Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Biocell
versión impresa ISSN 0327-9545
Biocell v.31 n.3 Mendoza sept./dic. 2007
Karyological and electrophoretic differences between Pomacea flagellata and P. patula catemacensis (Caenogastropoda: Ampullariidae)
María Esther Diupotex-Chong*, Néstor J. Cazzaniga**, And Manuel Uribe-Alcocer*
* Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Circuito Exterior, Ciudad Universitaria, Coyoacán 04510. México, D.F.
** Departamento de Biología, Bioquímica y Farmacia, Universidad Nacional del Sur. San Juan 670, 8000 Bahía Blanca. Argentina.
Address correspondence to: Dra. María Esther Diupotex-Chong. Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Circuito Exterior, Ciudad Universitaria, Coyoacán 04510, MEXICO, D.F. E-mail: medc@mar.icmyl.unam.mx
ABSTRACT: The widespread Mexican apple snail Pomacea flagellata (Say 1827) and the strictly endemic "tegogolo" P. patula catemacensis (Baker 1922) (restricted to Lake Catemaco), are the only known American Ampullariidae that have haploid complements n=13. Pomacea patula catemacensis has suffered a critical reduction in abundance due to immoderate fishing for human consumption. Chromosome slides were obtained from colchicine-injected Pomacea snails collected from nine locations along the coastal zone of the Gulf of Mexico, including Lake Catemaco, for use in principal component analysis (PCA). Total proteins in foot homogenates were analyzed through isoelectric focusing (IEF) and native-PAGE electrophoresis on polyacrylamide gels. The chromosome number 2n=26 was confirmed for snails from all locations, with a uniform 9 m + 4 sm formula. However, P. patula catemacensis showed significantly larger chromosomes (absolute size) than any population of P. flagellata. Pomacea patula catemacensis also differed from all populations of P. flagellata in a PCA with standardized data, i.e., independently of the absolute size difference between species. Proteins with an acid isoelectric point were dominant in the foot of both species. The electrophoresis analysis showed that P. flagellata has 17 protein bands, with an upper bound at IEF=7.6, while P. patula catemacensis has only 15 bands, with an upper bound at IEF=7 and a more evenly spaced band pattern. Molecular weights ranged from 40 to approximately 130 kDa in both species. Proteins with high values (>94 kDa) were the most abundant. Pomacea patula catemacensis showed a band of 93 kDa, which was absent from all specimens of P. flagellata. Samples of P. flagellata did not cluster according to any geographical pattern in the statistical analyses, nor did they show any taxonomically useful differences in their electrophoretic patterns that merit sub-specific discrimination.
Key words: Apple snails; Endangered species; Isoelectric focusing; Karyotype; Mexico; Native-PAGE electrophoresis.
Introduction
Most species of apple snails (Ampullariidae) have haploid complements of n=14 chromosomes (Choudhury and Pandit, 1997). Among the species living in the Americas, only the two species of Pomacea Perry 1810 thriving in Mexico, Pomacea flagellata (Say 1827) and P. patula catemacensis (Baker 1922), are known to be exceptions, with haploid complements of n=13 chromosomes (Diupotex, 1994; Diupotex-Chong et al., 2004).
Pomacea flagellata is a widespread species that is distributed from Mexico and Central America to northern Colombia (Pain, 1964), while P. patula catemacensis, also a member of the "Pomacea flagellata group", is confined to its type locality, Lake Catemaco, in southeastern state of Veracruz, Mexico (Naranjo-García, 2003; Naranjo-García and García-Cuba, 1986). The shared unusual number of chromosomes and the restricted geographical location of P. patula catemacensis within the natural realm of P. flagellata induced us to look further into the consistency of the differences between these two taxa.
The quantitative differences in chromosome morphology of various Pomacea populations along the coastal zone of the Gulf of Mexico were examined, and the possible specific discrimination among species, were analysed using electrophoretic techniques. Results confirmed that the two taxa, P. flagellata and P. patula catemacensis, which previously had been discriminated only by shell and anatomical features, are also clearly distinguished by karyological and electrophoretic characteristics.
Materials and Methods
Specimens were collected from nine locations along the coastal zone of the Gulf of Mexico: 1) state of Veracruz: Catemaco, Tlacotalpan, Alvarado, and three sampling points along the Misantla River (M1: near the river mouth, M2: mid course, and M3: near the headwaters), 2) state of Tabasco: near El Espino and San Miguel, and 3) state of Campeche: lake El Vapor adjacent to Laguna de Términos. The coastal zone of the Gulf of Mexico is characterized by low land and shallow clean water bodies of 2-5 ppm salinity, a mean water temperature of 26ºC, and a mean pH of 7.5 (Pérez-Rojas and Torres-Orozco Bermeo, 1992). The climate is humid and warm, and the prevailing vegetation is evergreen rainforest.
The specimens were kept alive in the laboratory in filtered and aerated water at room temperature, and were fed Vallisneria sp. Some specimens were sacrificed, dissected, and immediately frozen at -30ºC for a further study of protein patterns.
Five specimens from each sampling site were injected, 45 min before sacrifice, with 1 ml of colchicine 0.04% per 50 g of animal weight, for the chromosome study. Samples of fresh gonadic tissue were cut into 3 mm thick pieces, mashed and stained with 2% acid orcein, in agreement with Griffin et al. (1997), in order to determine the haploid complement. Gill tissue was also excised, cut into 3 mm thick slices, and put in Petri dishes in a hypotonic solution (0.075M KCl) for 2 h. The sediment was fixed in a 3:1 mixture of methanol and acetic acid after maceration and centrifugation at 1500 rpm for 10 min. The sequence of centrifugation and fixation was repeated for three periods of 5 min and the samples were stored for 24 h at 4ºC in the same fluid.The samples were treated according to the method of Kligerman and Bloom (1977), modified by Coullin and Pellestor (1997). Cell desquamation was performed in 60% glacial acetic acid. The sediment was resuspended and three drops of the liquid for each specimen were transferred to each of five slides at 50ºC. The slides were air-dried during 24 h before staining with Giemsa solution (Sigma) in a pH 6.4 phosphate buffer. The slides were washed in distilled water, clarified with xylene, and mounted with Canada balsam.
Metaphase plates were selected and photographed with a Zeiss phase contrast microscope. The plates were represented in ideograms according to the criteria established by Al-Aish (1969) and Levan et al. (1964). The chromosome sets used were those of adequate quality for statistical evaluation, with whole uniformly stained mitotic fields, non-superposed or shrunken chromosomes, and all chromosomes suitable for accurate measurement. Thus, the number of sets differs among populations in the analysis. Statistical comparisons were possible as all materials were processed under the same methodological protocol, by the same person (the senior author) and in a short time period, minimizing the effects of a differential contraction of the chromosomes, staining differences, and variations due to conservation. Homology of the chromosomes according to their order from the longest to the shortest in each ideogram was assumed.
The descriptive parameter calculations have been described by Diupotex-Chong et al. (2004). The absolute length of the chromosomes was compared by oneway ANOVAs and Tukey's HSD multicomparison test. Karyotype asymmetry was assessed through the indices defined by Romero-Zarco (1986): A1 estimates the intrachromosomal asymmetry as

where n is the number of homologous chromosome pairs, bi is the average length for short arms, and Bi is the average length for long arms in every chromosome pair (i). A2 depicts the interchromosomal asymmetry as the ratio of the standard deviation to the mean length of the chromosomes.
A principal component analysis (PCA) was applied to a matrix of mean relative lengths of the short (bi) and long (Bi) arms. In this analysis, 13 representative metaphases of P. patula catemacensis from Lake Catemaco and 52 representative metaphases of P. flagellata from Tlacotalpan (5), Alvarado (5), Misantla-1 (4), Misantla-2 (5), Misantla-3 (4), San Miguel (6), El Espino (13) and El Vapor (10) were included.
Foot flesh samples of specimens from each sampling location were macerated in an Ultraturax homogenizer with a Tris-HCl damper (0.05 M, pH 8) for a minimum of 7 min in short intervals of less than 1 min each at low temperature (4ºC) in order to avoid denaturalization of proteins. The extracts were centrifuged at 30,000 g and 4ºC for 30 min in a Bechman J2-21 centrifuge. The supernatant was dialyzed with a Tris-HCl damper (0.05 M, pH 8) for 9 h in a continuous dialysis system (Arreguín and Taboada, 1968). The protein contents of the extracts were quantified and concentrated to a single dilution in order to be able to make comparisons. Quantification was completed by the method described by Smith et al. (1985) which uses bicinchoninic acid (BCA; Protein Assay Reagent) and a spectrophotometer at 562 nm.
Electrophoretic studies were performed on samples from the six locations in the state of Veracruz: Catemaco, Tlacotalpan, Alvarado, and three locations along the Misantla River (M1, M2, and M3).
Isoelectric focusing (IEF) was carried out with a Pharmacia's Phast System equipment on 35 x 43 x 50 mm microplates, for a gradient of pH 3-9 calibrated with a Pharmacia electrophoresis calibration kit, running 1 mg of protein in each lane. The band position was read with a laser LKB 2202 Ultro Scan densitometer.
Native-PAGE electrophoresis on homogeneous 20% polyacrylamide gels was used to analyze the composition of native proteins and estimate their molecular weight on 45 x 43 x 50 mm microplates at 500 volts, 10 mA rising voltage and 15ºC. Reference samples of known molecular weight from 14.4 to 94 kDa (Pharmacia electrophoresis calibration kit) were used, and 1 mg of protein was run in each lane. Fixing, staining and lightening techniques were performed according to Neuhoff et al. (1985). The position of the bands was read with a laser LKB 2202 Ultro Scan densitometer.
Five collections of shells were deposited as voucher specimens in the Museo de La Plata, Argentina, under the following collection numbers: Pomacea patula catemacensis (Baker 1922), Lake Catemaco, Veracruz, MLP 11670; Pomacea flagellata (Say 1823), San Miguel, Tabasco, MLP 11929; Tlacotalpan, Veracruz, MLP 11925; Alvarado, Veracruz, MLP 11926; and El Vapor, Campeche, MLP 11924.
Results
Chromosome numbers 2n=26 were confirmed for the apple snails of all locations, and haploid complement n=13 was confirmed for gonadic tissues. Both Pomacea patula catemacensis and P. flagellata showed a fundamental number of FN=52. Neither negative heteropicnosis nor chromosomal heteromorphism were observed, and there was no visual evidence of sexual chromosomes.
The chromosomes of P. patula catemacensis (Fig. 1; absolute mean length = 4.94 mm ± 0.84 SD) were significantly longer than those of P. flagellata (Fig. 2; absolute mean length = 4.03 mm ± 0.58 SD): F=7.19, d.f.=8, p<0.0001 (data were transformed to ln(x) to improve homocedasticity before comparison).The mean chromosome length was not significantly different among populations of P. flagellata (Table 1).

FIGURE 1. A representative metaphase plate of Pomacea flagellata (Say, 1823).

FIGURE 2. A representative metaphase plate of Pomacea patula catemacensis (Baker, 1930).
TABLE 1.
Chromosomal absolute lengths (mm) for nine Mexican populations of Pomacea (two species) from the coastal zone of the Gulf of Mexico.All localities refer to Pomacea flagellata (Say 1823) except Lake Catemaco which is Pomacea patula catemacensis (Baker 1922). Results of a Tukey’s HSD multicomparison test are included(critical values: Tukey0.05 =5.27; Tukey0.01 =6.67).
The chromosome formula was 9 m + 4 sm for all the studied populations (Table 2). Figure 3 shows the respective ideograms.
TABLE 2.
Chromosomal measurements for nine Mexican populations of Pomacea (two species) from the coastal zone of the Gulf of Mexico. All localities refer to Pomacea flagellata (Say 1823) except Lake Catemaco which is Pomacea patula catemacensis (Baker 1922). Data on each cell is mean absolute length in mm, mean arm ratio in brackets, and chromosome classification following Levan et al. (1964): m, metacentric and sm, submetacentric chromosomes.
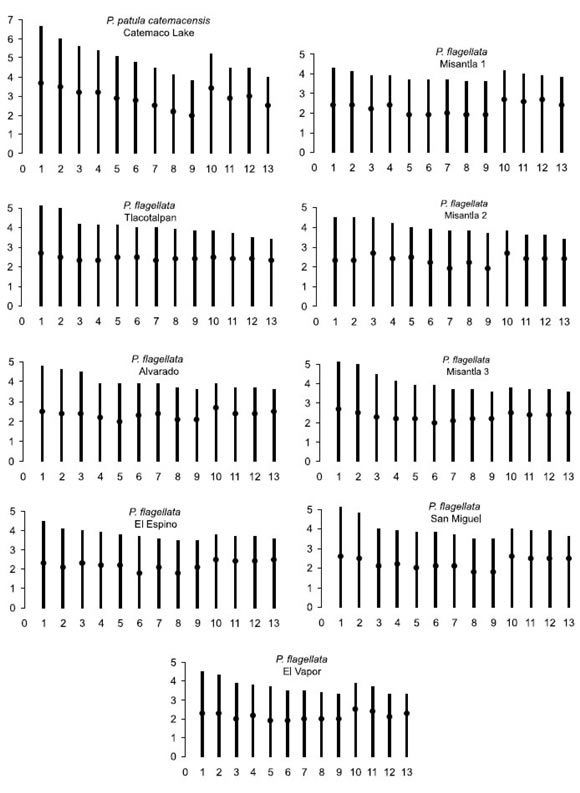
FIGURE 3. Ideograms of the haploid complement of nine Pomacea populations (two species) from the coastal zone of the Gulf of Mexico. Ordinate values are absolute lengths of the chromosomes (in mm).
Karyotype asymmetry of the two species corresponded to category A2 (Stebbins, 1971) for two reasons: a) the ratio of the largest to the smallest chromosome in the karyotype was <2 (1.76 for P. patula catemacensis and from 1.19 (Misantla-1) to 1.53 (Tlacotalpan) for P. flagellata), and b) less than 50% of the chromosomes showed a mean arm proportion >2:1. Only 1 of the 13 chromosomes (7.69%) in most populations, except in Tlacotalpan and Alvarado (two chromosomes with mean arm proportion >2:1, i.e. 15.38%), and Misantla-2 (four of these chromosomes, i.e. 30.77%).
Figure 4 shows a scatter plot of the intra(A1) and inter-chromosomic (A2) asymmetry indices for the nine studied populations. A1 was 0.29 and A2 was 0.17 for P. patula catemacensis, the latter value being the highest among the studied populations. A1 ranged from 0.23 to 0.33 andA2 ranged from 0.06 to 0.14 for P. flagellata. There was no significant correlation between A1 and A2 (r=0.207, p>0.1) or any clear pattern derived from the geographical origin of the specimens. It should be noted that the variability recorded for the three samples of P. flagellata from the Misantla River (M1, M2 and M3: Fig. 4, circle) enfolds the variation of most populations of this species, except for those from San Miguel (SM) and Tlacotalpan (T) that scored the lowest and highest values of A1, respectively.
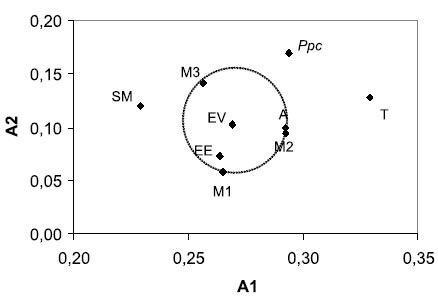
FIGURE 4. Scatter plot of the karyotype asymmetry indices (Romero-Zarco, 1986) for nine Pomacea populations (two species) from the coastal zone of the Gulf of Mexico. A1: intrachromosomal asymmetry index; A2: interchromosomal asymmetry index. Ppc identifies Pomacea patula catemacensis (Baker 1922) from Lake Catemaco. The remaining references are for Pomacea flagellata (Say 1827) from: Alvarado (A), El Espino (EE), El Vapor (EV), three localities along the Misantla River (M1: river mouth, M2: mid course, M3: headwaters), San Miguel (SM) and Tlacotalpan (T).
Before we performed a principal component analysis, we standardized the lengths of the chromosome arms as relative lengths of the total extent of the chromosome complement to avoid the effect of the absolute size difference between species. Even so, P. patula catemacensis differed from all populations of P. flagellata. Most populations of P. flagellata scored low on axis 2; only the snails from the middle course of the Misantla River (M2) are located near P. patula catemacensis at high values on axis 2. This is positively correlated with the length of the long arms in chromosomes 3 and 4 (B3, B4) and the length of the short arm in chromosome 7 (b7), and negatively correlated with B5, B7 and B13 (Fig. 5); on axis 3, P. patula catemacensis and P. flagellata from Misantla-2 were the populations that received the highest and the lowest scores respectively (Fig. 5). Axis 3 is positively correlated to B1 and B2 and negatively correlated with B13. The three samples from the Misantla River were located far from each other on these charts, which represents a remarkably high variability for co-specific populations from a single basin, and exceeds the observed variation among populations from different localities.


FIGURES 5-6. Plots of the Principal Component Analysis on the standardized lengths of the chromosome arms for nine Pomacea populations from the coastal zone of the Gulf of Mexico. Black square: Pomacea patula catemacensis (Baker 1922), grey squares: Pomacea flagellata (Say 1827). Other references as in figure 2.
Out of the five tested protein concentrations (1, 1.5, 2, 3 and 5 mg/ml) with the isoelectric focusing tests, the best-defined bands were obtained with the 3 mg/ml concentration. Proteins with acid isoelectric points were dominant in the two electrophoretic patterns, and the most intense bands were located around the 5.8, 5, 4 and 3.5 isoelectric points. One of the bands below the lowest IEF marker (3.5) was also quite intense (Fig. 7). The main differences between the two specific patterns were that P. flagellata showed 17 bands and the highest upper bound at isoelectric point pI 7.6, which was relatively far from the next five bands and concentrated between isoelectric points 6.6 and 6.15. The IEF on P. patula catemacensis showed only 15 bands, with the upper bound at isoelectric point pI 7 and a more evenly spaced band pattern.
The molecular weights of the proteins on the polyacrylamide gels are shown in figure 8. Most of the identified proteins had a molecular weight of 40 to >94 kDa (probably up to 130 kDa), and the most abundant proteins were those with the highest values. Pomacea flagellata showed 12 bands, while P. patula catemacensis showed 13 bands. Both patterns were very similar, except for a band of 93 kDa in the P. patula catemacensis specimens from Lake Catemaco, which was absent in all samples of P. flagellata.

FIGURE 7. Isoelectric focusing (IEF) on a gradient of pH 3-9 for some Pomacea populations from the coastal zone of the Gulf of Mexico. All localities refer to Pomacea flagellata (Say 1823) except Lake Catemaco which is Pomacea patula catemacensis (Baker 1922). The order of lanes is as follows: (1) protein standards, (2) Misantla-3, (3) Misantla-1, (4) Misantla-2, (5) protein standards, (6) Alvarado, (7) Tlacotalpan, and lane 8 is Lake Catemaco.
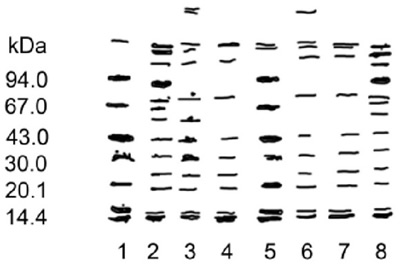
FIGURE 8. A representative microplate with results of native PAGE electrophoresis on homogeneous 20% polyacrylamide gels for some Pomacea populations from the coastal zone of the Gulf of Mexico. All localities refer to Pomacea flagellata (Say 1823) except Lake Catemaco which is Pomacea patula catemacensis (Baker 1922). The order of lanes is as follows: (1) molecular weight standards, (2) Misantla-3, (3) Misantla-1, (4) Misantla-2, (5) protein standards, (6) Alvarado, (7) Tlacotalpan, and (8) Lake Catemaco.
Discussion
Apple snails have variable morphologies (Pain, 1964; Rangel-Ruiz, 1988; Estebenet et al., 2006), genetics and molecular characteristics (Keawjam and Upatham, 1990; Thaewnon-Ngiw et al., 2003).
The most common ampullariid snail in Mexico is Pomacea flagellata, which has countless local forms that have received many specific names. Pain (1964: 224) said that this species is "variable to an extent which fills the investigator with dismay".
Pomacea flagellata has a distribution range that spans from the state of Veracruz and throughout the Mexican Gulf slope and Yucatan Peninsula, south to Panama and northern Colombia (Naranjo-García, 2003). In contrast, P. patula catemacensis is restricted to a single lake. Given that apple snail species commonly have wide distribution ranges, the strict endemism of P. patula catemacensis in Lake Catemaco is a remarkable exception.
Lake Catemaco is a relatively isolated 7250 hectare water body that lies in a volcanic crater at an altitude of 333 m, in the volcanic Los Tuxlas area, within the Papaloapan River basin. It is the most productive natural lake in Mexico, with approximately 1800 tons of fishing products harvested per year (Pérez-Rojas and Torres-Orozco Bermeo, 1992). Pomacea patula catemacensis is an important source of food for human consumption (26.6% of the local fishery production). However, overexploitation has resulted in a dramatic reduction in the abundance of this apple snail, from over 5000 tons between 1980 and 1989 to only 24 tons in 2001 (Carreón-Palau et al., 2003). At present, and according to the criteria of IUCN (2001), the endemic P. patula catemacensis may be considered "endangered" or "critically endangered", and measures to control fishing activities in the lake are the subject of legislative concern (Ochoa-Muñoz, 2006).
Pomacea patula catemacensis was originally described as Ampullaria patula catemacensis by Baker (1922), based only on shell characteristics. Its identity as a well defined taxon was reaffirmed recently by Carreón-Palau et al. (2003) on the basis of genital anatomy. The results presented in this study provide additional evidence that support these previous findings. Indeed, despite their morphological heterogeneity, all populations of P. flagellata have similar karyotypes and common patterns of protein bands (IEF and molecular weights) that differ from those of Pomacea patula catemacensis. The chromosomes of P. patula catemacensis were found to be significantly longer, and a multivariate analysis with standardized data clearly separated the two species in this study.
Landa and Nader (1991) reported for Pomacea flagellata a chromosome number of 2n=16, while Diupotex (1994) found a modal number of 2n=26 for the same species. This latter report is consistent with our findings for eight populations collected along the coastal zone of the Gulf of Mexico. Data published by Landa and Nader (1991) have not been confirmed.
After placing more than 30 species names within the synonymy of Pomacea flagellata, Pain (1964) recognized only four morphs as probable subspecies. However, these morphs do not seem to represent actual examples of independent geographic variation, and Rangel-Ruiz (1988) stated the inconvenience of retaining such trinomials. Our samples of P. flagellata did not cluster according to any geographical pattern in the statistical analyses. The chromosomes were found to vary among the three samples collected from the Misantla River (M1, M2, M3), and the karyotype and electrophoretic results did not lead to a subspecific discrimination.
Diupotex-Chong et al. (2004) stated that P. patula catemacensis has a more asymmetric karyotype than P. flagellata. The results obtained here verify this previous finding for the inter-chromosomal asymmetry index (A2), but not for the intra-chromosomal asymmetry index (A1), given that the P. flagellata samples from Alvarado and Misantla-2 are almost asymmetric and the snails from Tlacotalpan are much more asymmetric than P. patula catemacensis.
The presence of sexual chromosomes in the Gastropoda is rare. Only about 7% of the 230 species studied of Caenogastropoda were reported to have a chromosomal system of sex determination (Patterson, 1969; Thiriot-Quiévreux, 2003). Most previous authors found no evidence of sexual heterosomes in species of Ampullariidae (Choudhury and Pandit, 1997; Diupotex, 1994; Diupotex-Chong et al., 2004; Kawano et al., 1990; Mercado-Laczkó and Lopretto, 1998; Yaseen et al., 1991). Only von Brand et al. (1990) described the existence of male XY heterogamety in Pomacea canaliculata from Japan. Our extensive sampling along the Mexican coast does not support the hypothesis that sex is chromosomally determined in P. flagellata and P. patula catemacensis.Yusa and Suzuki (2003) suggested that a polyfactorial genetic determination of sex for P. canaliculata was the most likely mechanism to explain their findings. Also, more recent studies suggest the involvement of either a small number of sex-determining genes or a more complicated system such as sexratio or sex-determining polygenes that act non-additively (Yusa, 2005).
Acknowledgments
The authors thank Mario Alejandro Gómez-Ponce for his help in the field and for keeping the Pomacea specimens alive in the laboratory. Snail species identif ication was confirmed by the Laboratorio de Malacología, Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. NJC is a staff researcher of the Comisión de Investigaciones Científicas de la Provincia de Buenos Aires, Argentina.
References
1. Al-Aish M (1969). Human chromosome morphology studies on normal chromosome characterization, classification and karyotyping. Can J Genet Cytol. 11: 370-381. [ Links ]
2. Arreguín B, Taboada J (1968). Un aparato de diálisis continuo. Boletín del Instituto de Química, Universidad Nacional Autónoma de México 20: 95-97. [ Links ]
3. Baker HB (1922). The Mollusca collected by the University of Michigan Walker expedition in southern Veracruz, Mexico. Occasional Papers of the Museum of Zoology, University of Michigan 106: 162. [ Links ]
4. Carreón-Palau L, Uria-Galicia E, Espinosa-Chávez F, Martínez-Jerónimo F (2003). Desarrollo morfológico e histológico del sistema reproductor de Pomacea patula catemacensis (Baker 1922) (Mollusca, Caenogastropoda: Ampullariidae). Rev Chilena de Historia Natural 76: 665-680. [ Links ]
5. Choudhury RE, Pandit RK (1997). Chromosomes of three prosobranch gastropods from Viviparidae, Pilidae and Cyclophoridae (Order: Mesogastropoda). Caryologia 50: 303-315. [ Links ]
6. Coullin P, Pellestor F (1997). Rapid characterization of human chromosomes in hybrid cell lines by primed in situ (PRJNS) labelling. Somat Cell Mol Genet. 23: 159-163. [ Links ]
7. Diupotex ME (1994). Karyological analysis and "G" bands of Pomacea flagellata from southeast of Mexico. Western Society of Malacologists, Annual Report 26: 5-7. [ Links ]
8. Diupotex-Chong ME, Cazzaniga NJ, Hernández-Santoyo A, Betancourt-Rule JM (2004). Karyotype description of Pomacea patula catemacensis (Caenogastropoda, Ampullariidae), with an assessment of the taxonomic status of Pomacea patula. Biocell 28: 279-285. [ Links ]
9. Estebenet AL, Martin PR, Burela S (2006). Conchological variation in Pomacea canaliculata and other South American Ampullariidae (Caenogastropoda, Architaenioglossa). Biocell 30: 329-335. [ Links ]
10. Griffin DK, Millie EA, Reline RW, Hassold TJ, Zaragoza MV (1997). Cytogenetic analysis of spontaneous abortions: comparison of techniques and assessment of the incidence of confined placental mosaicism. Am J Med Genet. 72: 297-301. [ Links ]
11. IUCN (The World Conservation Union) (2001). The IUCN Red List Categories. 2001 Categories & Criteria (version 3.1). http:// www.redlist.org/info/categories_criteria2001.html#categories. [ Links ]
12. Kawano T, Gomes-Simoes LC, Correa FMA (1990). Chromosomes of Pomacea sp. (Perry, 1811) (Mesogastropoda, Mollusca). Rev Bras Genet. 13: 675-685. [ Links ]
13. Keawjam RS, Upatham ES (1990). Shell morphology, reproductive anatomy and genetic patterns of three species of apple snails of the genus Pomacea in Thailand. J Med Appl Malacol. 2: 45-57. [ Links ]
14. Kligerman AD, Bloom SE (1977). Rapid chromosome preparations from solid tissues of fishes. Journal of the Fisheries Research Board of Canada 34: 266-269. [ Links ]
15. Landa GP, NADER GB (1991). Número cromosómico de Pomacea flagellata Say, 1827 (Gastropoda: Pilidae). XI Congreso Nacional de Zoología, Mexico, October 28-31, 1991. Programa y Resúmenes, p. 19. [ Links ]
16. Levan AK, Fredga A, Sandberg A (1964). Nomenclature for centromeric position on chromosomes. Hereditas 52: 201-220. [ Links ]
17. Mercado-Laczkó AC, Lopretto EC (1998). Estudio cromosómico y cariotípico de Pomacea canaliculata (Lamarck, 1801) (Gastropoda: Ampullariidae). Revista del Museo Argentino de Ciencias Naturales "Bernardino Rivadavia", Hidrobiología 8: 15-20. [ Links ]
18. Naranjo-García E (2003). Moluscos continentales de México: Dulceacuícolas. Rev Biol Trop. 51 (Suppl. 3): 495-505. [ Links ]
19. Naranjo-García E, García-Cuba A (1986). Algunas consideraciones sobre el género Pomacea (Gastropoda: Pilidae) en México y Centroamérica. Anales del Instituto de Biología, Universidad Nacional Autónoma de México, Serie Zoología 56: 603-606. [ Links ]
20. Neuhoff V, Stamm R, Eibl H (1985). Clear background and highly sensitive protein staining with Coomassie blue dyes in polyacrylamide gels: A systematic analysis. Electrophoresis 6: 427-448. [ Links ]
21. Ochoa Muñoz LI (2006). Proyecto de Norma Oficial Mexicana PROY-NOM-041-PESC-2004, Pesca responsable en el lago de Catemaco, ubicado en el estado de Veracruz. Especificaciones para el aprovechamiento de los recursos pesqueros. República de México, Secretaría de Agricultura, Ganadería, Desarrollo Rural, Pesca y Alimentación. Diario Oficial, Primera Sección, Jueves 2 de marzo de 2006. [ Links ]
22. Pain T (1964). The Pomacea flagellata complex in Central America. J Conchol. 25: 224-231. [ Links ]
23. Patterson CM (1969). Chromosomes of mollusks. Proceedings of the Symposium on Molluscs, II, Marine Biology Association of India 1968: 635-686. [ Links ]
24. Pérez-Rojas A, Torres-Orozco Bermeo R (1992). Geomorfología y batimetría del Lago Catemaco, Veracruz, México. Anales del Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México, 19: 19-24. [ Links ]
25. Rangel-Ruíz RJ (1988). Estudio morfológico de Pomacea flagellata (Say, 1827). (Gastropoda: Ampullariidae) y algunas consideraciones sobre su taxonomía y distribución geográfica en México. Anales del Instituto de Biología, Universidad Nacional Autónoma de México, Serie Zoología 58: 21-34. [ Links ]
26. Romero-Zarco C (1986). A new method for estimating karyotype asymmetry. Taxon 35: 526-530. [ Links ]
27. Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985). Measurement of protein using bicinchoninic acid. An Biochem. 150: 76-85. [ Links ]
28. Stebbins GL (1971). Chromosomal evolution in higher plants. Edward Arnold Ltd., London. [ Links ]
29. Thaewnon-Ngiw B, Klinbunga S, Phanwichien K, Sangduen N, Lauhachinda N, Menasveta P (2003). Genetic diversity of introduced (Pomacea canaliculata) and native (Pila) apple snails in Thailand revealed by randomly amplified polymorphic DNA (RAPD) analysis. ASEAN Journal of Science and Technology for Development 20: 289-306. [ Links ]
30. Thiriot-Quiévreux C (2003).Advances in chromosomal studies of gastropod molluscs. Journal of Molluscan Studies 69: 187-201. [ Links ]
31. von Brand E, Yokosawa T, Fujio Y (1990). Chromosome analysis of apple snail Pomacea canaliculata. Tohoku Journal of Agricultural Research 40: 81-89. [ Links ]
32.Yaseen AE, Ebaid ABM, Kawashti IS (1991). Studies on the chromosomal analysis and shell measurements of two species, Bellamya unicolor and Lanistes bolteni (Prosobranchiata: Gastropoda). Cytologia 56: 503-509. [ Links ]
33. YusaY (2005) Genetics of sex-ratio variation inferred from parentoffspring regressions and sib correlations in the apple snail Pomacea canaliculata. Heredity 96: 100-105. [ Links ]
34. Yusa Y, Suzuki Y (2003). A snail with unbiased population sex ratios but highly biased brood sex ratios. Proceedings of the Royal Society of London, series B 270: 283-287. [ Links ]
Received on September 28, 2006. Accepted on June 20, 2007.














