Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Biocell
Print version ISSN 0327-9545
Biocell vol.30 no.3 Mendoza Ago./Dec. 2006
MDCK cells express serotonin-regulable 11ß-hydroxysteroid dehydrogenase type 2
Marisa L. Zallocchi*1, Maria C. Damasco1, Juan C. Calvo1,2, Carlos P. Lantos1 and Laura B. Matkovic'1
1 Departamento de Química Biolígica, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires and PRHOM-CONICET. Buenos Aires, Argentina.
2 Instituto de Biología y Medicina Experimental (CONICET). Buenos Aires, Argentina.
*For the definition of the terms neurotransmitter and neurohormone, we adopt the terminology of David O. Norris in Vertebrate Endocrinology 3rd, Edition Academic Press. Neurotransmitter: Chemical regulators produced and secreted by neurons into synapses that produce effects on the postsynaptic and/or presynaptic cells. Neurohormones: Neural regulators that are released, like hormones, into the blood.
Address correspondence to: Dra. Laura Beatriz Matkovic'. Departamento de Química Biolígica, Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires. Pabellón II, Ciudad Universitaria. (1428) Buenos Aires, ARGENTINA. Fax: (+54-11) 4576 3342. E-mail: lmat@qb.fcen.uba.ar
ABSTRACT: Prior to this work, we found that adrenal as well as extra-adrenal factors activate the response of renal 11ß-hydroxysteroid dehydrogenase 2 to stressful situations. These results -showing ways through which the organism hinders the pathological occupation of mineralocorticoid receptors by glucocorticoids leading to sodium retention and hypertension- prompted the present study on the nature of the above-mentioned extraadrenal factors. Serotonin was chosen because of its properties as a widely distributed neurohormone, known to interact with glucocorticoids at many sites, also exhibiting increased levels and effects under stressful situations. We studied serotonin effects on 11b-hydroxysteroid dehydrogenase 2 activity in a cell line derived from distal nephron polarized-epithelium, employing 3H-corticosterone as substrate. The end-product, 3H-11-dehydrocorticosterone was separated from the substrate by HPLC and quantified. Serotonin stimulated 11ß-hydroxysteroid dehydrogenase 2 activity only at 2nM and 25pM, the magnitude of the response depending also on substrate concentration. The stimulation was blocked by the specific inhibitors methiothepin and ketanserin. We postulate that the organism partially prevents renal mineralocorticoid receptor occupancy by glucocorticoids, circulating at enhanced levels under stressful situations, through serotonin-mediated catabolic regulation of the 11ß- hydroxysteroid dehydrogenase 2 activity. Given many, mostly positive, interactions between both hormones, this might eventually pave the way to studies on a new regulatory axis.
Keywords: Serotonin; 11b-HSD2; Stress hormones; Mineralocorticoid; Glucocorticoid.
Introduction
It is now well established that renal 11ß- hydroxysteroid dehydrogenase type 2 (11ß-HSD2) is one of the factors protecting the mammalian organism against the pathological retention of sodium and water (Funder, 1997; Young and Funder, 2002). This is achieved through the selective catabolism of glucocorticoids, which are 11ß-hydroxysteroids, to inactive 11-ketosteriods (Edwards et al., 1988; Funder et al., 1988; Rusvai and Naray-Fejes-Toth, 1993; Zhou et al., 1995; Farman and Bocchi, 2000). This inactivation protects MR (mineralocorticoid receptors), for which both corticosteroid subfamilies have equal affinity, from the illegal occupation by glucocorticoids circulating at 100 to 1000 fold higher levels than aldosterone (Funder et al., 1988; Edwards et al., 1988; Farman and Bocchi, 2000). Thus, said inactivation confers tissue-specificity to mineralocorticoids.
Stress activates glucocorticoid biosynthesis and blood levels; these hormones, in turn, mediate the organism's protective response against noxious stimuli inherent to stress through a variety of anabolic and catabolic events known as the "pleiotropic" glucocorticoid effect (McEwen, 2003; Sapolsky et al., 2000; Yeager et al., 2004). Accordingly, an increase in circulating glucocorticoids coinciding with a decrease in their tissueconcentration at the site of MR is of homeostatic relevance (Funder et al., 1988; Edwards et al., 1988; Farman and Bocchi, 2000).
Recently, it has been shown that certain stressful situations, besides activating the hypothalamic-pituitaryadrenal (HPA) axis, increase renal 11 ß-HSD2 activity (Igarreta et al., 1999; Zallocchi et al., 2004a, b). Interestingly, some of the corresponding kinetic alterations of the isoenzyme could be found not only in intact, but also in adrenalectomized rats. Those stress-conditions were found to stimulate renal 11ß-dehydrogenase 2 (11ß-HSD2) activity not only through the activation of the HPA axis but also through extra-adrenal factors.
When we decided to investigate those new stressrelated factors, the choice of serotonin (5HT) was motivated by the following previous findings: a) the by now well studied interaction of this indolamine with corticoids in the brain and along the HPA axis, specially the modulation of 5HT receptor responsiveness to different concentrations of circulating corticosterone (Contesse et al., 2005; Hesen and Joels, 1996; Lefebvre et al., 2001; McKittrick and McEwen, 1996; Chaouloff, 2000; Laplante et al., 2002; Seckl, 1996; Jorgensen et al., 1999); b) the local release of 5HT during stress situations not only at neural levels but also in other tissues such as the adrenal gland (Lefebvre et al., 2001; Chaouloff, 2000; McKittrick and McEwen, 1996); c) the properties of 5HT as a widely distributed neurohormone*, in addition to those of a neurotransmitter (McKittrick and McEwen, 1996; Sanders-Bush and Mayer, 2001); and d) the synthesis and/or capture of 5HT by many tissues, including extra-adrenal ones, such as the nephron (Berndt et al., 2001; Gross et al., 2000; Wang et al., 2001).
For our investigations we chose a distal nephronderived dog-epithelial cell line (MDCK) (Gaush et al., 1966; Gstraunthaler et al., 1985) possessing most of the characteristics of the distal nephron, especially 5HT receptors (Schoeffter and Bobirnac, 1995) and ATPase units (Gstraunthaler et al., 1985; Oberleithner et al., 1990; Simmons, 1982), the latter functionally associated to MR in that renal structure (Oberleithner et al., 1990). Most important, MDCK cells expresses endogenous 11ß-dehydrogenase 2 but not its reductase activity (Korbmacher et al., 1989).
In the present study, therefore, we decided to test the hyphotesis that 5HT, acting at distal level, is a physiological modulator of renal 11 ß-HSD2. We showed that MDCK cells respond to 5HT with an increase in 11ß- HSD2 activity. Moreover, this stimulation depends not only on 5HT but also on glucocorticoid concentrations.
Together these data indicate that 5HT stimulates renal 11 ß -HSD2 activity through a paracrine mode of action.
Materials and Methods
The MDCK (Madin-Darby canine kidney) cell line was from ABAC (Asociaci.n Banco Argentino de C.lulas, INEVH, Pergamino, Buenos Aires, Argentina). All sterile cell culture disposable plasticware was from Renner GmbH, Germany. Liquid cell culture products were obtained from Life Technologies (Cergy-Pontoise, France). Fluorescent silica gel plates were from Merck- Darmstadt (Federal Republic of Germany). Tritiated corticosterone ([3H1,2,6,7]corticosterone, specific activity = 70 mCi/mmoles), was from New England Nuclear, Life Sciences Products, Boston, MA., USA. All other reagents were either from Merck-Darmstadt (Federal Republic of Germany), Sigma (St. Louis, MO., USA.) or Aldrich (Milwaukee, WI, USA).
Cell culture
MDCK cells were maintained in Dulbecco's Modified Eagle Medium (DMEM), supplemented with 10% fetal bovine serum, 100 IU penicillin/mL and 100 mg streptomycin/mL. The cells were subcultured twice a week at a split ratio of 1:3 with 0.25% trypsin. Three hours prior to experiments the medium was replaced by fresh serum-free medium.
Detection, characterization and quantitative determination of 11-dehydrocorticosterone as reaction product
A routine determination of 11Β-HSD2 activity consisted of incubating intact MDCK cells with [3H1,2,6,7]corticosterone during 75 minutes -a time interval corresponding to initial velocities (Figure 2A)- followed by extraction of the media with ethyl acetate and evaporation to dry. Extracts were resuspended in methanol and resolved by HPLC on a reverse phase C- 18 mBondapak (5 mm) 25 cm x 4.6 mm column, using a mobile phase of methanol:H2O (3:2), at a flow rate of 0.8 mL/min. Standards added after the incubation were UV-detected at 254 nm and integrated in a Spectra-Physics Instrument. Incubations consistently yielded two radioactive fractions associated to corticosterone and 11-dehydrocorticosterone.
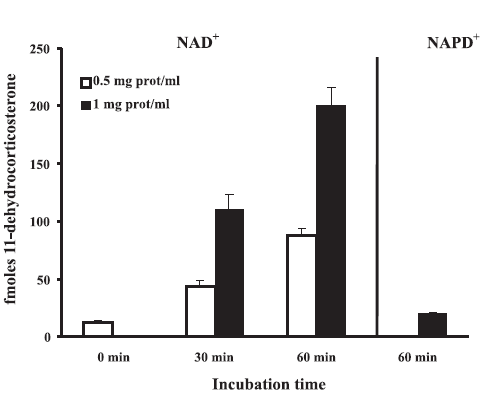
FIGURE 1. 11Β-HSD2 activity in homogenates from MDCK cell. 11Β-HSD activity was measured in the presence of 12 nM [3H1,2,6,7]corticosterone at 37ºC. For 11Β-HSD2 activity, 400![]() NAD+ was added to MDCK homogenates (0.5 mg protein/ ml or 1 mg protein/ml) and the samples were incubated for 0, 30 or 60 min. For 11Β-HSD1 activity, 400
NAD+ was added to MDCK homogenates (0.5 mg protein/ ml or 1 mg protein/ml) and the samples were incubated for 0, 30 or 60 min. For 11Β-HSD1 activity, 400![]() NADP+ was added to MDCK homogenates (1 mg protein/ml) and the samples were incubated for 60 min. Steroid conversion (indicating 11Β-HSD activity) was expressed as total fmoles of 11-dehydrocorticosterone. Values represent the mean
NADP+ was added to MDCK homogenates (1 mg protein/ml) and the samples were incubated for 60 min. Steroid conversion (indicating 11Β-HSD activity) was expressed as total fmoles of 11-dehydrocorticosterone. Values represent the mean ![]() SE of three independent experiments.
SE of three independent experiments.

FIGURE 2A. Time course studies. Intact cells were incubated with 10 nM [3H1,2,6,7]corticosterone between 0 to 110 min. Values represent the mean ![]() SE of three independent experiments. 11Β-HSD2 activity was expressed as pmoles of 11-dehydrocorticosterone per mg of protein.
SE of three independent experiments. 11Β-HSD2 activity was expressed as pmoles of 11-dehydrocorticosterone per mg of protein.
Following this protocol, the possibilities of less polar radiometabolites of 3H-corticosterone are limited to 11-dehydrocorticosterone, 5a-dihydrocorticosterone and 5Β-dihydrocorticosterone. Of these, 5α-dihydrocorticosterone had been detected and characterized by mass spectrometry in addition to 11-dehydrocorticosterone and polar metabolites (Hoyer et al., 1984). To exclude an eventual mixture of the three radio-metabolites in the 11-dehydrocorticosterone fraction, an experiment was carried out in which media-extracts from cell-incubations were divided into two aliquots: one aliquot was resolved on HPLC and the other one by TLC (Igarreta et al., 1999). Standards of radioinert corticosterone, 11-dehydrocorticosterone, 5a-dihydro-corticosterone and 5Β-dihydrocorticosterone were run in parallel. The first two standards were detected with UV light, and the two reduced standards by spraying acidified ammonium molybdate. Both reduced standards migrated nearer to corticosterone than did 11-dehydrocorticosterone, the 5a isomer was slightly more polar than the 5Β isomer.
Characterization of 11β-hydroxysteroid dehydrogenase activity in MDCK cells.
Cell homogenates. 11Β-HSD activity in MDCK homogenates was measured according to Leckie et al. (1995) with minor modifications. Briefly, samples were diluted to the appropriate final protein concentration (0.5 or 1 mg/mL) and 11Β-HSD activity was determined in the presence of 400m NAD+ (11Β-HSD2) or NADP+ (11Β-HSD1) and 12 nM [3H1,2,6,7]corticosterone, at 37ºC for 0, 30 or 60 min (11Β-HSD2) or 60 min (11Β-HSD1). Protein concentration was determined by the method of Bradford (1976). Reactions were terminated by addition of ethyl acetate containing excess of unlabelled corticosterone and 11-dehydrocorticosterone as standards. The steroids were then separated by TLC according to Igarreta et al. (1999). 11Β-HSD activity was expressed as fmoles of 11-dehydrocorticosterone per total protein and per total incubation time.
Cell suspensions. Subconfluent cell monolayers were trypsinized, centrifuged and resuspended in fresh serum-free medium. Intact cells were incubated in 24- well plates (4 x 106 cells per well) at 37ºC in 500 mL of DMEM containing [3H1,2,6,7]corticosterone and 8% glycerol. The reaction was stopped by centrifugation and addition of 1 mL ethyl acetate containing 0.25 mg/mL of unlabelled corticosterone and 11-dehydrocorticosterone to the supernatant. Steroids present in the organic layer were separated by HPLC and analyzed by counting the radioactivity associated to the UV-absorbent standards in a scintillation counter.
The amount of 11-dehydrocorticosterone generated per well was standardized for the amount of protein, determined by the Bradford method (1976).
For time-course studies, cell suspensions were incubated with 10 nM [3H1,2,6,7]corticosterone for 0 to 110 minutes. Kinetic parameters were determined using an initial range of corticosterone concentrations between 1 and 40 nM, with no addition of unlabeled corticosterone for concentrations below 10 nM and, for higher concentrations, 10 nM of the tritiated form plus the necessary unlabeled steroid. The KM and maximum velocity (Vmax) were established using Lineweaver-Burk plots (data points were fitted by a least square fit program). Inhibition studies were carried out incubating cell suspensions in the presence of 10-6-10-8 M carbenoxolone (CBX), with 10 nM [3H1,2,6,7]corticosterone for 75 min.
Serotonin stimulation
Cell suspensions were incubated in 24-well plates (4 x 106 cells per well), during 75 min at 37ºC with 10 nM [3H1,2,6,7]corticosterone and different concentrations of 5HT (10 pM to 10 nM). This range of 5HT concentrations was chosen taking into account previous reports on serotonin released in the central nervous system and in the adrenal cortex (Dinan, 1996; Berndt et al., 2001; Lefebvre et al., 2001).
Specificity of 5HT action was determined with methiothepin and ketanserin (Schoeffter and Bobirnac, 1995). Cells were preincubated with 1 mM of each antagonist during 15 min and then incubated for 75 min in the presence of 2 nM or 25 pM 5HT.
Statistical analysis
Values were presented as means + SE. Differences between multiple groups were evaluated by ANOVA followed by Dunnett's test or by Tukey-Kramer's test, as corresponded. Statistical significance was set at p<0.05.
Results
Characterization of 11β-HSD2 activity in MDCK cells
Homogenates were only used for cofactor dependency according to Leckie et al. (1995), at a protein concentration of 0.5 mg/mL and 1 mg/mL over a period of 30 or 60 min (Fig. 1). Results obtained show a clear cofactor preference for NAD+ over NADP+. As expected, there was no 11Β-HSD activity when MDCK homogenates were incubated in the absence of NAD+ or NADP+ (data not shown).
In spite of dogs being cortisol secreters, we used corticosterone as substrate. This glucocorticoid had been employed repeatedly as substrate in many distal nephron cell lines stemming from mammals with 17- hydroylating adrenals. This criterion had been adopted by previous authors because 11Β-HSD2 has a greater affinity for corticosterone than cortisol even in species in which cortisol is the major glucocorticoid such as pigs, sheep and even humans (reviewed in Leckie et al.). Our KM value of around 13 found for the isoenzyme coincides with values reported by former authors under similar experimental conditions (Leckie et al., 1995; Agarwal, 2000; Odermatt et al., 2001).
Indeed, our kinetic studies using intact cells showed a KM for corticosterone of 12.8 + 0.8 nM and a V max of 11.4 + 0.9 fmoles/mg x min (Figs. 2B and 2C). Carbenoxolone inhibited 11Β-HSD2 activity in intact cells with an IC50 of approximately 0.5 x 10-7 M (Fig. 2D).

FIGURE 2B, 2C. Kinetic studies. Intact MDCK cells were incubated with 1 to 40 nM corticosterone at 37ºC during 75 min. Kinetic parameters were calculated according to Lineweaver-Burk linear transformation. Values represent the mean ![]() SE of four independent experiments.
SE of four independent experiments.

FIGURE 2D. Inhibition of 11Β-HSD2 activity. Intact MDCK cells were incubated in the presence of increasing concentrations of CBX during 75 min with 10 nM [3H1,2,6,7]corticosterone at 37ºC. 11Β-HSD2 activity was expressed as pmoles of 11-dehydrocorticosterone per mg of protein during 75 min. Values represent mean ![]() SE of three independent experiments. CBX: carbenoxolone.
SE of three independent experiments. CBX: carbenoxolone.
Together NAD+ dependence, a nanomolar KM for corticosterone, drug-inhibition and the univocal characterization of the end-product (see Materials and Methods) along with the distal origin of the cell line (Gaush et al., 1966; Gstraunthaler et al., 1985), these results confirm that the activity in MDCK cells is due to the 11Β-HSD2 isoform.
Effect of serotonin on 11β-HSD2 activity
The effect of 5HT was assayed at 10 nM corticosterone and at 5HT concentrations between 10 pM and 10 nM. Clearly, the response to this activator showed two peaks, one at 25 pM (20 pM to 30 pM) and another one at 2 nM (Fig. 3).

FIGURE 3. Effect of serotonin on 11Β-HSD2 activity. Intact MDCK cells were incubated with 10 nM [3H1,2,6,7]corticosterone at 37ºC during 75 min without serotonin (Control) or with 10 pM to 10 nM 5HT. 11Β-HSD2 activity was expressed as pmoles of 11Β-dehydrocorticosterone per mg of protein during 75 min. Values represent the mean ![]() SE of five independent experiments. **p<0.01 vs Control, Dunnett's Test. 5HT: serotonin
SE of five independent experiments. **p<0.01 vs Control, Dunnett's Test. 5HT: serotonin
Specificity of this activation was assessed using methiothepin and ketanserin, 5HT1/7 receptor and 5HT2 receptor antagonists, respectively. Figure 4 shows that both antagonists reduced the 5HT effect to control values. Both were devoid of intrinsic activity.
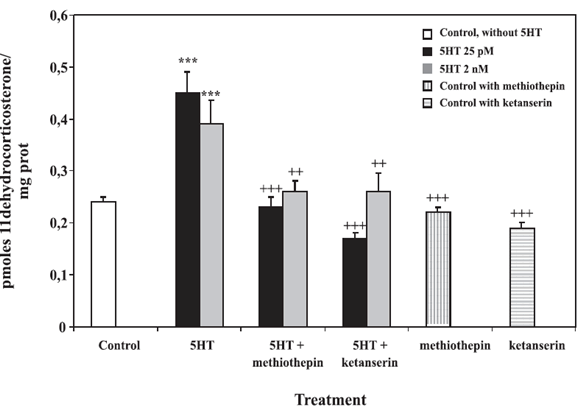
FIGURE 4. Effect of 5HT antagonists. Intact MDCK cells were preincubated at 37ºC during 15 min with 1 mM methiothepin or ketanserin. [3H1,2,6,7]corticosterone (10 nM) and 5HT (25 pM or 2 nM) were then added and incubated with the cells during 75 min at 37ºC. 11Β-HSD2 activity was expressed as pmoles of 11-dehydrocorticosterone per mg of protein during 75 min. Values represent the mean ![]() SE of five independent experiments. ***p<0.001 vs Control; ++p<0.01, +++p<0.001 vs 5HT, Tukey-Kramer's Test.
SE of five independent experiments. ***p<0.001 vs Control; ++p<0.01, +++p<0.001 vs 5HT, Tukey-Kramer's Test.
The magnitude of the 5HT effect was modulated by different corticosterone concentrations. Figure 5 depicts experiments with MDCK cell suspensions, increasing corticosterone concentrations and 25 pM, 2 nM or 10 nM 5HT. These 5HT values according to Figure 3, correspond to activating and non-activating concentrations of the indolamine. At low substrate concentration (4 nM corticosterone), 5HT -at whatever concentrationfailed to activate the enzyme. Near the KM, only 25 pM and 2 nM 5HT efficiently increased enzyme activity. As expected by the results presented in Figure 3, the highest concentration of 5HT (10 nM) failed to stimulate enzyme activity at any substrate concentration.
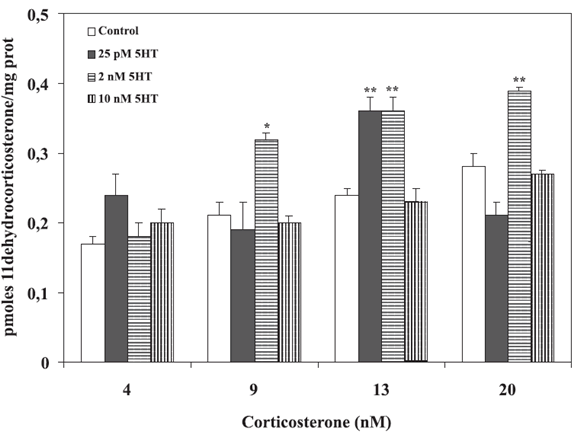
FIGURE 5. Effect of corticosterone on 5HT stimulation. Intact MDCK cells were incubated at 37ºC during 75 min with different concentrations of [3H1,2,6,7]corticosterone and 25 pM, 2 nM or 10 nM of 5HT. 11Β-HSD2 activity was expressed as pmoles of 11-dehydrocorticosterone per mg of protein during 75 min. Values represent the mean ![]() SE of three independent experiments. *p<0.05, **p<0.01 vs each Control, Dunnett's Test.
SE of three independent experiments. *p<0.05, **p<0.01 vs each Control, Dunnett's Test.
Discussion
Our results with MDCK cells cultured in DMEM show a feeble 11Β-HSD2 activity, thereby confirming similar findings by Korbmacher et al. (1989) and Leckie et al. (1995). Both groups focused their work on other cell lines and considered their assays with MDCK cells as pilot experiments. However, they observed an oxidative 11Β-HSD activity.
In the Introduction to the present work we explained the hypothesis that led us to assay 5HT as a renal 11Β- HSD2 modulator, based on a series of interactions between stress, glucocorticoids and extra-adrenal factors in enzyme control (Igarreta et al., 1999; Zallocchi et al., 2004 a and b), thus allowing for the assumption of a paracrine control of glucocorticoid inactivation in the distal segment by some exogenous factor(s).
The results obtained confirm this hypothesis. 5HT activates the glucocorticoid-catabolizing enzyme 11Β- HSD2 in a cell line often used as a model for events occurring in the distal nephron of mammals (Gekle et al., 2001; Oberleithner et al., 1990).
The MDCK cell line is endowed with 11Β-HSD2 activity as seen by the cofactor dependency (Fig. 1), the KM value for corticosterone and its characteristic pharmacological inhibition by carbenoxolone (Fig. 2), along with the univocal characterization of the end-product.
The interaction of 5HT with glucocorticoids has been studied in the central nervous system (CNS) and the HPA axis, suggesting a highly organized network between both stress hormones at the nervous, neuroendocrine, and endocrine levels (Azmitia and Segal, 1978; Jorgensen et al., 1999; McKittrick and McEwen, 1996; Tafet et al., 2001). We present here a novel role for 5HT in this network, now a catabolic one, at a distinct peripheral site at which the biological inactivation of glucocorticoids is of homeostatic relevance. As shown in Figure 3, two stimulation peaks could be evidenced, one at 2 nM 5HT concentration and a second equally intense peak at the very low 25 pM concentration of the indolamine.
As one of several alternatives, this multiphasic response in 11Β-HSD2 activity could be explained taking into account the nature of the 5HT receptors involved. The 5HT1D receptor present in MDCK cells (Schoeffter and Bobirnac, 1995) belongs to the superfamily of Gprotein- coupled receptors (GPCRs) which interact with heterotrimeric Gi proteins negatively couple to adenylyl cyclase (McKittrick and McEwen, 1996; Sanders-Bush and Mayer, 2001; Schoeffter and Bobirnac, 1995). Several recent studies have reported interactions between different GPCRs, demonstrating that GPCRs could exist as monomers, homo- or hetero-dimers or homo- or heterooligomers, these conformational states are susceptible to modifications by agonists (see a recent review by Devi, 2001; Tao et al., 2004). Likewise, the 5HT1D receptor occurs as a monomer, homodimer or heterodimer in the plasma membrane of transfected cells (Xie et al., 1999). Thus, the stimulation of 11Β-HSD2 would not only depend on 5HT binding, but also on the conformational state of the receptor depending on 5HT concentration.
The hypothesis implicates two discrete stimulation patterns in which the ligand would act as an allosteric effector, circumscribed to a "more sensitive" (low capacity), monomeric state -and a "less sensitive" (higher capacity) oliogomeric state of the receptor for the indolamine ligand, with a broad zone around 102 picomoles. In this broad zone, conversions to 11-dehydrocorticosterone equal control values. This, in turn, suggests lack of activation of receptor-affinity for serotonin.
According to alternative hypotheses, additional activations of 11Β-HSD2 activity might occur not only at the 5HT- membrane receptor, but also at cellular loci nearer to the isoenzyme itself.
A reciprocal interaction between 5HT and corticosteroids exist at different physiological levels. For example 5HT regulates the release of the HPA peptides CRH and ACTH, together with the adrenal steroid cortisol (Azmitia and Segal, 1978; Jorgensen et al., 1999; McKittrick and McEwen, 1996; Tafet et al., 2001; Lefebvre et al., 2001). On the other hand, corticoids stimulate the synthesis of 5HT in the CNS (Dinan, 1996) decreasing, at the same time, the number of 5HT receptors (de Kloet et al., 1986). Thus whilst 5HT regulates the HPA, the HPA in turn regulates the serotonergic activity.
Given the complex inter-relationship between these two systems, and using the two stimulatory concentrations of 5HT (Fig. 3), we decided to study its effect on 11Β-HSD2 activity in the presence of increasing corticosterone concentrations. Figure 5 shows that 25 pM and 2 nM 5HT effectively stimulated the enzyme at corticosterone concentrations of 12 nM or above. However, while 25 pM 5HT showed a unique peak at 12 nM corticosterone, 2 nM 5HT stimulated 11Β-HSD2 activity between 8 nM and 20 nM corticosterone concentrations. These results may be related to chronobiological aspects of indolamine and corticoid secretions, suggesting the existence of divergent signalling pathways.
5HT-mediated 11Β-HSD2 stimulation was inhibited by the presence of methiothepin, a 5HT1 receptor antagonist, or ketanserin, a 5HT2A, 5HT2C and 5HT1D antagonist, showing the specificity of the response (Fig. 4) (Schoeffter and Bobirnac, 1995; Xie et al., 1999; McKittrick and McEwen, 1996).
The results show for the first time a glucocorticoid- 5HT interaction at an anatomical site belonging neither to the CNS nor the HPA axes. Further, they suggest that 5HT, which under stressful circumstances circulates at increased levels (see for example Tafet et al., 2001), contributes to stimulate at a crucial site the inactivation of another enhanced "stress hormone", the glucocorticoid corticosterone. In line with results and concepts reported by Edwards et al. (1988) and Funder (1988; 1997), Young and Funder (2002), 5HT might thereby help to protect the organism against pathological sodium and water retention, as well as other corticoidderived pathologies following stress (Pacak and Palkovits, 2001; Sapolsky et al., 2000; McEwen, 1998, 2003; Yeager et al., 2004). Given the manifold, mostly positive interactions between both hormones, the 5HTenhancement of catabolic 11-ΒHSD activity could eventually pave the way to the study of a new regulatory axis in addition to HPA and the immunoneuroendocrine axes.
Acknowledgments
This work was supported by grants from the Consejo Nacional de Investigaciones Científicas y Técnicas (PRHOM-CONICET), the University of Buenos Aires and Fundación Alberto Roemmers. The authors wish to thank Prof. Dr. Rodolfo Pedro Rothlin (Facultad de Medicina - UBA) and Prof. Dr. Lidia Szczupak (Facultad de Ciencias Exactas y Naturales - UBA) who generously provided the indolamine 5HT and the 5HT antagonists, respectively. We also thank Sandra Rumille for technical assistance.
References
1. Agarwal A (2000). Expression of HSD11K (NAD+ dependent 11b-hydroxysteroid dehydrogenase) promoter constructs in renal cell lines. Endocr Res 26: 289-302. [ Links ]
2. Azmitia E, Segal M (1978). An autoradiographic analysis of the differential ascending projections of the dorsal and median raphe nuclei in the rat. J Comp Neurol 179: 641-667. [ Links ]
3. Berndt T, Liang T, Tyce G, Knox F (2001). Intrarenal serotonin, dopamine, and phosphate handling in remnant kidney. Kidney Int 59: 625-630. [ Links ]
4. Bradford M (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilising the principle of protein dye binding. Anal Biochem 72: 248-254. [ Links ]
5. Chaouloff F (2000). Serotonin, stress and corticoids. J Psychopharmacol 14: 139-151. [ Links ]
6. Contesse V, Reznik Y, Louiset E, Duparc C, Cartier D, Sicard F, Laquerriere A, Parmentier F, Kuhn J, Vaudry H, Lefebvre H (2005). Abnormal sensitivity of cortisol-producing adrenocortical adenomas to serotonin: in vivo and in vitro studies. J Clin Endocrinol Metab (Epub ahead of print). [ Links ]
7. De Kloet E, Sybmesma H, Reul H (1986). Neuroendocrinol 42: 513-521. [ Links ]
8. Devi L (2001). Heterodimerization of G-protein-coupled receptors: pharmacology, signalling and trafficking. TRENDS Pharmacol Sci 22: 532-537. [ Links ]
9. Dinan T (1996). Serotonin and the regulation of hypothalamic-pituitary- adrenal axis function. Life Sci 58: 1683-1694. [ Links ]
10. Edwards C, Stewart P, Burt D, Brett L, McIntyre M, Sutanto W, de Kloet E, Monder C (1988). Localisation of 11b-hydroxysteroid dehydrogenase tissue specific protector of the mineralocorticoid receptor. Lancet 2: 986-989. [ Links ]
11. Farman N, Bocchi B (2000). Mineralocorticoid selectivity: Molecular and cellular aspects. Kidney Int 57: 1364-1369. [ Links ]
12. Funder J (1997). Aldosterone, salt and cardiac fibrosis. Clin Exp Hypertens 19: 885-899. [ Links ]
13. Funder J, Pearce P, Smith R, Smith A (1988). Mineralocorticoid action: target tissue specificity is enzyme, not receptor mediated. Science 242: 583-585. [ Links ]
14. Gaush C, Hard W, Smith T (1966). Characterization of an established line of canine kidney cells (MDCK). Proc Soc Exp Biol Med 122: 931-935. [ Links ]
15. Gekle M, Freudinger R, Mildenberger S, Sauvant C (2001). Determination of basolateral Na+/H+ exchange activity in MDCK cells using a multiwell-multilabel reader. Anal Biochem 296: 174-178. [ Links ]
16. Gross J, Berndt T, Knox F (2000). Effect of serotonin receptor antagonist on phosphate excretion. J Am Soc Nephrol 11: 1002- 1007. [ Links ]
17. Gstraunthaler G, Pfaller W, Kotanko P (1985). Biochemical characterization of renal epithelial cell cultures (LLC-PK1 and MDCK). Am J Physiol 248: F536-F544. [ Links ]
18. Hesen W, Joels M (1996). Modulation of 5HT1A responsiveness in CA1 pyramidal neurons by in vivo activation of corticosteroid receptor. J Neuroendocrinol 8: 433-438. [ Links ]
19. Hoyer G, Tsiakiras D, Siebe H, Hierholzer K (1984). Corticosteroid metabolism in isolated rat kidney in vitro. III. Structure analysis of lipid soluble metabolites of corticosterone. Pflugers Arch 400: 377-380. [ Links ]
20. Igarreta P, Calvo JC, Damasco MC (1999). Activity of renal 11bhydroxysteroid dehydrogenase 2 (11bHSD2) in stressed animals. Life Sci 64: 2285-2290. [ Links ]
21. Jorgensen H, Knigge U, Kjaer A, Warberg J (1999). Adrenocorticotropic hormone secretion in rats induced by stimulation with serotonergic compounds. J Neuroendocrinol 11: 283-290. [ Links ]
22. Korbmacher C, Schulz W, Konig M, Siebe H, Lichtenstein I, Hierholzer K (1989). Renal epithelial cell lines (BSC-1, MDCK, LLC-PK1) express 11 beta-hydroxysteroid dehydrogenase activity. Biochim Biophys Acta 1010: 311-317. [ Links ]
23. Laplante P, Diorio J, Meaney M (2002). Serotonin regulates hippocampal glucocorticoid receptor expression via a 5-HT7 receptor. Brain Res Dev Brain Res 139: 199-203. [ Links ]
24. Leckie C, Chapman K, Edwards C, Seckl J (1995). LLC-PK1 cells model 11b-hydroxysteroid dehydrogenase type 2 regulation of glucocorticoid access to renal mineralocorticoid receptors. Endocrinology 136: 5561-5569. [ Links ]
25. Lefebvre H, Compagnon P, Contesse V, Delarue C, Thuillez C, Vaudry H, Kuhn J (2001). Production and metabolism of serotonin (5-HT) by the human adrenal cortex: paracrine stimulation of aldosterone secretion by 5-HT. J Clin Endocrinol Metab 86: 5001-5007. [ Links ]
26. McEwen B (2003). Interacting mediators of allostasis and allostatic load: towards an understanding of resilience in aging. Metabolism 52: 10-16. [ Links ]
27. McEwen B (1998). Protective and damaging effects of stress mediators. New Engl J Med 338: 171-179. [ Links ]
28. McKittrick C, McEwen B (1996). Regulation of serotoninergic function in the CNS by steroid hormones and stress In: Stone T, editor: CNS: Neurotransmitters and Neuromodulators. Neuroactive Steroids. New York: CRC Press Inc. pp 37-76. [ Links ]
29. Oberleithner H, Steigner W, Silbernagl S, Vogel U, Gstraunthaler G, Pfaller W (1990). Madin-Darby canine kidney cells. III. Aldosterone stimulates an apical H+/K+ pump. Pflugers Archiv 416: 540-547. [ Links ]
30. Odermatt A, Arnold P, Frey F (2001). The intracellular localization of the mineralocorticoid receptor is regulated by 11b-hydroxysteroid dehydrogenase type 2. J Biol Chem 276: 28484-28492. [ Links ]
31. Pacak K, Palkovits M (2001). Stressor specificity of control neuroendocrine responses: implication for stress-related disorders. Endocr Rev 22: 502-548. [ Links ]
32. Rusvai E, Naray-Fejes-Toth A (1993). A new isoform of 11b- hydroxysteroid dehydrogenase in aldosterone target cells. J Biol Chem 268: 10717-10720. [ Links ]
33. Sanders-Bush E, Mayer S (2001). 5-hydroxytriptamine (serotonin): receptor agonists and antagonists. In: Hardman JG, Limbird LE, Gilman AG, editors: Goodman & Gilman's The Pharmacological Basis of Therapeutics. pp 269-290. [ Links ]
34. Sapolsky R, Romero L, Munck A (2000). How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr Rev 21: 55- 89. [ Links ]
35. Schoeffter P, Bobirnac I (1995). 5-hydroxytryptamine 5HT1D receptors mediating inhibition of cyclic AMP accumulation in Madin-Darby canine kidney (MDCK) cells. Naunyn- Schmiedebergs Arch Pharmacol 352: 256-262. [ Links ]
36. Seckl J (1996). Expression and properties of corticosteroid receptors in the CNS. In: Stone T, editor: CNS: Neurotransmitters and Neuromodulators. Neuroactive Steroids. New York: CRC Press Inc. pp 1-21. [ Links ]
37. Simmons NL (1982). Cultured monolayers of MDCK cells: a novel model system for the study of epithelial development and function. Gen Pharmacol 13: 287-291. [ Links ]
38. Tafet G, Toister-Achituv M, Shinitzky M (2001). Enhancement of serotonin uptake by cortisol: a possible link between stress and depression. Cogn Affect Behav Neurosci 1: 96-104. [ Links ]
39. Tao Y, Johnson N, Segaloff D (2004). Constitutive and agonistdependent self-association of cell surface human lutropin receptor. J Biol Chem 279: 5904-5914. [ Links ]
40. Wang T, Berndt T, Gross J, Peterson M, So M, Knox F (2001). Effect of inhibition of MAO and COMT on intrarenal dopamine and serotonin and on renal function. Am J Physiol Regul Integr Comp Physiol 280: R248-R254. [ Links ]
41. Xie Z, Lee S, O'Dowd B, George S (1999). Serotonin 5-HT1B and 5-HT1D receptors form homodimers when expressed alone and heterodimers when co-expressed. FEBS Lett 456: 63-67. [ Links ]
42. Yeager MP, Guyre PM, Munck AU (2004) Glucocorticoid regulation of the inflammatory response to injury. Acta Anaesthesiol Scand. Aug;48(7): 799-813. [ Links ]
43. Young M, Funder J (2002). Mineralocorticoid receptors and pathophysiology roles for aldosterone in the cardiovascular system. J Hypertens 20: 1465-1468. [ Links ]
44. Zallocchi M, Matkovic' L, Calvo JC, Damasco MC (2004a). Adrenal gland involvement in the regulation of renal 11b- hydroxysteroid dehydrogenase 2. J Cell Biochem 92: 591-602. [ Links ]
45. Zallocchi ML, Matkovic' L, Damasco MC (2004b). Adrenal 11- beta hydroxysteroid dehydrogenase activity in response to stress Can J Physiol Pharmacol. Jun; 82(6): 422-425. [ Links ]
46. Zhou M, Gomez-Sanchez E, Cox D, Cosby D, Gomez-Sanchez C (1995). Cloning, expression, and tissue distribution of the rat nicotinamide adenine dinucleotide-dependent 11 betahydroxysteroid dehydrogenase. Endocrinology 136: 3729-3734. [ Links ]
Received on November 7, 2005.
Accepted on June 16, 2006.














