Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Biocell
Print version ISSN 0327-9545
Biocell vol.33 no.1 Mendoza Jan./Apr. 2009
Cytogenetical and ultrastructural effects of copper on root meristem cells of Allium sativum L.
Donghua Liu1*, Wusheng Jiang2, Qingmin Meng1, Jin Zou1, Jiegang Gu1 and Muai Zeng1
1.Department of Biology, Tianjin Normal University, Tianjin 300387, P.R. China.
2.Library of Tianjin Normal University, Tianjin 300387, P.R. China.
Address correspondence to: Donghua Liu. Department of Biology, Tianjin Normal University, Tianjin 300387, P.R. CHINA. E-mail: donghua@mail.zlnet.com.cn
ABSTRACT: Different copper concentrations, as well as different exposure times, were applied to investigate both cytogenetical and ultrastructural alterations in garlic (Allium sativum L.) meristem cells. Results showed that the mitotic index decreased progressively when either copper concentration or exposure time increased. C-mitosis, anaphase bridges, chromosome stickiness and broken nuclei were observed in the copper treated root tip cells. Some particulates containing the argyrophilic NOR-associated proteins were distributed in the nucleus of the root-tip cells and the amount of this particulate material progressively increased with increasing exposure time. Finally, the nucleolar material was extruded from the nucleus into the cytoplasm. Also, increased dictyosome vesicles in number, formation of cytoplasmic vesicles containing electron dense granules, altered mitochondrial shape, disruption of nuclear membranes, condensation of chromatin material, disintegration of organelles were observed. The mechanisms of detoxification and tolerance of copper are briefly discussed.
Key words: Garlic; Organelle; Cytogenetical and ultrastructural alterations; Mitosis.
Introduction
Copper is an essential nutrient for plants; it plays an irreplaceable role in the function of a large number of enzymes catalyzing oxidative reactions in a variety of metabolic pathways (Lolkema and Vooijs, 1986; Marschner, 1995; Quartacci et al., 2000). Plants respond to both the deficiency and the excess of metals (Kukkola et al., 2000). Various copper sources, including industrial and domestic wastes, agricultural practices, copper mine drainage, copper-based pesticides, and antifouling paints, have contributed to a progressive increase in copper concentrations in several environments (Andrade et al., 2004). High copper levels in soil can be phytotoxic, causing deleterious effects both morpho-logically and physiologically (Liu et al., 1995; Ke et al., 2007; Meng et al., 2007; Tanyolaç et al., 2007). Copper can lower the mitotic index, inhibit cell divi-sion and induce chromosomal aberrations (Liu et al., 1994; Jiang et al., 2001). Studies on the accumulation and subcellular localization of heavy metals in plants have been reported recently by energy dispersive X-ray spectroscopy (Kupper et al., 2000; Monni et al., 2002; Liu et al., 2007; Sahi et al., 2007), synchrotron radiation X-ray fluorescence spectroscopy (Shi et al., 2004) and electron energy loss spectroscopy (Liu and Kottke, 2004a; Rau et al. 2006). These techniques can provide essential information on subcellular localization and accurate elemental analysis of heavy metals. However, few investigations on toxic effects of Cu2+ on cell division, nucleolus, and on ultrastructural alterations in constructing anti-copper system in cells under copper stress have been reported.
Allium sativum L. is a potentially useful plant to study the absorption and accumulation of heavy metals (Jiang and Liu, 2001; Liu and Kottke, 2003). In the current work we combined both cytogenetical and ultra-structural effects of the different copper concentrations on the root meristem cells of A. sativum, and to provide essential information on mechanisms of detoxification and tolerance.
Materials and Methods
Plant material, growth conditions and metal treatments
Healthy and equal-sized garlic cloves (Allium sativum L.) were chosen from bulbs showing no growth of either green leaves or roots. The experimental set up was similar to that of Fiskesjö (1988): 12 garlic cloves with the dry scales removed were used in each series, and were washed and rooted in tap water for 36 h. Afterwards, 10 of these rooted cloves were selected and directly placed in a container with either 10-5 M, 10-4 M or 10-3 M copper solution (provided as copper sulfate, CuSO4 · 5H2O), respectively. Controls were grown on tap water. The test liquids were changed regularly every 24 h, seedlings were grown in a greenhouse equipped with supplementary 15-h light/9-h dark at 18-20ºC diurnal cycle.
Cytogenetical assay
Twenty root tips in each treatment group were washed with tap water and distilled water, and cut at 24, 48 and 72 h, respectively. They were fixed in ethanol + acetic acid (3:2) for 4 to 5 h and hydrolysed in 1 M hydrochloric acid + 95% ethanol + acetic acid (5:3:2) for 6 min at 60°C. For the observation of chromosomal morphology, 10 root tips were squashed in a carbol-fuchsin solution (Li, 1982) and for the observation of nucleo-lus changes, the others were squashed in 45% acetic acid, drying, and on day 2 staining with silver nitrate (Li et al., 1990; Liu and Jiang, 1991). Data for root length were analysed with standard statistical software (SigmaPlot 9.0).
Ultrastructure
For transmission electron microscopy observation, according to the results from cytogenetic investigation, seedlings treated at 10-4 M copper for 1 h, 2 h, 4 h, 8 h, 12 h, 24 h, 36 h, 48 h and 72 h.
The terminal 1-3 mm portion from each root in both the control and treated groups was fixed in a mixture of 2% formaldehyde and 2.5% glutaraldehyde in 0.2 M phosphate buffer (pH 7.2) for 2 h and, and then thoroughly washed in the same buffer, before post-fixation in 2% osmium tetroxide in the same buffer for 2 h. They were then dehydrated in an acetone series and embedded in Spurr's resin. For ultrastructural observations, ultrathin sections of 75 nm thickness were cut on an ultramicrotome (Leica EM UC6, Germany) with a diamond knife, and were mounted on copper grids with 300 square mesh. The sections were stained with 2% uranyl acetate for 50 min and lead citrate for 15 min. They were examined under a transmission electron microscope (JEM-1230, Joel Ltd., Tokyo, Japan).
Cytochemical tests
The Gomori-Swift reaction was used to detect cysteine-rich proteins. For such purpose, 100 nm thick sections of roots prepared as described above, were cut and mounted on gold grids.
Solution A containing 5 ml of 5% silver nitrate and 100 ml of 3% hexamethylenetetramine and solution B consisting of 10 ml 1∞ 44% boric acid and 100 ml 1∞ 9% borax were prepared. The final stain was obtained by mixing 25 ml of A, 5 ml of B and 25 ml of distilled water. The grids were floated in the silver methenamine solution for 90 min at 45ºC in the dark, and were then washed four times for 2 min. The grids were then floated on 10% sodium thiosulphate solution for 1 h at room temperature to dissolve metallic silver and rinsed in deionized water four times for 2 min. Finally, the sections were stained with uranyl acetate and lead citrate (Swift, 1968).
Controls were carried out to block S-H group and S-S group by the reduction of disulfide bonds in benzylmercaptan followed by alkylation of S-H groups in iodacetate boric acid. The procedures were described by Swift (1969) and Liu and Kottke (2004b).
Results
Cytogenetical investigation
Observations of roots treated with different levels of Cu2+ revealed structural damage that were not observed in the root cells of control plants.
Mitotic index: The mitotic index reflects the frequency of cell division and it is regarded as an important parameter. The mitotic index decreased progressively as a function of increased copper concentration and exposure time (Table 1).
TABLE 1. Effect of CuSO4 on cell division in the root tip cells of A. sativum
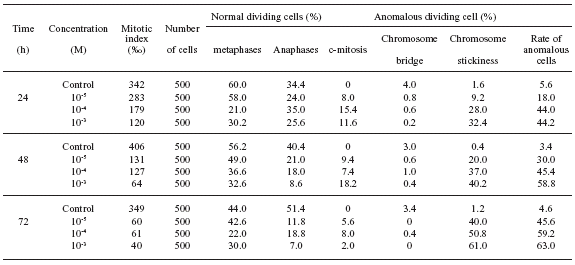
Chromosomal aberrations: C-mitosis was observed in the root tip cells of all treated groups after treatment with Cu2+ (Fig. 1a). Anaphase bridges in-volving one or more chromosomes (Fig. 1b-c) were found after the Cu treatment. Anaphase bridges exhibiting stickiness were observed only in the treatment with 10-3 M Cu (Fig. 1d). This type of toxic effect is most likely irreversible. The cells with chromosome stickiness were noted (Fig. 1e), and the frequency of cells progressively increased with increasing Cu2+ concentration. Some broken nuclei in the root tip cells treated with Cu (10-3 M) were observed (Fig. 1f).

Figure1. The effects of Cu2+ on cell division in root tips of Allium sativum. a: C-metaphase (10-4 M Cu2+, 24 h); b-d: Chromosome bridges (b, 10-4 M Cu2+, 24 h; c, 10-3 M Cu2+, 24 h; d, 10-3 M Cu2+, 48 h); e: Chromosome stickiness (10-3 M Cu2+, 48 h ); f: Nucleus disintegration (10-3 M Cu2+, 72 h ). Scale = 10 μm.
Nucleolar organiser regions (NORs) are defined as nucleolar components containing a set of argyrophilic proteins, which are selectively stained by silver methods. After silver-staining, the NORs can be easily identified as black dots exclusively localized throughout the nucleolar area, and are called "AgNOR proteins" (Trerè, 2000). Normally, the diploid nucleus of A. sativum contains one or two nucleoli (Fig. 2a). The effects of copper on nucleoli varied with the different concentrations of copper sulfate solutions used. Two phenomena were observed after these treatments. Firstly, after 48 h treatment with 10-4 M copper, some particulates containing the argyrophilic NOR-associated proteins were observed together with the main nucleolus/nucleoli in the nucleus of some roottip cells (Fig. 2b). The amount of the particulates increased progressively and nearly occupied the whole nucleus when the copper concentration increased to 10-3 M (Fig. 2c). Secondly, in concentrations of 10-3 M copper, the AgNOR proteins were extruded from the nucleus into the cytoplasm (Fig. 2d). The nucleolar proteins in the cytoplasm increase progressively (Fig. 2e) and aggregate into irregular shapes with longer duration of the treatment (Fig. 2f).

Figure 2. Effects of Cu2+ on nucleoli in root tip cells of Allium sativum. a: Control cell (48 h); b: Some particulate containing the argyrophilic NOR-associated proteins scattered in the nuclei (10-4 M Cu2+, 48 h); c: More and more particles occupied the whole nucleus (10-3 M Cu2+, 48 h); d-f: The particles scattered in the cytoplasm (d, 10-3 M Cu2+, 48 h; e, 10-3 M Cu2+, 48 h; f, 10-3 M Cu2+, 72 h). Scale = 10 μm
Ultrastructural investigation
A typical ultrastructure was exhibited in control cells. Plasma membrane was unfoled with a uniform shape in all parts. The numerous organelles were im-mersed in cytoplasm. Endoplasmic reticulum is mostly composed of parallel cisternae. Dictyosomes were also found in cytoplasm (Fig. 3a). The nucleus with well-stained nucleoplasm and distinct nucleolus was located in the center of cells, whereas a large vacuole or several vacuoles are distributed in root meristem cells.

Figure 3. TEM micrographs showing toxic effects of Cu on ultrastructure of the root meristematic cells of Allium sativum. a: Control cells showing well developed root tip cells. b-d: The ultrastructural changes of root meristematic cells treated with 10-4 M Cu from 1 h - 72 h. b: Dictysome vesicles obviously increased in the root meristematic cells exposed to 10-4 M Cu for 1 h. c: Vesiculated ER was distributed in cytoplasm near cell wall (10-4 M Cu, 4 h). d: The electron dense granules were accumulated in vacuoles (10-4 M Cu, 4 h). C = cytoplasm, CM = cytoplasm membrane, CW = cell wall, D = dictyosome, ER = endoplasmic reticulum, EDG = electron dense granules, M = mitochondria, N = nucleus, V = vacuole, Ve = vesicle. Bar = 0.25 μm.
Root tip cells exposed to 10-4 M copper solutions showed several ultrastructural alterations. Visible symptom of copper toxicity was noted after copper treatment for 1 h. The dictyosome vesicles markedly increased, which seems as a compact mass of vesicles in the cytoplasm (Fig. 3b). Endoplasmic reticulum with dilation of flattened cisternae appeared in cytoplasm after 2 h copper treatment, and these flattened cisternae were broken up into small closed vesicles (Fig. 3c), in which electron dense granules were occasionally observed in it when root cells exposed to 10-4 M copper for 4 h (Fig. 3d). Any degenerative changes were not showed in the other organelles in relation to control cells. However, after 8 h copper treatment, larger vacuoles were rapidly formed by migrating to and fusing small vesicles derived from endoplasmic reticulum, and more electron dense granules in the vacuoles were accumulated (Fig. 4a). This tendency was more pronounced in the cells with prolonging copper treatment time. After 12 h copper treatment, abundance of parallel arrays of rough endoplasmic reticulum with regularly cisternae was also observed in cytoplasm (Fig. 4b). Due to an extension of cisternae and a loss of matrix density, alterations of mitochondria shapes were gradually showed in figure 4c. Electrondense granules were taken into cells by means of invaginations of the plasma membrane. The invaginations form small vesicles that are pinched off the plasma membrane and carried, with their enclosed the material, into the cytoplasm, which appeared after 24 h treatment (Fig. 4c). The most significant ultrastructural changes were noted after 36-72 h of root cells treatment, revealing disruption of nuclear membranes, a highly condensed chromatin material and disintegration of organelles, which led to death of some copper treated cells after 72 h.

Figure 4. TEM micrographs showing toxic effects of Cu on ultrastructure of the root meristematic cells of Allium sativum. a: The vacuole increased by fusing small vesicles from ER and dictysomes in cytoplasm. b: Abundance of parallel arrays of rough ER with regularly cisternae exhibited in cytoplasm (10-4 M Cu, 12 h). c: Endocytosis of plasma membranes appeared. Arrow shows reduction of mitochondria. (10-4 M Cu, 24 h). d-f: Cytochemical test of the root meristematic cells of A. strvum. d: Without showing Gomori-Swift reaction in the cell of control. e: Metallic silver grains with Gomori-Swift positive reaction localized in the cytoplasm and vesicles (10-4 M Cu, 4 h). f: Showing metallic silver grains distribution in cell wall, in cytoplasm as well as vesicles (10-4 M Cu, 8 h). CM = cytoplasm membrane, CW = cell wall, D = dictyosme, ER = endoplasmic reticulum, EDG = electron dense granules, M = mitochondria, MSG = metallic silver granules, MT = microtubule, N = nucleus, V = vacuole, Ve = vesicle. Bar = 0.25 μm.
Cytochemical test
Cytochemical test were conducted to discover Cu2+ localization in root cells using the Gomori-Swift reaction which is highly sensitive and allows the detection of cysteine-rich proteins. In control root cells metallic silver grains as a result of positive reaction were not found (Fig. 4d). Swift reaction showed the presence of cysteinerich proteins in root cells exposed to 10-4 M copper. Traced amounts of silver grains were, at early stage, noted in the cell walls of root cells at copper treatment for 2 h, and then they appeared in cytoplasm and small vesicles near the cell walls (Fig. 4e). In vesicles and cytoplasm, the number of cysteine-rich protein increased as a consequence of excessive copper-exposure time from 4 h to 72 h, whereas, metallic silver grains in cell walls do not increased remarkably (Fig. 4f).
Discussion
The results in the present investigation indicated that copper affects Allium sativum at concentrations from 10-5 to 10-3 M. These are in agreement with the findings of Liu et al. (1994) for the effect of Cu2+ on tip cells of A. cepa. However, this results were with a few differences when comparison with the findings of Jiang et al. (2001) for the effect of Cu2+ on Zea mays. For instance, 1) there is not so many nucleolar particles scattered in the nuclei and so much nucleolar material released from nuclei into cytoplasm in Z. mays; 2) Copper toxicity on the nucleoli in root tip cells of A. sativum is stronger than those of Z. mays, which shows that A. sativum and A. cepa is more sensitive to copper compared with Z. mays.
Some reports revealed that copper caused the inhibition of root elongation by metal interference with cell division, including inducement of chromosomal aberrations and abnormal mitosis (Agarwal et al., 1987; Kahle, 1993; Jiang et al., 2001). Quzounidou (1994) stated that copper accumulation influences the tissue distribution of Ca, Mg, Fe and K. Jensén and Adalsteinsson (1989) also indicated that copper ions tend to displace Ca2+ ions from exchange sites and are strongly bound in root-free space. It is well known that the nucleolus is the metabolic center of RNA. The integrity of the nucleolus depends on the existence of Ca2+ (Wang, 1988). Because of the low level of free Ca2+ in the cells, calmodulin does not activate Ca-AT-Pase (Xu, 1985), which leads to failure in regulation of calcium concentration and disturbance or inhibition of various cellular metabolic processes.
The results from the present investigation revealed that excess copper caused adverse effects on garlic meristem cells. The plasma membrane and main organelles participated the establishment of antiheavy metal toxic systems in the cells. The response to Cu2+ toxicity occurs in 1 h after copper treatment, resulting in an enlargement of dictyosomes. Usually, cisternae of endoplasmic reticulum are arranged in parallel in control cells. However, vesiculated endoplasmic reticulum could be observed markedly in cytoplasm. The vesicles derived from dictysomes and endoplasmic reticulum were formed into bigger vacuoles by fusing from each other. The feature of vacuolar compartmentation is the most obvious ultrastructure evidence of cells when cells resist heavy metals toxicity, which is in agreement with the work reported by Ouzounidou et al. (1995), Sresty and Madhava Rao (1999). It is very interesting that at copper treatment for 12 h, a large amount of endoplasmic reticulum with regular cisternae again appeared in the cytoplasm, similar to that in the control cells. One possible explanation is that target of toxic copper is at the molecular level and effects are always reflected in the structure of the cell and its organelles. Once excess Cu2+ ions entered cytosol, the synthesis of new proteins of endoplasmic reticulum involved in heavy metal tolerance is stimulated. Then, the vesicles from endoplasmic reticulum carried the proteins, which bind copper by formation of stable metalphytochelatin complexes. In this way, free metal ions in cytosol decreased. Be-sides, some vesicles from endoplasmic reticulum carried polysaccharide and they were transferred to the dictyosomes where they are modified prior to secretion (Raven et al., 1986). Secretory vescles derived from cisternae of dictyosomes migrate to the plasma membrane and repair the damaged plasma membrane and discharging their contents in the region of the wall. Plants have a range of potential mechanisms at the cellular level that might be involved in the detoxification and thus tolerance to heavy metal stress (Hall, 2002). The results from the present investigation suggest that endoplasmic reticulum and dictysomes may participate in heavy metal detoxification in cells.
In this context, it is interesting that cysteinerich proteins were found localized both in the cytoplasm and in cell walls and vesicles of copper treated cells (Fig. 4f). Rau et al. (2006) indicated that electron energy loss spectra demonstrated copper chelation by SH-groups of glutathione in the cytoplasm and the copper-binding as phosphates in vacuoles. Copper accumulation was associated with a reduced sulfur level in the electron-dense-precipitates of the cytoplasm and an increasing phosphorus level in the vacuole. Morelli and Scarano (2004) reported that phytochelatin synthesis started in the marine diatom P . tricornutum after copper exposure for 15 min, and phytochelatin accumulation in the cell reached a maximum rate at the treatment for 7 h. Data from size exclusion chromatography analyzed that an amount close to 60% of the total intracellular copper was bound to phytochelatin at seventh hour of exposure.
The evidence reviewed in this investigation and formed works strongly suggest that 1. Cell walls, a first barrier against copper stress, can immobilize some copper ions, therefore prevent contact with the sensitive plasmalemma and cytoplasmic components. The results here exhibited that the presence of cysteine-rich proteins were identified in cell walls exposed to copper. 2. Active endocytosis of plasma membrane and active secetory of vesicles from dictyosomes and endoplasmic reticulum appear at low concentration of heavy metals, which reflects the morphological feature during detoxification and tolerance to heavy metals. 3. At first stage, electron dense granules containing copper distribute in the cytoplasm and vesicles (Liu and Kottke, 2004b). However, vacuoles finally are one of main storage sites of heavy metals. 4. With the increased level of heavy metal ions in cytosol, cell and its organelle were seriously injured and lost their functions of anti-heavy metal toxicity, leading to some cells death.
Acknowledgements
This project was supported by the National Natural Science Foundation of China. The authors wish to express their appreciation to the reviewers for this paper.
References
1. Agarwal K, Sharma A, Talukder G (1987). Copper toxicity in plant cellular systems. Nucleus 30: 131-158. [ Links ]
2. Andrade LR, Farina M, Amado Filho GM (2004). Effects of copper on Enteromorpha flexuosa (Chlorophyta) in vitro. Ecotoxicology and Environmental Safety 58: 117-125. [ Links ]
3. Fiskesjö G (1988). The Allium test - an alternative in environmental studies: the relative toxicity of metal ions. Mutation Research 197: 243-260. [ Links ]
4. Hall JL (2002). Cellular mechanisms for heavy metal detoxification and tolerance. Journal of Experimental Botany. 53: 1-11. [ Links ]
5. Jensén P, Adalsteinsson S (1989). Effects of copper on active and passive Rb+ influx in roots of winter wheat. Physiologia Plantarum 75: 195-200. [ Links ]
6. Jiang W, Liu D, Liu X (2001). Effects of copper on root growth, cell division, and nucleolus of Zea mays. Biologia Plantarum 44: 105-109. [ Links ]
7. Jiang WS, Liu DH (2001). Hyperaccumulation of cadmium by roots, bulbs and shoots of Allium sativum L. Bioresource Technology 76: 9-13. [ Links ]
8. Ke WS, Xiong ZT, Xie MG (2007). Accumulation, subcellular localization and ecophysiological responses to copper stress in two Daucus carota L. popularions. Plant and Soil 292: 291-304. [ Links ]
9. Kukkola E, Rautio P, Huttune S (2000). Stress indications in copper- and nickel-exposed Scots pine seedlings. Environmental and Experimental Botany. 43: 197-210. [ Links ]
10. Kupper H, Lombi E, Zhao FJ McGrath SP (2000). Cellular compartmentation of cadmium and zinc in relation to other elements in the hyperaccumulator Arabidopsis halleri. Planta 221: 75-84. [ Links ]
11. Li MX (1982). Introducing a good stain used for nuclei and chromosome. Biological Bulletin 5: 53. [ Links ]
12. Li MX, Zhang Z P, Yuan JS, Pu XL (1990). An improved method for Ag-NOR staining and its application to plant chromosomes. Chinese Bullin of Botany 7: 56-59. [ Links ]
13. Liu D, Kottke I, Adam D (2007). Localization of cadmium in the root cells of Allium cepa by energy dispersive X-ray analysis. Biologia Plantarum 51: 363-366. [ Links ]
14. Liu DH, Jiang WS (1991). Effects of Al3+ on the nucleolus of Allium cepa root-tip cells. Hereditas 115: 213-219. [ Links ]
15. Liu DH, Jiang WS, Lu C, Zhao FM, Hao YQ, Guo L (1994) Effects of copper sulfate on the nucleolus of Allium cepa root-tip cells. Hereditas 120: 87-90. [ Links ]
16. Liu DH, Jiang WS, Wang W, Zhai L (1995). Evaluation of metal ion toxicity on root tip cells by the Allium test. Israel Journal of Plant Sciences 43:125-133. [ Links ]
17. Liu DH, Kottke I (2003) Subcellular localization of Cd in the root cells of Allium sativum by electron energy loss spectroscopy. Journal of Biosciences 28: 471-478. [ Links ]
18. Liu DH, Kottke I (2004a). Subcellular localization of copper in the root cells of Allium sativum by electron energy loss spectros-copy (EELS). Bioresource Technology 94: 153-158. [ Links ]
19. Liu DH, Kottke I (2004b). Subcellular localization of cadmium in the root cells of Allium cepa by electron energy loss spectros-copy and cytochemistry. Journal of Biosciences 29: 330-335. [ Links ]
20. Lolkema PC, Vooijs P (1986). Copper tolerance in Silene cucubalus: subcellular distribution of copper and its effects on chloroplasts and plastocyanin synthesis. Planta 167: 30-36. [ Links ]
21. Marschner H (1995). Mineral nutrition of higher plants. 2ed Edition. Academic Press, London. [ Links ]
22. Meng QM, Zou J, Zou JH, Jiang WS, Liu DH (2007). Effect of Cu2+ concentration on growth, antioxidant enzyme activity and malondialdehyde content in Garlic (Allium sativum L.). Acta Biologica Cracoviensia Series Botanica 49: 95-101. [ Links ]
23. Monni S, Bucking H, Kottke I (2002). Ultrastructural element localization by EDXS in Empetrum nigrum. Micron 33: 339-351. [ Links ]
24. Morelli E, Scarano G (2004). Copper-induced changes of non-protein thiols and antioxidant enzymes in the marine microalga Phaeodactylum tricornutum. Plant Science 167: 289-296. [ Links ]
25. Quzounidou G, Èiamporová M, Moustakas M, Karataglis S (1995). responses of maize (Zea mays L.) plants to copper stress - I. Growth, mineral content and ultrastructure of roots. Environmental and Experimental Botany 35: 167-176. [ Links ]
26. Quartacci MF, Pinzino C, Sgherri CLM, Vecchia FD, NavariIzzo F (2000). Growth in excess copper induces changes in the lipid composition and fluidity of PSII-enriched membranes in wheat. Physiologia Plantarum 108: 87-93. [ Links ]
27. Rau S, Miersch J, Neumann D, Weber E, Krauss GJ (2006). Bio-chemical responses of the aquatic moss Fontinalis antipyretica to Cd, Cu, Pb and Zn determined by chlorophyll fluorescence and protein levels. Environmental and Experimental Botany 59: 299-306. [ Links ]
28. Raven PH, Evert RF, Eichhorn SE (1986). Biology of plants. Worth Publishers, Inc., New York, pp. 28-29. [ Links ]
29. Sahi S V, Israr M, Srivastava AK, Gardea-Torresdey JL, Parsons JG (2007). Accumulation, speciation and cellular localization of copper in Sesbania drummondii. Chemosphere 67: 2257-2266. [ Links ]
30. Shi J Y, Chen Y X, Huang Y Y, He W (2004). SRXRF microprobe as a technique for studying elements distribution in Elsholtzia splendens. Micron 35: 557-564. [ Links ]
31. Sresty TVS, Madhava Rao KV (1999). Ultrastructural alterations in response to zinc and nickel stress in the root cells of pigeonpea. Environmental and Experimental Botany 41: 3-13. [ Links ]
32. Swift JA (1968). The electron histochemistry of cysteine-containing proteins in thin transverse sections of human hair. Journal Royal Microscopical Society 88: 449-460. [ Links ]
33. Swift JA (1969). The electron histochemical demonstration of cystine-containing proteins in the guinea pig hair follicle. Histochemie 19: 88-98. [ Links ]
34. Tanyolaç D, Ekmekçi Y, Ûnalan ª (2007). Changes in photochemical and antioxidant emzyme activities in maize (Zea mays L.) leaves exposed to excess copper. Chemosphere 67: 89-98. [ Links ]
35. Trerè D (2000). AgNOR staining and quantification. Micron 31: 127-131. [ Links ]
36. Wang Y (1988). General Cell Biology. - Shanghai Scientific and Technical Publishing House, Shanghai, P. R. of China, p. 301-308. [ Links ]
37. Xu Y (1985). Structure and function of calmodulin (I). Acta Biochimica et Biophysica Sinica 1: 22-27. [ Links ]
Received: March 18, 2008.
Final version received: December 19, 2008.
Accepted: December 20, 2008.














