiation of the tapetum in Avena. I. The cell surface. J Cell Sci 25: 125-138. [ Links ]
27. Theunis CH, Conchie CA, Knox RB (1985). Three dimensional reconstruction of the generative cell and its wall connection in mature bicellular pollen of Rhododendron. Micron Mic Acta 16: 225-231. [ Links ]
28. Tiwari S (1994). An intermediate -voltage electron microscopic study of freezer -substituted generative cell in pear (Pyrus communis L.): features with relevance to cell- cell communication between the cells of a germinating pollen. Sex Plant Reprod 7: 177-186. [ Links ]
29. Ubisch G (1927). Zur Entewicklungsgeschichte der Antheren. Planta 3: 490-495. [ Links ]
30. Vijayaraghavan MR, Chaudhry B (1993). Structure and development of orbicules in the tapetum of Prosopis juliflora (Leguminosae, Mimosoideae). Phytomorphology 43: 41-48. [ Links ]
31. Wang Y-F, Chen S-H (2001). Pollen Flora of Yuenyang Lake Nature Preserve, Taiwan (II). Taiwania 46: 167-191. [ Links ]
Received on November 24, 2004.
Accepted on May 16, 2005.














