Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Biocell
Print version ISSN 0327-9545
Biocell vol.32 no.1 Mendoza Jan./Apr. 2008
Antioxidant capacity of vitamin C in mouse liver and kidney tissues
Alicia Weyers, Laura I. Ugnia, Hugo García Ovando, Nora B. Gorla*
Facultad de Agronomía y Veterinaria, Universidad Nacional de Río Cuarto, (5800) Río Cuarto, Córdoba, Argentina.
*CONICET.
Address correspondence to: Dra. Nora B. Gorla. Facultad de Agronomía y Veterinaria, Universidad Nacional de Rio Cuarto (UNRC), (5800) Río Cuarto, Córdoba, ARGENTINA. FAX: (+054-358) 468 0280. E-mail: ngorla@ayv.unrc.edu.ar
ABSTRACT: In the present study, the antioxidant capacity of vitamin C was examined in the liver and the kidney tissues of mice with or without ciprofloxacin (CFX) treatment. The antioxidant capacity of the vitamin was evaluated in terms of lipid hydroperoxides (LOOH) and thiobarbituric acid reactive substances (TBARs). The experimental design was 15 days of water (control and CFX groups) or vitamin C (vitamin C and vitamin C plus CFX groups) in drinking water. One dose of CFX was injected, 15 minutes before sacrifice, in the corresponding mice.
The initial nmol of lipid hydroperoxides/g of tissue were 137 ± 11 in the kidney and 145 ± 15 in the liver, and the nmol of TBARs were 13 ± 0.7 and 12 ± 0.6, respectively. Pre-treatment with vitamin C reduced the levels of LOOH in the liver to 45 ± 11 (p<0.01) and vitamin C with CFX injection to 54 ± 9 (p<0.01). Vitamin C treatment also reduced the LOOH levels in the kidney roughly duplicated by CFX. Through the TBARs method we have not observed these effects.
Quantification of LOOH is more sensitive than that of TBARs for estimating lipid peroxidation. CFX is used especially for urinary infections and can produce oxidative stress in the kidney. Pre-treatment with vitamin C may ameliorate this stress and also may improve the oxidative balance in the liver.
Key words: Lipid peroxidation; Vitamin C; Ciprofloxacin; Mice
Introduction
Oxidative stress is the term used to describe the condition of oxidative damage that results when the critical balance between free radical generation and antioxidant defenses are unfavorable (Machlin, 1987; Sies, 1991). A well-recognized result of oxidant injury is peroxidation of lipids. An antioxidant is any substance that can delay or prevent cellular oxidation damage caused by free radicals and may act in numerous ways, ranging from prevention of their formation to their interception once formed. Antioxidants may be broadly classified into enzymatic (superoxide dismutase, catalase, and glutathione or their precursors) or non enzymatic (vitamins C, E, ß-carotene, and allopurinol) (Jankov et al., 2001).
A potential role for vitamin C, vitamin E, and carotenoids in modifying the risk of conditions that may result from oxidative stress has stimulated intense research efforts, increased interest in micronutrients supplements, and heightened consumers interest in these compounds (Rock et al., 1996).
A widespread perception is that the use of antioxidant therapies is a good therapeutic objective. This idea is based on evidence in very few human disease processes. Certainly, antioxidant interventions have been effective in various animal disease models (Jankov et al., 2001). Several reports have suggested that vitamin C has antioxidant properties to protect against oxidative damage caused by free radicals (Gangadharan et al., 2001; Bindhumol et al., 2003). Trials have been conducted to examine the effect of antioxidants such as vitamin C, vitamin E, or others in preventing or ameliorating some of the pathologies of diseases in which free radicals have been implicated (Liu et al, 1998).
The purpose of this study was to examine the effects of 15 days of dietary vitamin C on the oxidative status of the liver and the kidney tissues of mice, under natural conditions and with a single dose antimicrobial injection with ciprofloxacin (CFX). We have previously reported that administration of this fluoroquinolone (10 mg/kg) produced maximal concentration of lipid hydroperoxides (LOOH) after 15 minutes (Weyers et al., 2002). The production of LOOH, mediators of lipid peroxidation, and thiobarbituric acid reactive substances (TBARs), products of lipid peroxidation, were quantified in the liver and the kidney tissues of mice maintained with vitamin C in drinking water during 15 days, with or without one injection of CFX 15 minutes before sacrifice.
Materials and methods
Animals and treatment
Eight- to twelve-week-old male and female Balb C mice (total n= 20) were used in the experiments. Animals were distributed into four groups. The control group was administered with water for 15 days. The second group received vitamin C (100 mg/l) in drinking water, which provided 1 g/day of vitamin C, during 15 days. The mice in the third group had access to water and were only administered with CFX 15 min before they were sacrificed. The mice in the fourth group received vitamin C dissolved in water, during 15 days, and 15 min CFX. The food, the autoclaved potable water, and the vitamin C dissolved in this water were provided to the mice ad libitum , in dark bottles. Vitamin C was Redoxon drops (Roche, 200 mg/ ml).
At the end of the experimental period (15 days), 10 mg/kg CFX single dose (Parafarm), 0.5 ml/25 g of body weight, was intraperitoneally injected and mice were sacrificed after 15 min. The livers and kidneys were removed and stored at -20ºC until analyzed. The organs were homogenized in 3 parts (w/v) of buffer phosphate (pH 7.2-7.4) in a tissue homogenizer with a teflon pestle at 4ºC.
Analysis of lipid hydroperoxides levels
After the tissues were homogenized, the lipids were extracted twice with chloroform/ methanol (2:1, v/v) containing 0.03% of butylated hydroxytoluene (BHT). The organic layers were evaporated to dryness and the lipid extract was dissolved in 300 μl chloroform: methanol (1: 2, v:v) for LOOH determination. LOOH levels were evaluated through the reaction with 1-napthyldiphenilphosphine (NDPP) into its oxide (ONDPP) in a black tube at 60ºC during 60 min (Tokumaru et al., 1995). ONDPP generated stoichiometrically was quantified by high performance liquid chromatography, reverse-phase ODS C18 column at 292 nm. The mobile phase was methanol: water (90: 10) and flow rate 1.0 ml/ min. The results were expressed as nmol of LOOH/ g of tissue. LOOH values were calculated using tert-butyl hydroperoxide as a standard with a linear detection from 0.0312 to 1.0 mM and a coefficient of correlation of 0.991.
TBARs concentration
As a marker of lipid peroxidation products, the hepatic and the renal TBARs contents were measured according to the original method of Buege and Aust (1978) modified for tissues by Marcincak et al. (2003). Six hundred microliters of 30% (w/v) liver and kidney homogenate was mixed with BHT as an antioxidant and trichloroacetic acid 15%, vortexed, and incubated for 30 min at 90ºC. After cooling and centrifuging the mixture for 10 min at 2500 rpm, 600 ul of supernatant were vortexed with an equal volume of ClH 0.25 N and thiobarbituric acid 0.375%. The reaction mixture was then heated again for 30 min at 90ºC, extracted with two volumes of butanol, and centrifuged. With malondialdehyde (MDA) (Sigma) as the standard, the colored layer was measured at 532 nm. The TBARs concentrations (measured as MDA) were calculated as nmol/g of tissue.
Statistical analysis
ANOVA and Tuckey as a post-test were performed to analyze the statistical differences between groups (p≤0.05) (PRISM, 1997).
Results
The results of the LOOH levels and TBARs determinations in liver and kidney tissues of mice after 15 days of vitamin C administration in drinking water and with or without ciprofloxacin treatment are shown in Table 1. TBARs concentrations, expressed as nmol of TBARs/g of tissue, were 13 ± 0.7 in kidney and 12± 0.6 in liver in the control group. The group treated with CFX did not significantly differ from the control group. The two vitamin C groups (with and without CFX) had very similar results as the control group.
TABLE 1. Lipid hydroperoxides (LOOH) and MDA (equivalent to TBARs) levels in kidney and liver of mice after 15 days of vitamin 100 mg/Kg in drinking water and ciprofloxacin 10 mg/Kg io single dose.
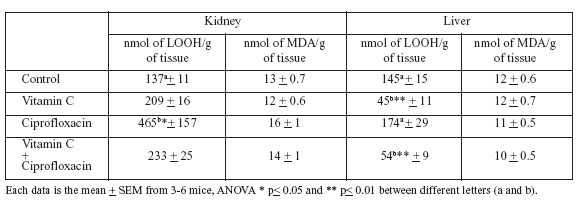
On the other hand, LOOH levels in the liver and the kidney tissues of mice in the control group showed 137 ± 11 nmol of LOOH/g of kidney tissue and 145± 15 nmol of LOOH/g of liver tissue. Mice in the group administered vitamin C showed 209 ± 16 nmol of LOOH/g of kidney tissue and 45 ± 11 nmol of LOOH/g of liver tissue (p<0.01). CFX 10 mg/kg injection 15 min after administration produced oxidative stress in the kidney to 465 ± 167 nmol of TBARs/ g of tissue.
Fifteen days of vitamin C administration induced a significant difference in the natural levels of LOOH in the liver tissues, but not a significant difference in the kidney tissues. The pre-treatment with vitamin C reduced the levels of LOOH in the liver to 45 ± 11 (p<0.01), and in the mice with 15 min CFX vitamin C treatment reduced the levels of LOOH in the liver to 54 ± 9 (p<0.01) nmol of LOOH/g of tissue. The difference in the levels of LOOH in the kidney between these two treatments (vitamin C with and without CFX) was 465 ± 157 compared to 233 ± 25 nmol of LOOH/g of tissue (p<0.05). The pre-treatment with vitamin C resulted in 12 ± 0.7 nmol of TBARs/g of liver tissue, and with the addition of the CFX injection, 10 ± 0.5 nmol of TBARs/ g of liver tissue. The TBARs levels for the kidney tissue had similar values to those of the liver tissue. The various treatments did not result in different TBARs levels as they did for LOOH levels.
Discussion
LOOH levels in the liver and the kidney tissues of mice were detected by the NDPP method (Tokumaru et al., 1995), which has proven to be sensitive and reliable for determining oxidative stress in mammalian organs (Weyers et al., 2001).
The LOOH content was significantly lower in the groups treated with vitamin C than in the control group. Many studies have associated LOOH with serious pathological conditions of oxidative mechanisms, early induced tissue damage (Cheeseman, 1993; Coyle and Puttfarcken, 1993; Díaz Velez et al., 1996), and normal aging process (Pratico et al., 1995).
It is recognized that, primarily, the liver and, secondarily, the kidney are the major organs of biotransformation in animals. Similar to the oxidative effect of CFX, which is more notable in the kidney than in the liver, the physiological protective capacity was less efficient in the kidney than in the liver, which was indicated by the slight induced levels of LOOH. The kidney was more sensitive than the liver to the oxidative effect, as has been previously reported (Weyers et al., 2002).
In this study, the levels of LOOH in the liver were significantly lower in the group treated with vitamin C and in the group treated with vitamin C plus CFX, decreasing the oxidative stress induced by ciprofloxacin (p<0.01). A vitamin C supplement can improve the oxidative status in the liver and is diminishes the lipid peroxidation induced in the mouse kidney. This possibility is exciting and may stimulate new research in this area. TBARs are products of lipid peroxidation and were quantified using MDA as the standard. Although the reactivity of TBARs is not only limited to MDA, this method is one of the most commonly used for detecting oxidative modification of lipids, and hence oxidative stress. The TBARs method did not demonstrate the effects observed by NDPP method, perhaps due to a different sensibility of the technique or to different end points of detection, that is products (TBARs method) versus intermediates of lipid peroxidation (NDPP method) respectively.
The present results indicate that in the doses used, pre-treatment with vitamin C selectively decreases the oxidative stress in the mouse liver. It would be interesting to evaluate in a future study if the antibiotic effect of ciprofloxacin changes in the presence of vitamin C. The interest in vitamin C and antioxidants stems from the growing understanding that a substantial number of free radicals are generated as an ordinary process of oxygen metabolism and that a wide range of conditions are associated with either an excess generation of free radicals or an inadequate antioxidant defense system (Cheeseman and Slater, 1993). Several clinical trials involving vitamin C, vitamin E, and beta carotene supplements were underway, predominantly as cancer chemoprevention, in an attempt to demonstrate that the presumed imbalance could be reversed and the conditions that were possibly due to oxidative stress could be rectified (Malone, 1991; Rock et al., 1996). In the present work, vitamin C could reduce the CFX induced oxidative stress in kidney and liver, and the physiological oxidative level in the liver could also been reduced.
Adequate doses of micronutrients are another area of interest and controversy. The adequate intake in humans was estimated to be either 200 mg/day from 5 servings of fruits and vegetables or 100 mg/day of vitamin C to prevent deficiency with a margin of safety (Levine et al., 1999). Maximal absorption is attained by the ingestion of several spaced doses of less than 1 g throughout the day, similar to the schedule in our study, rather than the ingestion of a single mega dose (Rock et al., 1996). Quantification of LOOH through NDPP oxidation remains a sensitive method for estimating lipid peroxidation, and adequate doses of vitamin C can be considered as a good an effective in vivo antioxidant pre-treatment for diminishing lipid natural or induced hydroperoxides levels in different organs.
Acknowledgements
We are indebted to Oscar Masciarelli, Santiago Saldaño, and Alejandro Tirone for their technical assistance. We greatly appreciate the initial collaboration of Dr. Carlos Chesta in the NDPP synthesis. This study was supported by the Secretaría de Ciencia y Técnica, Universidad Nacional de Río Cuarto.
References
1. Bindhumol V, Chitra KC, Mathur PP (2003). Bisphenol A induces reactive oxygen species generation in the liver of male rats. Toxicology 188: 117-124. [ Links ]
2. Buege J, Aust S (1978). Microsomal lipid peroxidation. Method Enzymol 52: 302-310. [ Links ]
3. Cheeseman KH (1993). Lipid peroxidation and cancer. In: DNA and Free Radicals. B. Halliwell and O.I. Aruoma Eds, New York: Ellis Horwood, pp. 109-144. [ Links ]
4. Cheeseman KH, Slater TF (1993). An introduction to free radical chemistry. Br Med Bull. 49: 281-493. [ Links ]
5. Coyle JT, Puttfarcken P (1993). Oxidative stress, glutamate, and neurodegenerative disorders. Science 262: 680-695. [ Links ]
6. Díaz Velez CR, García Castañeiras S, Mendoza Ramos E, Hernandez Lopez E (1996). Increased malondialdehyde in peripheral blood of patients with congestive heart failure. Am Heart J 131: 146-152. [ Links ]
7. Gangadharan B, Murugan MA, Mathur PP (2001). Effect of methoxychlor on antioxidant system of goat epididymal sperm in vitro. Asian J Androl 3: 285-288. [ Links ]
8. Jankov RP, Negus A, Tanswell AK (2001). Antioxidants as Therapy in the Newborn: Some Words of Caution. Pediatric Research 50: 681-687. [ Links ]
9. Levine M, Rumsey SC, Daruwala R, Park JB, Wang YH (1999). Criteria and recommendations for vitamin C intake. J Am Med Assoc 281: 1415-1423. [ Links ]
10. Liu T, Stern A, Roberts LJ, Morrow JD (1998). The isoprostanes: Novel prostaglandin- like products of the free radical- catalyzed peroxidation of arachidonic acid. J Biomedical Science. 6: 226-235. [ Links ]
11. Machlin LJ (1987). Free radical tissue damage: protective role of antioxidant nutrients. FASEB J 1: 441-446. [ Links ]
12. Malone WF (1991). Studies evaluating antioxidants and beta- carotene as chemopreventives. Amer J Clin Nutr 53 (suppl): 305S-313S. [ Links ]
13. Marcincak S, Sokol J, Turek P, Rozanska H, Dicakova Z, Mate D, Popedka P, Kozim P (2003). Comparative evaluation of analytical techniques to quantify malondialdehyde in broiler meat. Bull Vet Inst Pulawy 47: 491-496. [ Links ]
14. Pratico D, Reilly M, Lawson J, Delanty N, Fitzgerald GA (1995). Formation of 8-iso-prostaglandin F2by human platelets. Agents Actions 45 (suppl): 27-31. [ Links ]
15. PRISM (1997). GraphPad Software, Inc., San Diego, CA 92121 USA. [ Links ]
16. Rock CL, Ajacob R, Bowen P (1996). Update of the biological characteristics of the antioxidant micronutrients: vitamin C, vitamin E, and the carotenoids. J Am Dietetic Association 96: 693-702. [ Links ]
17. Sies H (1991). Oxidative stress: Introduction. In: Oxidative Stress: oxidants and Antioxidants. H. Sies Ed, New York, NY, Academic Press. [ Links ]
18. Tokumaru S, Tsukamoto I, Iguchi H, Kojo S (1995). Specific and sensitive determination of lipid hydroperoxides with chemical derivatization into 1-naphthyldiphenylphosphine oxide and high-performance liquid chromatography. Anal Chim Acta 307: 7-10. [ Links ]
19. Weyers A, Gorla N, Ugnia L, García ovando H, Chesta C (2001). Increase of tissue lipid hydroperoxides as determination of oxidative stress. Biocell 25(1): 11-15. [ Links ]
20. Weyers A, Ugnia L, García Ovando H, Gorla N (2002). Ciprofloxacin increases hepatic and renal lipid hydroperoxides levels in mice. Biocell 26(2): 225- 228. [ Links ]
Received on December 11, 2006.
Accepted on October 26, 2007.














