Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Biocell
versión impresa ISSN 0327-9545
Biocell v.29 n.3 Mendoza ago./dic. 2005
TNFa increases in vitro migration of human HPV18-positive SW756 cervical carcinoma cells
K. Hidalgo1, I. G. Rojas2, A. B. Penissi3, and M. I. Rudolph1
1Faculty of Biological Sciences Universidad de Concepción, Concepción, Chile.
2College of Dentistry, Universidad de Concepción, Concepción, Chile.
3IHEM-CONICET, Faculty of Medical Sciences, Universidad Nacional de Cuyo, Mendoza, Argentina.
Address correspondence to: Maria Isolde Rudolph G. Ph.D. Departmento de Farmacología, Facultad de Ciencias Biológicas, Universidad de Concepción. Casilla 160-C. Concepción, CHILE. FAX: (+56-41) 245975. E-mail: mrudolph@udec.cl
ABSTRACT: TNFa has been associated with both, tumor survival and apoptosis. This cytokine is also involved in promoting cell migration during wound healing and tumorigenesis. SW756 is a HPV18-positive cervical carcinoma cell line, which has been used to study different mechanisms of cervical cancer progression. An in vitro assay of scratch wound healing onto monolayers of SW756 cells was used to assess the effect of TNFa on cell migration into a wound space. It was found that SW756 cells have the ability to migrate, but not proliferate in response to scratch wounding in a serum-free medium supplemented with TNFa. RT-PCR analysis showed that SW756 cells express TNFa mRNA when incubated in medium with and without serum. Wound closure and migration rate of SW756 cells were significantly increased in the presence of serum-free media supplemented with TNFa (10 ng/mL) as compared to serum-free media, and media supplemented with either anti-TNFa antibody or both TNFa and anti-TNFa antibody (p<0.05). The results showed a stimulatory effect of TNFa on the migration of SW756 cervical carcinoma cells, suggesting a novel and important role for TNFa in cervical cancer progression.
Keywords: TNFa. Migration. Carcinoma. SW756. Wound healing assay.
Introduction
Tumor cell invasion of connective tissue and blood vessels is one of the key steps in the metastatic spread of cancer. Evidence for the role of TNFa in increased cell migration has been demonstrated in vitro in cultured keratinocytes, and in vivo in murine tumor promotion models (Scott et al., 2004). TNFa has also been identified as a chemotactic factor for inflammatory cells involved in metastasis, such as mast cells (Olsson et al., 2003). In cervical cancer, little is known of the role of TNFa in migration of transformed cells.
Epidemiological studies have demonstrated a causal relationship between chronic infection of the uterine cervix by human papillomavirus (HPV) and cervical neoplasia (De Boer et al., 2004). HPVs are a family of DNA viruses with over 150 genotypes. More than 40 of these genotypes infect the anogenital tract, causing a variety of abnormalities ranging from genital warts to invasive cancer. Certain types are considered more carcinogenic in humans, such as HPV16 and HPV18, which are associated with adenocarcinomas and squamous cell carcinomas of the cervix, respectively (Woodman et al., 2003). Recent genetic studies support the hypothesis that constitutionally determined TNFa polymorphisms might influence response to high risk HPV infection, and therefore, susceptibility to cervical neoplasia (Kirkpatrick et al., 2004).
SW756 is the code for an HPV18-positive cervical carcinoma cell line, which is used to study the different mechanisms of cervical cancer progression. It has been shown that SW756 cells do not synthesize b-interferon, an antiangiogenic and antimitotic cytokine (Berger and Hawley, 1997), in response to TNFa stimulation (Bachmann et al., 2002). However, this cell line is believed it might maintain its ability to synthesize TNFa through NK-κB activation (Altenburg et al., 1999), probably through TNFR1 (Park et al., 2003) or CD40R, both members of the TNF receptor family whose activation induces NK-κB translocation (Berverich et al., 1994). The SW756 cell line also produces high levels of IL-6, which in turn stimulates monocyte chemoattractant protein- 1 (MCP-1) production, however, due to the lack of sgp80, a key protein in the IL-6 signaling pathway, they are unresponsive to IL-6 stimulation (Hess et al., 2000; Smola-Hess et al., 2001).
In vitro wound healing assays have been used with multiple cell types and are a classic and commonly used method for studying cell migration and the biology underlying it. In this study we developed an in vitro assay of scratch wound healing to assess if TNFa had a significant effect on SW756 migration.
Materials and Methods
Cells, reagents and antibodies
The human cervical carcinoma SW 756 cell line was purchased from American Type Culture Collection (ATCC) (Rockville, MD, USA). Reagents were purchased from Sigma (St. Louis, MO, USA). Recombinant human soluble TNFa was purchased from Boehringer Manheim Biochemica (Manheim, Germany). Anti-TNFa Ab neutralizing rhTNFa (Clone B-C7) was purchased from Biosource International (Camarillo, CA, USA). Polyclonal rabbit antihuman Ki67 antibody and peroxidase- conjugated goat antirabbit IgG secondary antibody were obtained from Dako (Carpinteria, CA, USA). The primers for glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and TNFa were synthesized by Bios Chile (Santiago, Chile). Oligo-dT primer, dNTP mix, ribonuclease inhibitor RNasin and 15 U of AMV reverse transcriptase (AMV-RT) were purchased from Promega (Madison, WI, USA). Taq DNA polymerase, trizol and all culture media were was purchased from Gibco BRL (Gaithersburg, MD, USA). 3-3‘diaminobenzidine (DAB) was obtained from Chemicon International, Inc (Temecula, CA, USA).
Wound healing assay
Culture conditions were optimized to ensure that cell monolayers were homogeneous maintaining the cell viability prior to wounding. In order to reduce cell proliferation for the experiments, cell cultures were grown to confluency and deprived of serum, as previously described (Geimer and Bade, 1991; Hinz et al., 1999; Wang et al., 2002; Fumoto et al., 2003). Some incubation conditions were modified for optimization of the experimental model as described below. The objective was to obtain a reproducible measurement of the migration of the wound edge towards the wound space with minimum cell proliferation but without loss of cell viability over the experimental time period.
SW756 cells were grown at 37ºC under humidified atmosphere of 5% CO2 and 95% O2, in L-15 Leivobitz, supplemented with 10% fetal bovine serum and 100 U/ mL Penicillin and 100 μg/mL Streptomycin according to ATCC indications. For migration analysis cells were detached by trypsinization, resuspended in the same medium and plated in triplicate at 3x105 cells per well of a 24-well plate (area 7 cm2). After cell became confluent cells were starved in a RPMI serum free medium for 18 hrs, then one artificial wound per well was scratched into the monolayers with a sterile plastic 10 μL micropipette tip to generate a uniform wound that was devoid of adherent cells. After wounding, tissue culture medium was removed, and cells were washed at least twice in PBS to eliminate detached cells. Experiments were performed by incubating the cells with RPMI only (control) or supplemented with a) a saturating concentration of 10 ng/mL rhTNFa (Bechtel et al., 1996), b) 10 μg/mL anti- TNFa Ab, or c) a combination of both, to evaluate the effect of TNFa on cell migration into the wound space. Wound closure was monitored by digital photographs taken in a phase-contrast microscope (Nikon Diaphot 300, Tokyo, Japan) across the wound at the moment of wounding and at 24 hrs post-wounding. Micrographs were studied by videomicroscopy and image analysis.
Videomicroscopy and image analysis
The system consisted of a light microscope with phase-contrast illumination (Nikon 300, Japan) fitted with a TV camera (CoolSNAP-Pro, Media Cybernetics, MD. USA) and interfaced with a host computer, image processing (Image-Pro Express software, Media Cybernetics, MD. USA), and recording systems. The light microscope images were entered into the camera through a 4x objective and transferred to a color monitor. Before measuring, distance calibration was defined using a slide with a micrometric scale for microscopy (Rechert, Austria). Distances between the edges were measured by using the image analysis software at 50 μm intervals all along the wound. A minimum of 120readings were made on each well. Quantitative wound closure assays were performed in triplicate wells andeach experiment was repeated at least three times. This system provided sufficient resolution to determine the magnitude of wound closure and cell morphology at the wound margins. The results of the three experiments were pooled for statistical analysis.
RNA isolation, reverse transcription and polymerase chain reaction (PCR)
Lysis of SW756 cells was performed with TRIzol reagent, and total RNA was extracted according to the manufacturer's protocol. A solution containing 20 μg of total RNA, oligo-dT primer, dNTP mix, ribonuclease inhibitor RNasin, and 15 U of AMV reverse transcriptase (AMV-RT) in reaction buffer (250 mM Tris-HCl, pH 8.3; 250 mM KCl; 50 mM MgCl2; 50 mM DTT; 2.5 mM spermidine) was incubated at 42°C for 60 min. The solution was heated to 90°C for 5 min to inactivate AMVRT and then cooled at 4°C.
The following primer sets described by Baram et al. (2001) were synthesized: GAPDH sense 5´- CGGAGTCAACGGATTTGGTCGTAT, GAPDH antisense 5´-AGCCTTCTCCATGGTTGGTGAAGAC; and TNF-a sense 5´-CTGTACCTCATCTACTCC CAGGTC, TNF-a antisense 5´-AGACTCGGCAAAGTCGAGATAGT. The PCR was performed in a 25 μl reaction mixture (10 mM Tris-HCl, 50 mM KCl, 3 mM MgCl2, 0.2 mM of each dNTP, 0.1-1.4 μM of 5' and 3' primers of each gene, and 1 unit of Taq DNA polymerase. cDNA was amplified for 35 cycles (Amplitron II, Barnstead/ Thermolyne Corporation, Dubuque, IA, USA). Each cycle consisted of denaturation at 94°C for 45 sec, annealing at 55°C for 30 sec, and primer extension at 72°C for 1 min. Before amplification, reactions were incubated at 94°C for 3 min. After amplification, extension was performed at 72°C for 10 min. Samples were electrophoresed, visualized in an ultraviolet transilluminator, photographed, and analyzed with NIH Image 1.61 (Bethesda, MD, USA).
Cell proliferation analysis
Cells were plated in 24-well plates in triplicate at a density of 3x105 cells per well in the tissue medium described above for SW756. Then they were incubated in the serum free mediun with RPMI for 18 hrs and treated with TNFa (10 ng/mL). Cell counts were performed in duplicate at 24 hrs using a hemacytometer.
SW756 cells were grown on glass coverslips in a 12-well plate. After the wound was made and the cells were treated with TNFa (10 ng/mL) for 24 hrs, cells were washed with 10 mM Tris-HCl, pH 7.8 (TBS), fixed with paraformaldehyde for 30 min and permeabilized with TBS containing 0.1% Triton X-100 for 10 min. Subsequently, the permeabilized cells were treated with 10% H2O2 for 25 min and blocked in TBS/Tween 20 supplemented with bovine serum albumin for 1 hr and then incubated with (1:200) polyclonal rabbit anti-human Ki67 Ab for 24 hrs at room temperature and humidified atmosphere. Cells were then washed and incubated with (1:100) peroxidase-conjugated goat anti-rabbit IgG secondary Ab for 1 hr at room temperature. The reaction was developed with 3-3´-diaminobenzidine (DAB) and 3 mL/mL H2O2 in 50mM Tris (pH=7.6).
Trypan blue assay
For evaluation of cell monolayer damage produced by the 10 μL micropipette tip scratch wound and culture cell viability, SW756 cells were treated with 0.4% Trypan blue and immediately observed in a phase-contrast microscopy (Nikon Diaphot 300, Tokyo, Japan) all across the wound area to determine trypan blue exclusion (Yasui et al., 2003).
Statistical analysis
All data were tabulated and statistical tests were performed with JMP-IN 4.0.4 SAS Institute Inc. (Cary, NC, USA). All data are represented as the mean ± SEM of three independent experiments performed in triplicate. Significant statistical differences between groups were examined using unpaired ´t´ test and one way ANOVA and Tukey Kramer tests. The non-parametric Wilcoxon and Kruskal-Wallis tests were used when variables did not have a normal distribution. Differences were considered statistically significant when p<0.05.
Results
Effects of incubation with serum-free media on SW756 cell culture
Human SW756 cervical carcinoma cells positive for HPV-18 grew as monolayers, adherent to the culture flasks. Their proliferation rate was dependant on the initial cell concentration in each well. Optimum confluent monolayer cultures were obtained with an initial concentration of 3x105 SW756 cells/mL in 24- well flasks after 72 hrs of culture. Cells were incubated with L-15 Leivobitz culture media supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL Streptomycin, at 37ºC and 5% CO2.
Replacement of culture media by a serum-free media resulted in proliferation inhibition with no alteration of cell viability, as confirmed by hemacytometer counting and trypan blue exclusion at 42 hrs of culture in serum-free media. The lapse of serum free media of 42 hrs corresponds to the 18 hrs before wounding and 24 hrs post-wounding, according to the following experiments. In fact, viability was maintained near 97% and the number of cell counts was similar at 0 and at 42 hrs after incubation with serum-free media. In addition, loss of the proliferation marker Ki67 expression was observed in confluent cells incubated with serum-free media and in confluent cells incubated with serum-free media supplemented with 10 ng/mL TNFa, as compared to SW756 cells growing in full media of L-15 Leivobitz, supplemented with 10% fetal bovine serum (Fig. 1).
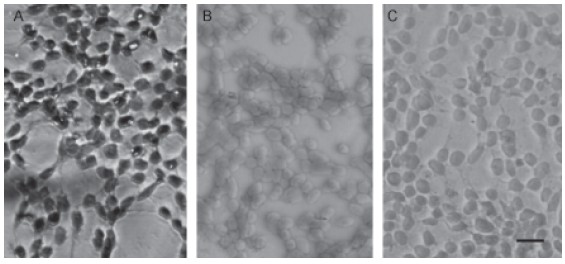
FIGURE 1. TNFa (10 ng/mL) does not promote expression of the proliferation marker Ki67 in SW756 cells incubated with serum-free medium. SW756 cells were grown on glass coverslips and Ki67 expression was detected by immunohistochemistry as described in Methods. Ki-67 protein is stained in black. A: Positive control: SW756 cells grown in L-15 Leivobitz medium, supplemented with 10% fetal bovine serum as described in methods. B: SW756 cells grown into confluence, incubated with a serum free medium for 18 hrs and then supplemented with 10 ng/ mL TNFa for 24 h. C: Negative control: obtained by omitting the first Ab. Scale bar: 10 μm.
SW756 cells express endogenous mRNA TNFa
To determine if SW756 express endogenous mRNA TNFa, and if this endogenous expression was affected by incubation with serum-free media, total RNA was extracted from SW756 cells incubated with serum supplemented media, and with serum-free media for 42 hrs, and subjected to RT-PCR to determine TNFa mRNA expression. The results show TNFa expression in SW756 cells after 42 hrs of serum starvation (Fig. 2).

FIGURE 2. SW756 cell expression of TNFa mRNA is independent of serum presence in the culture medium. SW756 cells were grown to confluency in L-15 Leivobitz medium, supplemented with 10% fetal bovine serum (0h) as described in Methods, incubated with a serum free medium for 42 hrs (24h). Total mRNA was isolated, and TNFa mRNA expression was detected by RT-PCR.
SW756 culture wounding
The size of the scratch wound created with a 10 μL micropipette tip ranged from 306 to 583 μm between wound edges. Damaged and non-viable cells were eliminated by PBS washing. Cell death as a result of scratch wounding was minimal and had no impact on cell viability at the wound edges as demonstrated by trypan blue exclusion. After 24 hrs of wounding a large amount of SW756 cells had already migrated into the wound space. Migrating cells exhibited an elongated morphology, with the longitudinal axis polarized toward the wound, which is a feature of well-organized migration. In addition, migrating cells presented similar size and morphology to growing cells before becoming confluent. In all the experiments the wound margin was observed rather diffuse due to active cell migration. At the wound edges uniform cell migration was observed. The same pattern was observed when TNFa was added to the medium (Fig. 3).

FIGURE 3. Effect of rhTNFa on SW756 cell migration. Representative phase-contrast images of cells migrating into the wounded area in an in vitro scratch wound healing assay. SW756 cells were grown into monolayers in triplicate in 24-well plates. Confluent cultures were starved for 18 hrs in RPMI, then scratched with a 10 mL pipette tip as described in the Methods section. Wound at time 0 (0h) for SW756 cells incubated with control, serum-free media (A) and with serum-free media supplemented with 10 ng/ml TNFa (C). (24h) 24hrs after wounding for SW756 cells incubated with control, serum-free media (B) and with control media supplemented with 10ng/ml TNFa (D). W= wound space, WE= Wound edge. Scale bar, 100 μm.
Effects of TNF a on wound closure and SW756 migration rate
SW756 cells, cultured as confluent monolayers in 24-well plates were wounded as described above, and incubated with serum-free media alone (control media), or serum-free media supplemented with either 10 ng/ mL of rhTNFa, 10 μg/mL of TNFa neutralizing antibody (anti-TNFa), or 10 ng/mL rhTNFa and 10 μg/ mL anti-TNF antibody (rhTNF-a/anti-TNFa).
First, changes in wound size (distance between wound edges) at 24 hrs post wounding were analyzed using the formula (wound size at 24 hrs/initial wound size) x 100. The results showed that addition of 10 ng/ mL rhTNFa significantly increased wound closure as compared to all the other treatment conditions, control media, and control media supplemented with anti-TNFa and TNFa/anti-TNFa (p<0.05 ANOVA and Tukey Kramer tests). However, a significant increase in wound closure was also observed in the cells treated with media supplemented with rhTNF-a/anti-TNFa antibody, as compared to cells treated with control media or control media supplemented with anti-TNFa antibody (p<0.05) (Fig. 4).
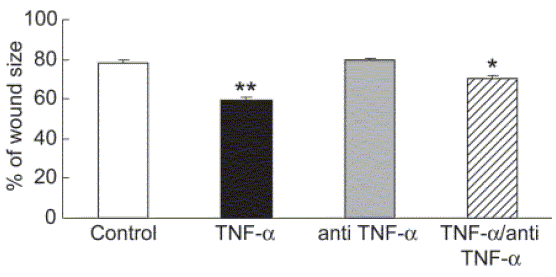
FIGURE 4. Effects of TNFa on wound closure of SW756 cells. Wound closure was monitored by digital photographs taken in a Nikon phase-contrast microscope across the wound at the moment of wounding and at 24 hrs after wounding. Wound closure was assessed as the distance between the wound edges, calculated and quantified using an Image Pro-Express software 4.01. The percentage of wound closure was evaluated using the formula (wound size at 24 hrs/ initial wound size) x 100. Data are shown as mean ± SEM from 3 independent experiments done in triplicate.
** P<0.05 increased wound closure for TNFa incubated cells as compared to control, antiTNFa, and antiTNF/TNFa (Anova and Tukey Kramer tests).
*P<0.05 increased wound closure for TNFa/antiTNFa as compared to control and antiTNFa, and reduced wound closure as compared to TNFa. (Anova and Tukey Kramer tests)
In order to assess the effects of TNFa on SW756 cell migration, the rate of cell migration was determined at 24 hrs using the formula: º (initial wound size - wound size at 24 hrs)/24. Between the time of wounding and 24 hrs after wounding, cells treated with 10 ng/mL rhTNFa supplemented media presented significantly higher migration rate as compared to cells treated with control media, control media with 10 μg/mL anti-TNFa, and control media with TNFa/anti-TNFa (p<0.05, Wilcoxon and Kruskal Wallis tests) (Fig. 5).
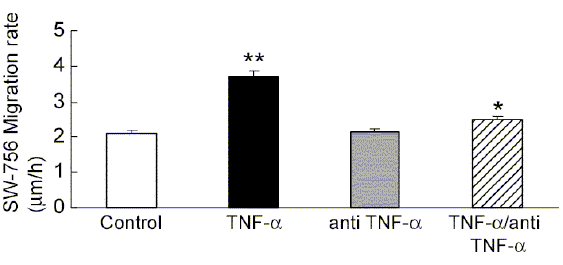
FIGURE 5. TNFa increases the migration rate of SW756 cells. The rate of migration (μm/ h) of SW756 cells into the wound space was evaluated as half the distance difference between wound edges at 0 and 24 h, divided by 24. The distance between the edges was monitored by digital photographs taken in a phase-contrast microscope across the wound at the moment of wounding and at 24 hrs post-wounding. Images were analysed using an Image Pro-Express software 4.01. Results are the mean ± SEM of three independent experiments performed in triplicate.
*P<0.001 for TNFa as compared to control, antiTNFa, and anti-TNF/TNFa (Wilcoxon and Kruskal Wallis).
** P<0.02 for TNFa/antiTNFa as compared to control (Wilcoxon and Kruskal Wallis).
Discussion
Initially, TNFa was thought as a potentially effective anticancer therapeutic agent, due to its ability to kill some tumor cell lines in vitro, as well as, certain types of tumors in vivo (Haranata et al., 1984; Palladino et al., 1987; Creasey et al., 1986; Hagari et al., 1995). However, in the recent years new biological activities of TNFa, which could promote tumor growth and invasion, were identified. TNFa, a key mediator of inflammation (Locksley et al., 2001), acted as a mitogenic growth factor for human follicular dendritic cell lines (Park et al., 2003), and in cervical fibroblasts of human uterus stimulated MMP-9 production, an enzyme with highly invasive activity and metastatic potential (Sato et al., 1996; Hofmann et al., 2000; Scott et al., 2004). In addition, TNFa acted on the endothelium stimulating angiogenesis (Norrby, 1996; Secchiero et al., 2004) and leukocyte recruitment (Walsh et al., 1991).
In vitro wound healing assays are useful for the study of the mechanisms involved in cell migration in multiple cell types (Lampugnani, 1999; Huang et al., 2003; Yarrow et al., 2004). Although it seems a simple assay, experimental conditions differ significantly depending on the type of cell line and the parameters to be assessed. The scratch wound healing assay used in these studies had the purpose of analyzing the role of TNFa on SW756 cell migration. It was found that SW756 carcinoma cell line, positive for HPV-18, in a serum-free environment, migrated in response to a scratch wound and was able to express endogenous TNFa mRNA. When exogenous TNFa was added to the serum-free media, this migration ability was significantly increased as measured at 24h after wounding. This TNFa effect on migration was partially, but significantly inhibited by the addition of an anti-TNFa antibody to the serum-free media. In addition, SW756 migration into the wound space, in the absence of exogenous TNFa, seemed to be independent of the endogenous TNFa produced by these cells, since addition of anti-TNFa to the serum free media did not affect cell migration. Therefore, other growth factors or cytokines produced in response to wounding could be involved in stimulating SW756 cell migration, which should be further analyzed.
When wounded or scratched, cell monolayers respond to the disruption of their cell to cell contacts with either apoptosis or a combination of proliferation and migration. This seems to depend on cell line sensitivity and the concentration of growth factors and cytokines at the wound margin (Firth and Putnins, 2004; Coomber and Gotlich, 1990; Zahm et al., 1997). Apoptosis in response to a scratch wound was reported in an epidermal keratinocyte cell line, and this effect was exacerbated by the presence of TNFa (Firth and Putnins, 2004). As shown in these results, in cultures with serum-deprived media, SW756 cells did not respond to the scratch wound with apoptosis, as confirmed by trypan blue exclusion assay, but with unidirectional and synchronized migration towards the empty wound space. This agrees with previous reports in which human ovarian (SK-OV- 3 and CaOV-3) and cervical (SiHa and HT-3) carcinoma cell lines were resistant to the cytolytic action of TNFa (Powell et al., 1998). Furthermore, in more recent studies it was shown that human gastric MKN45 and cervical HeLa carcinoma cells could become susceptible to the apoptotic effects of TNFa by a combination of TNFa and Sulindac, an inhibitor of NF-κB (Yasui et al., 2003).
Stimulation of cell migration in an in vitro wound healing assay is complex, and requires the coordination of numerous events. Several endogenous, as well as, exogenous factors have been implicated in cell migration. Reorganization of the actin cytoskeleton (Lauffenburger and Horwitz, 1996) as well as activation of myosin II chain play important roles in changes of cell polarity and directionality during migration (Fumoto et al., 2003). The mechanisms by which TNFa could mediate cell migration have not been elucidated. TNFa activation of the transcription factor NF-κB seems to be critical for smooth muscle cell migration (Wang et al., 2001) and also for its mitogenic effect (Peppel et al., 2005). However, TNFa induced mitogenesis requires co-stimulation with other growth factors (Peppel et al., 2005). Therefore, in a cell culture exposed to serumdeprived media, TNFa could induce migration, but not cell proliferation due to the lack of additional growth factors present in the serum. This was confirmed in these studies, where serum-starvation of SW756 cultures resulted in inhibition of cell proliferation without loss of the ability of the cells to migrate in response to a scratch wound.
The present study showed a stimulatory effect of exogenous TNFa on migration of the cervical carcinoma cell line SW756. Since carcinomas, an in particular cervical carcinoma, are infiltrated by high amounts of stromal cells with the ability to secrete TNFa, such as mast cells and macrophages (Rojas et al., 2005; Cabanillas et al., 2002; Coussens and Werb, 2001), increased TNFa production at the tumor stroma could promote cancer cell migration, and therefore tumor invasion and metastasis.
Acknowledgements
This work was supported by research grant from FONDECYT (1020458).
References
1. Altenburg A, Baldus SE, Smola H, Pfister H, Hess S (1999). CD40 Ligand-CD40 interaction induces chemokines in cervical carcinoma cells in synergism with IFN-g. J Immunol. 162: 4140- 4147. [ Links ]
2. Bachmann A, Hanke B, Zawatzky R, Soto U, van Riggelen J, zur Hausen H, Rösl F (2002). Disturbance of tumor necrosis factor alpha-mediated beta interferon signaling in cervical carcinoma cells. J Virol. 76: 280-291. [ Links ]
3. Baram D, Vaday GG, Salamon P, Drucker I, Hershkovitz R, Mekori YA (2001). Human mast cells release metalloproteinase-9 on contact with activated T cells: Juxtacrine regulation by TNFa. J Immunol. 167: 4008-4016. [ Links ]
4. Bechtel MJ, Reinartz J, Rox JM, Inndorf S, Schaefer BM, Kramer MD (1996). Upregulation of cell-surface-associated plasminogen activation in cultured keratinocytes by interleukin-1 beta and tumor necrosis factor-alpha. Exp Cell Res. 223: 395-404. [ Links ]
5. Berger LC, Hawley RG (1997). Interferon-beta interrupts interleukin-6-dependent signaling events in myeloma cells. Blood. 89: 261-71. [ Links ]
6. Berverich I, Shu GL, Clark FA (1994). Cross-linking CD40 on B cells rapidly activates nuclear factor-κB. J Immunol. 153: 4357. [ Links ]
7. Cabanillas A, Schalper JA, Nicovani SM, Rudolph MI (2002). Characterization of mast cells according to their content of tryptase and chymase in normal and neoplastic human uterine cervix. Int J Gynecol Cancer. 12: 92-98. [ Links ]
8. Coomber BL, Gotlich AI (1990). In vitro endothelial wound repair. Interaction of cell migration and proliferation. Arteriosclerosis 10: 215-222. [ Links ]
9. Coussens LM, Werb Z (2001). Inflammatory Cells and Cancer: Think Different!. J Exp Med 193: F23-F26. [ Links ]
10. Creasey AA, Reynolds MT, Laird W (1986). Cures and partial regression of murine and human tumors by recombinant human tumor necrosis factor. Cancer Res. 46: 5687-5690. [ Links ]
11. De Boer MA, Peters LA, Aziz MF, Siregar B, Cornain S, Vrede MA, Jordanota ES, Kolkman-Uljee S, Fleuren GJ (2004). Human papillomavirus type 16 E6, E7 and L1 variants in cervical cancer in Indonesia, Suriname and The Netherlands. Gynecol Oncol. 94: 488-494. [ Links ]
12. Firth JD, Putnins EE (2004). Keratinocyte growth factor 1 inhibits wound edge epithelial cell apoptosis in vitro. J Invest Dermatol. 122: 222-231. [ Links ]
13. Fumoto K, Uchimura T, Iwasaki T, Ueda K, Hosoya H (2003). Phosphorylation of myosin II regulatory light Caín is necessary for migration of Hela cells but not for localization of myosin II at the leading edge. Biochem J. 370: 551-556. [ Links ]
14. Geimer P, Bade EG (1991). The epidermal growth factor-induced migration of rat liver epithelial cells is associated with transcient inhibition of DNA synthesis. J Cell Sci. 100: 349- 355. [ Links ]
15. Hagari Y, Budgeon LR, Picket MD, Kreider JW (1995). Association of tumor necrosis factor-a gene expression and apoptotic cell death with regression of Shope papillomas. J Investig Dermatol. 104: 526-529. [ Links ]
16. Haranata K, Satomi N, Sakurai A (1984). Antitumor activity of murine tumor necrosis factor against transplanted murine tumors and heterotrasplanted human tumors in nude mice. Acta Med Scand. 29: 274-277. [ Links ]
17. Hess S, Smola H, Sandarabura de Silva U, Hadaschick D, Kube D, Baldus SE, Flucke U, Pfister H (2000). Loss of IL-6 receptor expresion in cervical carcinoma cells inhibits autocrine IL-6 stimulation: abrogation of constitutive monocyte chemoattractant protein-1 production. J Immunol. 165: 1939-1948. [ Links ]
18. Hinz B, Alt W, Johnen C, Herzog V, Kaiser H-W (1999). Quantifying lamella dynamics of cultured cells by SACED, a new computer- assisted motion analysis. Exper Cell Res 251: 234-243. [ Links ]
19. Hofmann UB, Westphal JR, van Muijen GN, Ruiter DJ (2000). Matrix metalloproteinases in human melanoma. J Invest Dermatol. 115: 337-244. [ Links ]
20. Huang C, Rajfur Z, Borchers C, Schaller MD, Jacobson K (2003). JNK phosphorylates paxillin and regulates cell migration. Nature 424: 219-223. [ Links ]
21. Kirkpatrik A, Bidwell J, van den Brule AJC, Meijer CJLM, Pawade J, Glew S (2004). TNF-a polymorphism frequencies in HPVassociated cervical displasia. Gynecol Oncol. 92: 675-679. [ Links ]
22. Lampugnani MG (1999). Cell migration into a wound area in vitro. Methods Mol Biol. 96: 177-182. [ Links ]
23. Lauffenburger DA, Horwitz AF (1996). Cell migration: a physically integrated molecular process. Cell 84: 359-369. [ Links ]
24. Locksley RM, Killen N, Lenardo MJ (2001). The TNF and TNF receptor superfamilies integrating mammalian biology. Cell 104: 486-501. [ Links ]
25. Norrby K (1996). TNF-alpha and de novo mammalian angiogenesis. Microvasc Res. 52: 79-83. [ Links ]
26. Olsson N, Taub DD, Nilsson G (2003). Regulation of mast cell migration by Th1 and Th2 cytokines: Identification of tumor necrosis factor-a and interleukin-4 as mast cell chemotaxins. Scand J Immunol. 59: 267-272. [ Links ]
27. Palladino MA, Refaat Shalaby M, Dramer SM, Ferraiolo BL, Baughman RA, Deleo AB, Crase D, marafino B, Aggarwal BB, Figari IS (1987). Characterization of the antitumor activities of human tumor necrosis factor alpha and the comparison with other cytokines: induction of tumor-specific immunity. J Immunol. 138: 4023-4032. [ Links ]
28. Park S-M, Park H-Y, Lee TH (2003). Functional effect of TNF-a on a human follicular dendritic cell line: persistent NK-kB activation and sensitization for Fas-mediated apoptosis. J Immunol. 171: 3955-3962. [ Links ]
29. Peppel K, Zhang L, Orman ES, Hagen P-O, Amalfitano A, Brian L, Freedman NJ (2005). Activation of vascular smooth muscle cells by TNF and PDGF; overlapping and complementary signa transduction mechanisms. Cardiovasc Res. 65: 674-682. [ Links ]
30. Powell CB, Scott JH, Collins JL (1998). Comparison of TNFa and TNFb cytolytic mechanisms in human ovarian and cervical carcinoma cell lines. Gynecol Oncol. 71: 258-265. [ Links ]
31. Rojas IG, Spencer ML, Martínez A, Maurelia MA, Rudolph MI (2005). Characterization of Mast cell Subpopulations in Lip Cancer. J Oral Pathol Med. 34: 268-273. [ Links ]
32. Sato T, Ito A, Ogata Y, Nagase H, Mori Y (1996). Tumor necrosis factor alpha induces pro-matrix metalloproteinase 9 production in human uterine cervical fibroblasts but interleukin 1 alpha antagonizes the inductive effect of TNFa. FEBS Lett. 392: 175-178. [ Links ]
33. Scott KA, Arnott CH, Robinson SC, Moore RJ, Thompson RG, Marshall JF, Balkwill FR (2004). TNF-a regulates epithelial expression of MMP-9 and integrin avb6 during tumour promotion. A role for TNF-a in keratinocyte migration?. Oncogene. 23: 6954-6966. [ Links ]
34. Secchiero P, Gonelli A, Carnevalle E, Corallini F, Rizzardi C, Zacchigna S, Melato M, Zauli G (2004). Evidence for a proangiogenic activity of TNF-related apoptosis-inducing ligand. Neoplasia 6: 364-373. [ Links ]
35. Siedle B, Garcia-Pineres AJ, Murillo R, Schulte-Monting J, Castro V, Rungeler P, Klaas CA, Da Costa FB, Kisiel W, Merfort I (2004). Quantitative structure-activity relationship of sesquiterpene lactones as inhibitors of the transcription factor NK-κB. J Med Chem. 47: 6042-54. [ Links ]
36. Smola-Hess S, Sandaradura de Silva U, Hadaschik D, Pfister HJ (2001). Soluble interleukin-6 receptor activates the human papillomavirus type 18 long control region in SW756 cervical carcinoma cells in a STAT-3 dependent manner. J Gen Virol. 82: 2335-2339. [ Links ]
37. Walsh LJ, Trinchieri G, Waldorf HA, Whitaker D (1991). Human dermal mast cells contain and release tumor necrosis factor a, which induces endothelial leukocyte adhesion molecule I. Proc Natl Acad Sci USA 88: 4220-4224. [ Links ]
38. Wang Z, Castresana MR, Newman WH (2001). NF-KappaB is required for TNF-alpha-directed smooth muscle cell migration. FEBS Lett. 508: 360-364. [ Links ]
39. Wang A, Nomura M, Patan S, Ware JA (2002). Inhibition of protein kinase Ca prevents endothelial cell migration and vascular tube formation in vitro and myocardial neovascularization in vivo. Circ Res. 90: 609-616. [ Links ]
40. Woodman CB, Collins S, Rollason TP, Winter H, Bailey A, Yates M, Young LS (2003). Human papillomavirus type 18 and rapidly progressing cervical intraepithelial neoplasia. Lancet 361: 40-43. [ Links ]
41. Yarrow JC, Perlman ZE, Westwood NJ, Mitchison TJ (2004). A high-throughput cell migration assay using scratch wound healing, a comparison of image-based readout methods. BMC Biotechnol. 4: 21-30. [ Links ]
42. Yasui H, Adachi M, Imai K (2003). Combination of tumor necrosis factor-a with sulindac augments its apoptotic potential and suppresses tumor growth of human carcinoma cells in nude mice. Cancer 97: 1412-1420. [ Links ]
43. Zahm JM, Kaplan H, Herard AL, Dorior F, Pierrot D, Somelette P, Puchelle F (1997). Cell migration and proliferation during in vitro wound repair of the respiratory epithelium. Cell Motil Cytoskeleton. 37: 33-43. [ Links ]
Received on February 25, 2005.
Accepted on May 27, 2005.














