Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Biocell
versión impresa ISSN 0327-9545
Biocell v.32 n.2 Mendoza jun./ago. 2008
Morphologic and morphometric analysis of testis of Pseudis limellum (Cope, 1862) (Anura, Hylidae) during the reproductive cycle in the Pantanal, Brazil
Adelina Ferreira1, Mahmoud Mehanna1,2, and Cynthia P. A. Prado3
1. Departamento de Biologia e Zoologia, Universidade Federal do Mato Grosso (UFMT), Brasil.
2. Departamento de Morfologia, Universidade Estadual Paulista Júlio de Mesquita Filho (UNESP- Campus de Botucatu), Brasil.
3. Departamento de Morfologia e Fisiologia Animal, Universidade Estadual Paulista Júlio de Mesquita Filho (UNESP-Campus de Jaboticabal), Brasil.
Address correspondence to: Prof. Dra. Adelina Ferreira. Departamento de Biologia e Zoologia, Instituto de Biociências, Universidade Federal do Mato Grosso (UFMT), CEP 78040-400, Cuiabá, MT, BRASIL. Fax: (+55-65)3615 8878. E-mail: adelina@ufmt.br
ABSTRACT: The spermatogenesis of Pseudis limellum, from the Southern Pantanal, Brazil, was studied from July 1995 to May 1996, through histological sections of the testis. The cells could be differentiated as: primary spermatogonia, large cells, generally with bilobed nucleus; secondary spermatogonia, smaller cells, with darker cytoplasm, chromatin of radial form; primary and secondary spermatocytes, differentiated according to the different stages of the nucleus during the successive cells divisions. Furthermore, we observed cells in process of morphologic differentiation: rounded spermatids much smaller, with nucleus containing chromatin in compacting process and cytoplasm reduction; elongated spermatids, generally parallel organized in well defined bundles, with the anterior region directed toward the periphery of the seminiferous tubule and the tail directed toward the lumen. Spermatozoa are free in the lumen of the seminiferous tubule. All the cells are organized as cysts, which are supported by a large amount of Sertoli cells. The spermatogenesis in P. limellum is very similar to that of other anurans, but peculiarities were observed regarding the organization of the germ cells, the great amount of free Sertoli cells in the lumen of testis collected in May, and the long cytoplasmatic extensions of the cells bearing pigments and involving the seminiferous tubule. The diameter of the seminiferous tubule (SD) exhibited an annual mean of 251.79 ± 37.57 μm. Spermatozoa number by seminiferous tubule (SN) exhibited an annual mean of 306.66 ± 39.83, also with higher and lower values at each month. Variations in SD and SN were not significantly correlated with climatic variables. In this species, reproduction occurs throughout the year in ponds and flooded areas, despite the seasonal climate
of the Pantanal. Although males varied in their annual reproductive activity, they were considered potentially reproductive in all months throughout the year.
Key words: Spermatogenesis; Male; Amphibia; Climatic influences
Introduction
The reproductive activity in anurans is correlated with the climatic conditions of the environment where they live. Changes in temperature, photoperiod, and rainfall affect spermatogenesis and establish continuous or discontinuous cycles (Jorgensen, 1992; Huang et al., 1997). Species inhabiting temperate regions show a defined annual gonadal cycle. Alternations in the activity and quiescence periods are related to the climatic annual cycles (Rastogi, 1976; Jorgensen et al., 1986). Species that inhabit tropical regions, with a hot and constant humid climate, exhibit continuous or potentially continuous cycles (Inger and Greenberg, 1963; Rastogi, 1976; Jorgensen et al., 1986; Montero and Pisanó, 1990; Prado et al., 2005). In Brazil, studies on the reproductive aspects of anurans describe mainly reproductive behaviors (e. g., Bastos and Haddad, 2002; Lima and Keller, 2003; Brasileiro and Martins, 2006), but few studies focus on the reproductive cycles under a morphological analysis (e.g., Oliveira et al., 2003; Costa et al., 2004).
The spermatogenesis process is very similar among the different groups of vertebrates. The most pronounced differences among them, including anurans, refer to the seminiferous tubules arrangement. Interesting features of the spermatogenesis of frogs were described based on light microscopy of members included in the family Hylidae (Hermosilla et al., 1983; Rastogi et al., 1988; Oliveira et al., 2002; Oliveira et al., 2003). This work describes some aspects of the seminiferous tubule morphology of the hylid frog Pseudis limellum.
Pseudis limellum adults are small size frogs, snoutvent length of 15.9 - 23.0 mm for females and 14.4 - 20.2 mm for males (Prado and Uetanabaro, 2000). They are typically aquatic (Gallardo, 1988), occurring in open habitats, such as the Chaco and Cerrado, but they are also found in forest habitats (Bosch et al., 1996). This species can withstand periods of drought in small volumes of water (Gallardo, 1988). Frost (2007) mentions its distribution only for Paraguay, Uruguay, Bolivia, and northern Argentina. Few studies were conducted on P. limellum biology (Kehr and Basso, 1990; Bosch et al., 1996; Prado and Uetanabaro, 2000), and no studies were made on gonad morphological aspects.
Herein we describe the annual reproductive cycle of males of P. limellum. We described spermatogenesis and verified testis histological variations along the year. Such variations were correlated with the seasonal climate of the Pantanal, where the species occurs in high densities throughout the year.
Materials and Methods
Study Area
The Pantanal is a floodplain with a markedly seasonal climate. Although seasonal floods are common in the region, they are not related to local rainfall but to drainage constraints, evidenced by the slow flow of waters (Amaral Filho, 1986). The exact period and intensity of floods may vary from year to year (Brown Jr., 1984). Climate is hot and humid, with a dry season extending from May to September and a rainy period from October to April. Rainfall, however, is low, with a total of 1,070 mm between July 1995 and June 1996 recorded for the municipality of Corumbá, state of Mato Grosso do Sul, 50 km apart from the sample locality. For the same period, mean monthly air temperature was 25.1ºC, varying from 21.4ºC to 27.7ºC (Soriano, 1997). Floods are common from January to April (Amaral Filho, 1986).
Specimens data
Adult males of Pseudis limellum (N = 36) were collected in the Southern Pantanal (19º34'S, 57º00'W), municipality of Corumbá, state of Mato Grosso do Sul, Brazil. Individuals were collected from July 1995 to May 1996, except for the months January and March 1996. Specimens were measured and prepared according to McDiarmid (1994).
Histological analysis
Testis were removed and fixed by immersion for overnight at 4ºC in 10% formalin, dehydrated in a graded series of ethanol, and embedded in methacrilate glycol resin (Historesin Leica®). The sections (2 μm) were stained with 1% toluidine blue, observed, and photographed under an Olympus BX 60 microscope. The recognition of cells was made according to Hermosilla et al. (1983).
Morphometric and statistical analysis
For an individual of each month, we measured the seminiferous tubule diameter (SD) from ten transversal oriented tubules, which were randomly selected from five different sections in each testis, next to the testes core. The software Image Pro-plus® was used during these analysis. Mean monthly diameter, mean annual variation, and standard deviation were calculated. Spermatozoa from ten tubules, exhibiting similar diameter and randomly selected from five different sections of each testis, were counted (SN - spermatozoa number), and mean monthly number, mean annual variation, and standard deviation were calculated.
Mean monthly air temperature and total rainfall for the years 1995 - 1996 were registered by the Climatic Station of the National Institute of Meteorology (INMET), in the municipality of Corumbá (19º05'S, 57º30'W; 130 m elevation), state of Mato Grosso do Sul, Brazil, and obtained from Soriano (1997). To verify monthly variation in SD and SN, one-way analysis of variance (ANOVA) was performed (Zar, 1999). To test for the influence of the climatic variables on the changes in SD and SN, a non-parametric analysis of correlation, Kendall's coefficient (Zar, 1999), was made among the variables monthly total rainfall, mean monthly air temperature, SD, and SN.
Results
Primary spermatogonia are characterized by their basal location within the seminiferous tubule, they are much larger than the other germ cells and their nuclei are frequently bilobed, with loose chromatin and a wellstained cytoplasm (Figs. 1-5). Secondary spermatogonia are smaller than the primary spermatogonia and characteristically form cysts; the cytoplasm is darker and the chromatin in the nuclei forms more evident clusters (Figs. 1, 3, 4, 6).

FIGURE 1. General aspect of the seminiferous tubule. Note the germ cells in a cystic organization, supported by Sertoli cells and free spermatozoa in the lumen. Melanomacrophage cells (mm) involving all seminiferous tubule. SC: Sertoli cell, G1: primary spermatogonia, G2: secondary spermatogonia, C1: primary spermatocyte, C2: secondary spermatocyte, T1: early spermatid, T2: late spermatid, Z: spermatozoa.

FIGURES 2-13. Sertoli cells (SC), primary spermatogonia (G1), secondary spermatogonia (G2), primary spermatocytes (C1), secondary spermatocytes (C2), early spermatids (T1), late spermatids (T2), spermatozoa (Z).

FIGURES 11 and 12. Elongated spermatids in along nuclear and chromatin compacting process. To observe the formation of the different regions of the spermatozoon: nucleus (n), middle piece (mp), residual cytoplasm (rc), flagellum (f), proacrosomal vesicle (av).
Primary spermatocytes are generally irregular spheres, with large, vesicular nuclei (Figs. 1-4, 7, 8). They divide into the secondary spermatocytes that are found in small quantities. The secondary spermatocytes are half the size of the primary spermatocytes and are found closer to the lumen of the seminiferous tubule. Their nuclei are usually dark and well stained and the cytoplasm may be thinner on one side (Figs. 1 and 3).
The spermatids, resulting from the second meiotic division, are smaller and have condensed irregular nuclei. The cysts of the spermatids resemble bundles. The rounded (or early) spermatids have spherical, extremely condensed nuclei, and may present a lateral bulge that is most likely the proacrosomal vesicle (Figs. 1-3, 9, 10). Long (or late) spermatids that are slightly thicker than the spermatozoa are also observed. They have long nuclei, and highly condensed chromatin (Figs. 1, 2, 4, 9-13).
The spermatozoa are extremely long and point towards the edge of the seminiferous tubule; they have a continuous nucleus with an anterior acrosome. The middle part of each spermatozoon is not visible but the tail can be seen in the lumen as an extension composed of gray filaments, approximately four times the length of the head. In all cross-sections of the testicle, the bundles of spermatozoa can be cut at right angles or tangents (Figs. 8-10, 13). The spermatids and the spermatozoa are dependent on external nourishment, which is why the heads always converge towards the cytoplasm of the Sertoli cells. These cells are relatively large, with basal columnar nuclei, evident nucleoli and slightly darker cytoplasm (Figs. 1-4, 7). Testicles collected in May showed Sertoli cells in the region of the lumen (Fig. 9).
During differentiation of the primary and secondary spermatocytes, several morphological variations occur, especially of the nucleus. During these changes the chromatin presents the most varied forms from relatively rough bulges to filaments arranged in adjacent rows (Fig. 8). The chromatin can also be arranged converging towards the edge of the nucleus. The granules of chromatin become small and aggregated to the filaments that are much shorter and denser (Fig. 7).
Monthly analysis of the spermatogenesis stages indicated no significant variations concerning the presence of germ cells in the seminiferous tubule and all spermatogenesis stages, including free spermatozoa, were observed in all months (Fig. 14a-14i). For all months studied, mean diameter of the seminiferous tubule (SD) was 251.79 ± 37.57 μm, ranging from 209.94 μm in December to 322.30 μm in May (Fig. 15). For the same period, spermatozoa number by seminiferous tubule (SN) showed an annual mean of 306.66 ± 39.83, with numbers varying from 249 in April to 377 in February. The statistical analysis revealed a significant variance in SD (ANOVA, F = 7.98, df = 89, P < 0.0001, N = 9), but not in SN (ANOVA, F = 0.91, df = 89, P = 0.51, N = 9). Neither SD nor SN correlated significantly with monthly rainfall and monthly air temperature (P> 0.05) (Fig. 15).
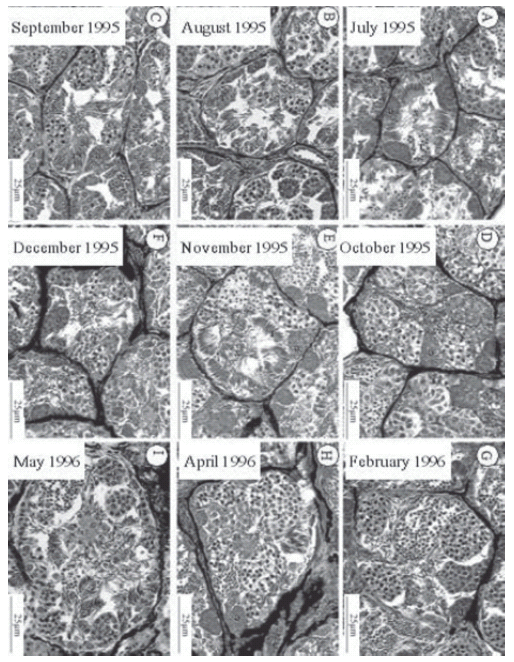
FIGURE 14 (A-I). Histological images showing the general aspect of the seminiferous tubule of L. limellum throughout the year. In all figures: spermatogonia (G), spermatocytes (C), spermatids (T) and spermatozoa (Z).

FIGURE 15. Monthly total rainfall (dashed line) and variation in the seminiferous tubule diameter in μm (SD: squares) and spermatozoa number (SN: triangles) in Lysapsus limellum in the southern Pantanal, Brazil, between 1995 and 1996.
Discussion
According to Oliveira et al. (2003), spermatogenesis occurs in structures called seminiferous tubules, where the germinal epithelium is organized into spermatocysts. In Scinax fuscovarius each cyst contains cells at the exact same stage of differentiation (Oliveira et al., 2003), which agrees with that observed in P. limellum. Several anuran species, mainly those with a discontinuous reproductive cycle, have seminiferous tubules with various cysts and all the cysts contain cells at the same stage of cellular differentiation (Rastogi, 1976; Jorgensen et al., 1986). However, anurans with a continuous reproductive cycle (Inger and Greenberg, 1963; Rastogi, 1976; Jorgensen et al., 1986; Montero and Pisanó, 1990), such as P. limellum, show various cysts within the seminiferous tubules with cells at different stages of differentiation.
Spermatogenesis in P. limellum occurs in a very similar way to that described for other anurans (Hermosilla et al., 1983; Rastogi et al., 1988; Oliveira et al., 2002; Oliveira et al., 2003). This is especially true if the structural characteristics of the cell are observed individually, irrespective of the cysts. The differentiation of primary spermatogonia into either pale or dark sub-groups is very common in anurans (Rastogi et al., 1985; Rastogi et al., 1988; Oliveira et al., 2003). However, in P. limellum this difference was not observed. The secondary spermatogonia are very similar to the descriptions for other anurans. According to Lofts (1974) and Rastogi et al. (1988), this is a period of intense cellular division, when the cysts are formed. They remain until the phase where the spermatids become more rounded. The main modifications between the primary and secondary spermatogonia are the acute reduction of the cytoplasm and the change from a lobe-shaped nucleus to a spherical shape, both of which are very similar to that observed by Rastogi et al. (1988) and Oliveira et al. (2003). The primary spermatocytes are much larger than the secondary spermatogonia and the cysts are more spread as described by Lofts (1974) and Rastogi et al. (1988). The secondary spermatocytes are not easily differentiated from the primary spermatocytes. Descriptions by Hermosilla et al. (1983) only suggest that there are differences between the cysts, where the cysts of the secondary spermatocytes present intercellular spaces. However, this was not observed in P. limellum. In this species intercellular spaces only occur in some cysts of the spermatids. These intercellular spaces had been described by Lofts (1974) as vacuoles, a term that is quite inappropriate if we consider the meaning and function of a vacuole in cellular terms. According to Lofts (1974), these clear spaces do not occur in the center of the rounded cysts of the spermatids as observed in P. limellum. The cellular differentiation of rounded spermatids into elongated spermatids marks an important stage in the conformation of the seminiferous tubule as these cells are no longer organized into cysts but form bundles, a characteristic confirmed by Lofts (1974), Rastogi et al. (1988) and Oliveira et al. (2003).
Data on the ultrastructure of spermiogenesis for ranids (Zirkin, 1971; Sprando and Russel, 1988), hylids (Lee and Kwon, 1992; Taboga and Dolder, 1993), and leiuperids (Amaral et al., 1999; referred as leptodactylids), allowed us to classify the observed cellular types. This was especially true for the initial spermatids that presented a structure similar to a proacrosomal vesicle, also observed in lizards and other anurans (Lee and Kwon, 1992; Taboga and Dolder, 1993; Ferreira and Dolder, 2002). The acrosomal vesicle is formed by the fusion of innumerable vesicles deriving from the Golgi complex. These vesicles suffer a series of modifications that culminate in the formation of the acrosome, responsible in the spermatozoon for enzymatic function and in some cases for the perforation of the ovule during fertilization (Ferreira and Dolder, 2002).
The flagella of the elongated spermatids and the free spermatozoa in the lumen of the seminiferous tubules are easily observed in P. limellum, however it is difficult to have an accurate idea of their length and morphology in the resolution of the optic microscope. The region of the anuran spermatozoon that shows most ultra structure variation is the flagellum. The flagellum of most vertebrates is made up of a central axoneme, with a 9+2 microtubule pattern associated to the peripheral proteins. Characteristics peculiar to anurans, such as flagella with two axonemes (Asa and Phillips, 1988), the presence of an axial rod (Taboga and Dolder, 1993), and an undulating membrane (Garda et al., 2002) have been described. These variations are used extensively as taxonomic characters.
Few data exist on the morphology and function of cells similar to the pigmented and dispersed cells in the testicle of P. limellum. These pigmented cells resemble cells of the monocytic phagocyte system, where pigmented granules can be observed in the interior of the cytoplasm (Knight et al., 1992; Rund et al., 1998).
Descriptions of the morphophysiology of Sertoli cells during spermatogenesis and spermiation in anurans are wanting in the literature. On the basis of our observations of the morphology of these cells in seminiferous tubules of animals collected in May, two hypotheses can be raised: (1) the Sertoli cells in these anurans can become loose from the germinal epithelium and are eliminated together with the spermatozoa during spermiation; or (2) these cells present large enough cytoplasmic extensions to fill the lumen of the seminiferous tubules, supporting spermatozoa at this period of the annual reproductive cycle. This could characterize a short discontinuity in the reproductive activity, without provoking a discontinuity in spermatogenesis. These hypotheses are preliminary and need to be confirmed through refined electronic microscopy analyses that may assist in the understanding of some aspects of the morphophysiology of the Sertoli cells in anurans and their activity throughout the annual reproductive cycle.
The few morphological analyses to establish reproductive cycles on anuran gonads revealed some patterns. The majority of anurans in temperate regions exhibit a discontinuous and synchronized reproductive cycle (Ritke and Lessman, 1994). This type of cycle possesses a periodic interruption that is under the control of an endogenous rhythm, independent of environmental factors. In tropical anurans, the cycles may vary, but usually the maximum of reproductive activities is associated with hot and humid conditions; reproductive cycles are generally continuous (Saidapur, 1983) or potentially continuous, according to temperature variations. In continuous cycles, the seminiferous tubule present cysts in various maturation stages (Lofts, 1974; Huang et al., 1997). In contrast, in discontinuous cycles, the germ cells mature uniformly in each seminiferous tubule (Lofts, 1974; Huang et al., 1997), but as testis presents many seminiferous tubules, all the stages of cell differentiation can be found (Inger and Greenberg, 1963). In P. limellum, the cycle seemed to be of the continuous type, since all germ cell stages could be found in the same seminiferous tubule and differentiation occurred simultaneously.
Concerning the morphological aspect of germ cells, the pattern observed for P. limellum is very similar to what was described for other species within the same family, such as Scinax ranki (Taboga and Dolder, 1991), Hyla japonica (Lee and Kwon, 1992), Hypsiboas pulchellaus, H. andinus (Montero and Pisanó, 1992), and Scinax fuscovarius (Oliveira et al., 2003). The pattern exhibited by P. limellum was also similar to that observed for leiuperids, such as Physalaemus marmoratus (Aoki et al., 1969, referred as P. fuscomaculatus) and P. cuvieri (Oliveira et al., 2002), and the calyptocephalellid Calyptocephalella gayi (Hermosilla et al., 1983, referred as Caudiverbera caudiverbera). The presence of a great amount of pigmented cells in the interior of the testis is observed in the gonads of males of some anuran species (Oliveira et al., 2002). These pigmented cells can be found in different organs, constituting an extracutaneous pigmentary system of unknown function (Zuasti et al., 1998). Studies on these cells describing morphology, pattern of distribution in the testis, and variations throughout the annual reproductive cycle are scarce in the literature.
All spermiogenic stages and spermatozoa were found in higher or lower frequency in the histological analysis of P. limellum testes, from July 1995 to May 1996. Temperature, rainfall, and photoperiod are important extrinsic factors regulating anuran reproduction (Duellman and Trueb, 1986). Our results, however, did not show any correlation between testes size variation and climatic condition variation. The seminiferous tubule diameter and spermatozoa number were almost constant throughout the year. One possible explanation for the absence of correlation is that the species present a continuous reproduction in the region, or alternatively, that our sample was inadequate to verify such correlation. Finally, the morphometric analysis of testes of P. limellum confirmed the histological analysis.
The reproductive activity of the anurans that inhabit seasonal environments is generally associated with the rainy period, mainly in the tropics (e. g., Donnelly and Guyer, 1994; Bertoluci and Rodrigues, 2002; Prado et al., 2005). However, it is not uncommon, especially in tropical areas, to find anurans reproducing year-round (e. g., Prado et al., 2000, 2005). Anuran gametogenic cycles were classified by Lofts (1974), Saidapur (1983) and Huang et al. (1997) as continuous, potentially continuous, or discontinuous. Mature spermatozoa and all the spermiogenic stages were observed in the testes of P. limellum throughout the year, characterizing spermatogenesis as continuous. Males can potentially reproduce throughout the year, despite the seasonal climate of the Pantanal. Prado and Uetanabaro (2000) observed gravid females throughout the year during a study carried out in the same locality. Based on the presence of gravid females and calling males, these authors found that reproduction of P. limellum occurs throughout the year in the southern Pantanal, corroborating our results on reproductive cycle.
Acknowledgements
The authors thank C. Oliveira (UNESP, Ibilce) and NEPA-UFMT for his support and the use of facilities. We thank M. Uetanabaro (UFMS) for invaluable help in obtaining the material. A. Ferreira acknowledges CNPq for the post-doc fellowship (proc. # 150007/03-5) and FAPEMAT (proc. # 0769/2006) for the financial support for research. C. P. A. Prado acknowledges FAPESP for the post-doc fellowship (proc. # 04/00709-0) and for the financial support granted to the Laboratório de Herpetologia, UNESP, Rio Claro, SP (Biota/FAPESP proc. # 01/13341-3).
References
1. Amaral Filho ZP (1986). Solos do Pantanal Matogrossense. In: Simpósio Sobre Recursos Naturais e Sócio-Econômicos do Pantanal. Ed. Embrapa-Pantanal, pp. 91-103. [ Links ]
2. Amaral MJLV, Fernandes AP, Bao SN, Recco-Pimentel SM (1999).An ultrastructural study of spermiogenesis in three species of Physalaemus (Anura, Leptodactylidae). Biocell 23: 211-221. [ Links ]
3. Aoki A, Vitale-Calpe R, Pisanó A (1969). The testicular interstitial tissue of the amphibian Physalaemus fuscomaculatus. Z Zellforsch Mikrosk Anat. 98: 9-16. [ Links ]
4. Asa CS, Phillips DM (1988). Nuclear shaping in spermatids of the thai leaf frog Megophrys montana. Anat Rec. 220: 287-290. [ Links ]
5. Bastos RP, Haddad CFB (2002). Acoustic and aggressive interactions in Scinax rizibilis (Anura, Hylidae) during the reproductive activity in southeastern Brazil. Amphibia-Reptilia 23: 97-104. [ Links ]
6. Bertoluci J, Rodrigues MT (2002). Seasonal patterns of breeding activity of Atlantic rainforest anurans at Boracéia, Southeastern Brazil. Amphibia-Reptilia 23: 161-167. [ Links ]
7. Bosch J, De La Riva I, Márquez R (1996). The calling behaviour of Lysapsus limellum and Pseudis paradoxa (Amphibia: Anura: Pseudidae). Folia Zoologica 45: 49-55. [ Links ]
8. Brasileiro CA, Martins M (2006). Breeding biology of Physalaemus centralis Bokermann 1962 (Anura: Leptodactylidae) in southeastern Brazil. J Nat Hist. 40: 1199-1209. [ Links ]
9. Brown Jr KS (1984). Zoogeografia da região do Pantanal Matogrossense. In: Simpósio Sobre Recursos Naturais e Sócio-Econômicos do Pantanal. Ed. Embrapa-Pantanal, pp: 137-178. [ Links ]
10. Caldwell JP (1987). Demography and life history of two species of chorus frogs (Anura: Hylidae) in South Carolina. Copeia 1: 114-127. [ Links ]
11. Costa GC, Vieira GHC, Teixeira RD, Garda AA, Colli GR, Báo SN (2004). An ultrastructural comparative study of the sperm of Hyla pseudopseudis, Scinax rostratus, and S. squalirostris (Amphibia: Anura: Hylidae). Zoomorphology 123: 191-197. [ Links ]
12. Crump ML (1974). Reproductive strategies in a tropical anuran community. Misc Publ Mus Nat Hist Univ Kansas 61: 1-68. [ Links ]
13. Donnelly MA, Guyer C (1994). Patterns of reproduction and habitat use in an assemblage of Neotropical hylid frogs. Oecologia 98: 291-302. [ Links ]
14. Duellman WE (1988). Patterns of species diversity in anuran amphibians in the American Tropics. Ann Miss Bot Gard. 75: 79-104. [ Links ]
15. Duellman WE, Trueb L (1986). Biology of Amphibians. New York, McGraw-Hill. [ Links ]
16. Ferreira A, Dolder H (2002). Ultrastructural analysis spermiogenesis in Iguana iguana (Reptilia: Sauria: Iguanidae). Eur J Morphol. 40: 89-100. [ Links ]
17. Frost DR (2007). Amphibian species of the world: an online reference. Version 5.1 (10 October, 2007). Amer Mus Nat Hist., New York. Database available at: http://research.amnh.org/herpetology/amphibia/ [ Links ]
18. Gallardo JM (1988). Anfibios Argentinos. Parte 2. Amphibia y Reptilia (conservación). In: Projecto Herpetofauna 1: 105-111. [ Links ]
19. Garda AA, Colli GR, Aguiar-Júnior O, Recco-Pimentel SM, Bao SN (2002). The ultrastructure of the spermatozoa of Epipedobates flavopictus (Amphibia, Anura, Dendrobatidae), with comments on its evolutionary significance. Tissue Cell 34: 356-364. [ Links ]
20. Haas A (2003). Phylogeny of frogs as inferred from primarily larval characters (Amphibia, Anura). Cladistics Intern J Willihennig Soc. 19: 23-89. [ Links ]
21. Hermosilla IB, Urbina AP, Cabrera JP (1983). Spermatogenesis in the chilean frog Caudiverbera caudiverbera (Linne, 1758) (Anura, Leptodactylidae). Bol Soc Biol Concepción 54: 103-115. [ Links ]
22. Huang WS, Lin JY, Yu JYL (1997). Male Reproductive cycle of the toad Bufo melanostictus in Taiwan. Zool St. 14: 497-503. [ Links ]
23. Inger RF, Greenberg B (1963). The annual reproductive pattern of the frog Rana erythraea in Sarawak. Phisil Zool 36: 21-33. [ Links ]
24. Jorgensen CB, Shakuntala K, Vijayakumar S (1986). Body size, reproduction and growth in a tropical toad, Bufo melanostictus, with a comparison of ovarian cycles in tropical and temperate zone anurans. Oikos 46: 379-389. [ Links ]
25. Jorgensen CB (1992). Growth and reproduction. In: Environmental physiology of the amphibians. Feder ME, Burggren WW. (Eds). Chicago, Illinois, Univ. Chicago Press, pp: 439-466. [ Links ]
26. Juncá FA (1998). Reproductive biology of Colostethus stepheni and Colostethus marchesianus (Dendrobatidae), with the description of a new anuran mating behavior. Herpetologica 54: 377-387. [ Links ]
27. Kehr AI, Basso NG (1990). Description of the tadpole of Lysapsus limellum (Anura, Pseudidae) and some considerations on its biology. Copeia 2: 573-575. [ Links ]
28. Knight KR, Vario G, Hamilton JA (1992). Regulation of pinocytosis in murine macrophages by colony-stimulating factors and others agents. J Leucoc Biol. 51: 350-359. [ Links ]
29. Lee YH, Kwon AS (1992). Ultrastructure of spermiogenesis in Hyla japonica (Anura, Amphibia). Acta Zool. (Stockh) 73: 49-55. [ Links ]
30. Lima AP, Caldwell JP, Biavati GM (2002). Territorial and reproductive behavior of an Amazonian dendrobatid frog, Colostethus caeruleodactylus. Copeia 1: 44-51. [ Links ]
31. Lima AP, Keller C (2003). Reproductive characteristics of Colostethus marchesianus from its type locality in Amazonas, Brazil. J Herpetol. 37: 754-757. [ Links ]
32. Lofts B (1974). Seasonal changes in the functional activity of the interstitial and spermatogenetic tissues of the green frog, Rana esculenta. Gen Comp Endoc. 4: 550-562. [ Links ]
33. McDiarmid RW (1994). Preparing amphibians as scientific specimens. In: Measuring and monitoring biological diversity. Standard methods for amphibians. Heyer WR, Donnelly MA, McDiarmid RW, Hayek LC and Foster MS. (Eds.). Smithsonian Institution, Washington, USA. [ Links ]
34. Montero R, Pisanó A (1990). Ciclo espermatogénico de dos especies de Telmatobius del noroeste argentino. Amphibia-Reptilia 11: 97-110. [ Links ]
35. Montero R, Pisanó A (1992). El ciclo espermatogénico de Hyla pulchella andina: um análisis numérico. Acta Zool Lilloana 41: 173-180. [ Links ]
36. Oliveira C, Zanetoni C, Zieri R (2002). Morphological observations on the testes of Physalaemus cuvieri (Amphibia, Anura). Rev Chil Anat. 20: 263-268. [ Links ]
37. Oliveira C, Vicentini CA, Taboga SR (2003). Structural characterization of nuclear phenotypes during Scinax fuscovarious spermatogenesis (Anura, Hylidae). Caryologia 56: 75-83. [ Links ]
38. Prado CPA, Uetanabaro M (2000). Reproductive biology of Lysapsus limellum Cope, 1862 (Anura, Pseudidae) in the Pantanal, Brasil. Zoocriaderos 3: 25-30. [ Links ]
39. Prado CPA, Uetanabaro M, Haddad CFB (2005). Breeding activity patterns, reproductive modes, and habitat use by anurans (Amphibia) in a seasonal environment in the Pantanal, Brazil. Amphibia-Reptilia 26: 211-221. [ Links ]
40. Prado CPA, Uetanabaro M, Lopes FS (2000). Reproductive strategies of Leptodactylus chaquensis and L. podicipinus in the Pantanal, Brazil. J Herpetol. 34: 135-139. [ Links ]
41. Rastogi RK (1976). Seasonal cycle in anuran (Amphibia) testis: The endocrine and environmental controls. Boll Zool. 43: 151-172. [ Links ]
42. Rastogi RK, Di Meglio M, Di Matteo M, Minucci S, Iela L (1985). Morphology and cell population kinetics of primary spermatogonia in the frog (Rana esculenta) (Amphibia, Anura). J Zool. (London) 207: 319-330. [ Links ]
43. Rastogi RK, Bagnara JT, Iela BL, Krasovich MA (1988). Reproduction in the mexican leaf frog, Pachymedusa dacnicolor. IV. Spermatogenesis: A light and ultrasonic study. J Morphol. 197: 277-302. [ Links ]
44. Ritke M, Lessman C (1994). Longitudinal study of ovarian dynamics in female Gray treefrogs (Hyla chrysoscelis). Copeia 1: 1014-1022. [ Links ]
45. Rund CR, Chiristiansen JL, Johnson JC (1998). In vitro culture of melanomacrophages from the spleen and liver of turtles: Comments on melanomacrophage morphology. Pigment Cell Research 11: 114-119. [ Links ]
46. Saidapur SK (1983). Patterns of testicular activity in Indian amphibians. Indian Rev Life Sci. 3: 157-184. [ Links ]
47. Soriano BMA (1997). Caracterização climática de Corumbá-MS. In: Departamento de Difusão Tecnológica. Boletim de pesquisa, Embrapa-Pantanal, Brasil, pp: 25. [ Links ]
48. Sprando RL, Russel LD (1988). Spermiogenesis in the bullfrog (Rana catesbiana): a study of cytoplasmic events including cell volume changes and cytoplasmic elimination. J Morphol. 198: 303-319. [ Links ]
49. Taboga SR, Dolder H (1991). Análise histológica da espermatogênese de Hyla ranki (Amphibia, Anura, Hylidae). Rev Bras Cien Morfol. 8: 66-71. [ Links ]
50. Taboga SR, Dolder H (1993). Ultrastructural analysis of the Hyla ranki spermatozoan tail (Amphibia, Anura, Hylidae). Cytobios 75: 85-92. [ Links ]
51. Zar J (1999). Biostatistical Analysis. 4a ed. Prentice Hall, Upper Saddle River, New Jersey. [ Links ]
52. Zirkin BR (1971). The fine structure of nuclei during spermiogenesis in the Leopard frog, Rana pipiens. J Ultrastruct Res. 34: 159-174. [ Links ]
53. Zuasti A, Jimenez-Cervantes C, Garcia-Borron JC, Ferrer C (1998). The melanogenic system of Xenopus laevis. Arch Histol Cytol. 61: 305-316. [ Links ]
Received on July 14, 2007.
Accepted on March 9, 2008.














