Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Biocell
versión impresa ISSN 0327-9545
Biocell v.33 n.1 Mendoza ene./abr. 2009
Studies on the reproductive and developmental biology of Cichlasoma dimerus (Percifomes, Cichlidae)
Matías Pandolfi*, Maximiliano M. Cánepa, Fernando J. Meijide, Felipe Alonso, Graciela Rey Vázquez, M. Cristina Maggese and Paula G. Vissio
Departamento de Biodiversidad y Biología Experimental. Facultad de Ciencias Exactas y Naturales. Universidad de Buenos Aires. Ciudad Universitaria (C1428EHA). Buenos Aires, Argentina.
*Address correspondence to: Matías Pandolfi. Departmento de Biodiversidad y Biología Experimental, Facultad de Ciencias Exactas y Naturales. Universidad de Buenos Aires, Ciudad Universitaria, (C1428EHA) Buenos Aires, ARGENTINA. E-mail: pandolfi@bg.fcen.uba.ar
ABSTRACT: Many characteristics of the South American teleost fish Cichlasoma dimerus (body size, easy breeding, undemanding maintenance) make it amenable to laboratory studies. In the last years, many of the fundamental aspects of its reproductive and developmental biology have been addressed in our laboratory. Rather recently, the immunohistochemical localization of pituitary hormones involved in reproduction and in background color adaptation has been described in both adult and developing individuals, and the role of FSH in ovarian differentiation has been established. These findings have been correlated with mapping of some of their brain-derived controlling hormones. The latter include brain-derived gonadotropins which were shown to be active in vitro in the control of pituitary hormone secretions. The emerging picture shows C. dimerus as an interesting species in which many of their basic features have already been investigated and which conform a solid platform for comparative studies correlating neurohormones, pituitary hormones and behavior, from the molecular to the organismic level.
Key words: Reproduction; Early development; Neuroendocrinology; Brain; Pituitary; Teleosts
1. Teleost fishes in physiological and developmental studies.
Teleost fishes represent the most numerous and diversified vertebrate group. Though fish physiology has been studied for many decades (Ball and Baker, 1969; Hoar, 1969; Conlon, 2000; Price et al., 2008), the use of very different species, together with the idea that differences were more important than similarities, gathered a lot of data, and sometimes non coherent ones. During the last years, however, several research groups have put the emphasis in the importance of using teleost fishes not for comparative studies within the class, but as general vertebrate physiological models, by taking advantage of some of their unique properties. One of these properties is an unusual characteristic of the fish nervous system: fish brains can remain alive and responsive for many hours after removal from the animal (Teyler et al., 1981). This has allowed researchers to use intactbrain preparations that have the advantage, compared to mammalian brain-slice preparations, of allowing afferent and efferent connections to remain functional (Wayne et al., 2005). Another interesting feature is the morphological arrangement of fish neuroendocrine systems: in teleost fishes there is no median eminence or portal system; therefore nerve fibers originating from the hypothalamus should end in close contact with pituitary cells. Immunocytochemical studies have shown that neurohormonal fibers and nerve endings are in close association with pituitary endocrine cells. Because of this special anatomical arrangement, teleosts are a unique experimental model for determining putative brain peptides or monoamines involved in the regulation of pituitary endocrine cells (Peter et al., 1990).
Also, over the past 15 years, the cyprinid zebrafish Danio rerio (Hamilton, 1822) and the adrianichthyid medaka Oryzias latipes (Temminck and Schlegel, 1846) have become very popular fish models, due to their transparency during embryonic and larval stages, for investigating the molecular genetics that control organ development. The by-product of this huge amount of work is that their genes have been characterized providing fundamental tools for physiological and developmental studies (Furutami-Seiki and Wittbrodt, 2004; Crollius and Weissenbach, 2009).
Cichlid fishes (Perciformes) are also becoming intensively studied in the field of social control of reproduction and the most studied species have been the African tilapia Astatotilapia burtoni (Günther, 1894) and Oreochromis mossambicus (Peters, 1858) (Fox et al., 1997; Ogawa et al., 2006). The South American cichlid fish Cichlasoma dimerus (Heckel, 1840) has been studied for several years in our laboratory, at the morphological, developmental, physiological, and molecular levels. The aim of the current review has been to present a comprehensive picture of all the data obtained in this species, and to put them in the context of contemporary, comparative fish physiology.
2. Cichlasoma dimerus (Fig. 1A) in the laboratory and in nature.

FIGURE 1. Some behavioral aspects of Cichlasoma dimerus under laboratory conditions. A: Fin display and body colors of an adult male of high social rank. B: Prespawning behavior: the female nibbles at the spawning substrate (a flat stone), while the male is on guard. C: Spawning behavior: the female rubs the genital papilla while depositing the last egg rows. D: Parental care of the clutch: the female is removing unfertilized or infected eggs. Scale bars = 20 mm.
It is a medium sized fish (12 cm standard length) which is easily maintained and bred in the laboratory, where it tolerates a wide range of water compositions and temperatures (10 to 30ºC, the optimal breeding temperature is 26ºC) (Meijide and Guerrero, 2000).
The natural range of this species encompasses the entire system of the Paraguay river, the lower Alto Paraná, and the rest of the Paraná river basin up to the vicinity of Buenos Aires city (Kullander, 1983). Locality records are known from four countries (Bolivia, Brazil, Paraguay and Argentina) where it inhabits a wide variety of both lentic and lotic environments. Its common names are "chanchita" (Spanish) and "acará" (Portuguese) (Staeck and Linke, 1995; Casciotta et al., 2005).
Its ground color is very variable, depending both on the specimen's "mood" and social status. Its body may vary from greenish to light or dark grey and it may also show golden-yellow and light blue reflections. It shows several dark-brown vertical bands and two blotches, one in the middle region of the trunk and the other in the caudal peduncle. The eyes may show a bright red border, particularly in reproductively active individuals (Fig. 1A). This species has a moderately developed sexual dimorphism, with the males growing larger than the females. In males, soft rays of the distal edge of the dorsal and anal fins may be extended as filaments (Alonso et al., 2007).
As many cichlids do, C. dimerus has highly organized breeding activities, as they may be observed in the laboratory. A few days after fish have been transferred to a new aquarium, territories are progressively established and defended by both males and females. The dominant pair will aggressively defend the prospective spawning site (usually a flat stone) and will start to display stereotyped prespawning activities, such as jerking and quivering, digging in the gravel, and nipping off and nibbling at the spawning substrate (Fig. 1B).
Spawning will eventually initiate, with the female rubbing her genital papilla over the substrate and the sticky eggs being deposited in rows. After the female lays its eggs, the male slides its erected genital papilla over them, thus achieving fertilization. At the end of the spawning process, which may last up to 90 min, some 1500 eggs have been deposited as a uniform rounded layer over the substrate (Fig. 1C).
Spawning is followed by a period of parental care during which the cooperative pair guards the eggs, both by fanning them (alternately beating the pectoral fins over the clutch) and by removing dead eggs, which are usually attacked by fungi (Fig. 1D). At a water temperature of 26ºC, the larvae hatch at the beginning of the third day and are transferred by both parents to a previously dug pit. Another five days pass until the larvae swim freely, and during this time they are recurrently transferred to several other pits. Mortality during the first month of development is typically high (only about 20% of hatchlings reach the juvenile stage). If provided with enough food and space, the young will grow rapidly, and may reach about 50 mm four months after hatching. A pair may spawn every 20 days during the extended breeding season (September to April).
The foregoing behavioral description was based on the works of Greenberg et al. (1965) and Polder (1971) in Aequidens portalegrensis, which is a synonym of C. dimerus. A comprehensive monograph on cichlid behavior by Baerends and Baerends-Van Roon (1950) is also available.
3. Embryonic and larval development.
The freshly spawned eggs of Cichlasoma dimerus are ovoid-shaped and light brown colored (Meijide and Guerrero, 2000). They have a smooth translucent vitelline envelope surrounded by a mucous layer which allows them stick to the substrate and to one another at the time of spawning.
A detailed description of C. dimerus development from oocyte activation until the beginning of the juvenile stage is found in Meijide and Guerrero (2000). The micropyle is cone-shaped and located at the egg's animal pole. Most of the egg volume is occupied by numerous bright, tightly packed yolk globules. Polarization of the cytoplasm towards the animal pole becomes evident within 60 min after fertilization, giving rise to a prominent dome-shaped cytoplasmic layer known as the blastodisc (Fig. 2A).

FIGURE 2. Embryonic and larval development of Cichlasoma dimerus. A-E. Transmitted light microscopy. F-J. Incident light microscopy. A: Zygote stage (1 h); B: Blastula stage (6 h); C: Yolk plug stage (22 h); D: 15-somite stage (30 h); E: Prehatching stage (53 h); F: Post-hatching stage (55 h). G: Opened-mouth stage (108 h). H: Free-swimming stage (190 h). I: Prejuvenile stage (25 days). J: Juvenile stage (45 days). Ag, adhesive glands; bd, blastodisc; bl, blastoderm; e, eye; ea, embryonic axis; fv, finfold vessels; g, gills; h, heart; i, intestine; m, melanophores; mi, micropyle; op, optic primordium; ov, otic vesicle; rp, primordial rays; s, somites; sb, swim bladder; ve, viteline envelope; y, yolk; yp, yolk plug. Scale bars: A-E = 300 μm; F-H = 500 μm; I-J = 1 mm.
Cleavage is typically meroblastic, resulting in a blastula with a blastodermal cellular region above the yolk mass (Fig. 1B). The subsequent gastrulation process is characterized by several morphogenetic movements that result in a rearrangement of the blastoderm relative to the yolk. Towards the end of the first day of development, about 75% of the yolk mass has been covered by the blastoderm and the elongated embryo's shape is discerned along an anterior-posterior axis. The "yolk plug" is the portion of uncovered yolk protruding in the neighbourhood of the vegetal pole (Fig. 2C).
Organogenesis initiates soon thereafter, the optic vesicles being one of the first organ primordia that become distinct. Towards 26 h, paired somites start developing sequentially in an antero-posterior direction, on both sides of the notochord. Otoliths become visible within the otic vesicles (Fig. 2D). Before hatching, the embryo shows conspicuous muscular contractions. The beating heart is placed beneath the head and blood flow is readily observed. Brain regions become distinct and the lenses are evident within the eyes. Rudiments of the adhesive glands are observed at the midbrain-hindbrain boundary (Fig. 2E).
Larval hatching occurs 54 h after fertilization. After hatching, yolk-sac larvae are readily transferred by their parents to a pit dug in the gravel. These larvae present three pairs of adhesive glands over the head, this feature being characteristic of substrate-brooding cichlids. The mouth is not yet formed, and the gut is narrow and straight. No fins are differentiated but a primordial fin fold is already developed dorso-ventrally in the sagittal plane. Melanophores are scattered over the oblong yolk-sac and extend along the junction of the body with the ventral fin fold (Fig. 2F).
During the fifth day of development, the mouth opens and mobile jaws can be observed. The yolk-sac has become smaller. Branchial arches are differentiated and soon thereafter gill filaments will start to develop. These larvae do not swim freely yet, but they rapidly swing their tails while still attached to the substrate by their adhesive glands (Fig. 2G).
The free-swimming stage is reached five days after hatching, i.e. during the eighth day of development. The oval-shaped, refractive swim bladder becomes visible. Adhesive glands have been regressing, and will completely disappear during the following days. Caudal fin rays are well developed, while while both dorsal and anal fins consist of thin, transparent membranes. Melanophores are more heavily distributed over the head and trunk (Fig. 2H). A few hours after the onset of free-swimming, these larvae start to feed from the environment, though the yolk is not yet totally consumed.
Larval development continues with the progressive differentiation of the dorsal, anal and pelvic fins and with the acquisition of a pigmentation pattern that gradually resembles that of the adult (Fig. 2I).
The juvenile stage is reached around day 45 of development and their body shape and pigmentation pattern are similar to those of the adult (Fig. 2J). The fins become fully developed and the adult complement of rays is completed. The scales are present over the greatest part of the body, which is almost completely covered with melanophores.
4. Gonadal development and sex differentiation.
C. dimerus is a gonochoristic fish, in which ovaries and testes develop directly from undifferentiated gonads. A detailed description of early gonadogenesis in C. dimerus, including ultrastructural features, is found in Meijide et al. (2005). As it is known for other teleosts (Bruslé and Bruslé, 1983), the extent of gonadal development in C. dimerus depends both on age and size, and gonadal sex differentiation will occur earlier in specimens that grow faster. Therefore, the ages corresponding to the developmental events described in the next paragraphs should be considered only as estimations.
On day 12 posthatching, paired gonadal primordia are present in the posterior region of the abdominal cavity, immediately below the kidney, and are suspended from the dorsal peritoneal wall by short mesenteries. The gonadal primordium consists of large, round to oval germ cells surrounded by somatic cells. Germ cells contain large and central euchromatic nuclei, with prominent nucleoli. Occasional mitotic activity is observed in these cells. Somatic cells are of varying shapes. They have heterochromatic nuclei and a small cytoplasm (Fig. 3A). Gonads remain undifferentiated by day 38. However, the number of germ cells has augmented, and as a result, gonads increase in size and several germ cells (not just one or two as in earlier stages) appear in cross sections (Fig. 3B).
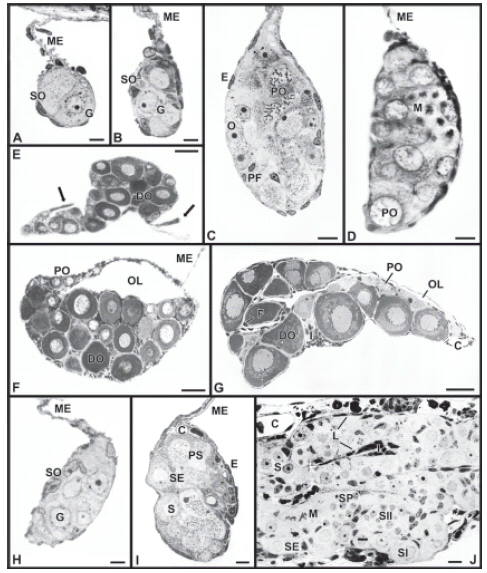
FIGURE 3. Gonadal development and sex differentiation in Cichlasoma dimerus. Toluidine blue. A: Primordial gonads on day 14 post-fertilization are characterized by the occurrence of a few germ cells with round nuclei, which are surrounded by some somatic cells. B: Sexually undifferentiated gonad on day 38. C: The appearance of meiotic figures is the first evidence of sexual differentiation in prospective females on day 42. D: Increased gonial mitotic activity accompanies the first cells undergoing meiosis towards day 50. E: Arrows indicate somatic cell outgrowths in a female on day 50, which will lead to the formation of the ovarian cavity. F: The ovarian cavity is already formed on day 65. Numerous basophilic diplotene oocytes are also observed. G: Ovarian follicles containing perinucleolar oocytes are observed by day 80. H: An undifferentiated gonad on a day 65 juvenile (a prospective male). I: Meiotic figures indicative of testicular differentiation are first seen on day 80 in a prospective male. J: Lobules are becoming distinct in a testis on day 100. Secondary spermatocytes (but no spermatids) are observed. Scale bars = 5 μm (A, B, H, I); 10 μm (C, D, J); 50 μm (E, F, G).; C, blood capillary; DO, diplotene oocyte; E, epithelial cell; F, follicle cell; G, germ cell; I, interstitial or stromal tissue; L, lobule; M, mitotic figure; ME, gonadal mesentery / mesovary / mesorchium; O, oogonium; OL, ovarian lumen; PF, prefollicle cell; PO, pachytene oocyte; PS, pachytene spermatocyte; S, spermatogonium; SE, Sertoli cell; SO, somatic cell; SP, spermatocyst; SI, spermatocyte I; SII, spermatocyte II.
Ovarian differentiation occurs at an earlier age than that of testicular differentiation, as it is usually observed in teleosts (Meijide et al., 2005). A prominent cytological feature marking the onset of gonadal differentiation is the appearance of meiotic figures in germ cells of the prospective ovary. By day 42, many oogonia have entered meiotic prophase and become growing primary oocytes, which in addition to a moderate oogonial and somatic cell proliferation result in an enlargement of the prospective ovary. For the first time, blood capillaries are observed in the gonadal dorsal region (Fig. 3C). The onset of ovarian meiosis is soon accompanied by an increase in germ cell mitoses. In addition, the ovary becomes either triangular or kidney-shaped in sections (Fig. 3D). A few days later, somatic reorganization of the developing ovary is evidenced by somatic cell proliferation, which forms appendix-like structures (Fig. 3E) which finally fuse to form the ovarian lumen. The ovarian cavity is formed completely by day 65, when numerous basophilic oocytes at the diplotene meiotic stage are also present (Fig. 3F). From days 80 to 100, the ovary consists mainly of follicles which contain characteristically large, perinucleolar oocytes with a basophilic cytoplasm: these previtellogenic oocytes are arrested at the diplotene stage of the first meiotic prophase and are characterized by the presence of multiple round nucleoli in the peripheral nucleoplasm. Each perinucleolar oocyte is covered by a continuous layer of flat follicle cells. Interstitial connective tissue is present both in the ovarian periphery and in the angular spaces between three or more follicles in the inner ovarian region (Fig. 3G).
In contrast to gonadal development in the female, signs of histological differentiation in the prospective males are not observed until day 72. Therefore, undifferentiated gonads on day 65 are certainly diagnosed as prospective testes. They retain the pearlike shape of undifferentiated gonads and are much smaller than the ovaries of females of the same age. Spermatogonia at this stage cannot be distinguished from the undifferentiated germ cells at day 38, and their most characteristic feature is a prominent nucleus with one or two nucleoli (Fig. 3H). By day 72, spermatogonial proliferation is occurring and blood vessels become evident in the dorsal region of the testis. On day 80, meiotic activity is apparent, and spermatocytes at early stages of meiotic prophase become numerous. Spermatocysts are formed by isogenic spermatocyte groups bound by the cytoplasmatic processes of Sertoli cells. Spermato-genesis progresses synchronously within each cyst. Spermatogonia are recognized as isolated cells, individually surrounded by the cytoplasm of a Sertoli cell (Fig. 3I). By day 100, the testis is divided into lobules composed by both Sertoli and germ cells, and which are separated by interstitial tissue. Spermatogenesis has reached the secondary spermatocyte stage (Fig. 3J).
Under laboratory conditions, mature gonads, i.e., ovaries containing full grown vitellogenic oocytes and testes containing mature spermatozoa can be observed in one year-old specimens, while the first spawnings are recorded towards month 16 after hatching.
5. The adult pituitary gland.
The gross morphology of C. dimerus pituitary gland is shown in figure 4. It is both an elongated and flat gland, in which the middorsal and caudal parts mostly consist of neurohypophyseal tissue. As it is found in other cichlids (e.g., Mattheij et al., 1971) the infundibular recess of the third ventricle is large and extends as branches that penetrate the adenohypophyseal tissue. The adenohypophysis is divided into the rostral pars distalis, the proximal pars distalis and the pars intermedia, the endocrine cells of these areas showing different tinctorial and immunohistochemical properties. The adenohypophysis is penetrated by neurohypo-physeal infoldings, particularly in the pars intermedia.

FIGURE 4. Sagittal section of the pituitary gland of an adult male Cichlasoma dimerus. Hematoxylin-eosin. The adenohypophyseal lobes (rostral -RPD- and proximal pars distalis -PPD-, and pars intermedia-PI) are indicated. The infundibular stalk (IS) is shown connecting the hypothalamus (HPT) to the pituitary (arrow). The neurohypophysis (NH) also shows its numerous infoldings into the pars intermedia, and in the rostral and proximal pars distalis too. Scale bar = 50 μm.
The distribution of endocrine cell types in the different adenohypophyseal lobes is summarized in figure 5. Information about the antisera used in this study and the molecular weights of the detected C. dimerus pituitary hormones (Western blot analysis) are summarized in Table 1.
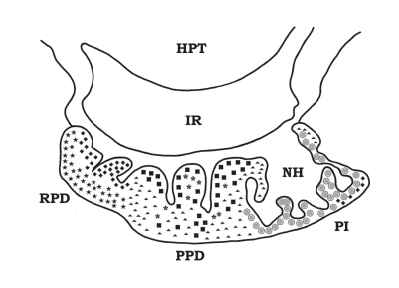
FIGURE 5. Distribution map of hormone-secreting pituitary cells, as shown by their immunoreactivity to specific antisera. ACTH cells (◆) and PRL (★) cells occur in the rostral pars distalis. GH (*), TSH (■) and GTH(▲) cells are mainly located in the proximal pars distalis, while SL (●) and a-MSH cells (◆) are located in the pars intermedia. HPT, hypothalamus; PI, pars Intermedia; RPD, rostral pars distalis; PPD, proximal pars distalis; NH, neurohypophysis; IR, infundibular recess. Redrawn from Pandolfi et al. (2001a).
TABLE 1. Antisera used in immunohistochemical and Western blot analysis and estimated molecular weight of Cichlasoma dimerus pituitary hormones.

Rostral pars distalis
This lobe of the adenohypophysis is invaded by narrow neurohypophyseal prolongations (Pandolfi et al., 2001a,b). In C. dimerus, two different cell types are typical of the rostral pars distalis, which are recognized by their immunoreactivity to antisera against prolactin and corticotropin (ir-PRL and ir-ACTH cells). Immunore-active PRL cells are relatively large cells which form a compact layer in the periphery of the rostral pars distalis, while ir-ACTH cells are arranged in narrow bands or small follicles along the neurohypophyseal branches but without forming a continuous layer. Immunocytochemical labeling with the ACTH antibody was also observed in the pars intermedia, where the cells reacting with a-melanocyte stimulating hormone (ir-aMSH) cells are located (see below). This cross-reactivity was likely due to the fact that both the corticotrops of the pars distalis and the melanotrops of the pars intermedia elaborate their distinctive hormones from the same large precursory molecule, namely propiomelanocortin (POMC) and that the whole a-MSH sequence is included in the ACTH mature molecule, so that the antisera against ACTH recognize a-MSH also (Eipper and Mains, 1981; Pandolfi et al., 2003).
Proximal pars distalis
In this region only some broad neurohypophyseal branches are present but they do not reach the ventral part of the pituitary. Four different cellular types are distinguished immunochemically with heterologous antisera against growth hormone, follicle-stimulating hormone, luteinizing hormone and thyrotropin (designated as ir-GH, ir-FSH, ir-LH and ir-TSH cells). Immunoreactive GH cells form a layer surrounding the centrally located neurohypophysis.
Particularly, the identification of ir-FSH cells using heterologous antisera was hindered by their low cross-reactivity with the endogenous FSH. Antisera raised against bFSH and bLH from the mummichog, Fundulus heteroclitus, were successfully used to recognize gonadotrops in several teleost fishes, since they recognize conserved regions of FSH and LH in teleosts (Shimizu et al., 2003a,b) and they were also successful in C. dimerus (Vissio et al., 1996; Pandolfi et al., 2001a). Gonadotrops are periodate reactive basophilic cells, which are localized mainly in the proximal pars distalis and the external border of the pars intermedia. Immunoreactive LH cells are located in central, ventral and marginal zones of the proximal pars distalis and in the external border of the pars intermedia. These cells are arranged in either cords or small groups along the whole proximal pars distalis and have conspicuous processes directed towards the neurohypophyseal infoldings and they showed a vacuolated cytoplasm (Pandolfi et al., 2006). Immunoreactive FSH cells are detected in central and ventral zones of the proximal pars distalis and also in the external border of the pars intermedia. Immunoreactive FSH cells are either spherical or polyhedrical cells, with a vacuolated cytoplasm, and they appear either isolated or 2-3 cells groups. In general, they are smaller and less numerous than ir-LH cells, and are more anteriorly located in the proximal pars distalis. Our immunocytochemical analysis revealed that FSH and LH are mainly produced in different cell popu-lations of the adult adenohypophysis in C. dimerus, as it was shown by both double-label immunohistochemistry and by the observation of adjacent sections (Pandolfi et al., 2006).
Finally, ir-TSH cells were seen forming discontinuous strands and small groups in the proximal pars distalis. However, their number was very small if compared with the other pituitary hormone cells of C. dimerus.
Pars intermedia
The small lobules and cell rows of the most caudal part of the pituitary gland are separated by numerous irregular branches of neurohypophyseal tissue. In adja-cent sections, somatolactin (SL) immunoreactivity was found in groups of periodate reactive cells which surround the most deeply located neurohypophyseal branches. In spite of the chemical similarities between GH, PRL and SL (Rand-Weaver et al., 1991; Takayama et al., 1991), no labeling with SL antisera was observed in either the rostral or the proximal pars distalis, where ir-GH and ir-PRL cells are located. Also, in adjacent sections, groups of periodate non-reactive cells can be detected in the pars intermedia, which corresponds to ir-aMSH cells.
Overview of our studies in the adult pituitary gland
A clear-cut lobar distribution of the common vertebrate pituitary hormones was found, with a localization of ir-PRL and ir-ACTH cells in the rostral pars distalis, the glycoprotein hormones ir-FSH, ir-LH and ir-TSH cells located in the proximal pars distalis (together with ir-GH cells), while the ir-SL cells (which are specific of fish) are located in the pars intermedia, where they are together with the non fish-specific ir-aMSH cells. In general, this lobar distribution would make the C. dimerus pituitary amenable to possibly interesting in vitro studies after microdissection and separate culture of the different lobes, since the chemically related PRL, GH and SL are produced in different lobes, as they are the chemically related ACTH and aMSH. Cells involved in the production of the glycoprotein family of gonadotropins and thyrotropin are located in a single lobe (the proximal pars distalis) but they would still be amenable to experimental manipulation with different putative releasing hormones.
6. The larval pituitary
The cellular organization of the pituitary gland during development in C. dimerus was described using immunocytochemical methods (Pandolfi et al., 2001b; 2006). Although several cytochemical investigations of pituitary cells ontogeny have been reported in teleosts (Mal et al., 1989; Cambre et al., 1990; Nozaki et al., 1990; Naito et al., 1993; Saga et al., 1993; Saga et al., 1999; Villaplana et al., 1997; Parhar et al., 1998), ours are the only studies performed in cichlid fishes. This has the unfortunate derivation, however, of the difficulty of making inter-familial or even inter-ordinal comparisons in such a diverse class as the Teleostei. In the following paragraphs, developmental stages are indicated as either hours or days after hatching.
Newly hatched larvae (1 h)
They show an incipiently differentiated brain, which is curved over the ventral part of the body, and a large yolk sac. At this stage, the brain and the pituitary gland cannot be distinguished from each other and no immunoreactive cells were detected.
Vitelline larvae (1.5 days)
The brain becomes straightened out and its ventricles are highlighted by the actively proliferating cells which surround them. Near the prechordal cartilage, which is in contact with the diencephalon, a mass of presumptive adenohypophyseal cells appears, but none of them is reactive to PRL, GH or SL antisera. At this stage ir-aMSH and ir-ACTH cells are detected in the posterior and anterior part of the developing pituitary, respectively. No immunoreactivity to either b-FSH, b-LH or b-TSH was detected.
Vitelline larvae (2 days)
The pituitary gland is recognized as a rounded structure hanging from the floor of the diencephalon and which is formed by morphologically similar epithelial cells. The neurohypophysis has not yet been differentiated. A few small ir-SL cells are scattered in the caudal part of the gland, showing strong immunoreactivity. Immunoreactive PRL cells also appear at this stage in the rostral area of the adenohypophysis. Some few ir-GH cells are immunostained weakly, and are distributed in the central area of the developing pituitary gland. At this stage, ir-aMSH and ir-ACTH cells are found in the posterior and anterior part of the developing pituitary, respectively.
Opened-mouth larvae (4 days)
The pituitary gland is located over a ventral cartilage plate, and it still shows no clearly delimited lobes. The detected pituitary cell types (ir-SL, ir-PRL and ir-GH) increase both in their number and their immunostaining intensity. A compact group of ir-PRL cells begins to form at the more rostral part of the gland, but these cells are not arranged in follicles as in adult animals. Both ir-SL and ir-GH cells begin to increase in number and size but still without forming compact groups. From this day on, a topographic segregation of the three cellular types in a rostral-caudal direction is observed, which precedes the adult lobar distribution.
Free-swimming larva (6 days)
The infundibular recess develops at the junction of the diencephalon and the pituitary gland. The adenohy-pophysis becomes larger but the neurohypophysis is not yet formed. The segregation of the endocrine cellular types (ir-ACTH, ir- aMSH, ir-GH, ir-PRL and ir-SL cells) is more evident at this stage, approximating their final localizations; also, however, some few immunore-active PRL, GH and SL cells remain isolated in different regions.
Odd-fins larva (14 days)
The neurohypophysis becomes distinct in the caudal part of the gland but it is still a thin structure and does not show the adult deep infoldings. The adenohy-pophysis becomes even larger and elongated in shape with ir-PRL and ir-ACTH cells occupying most of the rostral region (the prospective rostral pars distalis). The pars intermedia is distinct now, and is characterized by groups of ir-SL cells.
Prejuvenile larva (21 days)
The first pituitary ir-FSH cells are detected in the anterior region of the adenohypophysis. From this stage on, the number of ir-FSH cells augments steadily. By day 90, when this ontogenetic study was ended, the number of FSH-ir cells was still increasing.
Prejuvenile larva (26 days)
Two neurohypophyseal branches project into the adenohypophysis: a thicker one is directed towards the anterior adenohypophyseal regions while the other enters deep into the pars intermedia. At this time, a clear boundary between the pars distalis and the pars intermedia is seen, but there is still no clear demarcation between the rostral and the proximal pars distalis. Immunoreactive GH cells and SL-ir cells start to associate to the neurohypophyseal branches, both at the anterior adenohypophyseal regions and at the pars intermedia, respectively.
Juvenile (42 days)
The adult lobar topography is already distinct at this stage. Immunohistochemistry applied to adjacent sections showed that ir-SL cells are not periodate reactive, as they will be in the adult pituitary (Pandolfi et al., 2001a). All the pituitary cell types, except ir-LH cells, are present at this stage. Close to the onset of sexual differentiation, which will occur by this time of development, the number of ir-FSH cells increases and they can be observed along the proximal pars distalis and the external border of the pars intermedia.
Juvenile (60 days)
Immunoreactive LH cells are first seen in central zones of the proximal pars distalis (two and a half weeks after the appearance of the first signs of ovarian differentiation).
The foregoing results are summarized in Table 2.
TABLE 2. Ontogenetic expression of pituitary hormones in Cichlasoma dimerus.

Overview of our studies on pituitary ontogeny
Five immunoreactive cell types (PRL, GH, SL, ACTH, MSH) appear at very early stages of development, suggesting their functional importance for the fish larvae. Further studies focused on the ontogeny of hypothalamic factors that regulate their synthesis and secretion should still be performed. Gonadotrops become differentiated later: FSH cells appear prior to the onset of sexual differentiation, while LH cells appear during the sexual differentiation period. A possible role of FSH in determining ovarian differentiation in this species will be shown in section 8.
7. Neuroendocrine systems that control reproduction and body color in C. dimerus.
Gonadotropin-releasing hormone (GnRH) systems
The chemical structure of GnRH was initially discovered as a decapeptide which is present in the mammalian hypothalamus (Matsuo et al., 1971; Burgus et al., 1972) and is released in the portal system, and whose primary function was to induce pituitary gonadotropins release into the bloodstream. It was also found that it may act in neural tissue as a neuromodulator and/or neurotransmitter involved in eliciting reproductive behaviors (Ogawa et al., 2006) and neuroendocrine reactions (Parhar, 1997). In addition to that, there is evidence suggesting that GnRH may play a role in the release of other pituitary hormones such as PRL, GH and SL (Weber et al., 1997; Parhar, 1997; Stefano et al., 1999; Vissio et al., 1999).
Moreover, it was found that chemical forms other than the decapeptide also occurred in mammals and other vertebrates (Muske, 1993). At least three molecular GnRH forms exist in perciform species, which are identified after the species where they were originally found as sGnRH (salmon GnRH), sbGnRH (seabream GnRH) and cIIGnRH (chicken II GnRH). Their distribution patterns in the perciform central nervous system suggest the existence of three distinct neuronal populations located in (1) the olfactory bulbs, expressing sGnRH; (2) the preoptic area, predominantly expressing sbGnRH; and (3) the midbrain tegmentum, expressing cGnRH II. Based upon these observations and those of ontogenetic studies, it has been hypothesized that sGnRH neurons originate from the olfactory placode, cGnRH II neurons originate from a mesencephalic primordium, and two possible origins were proposed for sbGnRH neurons: the diencephalic floor and the olfactory placode (Parhar, 1997; Parhar et al., 1998; González-Martínez et al., 2002). This hypothetical triple origin of GnRH neuronal systems differs from that proposed for some fish and other vertebrates, according to which all forebrain and diencephalic GnRH neurons would originate from the olfactory placode (Schwanzel-Fukuda and Pfaff, 1989; Wray et al., 1989; Muske, 1993; Schwanzel-Fukuda, 1999).
In a recent study (Pandolfi et al., 2005), both antibodies against different perciform GnRH-associated peptides (GAPs), and riboprobes to different perciform GnRH and GAPs were used. These GAPs are useful markers of the different GnRH neuronal populations since they are produced from GnRH precursor molecules and they are colocalized within GnRH-expressing cells (Ronchi et al., 1992; Polkowska and Przekop, 1993). Furthermore, markedly different peptides are associated with the different GnRH types. Therefore, these proce-dures result in a more specific marking than that obtained with antibodies and riboprobes to the smaller GnRH molecules themselves, both for immunohistochemistry and for in situ hybridization, respectively. Results showed that distribution of both sGnRH and sbGnRH neuronal populations in the C. dimerus forebrain are largely overlapped along the olfactory bulb, the nucleus olfacto-retinalis, the preoptic area and the ventral telencephalon. It was also shown (Pandolfi et al., 2005) that sGAP immunoreactivity follows an antero-posterior gradient, with the most conspicuous immunoreactivity (both in terms of cell area and number) in the nucleus olfactoretinalis, and a lesser one in more posterior locations, as the preoptic area and the ventral telencephalon. In contrast, sbGAP cells show a postero-anterior gradient, with the most conspicuous immunoreactivity in the preoptic area and a weaker one in the nucleus olfacto-retinalis and olfactory bulb. The distribution of GnRH neuronal populations is summarized in Tables 3 and 4, and in figure 6.
TABLE 3. Localization, abundance and average diameter of GAP/GnRH-ir neurons in different brain zones.

TABLE 4. Timing of appearance (days after hatching) of ir-GAP/GnRH populations in different brain zones.


FIGURE 6. Distribution map of neurons producing the different types of GnRH found in the brain of Cichlasoma dimerus. The location of sGnRH, sbGnRH and cIIGnRH neurons was studied by immunoreactivity of their respective GnRH-associated peptides (GAPs). Insets A, B and C show details of sGnRH, sbGnRH, and cIIGnRH neurons at the same magnification; scale bar = 20 μm. Encephalic regions and brain nuclei are indicated as: ce, cerebellum; hpt, hypothalamus; mo, medulla oblongata; mb, midbrain; nor, nucleus olfacto retinalis; ob, olfactory bulb; oe, olfactory epithelium; on, olfactory nerve; ot, optic tectum; pit, pituitary; poa, preoptic area, tel, telencephalon; v, ventricle; vtel, ventral telencephalon. Redrawn from Pandolfi et al. (2005).
Overview of our studies on neuroendocrine systems that control reproduction (GnRH)
Our results showed that: (1) GnRH neuronal popu-lations in the forebrain (sGAP and sbGAP) show an overlapping pattern, (2) sGAP projections are mainly located in the forebrain and contribute to the pituitary innervation, with cGAPII projections being mainly distributed along the mid and hindbrain and not contributing to pituitary innervation, whereas sbGAP projections are restricted to the ventral forebrain, being the most important molecular form in relation to pituitary innervation; (3) sbGAP neurons have an olfactory origin; (4) GAP antibodies and GAP riboprobes are valuable tools for the study of various GnRH systems, by avoiding the cross-reactivity problems that occur when using GnRH antibodies and GnRH riboprobes alone.
MCH and a-MSH systems
Melanin-concentrating hormone (MCH) is a 17 amino acids cyclic peptide synthesized in the hypothalamus, which together with a-melanocyte-stimulating hormone (a-MSH) regulates the aggregation and dispersion of chromatophores in teleosts (Fujii, 1993). Other factors like norepinephrine, adenosine triphosphate (ATP), PRL and SL have also been reported as potential regulators of teleost skin color changes (Baker et al., 1986; Kiita et al., 1993; Zhu et al., 1999; Cánepa et al., 2006).
In adult C. dimerus, ir-MCH neurons are mainly located in anterior and midhypothalamic positions (Pandolfi et al., 2003) as they are also found in other teleost fishes (Baker and Bird, 2002). Two kind of ir-MCH cells are found in C. dimerus: magnocellular neurons located in the nucleus lateralis tuberis, and parvi-cellular neurons, which are less numerous ir-MCH cells, localized between the third ventricle and the lateral ventricular recess. Magnocellular neurons send their projections through the anterior and posterior infundibular stalk to the three adenohypophyseal lobes, but mainly to the pars intermedia. Other axons are also projected to the thalamus, preoptic area and telencephalon. Another group of small MCH-ir neurons located outside the nucleus lateralis tuberis show delicate immunoreactive nerve fibers that do not contribute to pituitary innervation (Pandolfi et al., 2003).
The distribution of ir-MCH magnocellular neurons and their fibers suggests two principal roles in C. dimerus, (1) as a neurohypophyseal hormone, and (2) as a hypothalamic regulator of pituitary hormones. The presence of ir-MCH fibers in the thalamus, preoptic area, ventral and dorsal telencephalon also suggests a potential role of MCH as a neuromodulator/neurotransmitter, as proposed for amphibians, sauropsids and mammals (Bittencourt et al., 1992; Cardot et al., 1994; Vallarino et al., 1989). The role of ir-MCH cell bodies located outside the nucleus lateralis tuberis, as well as the nerve fibers found in different brain regions is less clear, but they might be involved in the control of feeding, as it has been proposed for mammals (Qu et al., 1996).
In C. dimerus larvae, ir-MCH cells appear 4 days after hatching, in a proliferating zone located on the floor of the anterior hypothalamus (Pandolfi et al., 2003). At this stage, the MCH innervation can be detected as a dense bundle of fibers penetrating into the developing pituitary. The number of immunoreactive neurons and fibers increase with age and by day 30 (prejuvenile stage) cell bodies and fibers already show an adult arrangement in the nucleus lateralis tuberis. By day 6 post-hatching, another group of small ir-MCH neurons develops in the nucleus periventricularis posterior, located close to the third ventricle in the medial part of the hypothalamus. These cells have few and thin projections that do not seem to reach the pituitary. The number of these small ir-MCH neurons decrease with age and by day 42 they are not detectable anymore. In Sparus auratus, Mancera and Fernández Llebrez (1995) ob-tained similar results. Probably, these neurons differentiate in the ependymal layer and then migrate to the nucleus lateralis tuberis.
Involvement of SL in background color adaptation
SL is a pituitary hormone present at least in Actinopterygian fish and in the Sarcopterygian lung-fish, Protopterus annectens. This hormone is expressed in the pars intermedia and belongs to the GH/PRL family. A single main function of SL remains elusive, but it is thought to be involved in different regulatory processes, such as those controlling the stress response, circulating calcium and phosphate levels, fat metabolism, as well as some reproductive processes (Kawauchi and Sower, 2006). Furthermore, studies suggest that SL is involved in background color adaptation (Kawauchi and Sower, 2006). In C. dimerus, a melanin dispersing effect is observed in fish exposed during ten days to a black background, while a concentrating one is observed in those exposed to a white background. When we compared Western blot-detected pituitary SL content in fish adapted to either black or white backgrounds, we observed an important increase in those adapted to the black background. In addition, fish exposed to a black background had more SL-ir cells with a greater ir-area. On the other hand, fish chronically adapted to a black background showed a larger number of ir-aMSH cells, while fish adapted to a white background had more numerous ir-MCH neurons in the nucleus lateralis tuberis (Cánepa et al., 2006). Taken together, these results strongly suggest that SL interacts with both aMSH and MCH in the regulation of background color adaptation in C. dimerus.
Regulation of SL release by GnRH and MCH
As we have seen, many studies have focused on SL function, but there is scarce information on the regulation of its secretion. In cultured pituitaries of the rainbow trout, Oncorhynchus mykiss, Kakizawa et al. (1997) have shown that sGnRH, corticotropin releasing factor and serotonin stimulate SL release. Also, in the masu salmon, Oncorhynchus masou, Onuma et al. (2005) observed that the addition of sGnRH to primary pituitary cell cultures elevated SL mRNA expression.
In C. dimerus, we have shown by double label im-munofluorescence that ir-SL cells are in close contact with both ir-MCH and ir-GnRH fibers in the pars intermedia (Cánepa et al., 2008). We then performed in vitro pituitary cultures of intact in order to investigate a possible releasing or inhibiting function of MCH or GnRH on SL cells. We found that MCH stimulated SL release from pituitaries of male C. dimerus in a dose dependent manner; but no such an effect was observed in females. On the other hand, incubation with GnRH showed similar results than the MCH assay in male pituitaries. Although female pituitaries did not evidence significant differences between treatments, we observed a noticeable increasing tendency. In conclusion, our results show that SL, like other pituitary hormones, would be under hypothalamic control (MCH and GnRH) and that they are implicated in diverse physiological processes including background adaptation and reproduction (Cánepa et al., 2008).
8. The roles of pituitary and brain gonadotropins.
The gonadotropins follicle-stimulating hormone (FSH) and luteinizing hormone (LH) are glycoprotein hormones produced in the proximal pars distalis of C.dimerus (see section 5). In fishes, as in other vertebrates, these hormones are considered of central importance in the control of gametogenesis and gonadal steroidogenesis (Blázquez et al., 1998). In several tetrapod and teleost species, they are formed by two protein chains, namely the a and b subunits (Yoshiura et al., 1999; Yaron et al., 2001). The a-FSH and a-LH subunits have identical aminoacid sequences in a given species, while b subunits have different sequences, and are recognized by different specific receptors, and are thus responsible for their different actions (Suzuki et al., 1988; Tyler et al., 1991).
In salmonid species characterized for spawning only once a year, the different roles of FSH and LH have been studied in detail (Suzuki et al., 1988; Tyler et al., 1991). The functions of FSH, as well as the variations of its circulating levels during the reproductive cycle, are not completely understood in many non salmonid species examined so far, though FSH is generally considered as a vitellogenic hormone, while LH is generally related to gonadal maturation and steroidogenesis (Tyler et al., 1991).
Several developmental studies conducted in teleost fishes, showed varying plasma levels and different expression patterns of FSH and LH in the pituitary (Dufour et al., 2000). Taken together, these results suggest specific functions for both FSH and LH at the onset of sexual maturation and in the control of gametogenesis.
As we have mentioned above, the identification of FSH cells using heterologous antisera was very difficult to perform due to cross-reactions or low specificity between FSH molecules from different species (Vissio et al., 1996; Pandolfi et al., 2001a). Antisera raised against the mummichog Fundulus heteroclitus FSH and LH were successfully used to recognize GtH cells in several teleost fishes, because they recognized conserved regions of FSH and LH in teleosts (Shimizu et al., 2003a,b).
The localization, characterization and ontogeny of gonadotrops in the pituitary of Cichlasoma dimerus have been already addressed in section 6 of this review. In this species, pituitary ir-FSH cells appear before the onset of sexual differentiation, while ir-LH cells differ-entiate several days thereafter. This variation in the timing of immunoreactive FSH and LH expression was also observed in several teleost species (Mal et al., 1989; Nozaki et al., 1990; Saga et al., 1999; Magliulo-Cepriano et al., 1994). The results of these studies sug-gested that in salmonid species and in the platyfish (Schreibman et al., 1982; Parhar et al. 1995), as well as in C. dimerus (section 6), the differentiation of ir-FSH cells precedes that of ir-LH cells. In the pejerrey Odontesthes bonariensis, however, ir-LH cells are known to appear before ir-FSH cells (Miranda et al., 2001).
In salmonids, FSH, but not LH, is involved in the onset of gonadal growth and development (Saga et al., 1993). Also, FSH was shown to be involved in the onset of meiosis in an amphibian (Ito and Abe, 1999). Since ir-FSH was expressed in pituitary cells of sexually undifferentiated prejuvenile larvae of C. dimerus (30 days after hatching), we decided to study the effects of this hormone on larval gonadal explants. LH effects was not studied since ir-LH cells do not appear in the pituitary until day 60, i.e. two and a half weeks after the first indications of gonadal differentiation appear. When the undifferentiated gonadal explants were left for 15 days with no hormone in the control culture medium, no sexual differentiation occurred, while exposure to human recombinant FSH for the same period resulted in 80% of cases differentiating as prospective ovaries (N=80) (Pandolfi et al., 2006). These results reinforced our previous hypothesis about a temporal correlation between the innervation of pituitary cells by sbGnRH fibers and the subsequent release of FSH to circulation, and the onset of gonadal sex differentiation in both sexes (Pandolfi et al., 2002).
In the course of our anatomical study of the C. dimerus pituitary (Pandolfi et al., 2001a; 2006), ir-FSH and ir-LH fibers were detected in both the neurohypo-physis and the infundibular recess. These fibers were deriving from cell bodies in the parvicelullar and magnocellular preoptic nuclei, and in the hypothalamic nucleus lateralis tuberis. Also, the developmental appearance of both brain- and pituitary-derived gonadot-ropins was studied showing that ir-LH neurons appeared on day 15 after hatching, while ir-FSH pituitary cells and neurons appeared on day 21), and ir-LH pituitary cells appeared only on day 60. To corroborate these findings, specific PCR products of b-LH and b-FSH were amplified from cDNA obtained from pituitaries and the preoptic area and hypothalamus of adult C. dimerus during the reproductive season. The partial nucleotide sequences of mRNA from C. dimerus brain and from the preoptic-hypothalamic brain fragment consisted of approximately 258 bp for b-LH and 276 bp for b-FSH (Pandolfi et al., 2009).
Furthermore, we settled an individual pituitary in vitro culture system to study the possible modulatory effect of brain derived gonadotropins on pituitary hor-mone secretion. Whole pituitary explants were cultured with different concentrations of Fundulus heteroclitus LH or FSH and the culture media were analyzed by Western blot. Exogenous LH produced a dose-depen-dent increase in pituitary b-LH and SL release and an increase in b-FSH release. No effect was observed on both GH and PRL release. Exogenous FSH produced an inhibition in b-LH release, a dose-dependent increase in b-FSH and SL release, and no effect on PRL and GH release. This is the first work showing an effect of brain-derived gonadotropins on different vertebrate pituitary cells (Pandolfi et al., 2009).
9. Concluding remarks and future studies.
Cichlasoma dimerus is a very interesting fish species for the study of reproductive biology because of several characteristics already discussed in this review: (1) it is easy to maintain and breed under laboratory conditions; (2) it has a moderate size which allows extraction of tissue samples and pituitary dissection easily and fast; (3) it shows a very interesting reproductive behavior; (4) it is possible to specifically identify their pituitary endocrine cells and hormone-producing neuronal populations; (5) its brain anatomy is well known; and (6) most of the important reproductive events (sexual differentiation and maturity, annual reproductive cycle) have been already described.
Future research in this species will be aimed to understand how the brain influences behavior, and how behavior correlates with the expression of brain hormones. To analyze these interactions we will study reproduction, perhaps the most important process in an animal's life. All the presented data about neuroanatomy, neuroendocrinology, reproduction and development of this species will be extremely useful for us and any other research group interested in using C. dimerus as a vertebrate species which displays social interactions that would be manageable in the laboratory.
After several years of observations we realized that this species has a very sophisticated social system with features we can manipulate in the laboratory. Our current experiments are aimed at modifying the social circumstances, to analyze the consequences of our manipulations on behavior and on the previously characterized neuroendocrine systems. This species is also easy to observe in the field, so a lot of behavioral experiments could be performed not only for answering ecological and behavioral questions but also to address evolutionary issues (they have incredibly robust social interactions that are clearly important for natural selection).
The accessibility of its habitat enables observations in nature, something that is not as easy to achieve in "model species" such as medaka or zebrafish. For all these reasons and all the data gathered in this review we think that C. dimerus might turn into a valuable vertebrate model in the future.
References
1. Alonso F, Cánepa MM, Lo Nostro FL, Maggese MC, Pandolfi M (2007). Cichlasoma dimerus: a fish model for studying stress and social control of reproduction. Biocell 31 (Suppl): 153, abstract. [ Links ]
2. Baerends G P, Baerends-Van Roon JM (1950). An introduction to the study of the ethology of cichlid fishes. Behaviour, suppl. I, 242 pp. [ Links ]
3. Baker BI, Bird DJ, Buckingham JC (1986). Effects of chronic administration of melanin-concentrating hormone on corticotrophin, melanotrophin, and pigmentation in the trout. General and Comparative Endocrinology 63: 62-69. [ Links ]
4. Baker BI, Bird DJ (2002). Neuronal organization of the melaninconcentrating hormone system in primitive actinopterygians: evolutionary changes leading to teleosts. Journal of Comparative Neurology 442: 99-114. [ Links ]
5. Ball JN, Baker BI (1969). The pituitary gland: Anatomy and histophysiology. In: Fish Physiology Vol. 2, The endocrine system. (WS Hoar, DJ Randall, eds.) p. 1-110. Academic Press, New York. [ Links ]
6. Bittencourt JC, Presse F, Arias C, Peto C, Vaughan J, Nahon L, Vale W, Sawchenko (1992). The melanin-concentrating hormone system of the rat brain: an immuno- and hybridization histochemical characterization. Journal of Comparative Neu-rology 319: 218-245. [ Links ]
7. Blázquez M, Bosma PT, Fraser EJ, Van Look KJW, Trudeau VL (1998). Fish as models for the neuroendocrine regulation of reproduction and growth. Comparative Biochemistry and Physiology C 119: 345-364. [ Links ]
8. Bruslé J, Bruslé S (1983). La gonadogènese des poisons. Reproduction, Nutrition and Development 23: 453-491. [ Links ]
9. Burgus R, Butcher M, Amoss M, Ling N, Monahan M, Rivier J, Fellows R, Blackwell R, Vale W, Gullemin R (1972). Primary structure of ovine luteinizing hormone-releasing factor (LRF). Proceedings of the National Academy of Sciences (USA) 69: 278-282. [ Links ]
10. Cambre M, Mareels G, Corneillie S, Moons L, Ollevier F, Vandesande F (1990). Chronological appearance of the different hypophyseal hormones in the pituitary of sea bass larvae (Dicentrarchus labrax) during their early development: An immunocytochemical demonstration. General and Com-parative Endocrinology 77: 408-415. [ Links ]
11. Cánepa MM, Pandolfi M, Maggese MC, Vissio PG (2006). Involvement of Somatolactin in background adaptation of the cichlid fish Cichlasoma dimerus. Journal of Experimental Zoology 305: 410-419. [ Links ]
12. Cánepa MM, Pozzi AG, Astola A, Maggese MC, Vissio PG (2008). Effect of salmon melanin-concentrating hormone and mammalian gonadotrophin releasing hormone on somatolactin release in pituitary culture of Cichlasoma dimerus. Cell and Tissue Research 333: 49-59. [ Links ]
13. Cardot J, Fellman D, Bugnon C (1994). Melanin-concentrating hormone-producing neurons in reptiles. General and Comparative Endocrinology 94: 23-32. [ Links ]
14. Casciotta JR, Almirón AE, Bechara J (2005). Peces del Iberá. Hábitat y Diversidad. UNDP, Fundación Ecos, UNLP, UNNE. Glafikar. La Plata, Argentina. [ Links ]
15. Conlon JM (2000). Singular contributions of fish neuroendocrinology to mammalian regulatory peptide research. Regulatory Peptides 93: 3-12. [ Links ]
16. Crollius HR, Weissenbach J (2009). Fish genomics and biology. Genome Research 15: 1675-1682. [ Links ]
17. Dufour S, Huang YS, Rosseau K (2000). Puberty in teleosts: new insights into the role of peripheral signals in the stimulation of pituitary gonadotropins. In: Proceedings of the VI International Symposium on Reproductive Physiology of Fish. pp. 455-461. University of Bergen, Bergen. [ Links ]
18. Eipper BA, Mains RE (1981). Structure and biosynthesis of proadrenocorticotropin/endorphin and related peptides. Endocrine Reviews 1: 1-27. [ Links ]
19. Fox HE, White AW, Kao MH, Fernald RD (1997). Stress and dominance in a social fish. Journal of Neuroscience 17: 6463-6469. [ Links ]
20. Fujii R (1993). Cytophysiology of fish chromatophores. International Review of Cytology 143:191-255 [ Links ]
21. Furutami-Seiki M, Wittbrodt J (2004). Medaka and Zebrafish, an evolutionary twin study. Mechanism of Development. 121: 629-637. [ Links ]
22. González-Martínez D, Zamora N, Mañanos E, Saligaut D, Zauny S, Zohar Y, Elizur A, Kah O, Muñoz-Cueto JA (2002). Immunohistocemical localization of three different prepro-GnRHs (Gonadotrophin-releasing hormone) in the brain and pituitary of the European sea bass Dicentrarchus labrax using antibodies to the corresponding GnRH-associated peptides. Journal of Comparative Neurology 446: 95-113. [ Links ]
23. Greenberg B, Zijlstra JJ, Baerends GP (1965). A quantitative description of the behavior changes during the reproductive cycle of the cichlid fish Aequidens portalegrensis (Hensel). Proc. Kon. Ned. Akad. v. Wet., Ser C 68: 135-149. [ Links ]
24. Hoar WS (1969). Reproduction. In: Fish Physiology Vol. 3. Reproduction and growth (WS Hoar, DJ Randall, eds.) pp. 1-72. Academic Press, New York. [ Links ]
25. Ito R, Abe SI (1999). FSH-initiated differentiation of newt spermatogonia to primary spermatocytes in germsomatic cell reaggregates cultured within a collagen matrix. International Journal of Development Biology. 43: 111-116. [ Links ]
26. Kakizawa S, Ishimatsu A, Takeda T, Kaneko T, Hirano T (1997). Possible involvement of somatolactin in the regulation of plasma bicarbonate for the compensation of acidosis in rainbow trout. Journal of Experimental Biology 200: 2675-2683. [ Links ]
27. Kawauchi H, Sower SA (2006). The dawn and evolution of hormones in the adenohypophysis. General and Comparative Endocrinology 148: 3-14. [ Links ]
28. Kiita K, Makino M, Oshima N, Bern HA (1993). Effects of prolactins on the chromathophores of the tilapia Oreochromis niloticus. General and Comparative Endocrinology 92: 355-365. [ Links ]
29. Kullander SO (1983). A revision of the South American cichlid genus Cichlasoma. Swedish Museum of Natural History, Stockholm. [ Links ]
30. Magliulo-Cepriano L, Schreibman M P, Blüm V (1994). Distribution of variant forms of immunoreactive gonadotropin-releasing hormone and beta-gonadotropins I and II in the platyfish, Xiphophorus maculatus, from birth to sexual maturity. General and Comparative Endocrinology 94: 135-150. [ Links ]
31. Mal AO, Swanson P, Dickhoff WW (1989). Immunocytochemistry of the developing salmon pituitary gland. American Zoolo-gist 29: 94A. [ Links ]
32. Mancera JM, Fernández Llebrez P (1995). Development of melanin-concentrating hormone-immunoreactive elements in the brain of gilthead seabream (Sparus auratus). Cell Tissue Research 282: 523-526. [ Links ]
33. Matsuo H, Baba Y, Nair RMG, Arimura A, Schally AV (1971). Structure of the porcine LH and FSH-releasing hormone. I. The proposed amino acid sequence. Biochemical and Biophysical Research Communications 43: 1334-1339. [ Links ]
34. Mattheij JAM, Stroband H W, Kingma FJ (1971). The cell types in the adenohypophysis of the cichlid fish Cichlasoma biocellatum Regan, with speciall attention to its osmoregulatory role. Zeitschrift für Zellforschung 118: 113-126. [ Links ]
35. Meijide FJ, Guerrero GA (2000). Embryonic and larval development of a substrate-brooding cichlid Cichlasoma dimerus (Heckel, 1840) under laboratory conditions. Journal of Zoology 252: 481-493. [ Links ]
36. Meijide FJ, Lo Nostro FL, Guerrero GA (2005). Gonadal development and sex differentiation in the cichlid fish Cichlasoma dimerus (Teleostei, Perciformes): A light- and electron-microscopic study. Journal of Morphology 264: 191-210. [ Links ]
37. Miranda LA, Strüssmann CA, Somoza GM (2001). Immunocy-tochemical identification of GtH1 and GtH2 cells during the temperature-sensitive period for sex differentiation in pejerrey, Odontesthes bonariensis. General and Comparative Endocrinology 124: 45-52. [ Links ]
38. Muske LE (1993). Evolution of gonadotrophin-releasing hormone (GnRH) neuronal systems. Brain, Behavior and Evolution 42: 215-230. [ Links ]
39. Naito N, Jesus EG de, Nakai Y, Hirano T (1993). Ontogeny of pituitary cell types and the hypothalamo-hypophyseal relation-ship during early development of chum salmon, Oncorhynchus keta. Cell & Tissue Research 272: 429-437. [ Links ]
40. Nozaki M, Naito N, Swanson P, Dickhoff W W, Nakai Y, Suzuki K, Kawauchi H (1990). Salmonid pituitary gonadotrophs. II. On-togeny of GTH I and GTH II cells in the rainbow trout (Salmo gairdneri irideus). General and Comparative Endocrinology 77: 358-367. [ Links ]
41. Ogawa S, Akiyama G, Kato S, Soga T, Sakuma Y, Parhar IS (2006). Immunoneutralization of gonadotropin-releasing hormone type-III suppresses male reproductive behavior of cichlids. Neuroscience Letters 403: 201-5. [ Links ]
42. Onuma T, Ando H, Koide N, Okada H, Urano A (2005). Effects of salmon GnRH and sex steroid hormones on expression of genes encoding growth hormone/prolactin/ somatolactin family hormones and a pituitary-specific transcription factor in masu salmon pituitary cells in vitro. General and Comparative Endocrinology 143: 129-141. [ Links ]
43. Pandolfi M, Paz DA, Maggese MC, Meijide FJ, Vissio PG (2001a). Immunocytochemical localization of different cell types in the adenohypophysis of Cichlasoma dimerus (Teleostei, Perciformes). Biocell 25: 35-42. [ Links ]
44. Pandolfi M, Paz DA, Maggese MC, Ravaglia MA, Vissio PG (2001b). Ontogeny of immunoreactive somatolactin, prolactin and growth hormone secretory cells in the developing pituitary gland of Cichlasoma dimerus (Teleostei, Cichlidae). Anatomy and Embryology 203: 461-468. [ Links ]
45. Pandolfi M, Parhar IS, Ravaglia MA, Meijide FJ, Maggese MC, Paz DA (2002). Ontogeny and distribution of gonadotropin-releasing hormone (GnRH) neuronal systems in the brain of the cichlid fish Cichlasoma dimerus. Anatomy and Embryology 205: 271-281. [ Links ]
46. Pandolfi M, Cánepa MM, Ravaglia MA, Maggese MC, Paz DA, Vissio PG (2003). Melanin-concentrating hormone system in the brain and skin of the cichlid fish Cichlasoma dimerus: anatomical localization, ontogeny and distribution in comparison to a-melanocyte-stimulating hormone- expressing cells. Cell and Tissue Research 311: 61-69. [ Links ]
47. Pandolfi M, Muñoz Cueto JA, Lo Nostro FL, Downs JL, Paz DA, Maggese MC, Urbanski HF (2005). The GnRH systems of Cichlasoma dimerus (Perciformes, Cichlidae) revisited: A localization and developmental study using antibodies and riboprobes to the GnRH-associated peptides. Cell and Tissue Research 321: 219-232. [ Links ]
48. Pandolfi M, Lo Nostro FL, Shimizu A, Pozzi AG, Meijide FJ, Rey Vazquez G, Maggese MC (2006). Identification of immunoreactive FSH and LH cells in the cichlid fish Cichlasoma dimerus during the ontogeny and sexual differentiation. Anatomy and Embryology 211: 355-365. [ Links ]
49. Pandolfi M, Pozzi AG, Cánepa MM, Vissio PG, Shimizu A, Maggese MC, Lobo G (2009). Presence of b-FSH and b-LH transcripts in the brain of Cichlasoma dimerus (Perciformes; Cichlidae): effect of brain-derived gonadotropins on pituitary hormone release. Neuroendocrinology 89: 27-37. [ Links ]
50. Parhar IS, Iwata M, Pfaff DW, Schwanzel-Fukuda M (1995). Embryonic development of gonadotropin-releasing hormone neurons in the sockeye salmon. Journal of Comparative Neu-rology 362: 256-270. [ Links ]
51. Parhar IS (1997). GnRH in tilapia: Three genes, three origins and their roles. In: GnRH neurons: Gene to behaviour. Chapter 5 (IS Parhar, Y Sakuma, eds.). Brain Shuppan, Tokyo. [ Links ]
52. Parhar IS, Soga T, Ishikawa Y, Nagahama Y, Sakuma Y (1998). Neurons synthesizing gonadotropin-releasing hormone mRNA subtypes have multiple developmental origins in the medaka. Journal of Comparative Neurology 401: 217-226. [ Links ]
53. Peter RE, Habibi HR, Chang J P, Nahorniak CS, Yu KL, Huang Y P, Marchant TA (1990). Actions of gonadotropin-releasing hormone (GnRH) in the goldfish. Progress in Clinical and Biological Research 342: 393-398. [ Links ]
54. Polder JJW (1971). On gonads and reproductive behavior in the cichlid fish Aequidens portalegrensis (Hensel). Netherlands Journal of Zoology 21: 265-365. [ Links ]
55. Polkowska J, Przekop F (1993). Effect of protein deficiency on luteinizing hormone releasing hormone (LHRH), gonadotropoin releasing hormone associated peptide (GAP) and luteinizing hormone (LH) immunocytochemistry in the hypothalamus and pituitary gland of prepubertal ewes. Experimental and Clinical Endocrinology 101: 230-237. [ Links ]
56. Price AC, Weadick CJ, Shim J, Rodd FH (2008). Pigments, patterns, and fish behavior. Zebrafish 5: 297-307. [ Links ]
57. Qu D, Ludwig DS, Gammeltoft S, Piper M, Pelleymounter MA, Cullen MJ, Mathes WF, Pzrypek J, Kanarek R, Maratos-Flier E (1996). A role for melanin-concentrating hormone in the central regulation of feeding behavior. Nature 380: 243-247. [ Links ]
58. Rand-Weaver M, Baker JB, Kawauchi H (1991). Celular localization of somatolactin in the pars intermedia of some teleost fishes. Cell and Tissue Research 263: 207-215. [ Links ]
59. Ronchi E, Aoki C, Krey LC, Pfaff DW (1992). Immunocytochemical study of GnRH and GnRH-associated peptide in male Syrian hamsters as a function of photoperiod and gonadal alterations. Neuroendocrinology 55: 134-145. [ Links ]
60. Saga T, Oota Y, Nozaki M, Swanson P (1993). Salmonid pituitary gonadotrophs. III. Chronological appearence of GTH I and other adenohypophyseal hormones in the pituitary of the developing rainbow trout (Oncorhynchus mykiss irideus). General and Comparative Endocrinology 92: 233-241. [ Links ]
61. Saga T, Yamaki K, Doi Y, Yoshizuka M (1999). Chronological study of the appearence of adenohypophyseal cells in the ayu (Plecoglossus altivelis). Anatomy and Embryology 196: 227-234. [ Links ]
62. Schreibman M P, Margolis-Kazan H, Halpern-Sebold L (1982). Immunoreactive gonadotropin and luteinizing hormone releasing hormone in the pituitary gland of neonatal platyfish. General and Comparative Endocrinology 47: 385-391. [ Links ]
63. Schwanzel-Fukuda M, Pfaff DW (1989). Origin of luteinizing hormone-releasing hormone neurons. Nature 338: 161-164. [ Links ]
64. Schwanzel-Fukuda M (1999). Origin and migration of luteinizing hormone releasing hormone in mammals. Microscopy Research and Technique 44: 2-10. [ Links ]
65. Shimizu A, Tanaka H, Kagawa H (2003a). Immunocytochemical applications of specific antisera raised against synthetic fragment peptides of mummichog GtH subunits: examining seasonal variations of gonadotrophs (FSH cells and LH cells) in the mummichog and applications to other acanthopterygian fishes. General and Comparative Endocrinology 132: 35-45. [ Links ]
66. Shimizu A, Sakai T, Nashida K, Kagawa H (2003b). Universal antisera for immunocytochemical identification of two different gonadotrophs in acanthoptherygian fishes. Fish Physiology and Biochemistry 29: 275-287. [ Links ]
67. Staeck W, Linke H (1995). American Cichlids II. Large Cichlids. A Handbook for their Identification, Care and Breeding. Tetra-Verlag. Germany. [ Links ]
68. Stefano AV, Vissio PG, Paz DA, Somoza GM, Maggese MC, Barrantes G (1999). Colocalization of GnRH binding site with gonadotropin-, somatotropin-; somatolactin- and prolactin-expressing pituitary cells of the pejerrey, Odonthestes bonariensis, in vitro. General and Comparative Endocrinology 116: 133-139. [ Links ]
69. Suzuki K, Kawauchi H, Nagahama Y (1988). Isolation and characterization of two distinct gonadotropins from chum salmon pituitary glands. General and Comparative Endocrinology 71: 292-301. [ Links ]
70. Takayama Y, Ono M, Rand-Weaver M, Kawauchi H (1991). Greater conservation of somatolactin, a presumed pituitary hormone of the growth hormone/prolactin family, than of growth hormone in teleost fish. General and Comparative Endocrinology 83: 366-374. [ Links ]
71. Teyler TJ, Lewis D, Shashoua VE (1981). Neurophysiological and biochemical properties of the goldfish optic tectum maintained in vitro. Brain Research Bulletin 7: 45-56. [ Links ]
72. Tyler CR, Sumpter J P, Kawauchi H, Swanson P (1991). Involvement of gonadotropin in the uptake of vitellogenin into vitellogenic oocytes of the rainbow trout, (Oncorhynchus mykiss). General and Comparative Endocrinology 84: 291-299. [ Links ]
73. Vallarino M, Tranchand Bunel TD, Delbende C, Ottonello I, Vaudry H (1989). Distribution of the pro-opiomelanocortinderived peptides, alpha-melanocyte-stimulating hormone (aMSH), adrenocorticotropic hormone (ACTH), and beta-endorphin in the brain of the dogfish Scyliorhinus canicula: an immuno-cytochemical study. Journal of Experimental Zoology Suppl 2: 112-121. [ Links ]
74. Villaplana M, García Ayala A, García Hernández M P, Agulleiro B (1997). Ontogeny of immunoreactive cells in the pituitary of gilthead sea bream (Sparus aurata L., Teleostei). Anatomy and Embryology 196: 227-234. [ Links ]
75. Vissio PG, Paz DA, Maggese C (1996). The adenohypophysis of the swampeel, Synbranchus marmoratus: an immunocy-tochemical analysis. Biocell 20: 155-61. [ Links ]
76. Vissio PG, Stefano AV, Somoza GM, Maggese MC, Paz DA (1999). Close association of gonadotrophin-releasing hormone fibers and gonadotropins, growth hormone, somatolactin and prolactin expressing cells in pejerrey, Odonthestes bonariensis. Fish Physiology and Biochemistry 21: 121-127. [ Links ]
77. Wayne NL, Kuwahara K, Aida K, Nagahama Y, Okubo K (2005). Whole-cell electrophysiology of gonadotropin-releasing hormone neurons that express green fluorescent protein in the terminal nerve of transgenic medaka (Oryzias latipes). Biology of Reproduction 73: 1228-1234. [ Links ]
78. Weber GM, Powell JFF, Park M, Fischer WH, Craig AG, Rivier JE, Nanakorn U, Parhar IS, Ngamvongchon S, Grau EG, Sherwood NM (1997). Evidence that gonadotrophin-releasing hormone (GnRH) functions as a prolactin-releasing factor in a teleost fish (Oreochromis mossambicus) and primary structures of three native GnRH molecules. Journal of Endocrinology 155: 121-132. [ Links ]
79. Wray S, Grant P, Gainer H (1989). Evidence that cells expressing luteinizing hormone-releasing hormone mRNA in the mouse are derived from progenitor cells in the olfactory placodes. Proceedings of the National Academy of Sciences (USA) 86: 8132-8136. [ Links ]
80. Yaron Z, Gur G, Melamed P, Rosenfeld H, Levavi-Sivan B, Elizur A (2001). Regulation of gonadotropin subunit genes in tilapia. Comparative Biochemistry and Physiology. B: Compara-tive Biochemistry 129: 489-502. [ Links ]
81. Yoshiura Y, Suetake H, Aida K (1999). Duality of gonadotropin in a primitive teleost, Japanese eel (Anguilla japonica). General and Comparative Endocrinology 114: 121-131. [ Links ] [ Links ]
Received: July 17, 2008.
Final version received: October 23, 2008.
Accepted: February 9, 2009.














