Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Biocell
versión impresa ISSN 0327-9545
Biocell vol.36 no.2 Mendoza ago. 2012
ORIGINAL ARTICLES
Human umbilical artery smooth muscle exhibits a 2-APB-sensitive capacitative contractile response evoked by vasoactive substances and expresses mRNAs for STIM, Orai and TRPC channels
Ana Rocío Roldán Palomo1, Pedro Martín1, Alejandro Rebolledo1*, Nicolás Enrique1, Luis E. Flores2, Verónica Milesi1
1 GINFIV -Grupo de Investigación en Fisiología Vascular, Facultad de Ciencias Exactas, Universidad Nacional de La Plata. Argentina.
2 CENEXA -Centro de Endocrinología Experimental y Aplicada, (UNLP-CONICET La Plata, Centro Colaborador OPS/OMS), Facultad de Ciencias Médicas, Universidad Nacional de La Plata. Argentina.
*Address correspondence to:
Alejandro Rebolledo.
GINFIV, Departamento de Ciencias Biológicas, Facultad de Ciencias Exactas, Universidad Nacional de La Plata. Calles 47 y 115, La Plata (1900), Argentina. E-mail: ar@biol.unlp.edu.ar
ABSTRACT: After depletion of intracellular Ca2+ stores the capacitative response triggers an extracellular Ca2+ influx through store-operated channels (SOCs) which refills these stores. Our objective was to explore if human umbilical artery smooth muscle presented this response and if it was involved in the mechanism of serotonin- and histamine-induced contractions. Intracellular Ca2+ depletion by a Ca2+-free extracellular solution followed by Ca2+ readdition produced a contraction in artery rings which was inhibited by the blocker of Orai and TRPC channels 2-aminoethoxydiphenyl borate (2-APB), suggesting a capacitative response. In presence of 2-APB the magnitude of a second paired contraction by serotonin or histamine was significantly less than a first one, likely because 2-APB inhibited store refilling by capacitative Ca2+ entry. 2-APB inhibition of sarcoplasmic reticulum Ca2+ release was excluded because this blocker did not affect serotonin force development in a Ca2+-free solution. The PCR technique showed the presence of mRNAs for STIM proteins (1 and 2), for Orai proteins (1, 2 and 3) and for TRPC channels (subtypes 1, 3, 4 and 6) in the smooth muscle of the human umbilical artery. Hence, this artery presents a capacitative contractile response triggered by stimulation with physiological vasoconstrictors and expresses mRNAs for proteins and channels previously identified as SOCs.
Key words: Human umbilical artery; Capacitative response; TRPC channels; Serotonin; Histamine
Introduction
The human umbilical artery is a vessel of fundamental importance for fetal-placental blood flow. Since the human umbilical arteries are branches from the iliac arteries of the fetus, the properties of these blood vessels could offer information of the circulatory system of the newborn. Hence, the knowledge of the mechanisms implicated in the regulation of cytosolic Ca2+ concentration and therefore, contraction of their vascular smooth muscle cells (VSMC) is very important.
In VSMC Ca2+ influx through voltage-activated Ca2+ channels in response to membrane depolarization or to vasoactive agonists represents the most studied pathway. However, an increasing number of investigations in blood vessels have demonstrated that depletion of intracellular Ca2+ stores triggers a Ca2+ influx from the extracellular space through ionic channels present in the cellular membrane. This phenomenon is known as "capacitative"or "store-operated"Ca2+ entry and it is likely involved in the refilling of intracellular Ca2+ stores (Parekh and Penner, 1997; Putney et al., 2001; Bolotina and Csutora, 2005; Leung et al., 2008). This mechanism has not been previously studied in the human umbilical artery nor in other vessels of feto-placental circulation, and hence, its physiological relevance in agonist-induced contractions in this system is unknown.
Channels implied in the capacitative response have been named store-operated channels (SOCs) and it has been shown that they increase their open probability when the sarcoplasmic reticulum (SR) is depleted, producing an inward Ca2+ current (Albert et al., 2007; Parekh and Putney, 2005). Several reports in the literature propose as possible SOCs the transmembrane proteins named Orai (Frischauf et al., 2008; Hewavitharana et al., 2007; Soboloff et al., 2006; Deng et al., 2009; Baryshnikov et al., 2009; Peinelt et al., 2006; Roberts-Thomson et al., 2010) and the Transient Receptor Potential (TRP) channels family, especially those of the Canonical type (TRPC channels) (Leung et al., 2008; Parekh and Putney, 2005; Dietrich et al., 2006; Ng et al., 2010). Finally, the Stromal Interacting Molecules (STIM) types 1 and 2 located in the membrane of SR are Ca2+ sensors of SR Ca2+ content (Frischauf et al., 2008; Hewavitharana et al., 2007; Soboloff et al., 2006; Deng et al., 2009; Peinelt et al., 2006). When SR Ca2+ concentration diminishes, these proteins could activate SOCs present in the cellular membrane to allow the capacitative Ca2+ influx.
Although there is now much information about the structures involved in the capacitative response, further research is necessary to completely understand how these structures interact in order to produce SR refilling and, mainly, what is the physiological importance of this mechanism for each cellular type. Different kinds of experimental protocols have been used for this purpose. For instance, inducing SR depletion with Ca2+ chelating agents like BAPTA or EGTA in extracellular solutions followed by Ca2+ re-addition (Putney et al., 2001; Bird et al., 2008), blocking Ca2+ entry to the SR with sarcoplasmic reticulum Ca2+-ATPase (SERCA) inhibitors like thapsigargin or cyclopiazonic acid (CPA) (Putney et al., 2001; Bird et al., 2008), or directly inducing Ca2+ release from the SR by increasing intracellular inositol 1,4,5-trisphosphate concentration (Fasolato et al., 1993; Fasolato and Nilius, 1998; Parekh and Penner, 1995).
However, in vascular smooth muscle, SR Ca2+ release and depletion are physiologically produced by mechanisms involving activation of membrane receptors. Hence, in the present work we demonstrate using classical protocols that the human umbilical artery presents a capacitative response, and then that it can be activated by stimulation with serotonin (5-HT) and histamine, both of which produce tonic contractions by coupling to membrane receptors, phospholipase C (PLC) signaling and SR Ca2+ release and depletion. To strengthen these data, we demonstrate by the polymerase chain reaction (PCR) technique that the mRNAs for STIM, Orai and for the different subtypes of cationic TRPC channels involved in capacitative entry mechanisms are present in the smooth muscle cells of this artery.
Material and Methods
Sample collection
Umbilical cords were obtained after vaginal and caesarean deliveries performed in the Instituto Central de Medicina of La Plata, Argentina. They were placed in a transport solution of the following composition (in mM): 130 NaCl, 4.7 KCl, 1.17 KH2PO4, 1.16 SO4Mg, 24 CO3HNa, 2.5 Cl2Ca, pH 7.4 at 4ºC and immediately taken to our laboratory where they were stored at 4ºC and used within 24 hrs. All the vascular preparations were classified as surgical discard specimens and thus they were exempted from patient consent requirements.
Isometric force measurements
The vessels were placed in a Petri dish filled with Krebs Ringer bicarbonate solution (KRB, see composition later), cleaned of adherent connective tissue, and cut into 3-4 mm wide rings. Special care was taken in not damaging the inner endothelial layer or overdistending the vessel. The ring was gently suspended between two stainless steel wires in a water-jacketed organ bath kept at 37ºC and filled with a KRB with the following composition (mM): 130 NaCl, 4.7 KCl, 1.17 Na2HPO4, 1.16 MgSO4, 24.0 HCO3Na, 2.5 CaCl2, and 6.0 glucose, constantly bubbled with a mixture of 5% CO2 and 95% O2, giving a pH of 7.40. The lower wire was fixed to a vertical plastic rod immersed in the or gan bath, while the upper one was rigidly attached to a force transducer (Letica TRI-201). The preparations were then stretched to obtain a passive force of ≈ 2 grams. In some experiments we used as bath solutions a Ca2+-free KRB solution (KRB without CaCl2) or a Ca2+-free KRB with the addition of 1 mM of EGTA (see results for details).
Total RNA isolation and Reverse Transciptase Polymerase chain reaction (PCR)
Samples from seven umbilical arteries were used for this analysis. The endothelial layer was carefully eliminated by gently rubbing the inner surface with a cotton swab. Total RNA was obtained from homogenates using Trizol Reagent (Gibco-BRL, Rockville, MD, USA) (Chomczynski and Sacchi, 1987). The integrity of isolated RNA was checked by 1% agarose-formaldehyde gel electrophoresis. Possible contamination with protein or phenol was controlled by measuring the 260/280 nm absorbance ratio, while DNA contamination was avoided using 1 U/µl DNAase I (Gibco-BRL). RT-reaction was performed using the Super Script III reverse transcriptase (200 U/µl; Gibco-BRL) and total RNA (50 ng) as template. For PCR, specific primers were used based on the human cDNA sequence to detect TRPC 1, 3, 4, 5, 6 and 7 (TRPC 2 was not analyzed since it is a pseudogene (Parekh and Putney, 2005), STIM (1 and 2) and Orai (1, 2 and 3), as well as the von Willebrand factor (endothelial marker used to assure that endothelial cells were not present in our samples) and β-actin (positive control of the PCR). GenBank accession numbers, primer sequences used and expected size of PCR products are shown in Table 1. Possible contamination with genomic DNA was checked by performing PCR with or without the SuperScript III RT.
Table 1. Data regarding the different primers used for PCR experiments shown in figure 4: GenBank accession numbers, forward and reverse primers sequences, and size (in bps).

The cycling profile was the following: 3 min at 95ºC followed by 30 sec at 95ºC, 30 sec at 58ºC, and 1 min at 72ºC for 35 cycles and 10 min at 72ºC. PCR products were separated by electrophoresis on a 1.5% (w/v) agarose gel and stained with ethidium bromide.
Statistical analysis
Results were expressed as means ± SEM. Paired or unpaired Student's t-tests were used to evaluate statistically significant differences between two groups. ANOVA followed by the Tukey's test was used for multiple group comparisons. The level of statistical significancy was set at p<0.05.
Results
Presence of a capacitative response
We first used a classical protocol, such as exposition to a Ca2+-free extracellular medium and then Ca2+ readdition, to identify the presence of a capacitative response in the human umbilical artery, where this mechanism had not been described before. Hence, vessel rings were first incubated during 15 min at room temperature in a Ca2+-free Krebs Ringer bicarbonate (Ca2+-free KRB) solution containing 5 mM EGTA to eliminate all the extracellular Ca2+ that could remain attached to connective tissue. Then, they were mounted in the force transducer and exposed during 15 minutes to a KRB solution without Ca2+ (at 37ºC) in order to stabilize the temperature. Then, they were stretched to 2 grams and left for another 15 min in this Ca2+-free solution. Interestingly, during this time 32% of the vessel rings developed a small spontaneous and transient contraction of 42 ± 7 gF/gW (n=13), probably caused by SR release or leak, induced by the extracellular Ca2+-free solution. After that, Ca2+ was restituted to the bath reaching a final concentration of 2.5 mM. Immediately, the rings developed a rapid contraction which reached a maximal force (peak) and then stabilized at a minor force value (Fig. 1A). To see if all, or part, of this contraction could be inhibited by a blocker of capacitative Ca2+ entry, the same protocol was repeated in presence of 100 µM 2-aminoethoxydiphenyl borate (2-APB), which is widely used to study this mechanism (Bootman et al., 2002) (Fig. 1B). The peak force induced by addition of Ca2+ now decreased significantly respect to the control contraction, and it did not fall to a minor value. So, these results suggest that the exposition of artery rings to a Ca2+-free solution, during almost 45 minutes, is able to produce SR depletion and SOC activation.

Figure 1. The human umbilical artery exhibits a capacitative contractile response induced by a period of extracellular Ca2+ deprivation followed by Ca2+ readdition. Typical recordings of force development in artery rings subjected to a protocol of 30 min in a Krebs solution without Ca2+ followed by Ca2+ readdition in A: control conditions; B: with 100 µM 2-APB; C: with 100 µM 2-APB, 5 µM nifedipine, 200 µM Gd3+ and 5 µM KB-R7943 (KBR); E: with 5 µM nifedipine; F: with 5 µM KB-R7943; G: with 200 µM Gd3+; and H: with 5 µM nifedipine, 5 µM KB-R7943 and 200 µM Gd3+. gF/gW: grams of developed force / weight of wet tissue. D: mean ± SEM values of experiments shown in A (n=40), B (n=13) and C (n=7). I: mean ± SEM values of experiments shown in E (n=13), F (n=14), G (n=10) and H (n=6). In all cases, * indicates statistically significant differences from control conditions (p<0.05).
Afterwards, we tested if the remaining force could be inhibited by blocking other Ca2+ entry pathways that we know are present in smooth muscle cells of the human umbilical artery. In fact, if the artery rings were incubated with 100 µM 2-APB plus 5 µM nifedipine (a voltage-operated Ca2+ channel inhibitor), 200 µM Gd3+ (a non selective cationic channels blocker) and 5 µM KB-R7943(at this concentration, an inhibitor of the Na+/Ca2+ exchanger reverse mode), the contraction induced by Ca2+ restitution was abolished (Fig. 1C). When these were tested separately, all of them had a significant inhibitory effect but none produced a complete inhibition (Fig. 1E, F and G). Figure 1H shows results obtained using a combination of nifedipine, Gd3+ and KB-R7943 but not 2-APB, which would leave only the 2-APB-sensitive capacitative response as a Ca2+ entry mechanism to induce the artery ring contraction.
Vascular agonists and capacitative response
In human umbilical artery both 5-HT and histamine are potent contractile agonists acting through G-coupled 5-HT2 receptors and H1 receptors, respectively (Bertrand and St-Louis, 1999; Lovren et al., 1999). It is known that they produce activation of inositol 1,4,5-trisphosphate sensitive Ca2+ channels (InsP3 receptors) which allow Ca2+ release from sarcoplasmic reticulum (SR) and its consequent depletion (Tufan et al., 2003). Since this will cause a reduction in SR Ca2+ content, we hypothesized that it could trigger a capacitative Ca2+ entry. Therefore, we applied two consecutive stimuli with 1 µM 5-HT or 10 µM histamine separated by a 25 minutes period of agonist wash-out. The obtained results presented in Figure 2A and 2D show that for both vasoconstrictor agents, the second contraction had the same magnitude as the first one. However, when the same protocol was repeated in the presence of 100 µM 2-APB, the second contractions were significantly lower than the first ones (Fig. 2B and 2E). Also, the bars in Figure 2 are showing the mean force relationships between the force values of the first and second contraction for 5-HT (2C) and for histamine (2F) in both conditions.
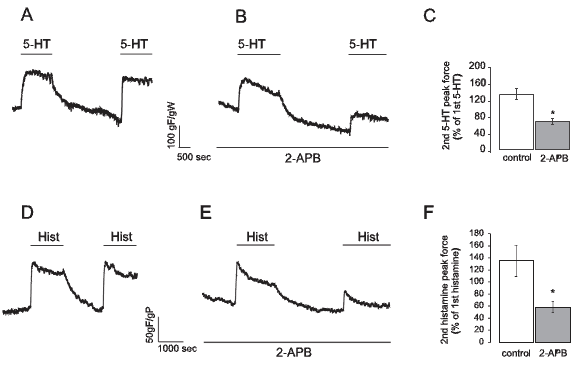
Figure 2. The capacitative contractile response participates in the mechanism of 5-HT and histamine force development in smooth muscle of the human umbilical artery. A: Typical recording of two 1 µM 5-HT-induced contractions separated by 25 min without 5-HT (gF/gW: grams of developed force / weight of wet tissue). B: Typical recording of an experiment similar to that shown in A but in the presence of 100 µM 2-APB. C: mean ± SEM values of experiments shown in A (n=10) and B (n=12). D: Typical recording of two 10 µM histamine-induced contractions separated by 25 min without histamine (gF/gW: grams of developed force / weight of wet tissue). E: Typical recording of an experiment similar to that shown in D but performed in the presence of 100 µM 2-APB. F: mean ± SEM values of experiments shown in D (n=10) and E (n=10). In all cases, * indicates statistically significant differences from controls (p<0.05).
It is known that 2-APB can also inhibit InsP3 receptors (Bootman et al., 2002; Maruyama et al., 1997), so the results presented in Figure 2B and 2E may be interpreted as a late effect of 2-APB on InsP3 receptors. In order to test this possibility, we performed a control experiment where 2-APB was tested on a contraction evoked by 5-HT on an arterial ring exposed to a Ca2+-free extracellular solution (with 1 mM EGTA), so only the contraction due to SR Ca2+ release would be observed. In this setting, stimulation with 1 µM 5-HT produced a significant transient contraction whose magnitude was not modified by 2-APB. Moreover, when extracellular Ca2+ was restored, a force development was observed which was now significantly diminished by the presence of 2-APB. Figure 3 shows typical recordings and mean values for these experiments.
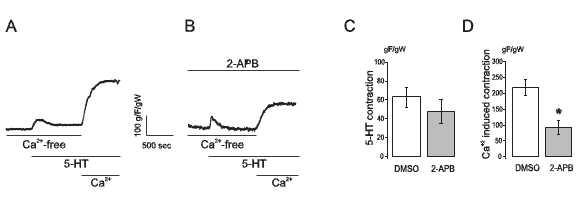
Figure 3. 2-APB does not interfere with 5-HT-induced Ca2+ release from the sarcoplasmic reticulum but it inhibits the capacitative contractile response in the human umbilical artery. A: Typical recording of 1 µM 5-HT contraction of artery rings in a Ca2+-free KRB followed by extracellular Ca2+ readdition. The chamber had an appropriate amount of the vehicle of 2-APB (DMSO) (gF/gW: grams of developed force / weight of wet tissue). B: Typical recording of an experiment similar to that shown in A but performed in the presence of 100 µM 2-APB. C: mean ± SEM values of peak 5-HT force of experiments shown in A (n=9) and B (n=10). D: mean ± SEM values of stable force after Ca2+ readdition as shown in A (n=9) and B (n=10).
Expression of mRNAs for structures implicated in capacitative response
STIM and Orai proteins and TRPC channels, are proposed in the literature to be involved in the capacitative response. Hence we explored the presence of the mRNA for these proteins in the human umbilical artery by the PCR technique. Although samples from 7 different arteries were processed in the same way (see Methods), the endothelial marker (von Willebrand factor) was not expressed by only two of them, so only the results from these samples were considered for conclusions about TRPC channels, STIM and Orai present in vascular smooth muscle (each of these two samples was analyzed twice). As shown in Figure 4, we found expression of mRNA for STIM 1 and 2, and for Orai 1, 2 and 3. The TRPC1, 3, 4 and 6 were also present in smooth muscle cells from the human umbilical artery, while we were not able to demonstrate the expression of TRPC5 and 7. The β-actin control was positive for all the PCRs performed.
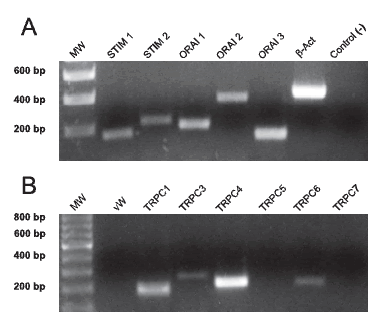
Figure 4. The PCR technique shows that smooth muscle cells of the human umbilical artery express mRNA for several structures implicated in the capacitative response. A: photograph of a typical PCR experiment showing the presence of mRNAs for STIM 1 and 2, and Orai 1, 2 and 3. Also shown are positive (with primer) and negative (without primer) results for β-actin. B: photograph of a typical PCR experiment showing the presence of mRNAs for TRPC 1, 3, 4 and 6, and the absence of TRPC 5 and 7 (TRPC 2 was not tested, see text for details). A negative result for von Willebrand factor (vW) indicates that there is no contamination of the sample with endothelial cells. A positive controls for b-actin was performed but is not shown.
Discussion
Our work presents results suggesting that the vasoconstrictors 5-HT and histamine produce an activation of the capacitative response in the human umbilical artery, which seems useful to allow a quick restoration of SR Ca2+ content. This conclusion is based on the results presented in Figure 2, where it can be seen that the vessels stimulated twice with these agonists evoked two similar contractions in control conditions, while in the presence of 2-APB the second contractions were significantly lower than the first ones. Our interpretation of these data is that during the first stimulation with the vasoconstrictor, the SR depletion caused by the agonist activated a capacitative response which would contribute to the SR refilling. When SOCs were inhibited by 2-APB the Ca2+ entry was diminished or inhibited and the SR did not completely refill after the first agonist addition, producing then a second contraction of smaller magnitude.
An alternative explanation for our results would be that the second 5-HT or histamine contraction was smaller than the first one because 2-APB had a late inhibitory effect on InsP3 receptors. However, we can exclude this possibility because when the stimulus on the SR was performed without extracellular Ca2+, 2-APB had no effect on the 5-HT induced contraction, but instead significantly diminished the force produced by Ca2+ re-addition.
Additionally, although we cannot completely exclude the possibility that the endothelium could have modified these responses, we have previously observed (unpublished data) that endothelial NO and protacyclins inhibition does not modify the contractile force induce by 5-HT. This suggests that the observed reduction of contractile force produced by 2-APB may be mostly due to inhibition of the smooth muscle capacitative response.
Our results are in accordance with the data presented by Gregory et al. (2001) in freshly isolated hepatocytes, where 2-APB (up to 100 µM) did not block InsP3 receptors but was able to inhibit the Ca2+ inflow when Ca2+ was restituted after vasopressin-induced SR depletion in a free-Ca2+ extracellular medium. Moreover, in cells incubated with normal extracellular Ca2+, they showed that 2-APB decreased Ca2+ entry once the SOC response was activated with vasopressin.
The degree of involvement of capacitative Ca2+ entry in the response to vasoconstrictors seems to be variable in different vessels and species. For instance, Moneer et al. (2005) propose that in A7r5 cells 5-HT produces extracellular Ca2+ entry only through a capacitative response. On the other hand, Guibert et al. (2004) showed the presence of a capacitative Ca2+ entry in small intrapulmonary arteries of the rat but provide evidence that it is independent of 5-HT-induced changes in cytosolic Ca2+ concentration and vessel diameter. However, the same researchers, showed that in human pulmonary artery a part of 5-HT-induced contractions was due to voltage-independent Ca2+ entry which could be mediated by SOCs (Rodat-Despoix et al., 2009). These results in human pulmonary artery are very similar to our findings in the umbilical artery, and it is interesting to note that both are human vessels in which 5-HT plays an important physiological role.
In order to further characterize the capacitative response in the human umbilical artery we also used a classical protocol of SR depletion induced by extracellular Ca2+ deprivation, and subsequent development of a contraction by Ca2+ readdition. This force development was partly inhibited by 2-APB, hence suggesting that part of this response was produced via SOCs due to the SR depletion. Similar results were shown by Potocnick et al. (2001) in skeletal muscle arterioles, where a capacitative Ca2+ influx produced by re-addition of extracellular Ca2+ following pre-treatment with an extracellular Ca2+-free solution (plus nifedipine) could be inhibited by 2-APB. They also demonstrated that 2-APB diminished the contractile response and (Ca2+)i transient evoked by phenylephrine but not by KCl. In vascular smooth muscle, Ca2+-permeable non-selective cationic channels able to be activated by Ca2+ store-depletion were also described in rabbit portal vein (Albert and Large, 2002), in human pulmonary artery (Golovina et al., 2001), in rabbit pial arterioles (Xu et al., 2006), in mouse and rabbit aorta (Trepakova et al., 2001) and in mouse pulmonary artery (Ng et al., 2009), among others.
It is interesting to note that in the human umbilical artery, 2-APB did not block all the force development induced by Ca2+ readdition. Instead, this was only eliminated by a combination of 2-APB and other antagonists of Ca2+ entry pathways (nifedipine, Gd3+ and KB-R7943). All of these blockers partially inhibited force development when added independently, meaning that there are multiple pathways which act in a concerted way to let Ca2+ into the cell after a period of extracellular Ca2+ deprivation. Some of these routes are related to the capacitative response, as evidenced by 2-APB effects (assuming that 2-APB is more selective for SOCs).
Our results suggest that smooth muscle cells in this artery express all the structures which many bibliographical data point as necessary for the production of a capacitative response. Namely, we present evidences that smooth muscle cells of the human umbilical artery express the mRNAs for STIM 1 and 2, for Orai 1, 2 and 3 and also for some of the members of the TRPC channels family, namely the 1, 3, 4 and 6 subtypes. The mRNAs for TRPC5 and 7 were found not to be present in these cells. According to the literature, the TRPC channel most likely to be a SOC in cells presenting capacitative response is the TRPC1 subtype, and to a lesser extent TRPC3, TRPC4 and TRPC6 (Leung et al., 2008; Parekh and Putney, 2005; Dietrich et al., 2006; Ng et al., 2010). Meng et al. (2007) report expression of mRNAs for TRPC 1, 3, 4 and 6 in cultured smooth muscle cells derived from the human umbilical artery. Our results may be considered a step further of those presented by these authors, since we used native cells, while they worked with cultured cells, where the pressure induced by growth factors in the culture media may have altered channel expression (Sandow and Hilton Grayson, 2009).
STIM 1 and 2, as well as Orai 1 and 3, seem to be broadly expressed in different tissues (Frischauf et al., 2008). However, Orai 2 is more expressed in the brain and less in other tissues (Frischauf et al., 2008). Both STIM 1 and 2, are able to activate the plasma membrane Orai proteins (Frischauf et al., 2008) and there are some recent reports that STIM1 could also interact with TRPC channels (Soboloff et al., 2006). Moreover, Liao et al. (2008) have reported that a STIM 1/Orai 1/TRPC 1 interaction is necessary to produce a capacitative response.
All these structures are expressed in the human umbilical artery smooth muscle, but it is beyond the scope of this work to establish if in these cells TRPCs, Orai, or a combination of both, are the target of STIM proteins. However, this subject merits further research in order to clarify the physiological roles of these structures in the human umbilical artery and other human vessels. This may provide more data for the understanding of vascular pathological processes affecting feto-placental blood flow.
In this work we have shown for the first time that smooth muscle of the human umbilical artery is able to evoke a capacitative contractile response inhibited by 2-APB, that stimulation with physiological vasoactive agonists which release Ca2+ from the SR is enough to induce this response, and that this artery expresses mRNA for STIM 1-2, Orai 1-3 and TRPC 1, 3, 4 and 6 channels which in the literature have been demonstrated to be involved in this mechanism.
Acknowledgements
The authors gratefully acknowledge excellent tech-nical assistance by Mr. Luciano Piccinini and Mr. Matías Vilche. They also wish to thank Mr. Pablo Urdampilleta, Ms. Anabel Poch and the staff of the Instituto Central de Medicina for the provision of umbilical cords. This work was financially supported by the grant PIP 0202 from the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Argentina.
References
1. Albert A P, Large WA (2002). A Ca2+ permeable non-selective cation channel activated by depletion of internal Ca2+ stores in single rabbit portal vein myocytes. Journal of Physiology 538: 717-728. [ Links ]
2. Albert A P, Saleh SN, Peppiatt-Wildman CM, Large WA (2007). Multiple activation mechanisms of store-operated TRPC channels in smooth muscle cells. Journal of Physiology 583: 25-36. [ Links ]
3. Baryshnikov SG, Pulina M V, Zulian A, Linde CI, Golovina VA (2009). Orai1, a critical component of store-operated Ca2+ entry, is functionally associated with Na+/Ca2+ exchanger and plasma membrane Ca2+ pump in proliferating human arterial myocytes. American Journal of Physiology (Cell Physiology) 297: C1103-1112. [ Links ]
4. Bertrand C, St-Louis J (1999). Reactivities to serotonin and histamine in umbilical and placental vessels during the third trimester after normotensive pregnancies and pregnancies complicated by preeclampsia. American Journal of Obstetrics and Gynecology 180: 650-659. [ Links ]
5. Bird GS, DeHaven WI, Smyth JT, Putney Jr. JW (2008). Methods for studying store-operated calcium entry. Methods 46: 204-212. [ Links ]
6. Bolotina VM, Csutora P (2005). CIF and other mysteries of the store-operated Ca2+-entry pathway. Trends in Biochemical Sciences 30: 378-387. [ Links ]
7. Bootman MD, Collins TJ, Mackenzie L, Roderick HL, Berridge MJ, Peppiatt CM (2002). 2-aminoethoxydiphenyl borate (2-APB) is a reliable blocker of store-operated Ca2+ entry but an inconsistent inhibitor of InsP3-induced Ca2+ release. FASEB Journal 16: 1145-1150. [ Links ]
8. Chomczynski P, Sacchi N (1987). Single-step method of RNA iso-lation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anals of Biochemistry 162: 156-159. [ Links ]
9. Deng X, Wang Y, Zhou Y, Soboloff J, Gill DL (2009). STIM and Orai: dynamic intermembrane coupling to control cellular calcium signals. Journal of Biological Chemistry 284: 22501-22505. [ Links ]
10. Dietrich A, Chubanov V, Kalwa H, Rost BR, Gudermann T (2006). Cation channels of the transient receptor potential superfamily: their role in physiological and pathophysiological processes of smooth muscle cells. Pharmacology & Therapeutics 112: 744-760. [ Links ]
11. Fasolato C, Hoth M, Penner R (1993). A GTP-dependent step in the activation mechanism of capacitative calcium influx. Journal of Biological Chemistry 268: 20737-20740. [ Links ]
12. Fasolato C, Nilius B (1998). Store depletion triggers the calcium release-activated calcium current (ICRAC) in macrovascular endothelial cells: a comparison with Jurkat and embryonic kidney cell lines. Pflugers Archives 436: 69-74. [ Links ]
13. Frischauf I, Schindl R, Derler I, Bergsmann J, Fahrner M, Romanin C (2008). The STIM/Orai coupling machinery. Channels (Austin) 2: 261-268. [ Links ]
14. Golovina VA, Platoshyn O, Bailey CL (2001). Upregulated TRP and enhanced capacitative Ca2+ entry in human pulmonary artery myocytes during proliferation. American Journal of Physiology (Heart and Circulatory Physiology) 280: H746-755. [ Links ]
15. Gregory RB, Rychkov G, Barritt GJ (2001). Evidence that 2-aminoethyl diphenylborate is a novel inhibitor of store-operated Ca2+ channels in liver cells, and acts through a mechanism which does not involve inositol trisphosphate receptors. Biochemical Journal 354: 285-290. [ Links ]
16. Guibert C, Marthan R, Savineau JP (2004). 5-HT induces an arachidonic acid-sensitive calcium influx in rat small intrapulmonary artery. American Journal of Physiology (Lung Cellular and Molecular Physiology) 286:L1228-1236. [ Links ]
17. Hewavitharana T, Deng X, Soboloff J, Gill DL (2007). Role of STIM and Orai proteins in the store-operated calcium signaling path-way. Cell Calcium 42: 173-182. [ Links ]
18. Leung F P, Yung LM, Ya o X, Laher I, Huang Y (2008). Store-operated calcium entry in vascular smooth muscle. British Journal of Pharmacology 153: 846-857. [ Links ]
19. Liao Y, Erxleben C, Abramowitz J, et al. (2008). Functional interactions among Orai1, TRPCs, and STIM1 suggest a STIM-regulated heteromeric Orai/TRPC model for SOCE/Icrac channels. Proceedings of the National Academy of Sciences USA 105:2895-2900. [ Links ]
20. Lovren F, Li X F, Lytton J, Triggle C (1999). Functional characterization and m-RNA expression of 5-HT receptors mediating contraction in human umbilical artery. British Journal Pharmacology 127: 1247-1255. [ Links ]
21. Maruyama T, Kanaji T, Nakade S, Kanno T, Mikoshiba K (1997). 2APB, 2-aminoethoxydiphenyl borate, a membrane-penetrable modulator of Ins(1,4,5)P3-induced Ca2+ release. Journal of Biochemistry 122: 498-505. [ Links ]
22. Meng F, To W, Kirkman-Brown J, Kumar P, Gu Y (2007). Calcium oscillations induced by ATP in human umbilical cord smooth muscle cells. Journal of Cellular Physiology 213: 79-87. [ Links ]
23. Moneer Z, Pino I, Taylor EJ, et al. (2005). Different phospholipase-C-coupled receptors differentially regulate capacitative and non-capacitative Ca2+ entry in A7r5 cells. Biochemical Journal 389: 821-829. [ Links ]
24. Ng LC, Airey JA, Hume JR (2010). The contribution of TRPC1 and STIM1 to capacitative Ca2+ entry in pulmonary artery. Advances in Experimental and Medical Biology 661: 123-135. [ Links ]
25. Ng LC, McCormack MD, Airey JA, et al. (2009). TRPC1 and STIM1 mediate capacitative Ca2+ entry in mouse pulmonary arterial smooth muscle cells. Journal of Physiology 587: 2429-2442. [ Links ]
26. Parekh AB, Penner R (1995). Depletion-activated calcium current is inhibited by protein kinase in RBL-2H3 cells. Proceedings of the National Academy of Sciences USA 92: 7907-7911. [ Links ]
27. Parekh AB, Penner R (1997). Store depletion and calcium influx. Physiological Reviews 77: 901-930. [ Links ]
28. Parekh AB, Putney JW Jr. (2005). Store-operated calcium channels. Physiological Reviews 85: 757-810. [ Links ]
29. Peinelt C, Vig M, Koomoa DL, et al. (2006). Amplification of CRAC current by STIM1 and CRACM1 (Orai1). Nature Cell Biology 8: 771-773. [ Links ]
30. Potocnik SJ, Hill MA (2001). Pharmacological evidence for capacitative Ca2+ entry in cannulated and pressurized skeletal muscle arterioles. British Journal of Pharmacology 134: 247-256. [ Links ]
31. Putney JW Jr, Broad LM, Braun FJ, Lievremont J P, Bird GS (2001). Mechanisms of capacitative calcium entry. Journal of Cell Science 114: 2223-2229. [ Links ]
32. Roberts-Thomson SJ, Peters AA, Grice DM, Monteith GR (2010). ORAI-mediated calcium entry: mechanism and roles, diseases and pharmacology. Pharmacology & Therapeutics 127: 121-130. [ Links ]
33. Rodat-Despoix L, Aires V, Ducret T, et al. (2009). Signalling pathways involved in the contractile response to 5-HT in the human pulmonary artery. European Respiratory Journal 34: 1338-1347. [ Links ]
34. Sandow SL, Hilton Grayson T (2009). Limits of isolation and culture: intact vascular endothelium and BKCa. American Journal of Physiology (Heart and Circulatory Physiology); 297: H1-H7. [ Links ]
35. Soboloff J, Spassova MA, Tang XD, Hewavitharana T, Xu W, Gill D (2006). Orai1 and STIM reconstitute store-operated calcium channel function. Journal of Biological Chemistry 281: 20661-20665. [ Links ]
36. Trepakova ES, Gericke M, Hirakawa Y, Weisbrod RM, Cohen RA, Bolotina VM (2001). Properties of a native cation channel activated by Ca2+ store depletion in vascular smooth muscle cells. Journal of Biological Chemistry 276: 7782-7790. [ Links ]
37. Tufan H, Ayan-Polat B, Tecder-Unal M, Polat G, Kayhan Z, O∂üπ E (2003). Contractile responses of the human umbilical artery to KCl and serotonin in Ca-free medium and the effects of levcromakalim. Life Sciences 72: 1321-1329. [ Links ]
38. Xu SZ, Boulay G, Flemming R, Beech DJ (2006). E3-targeted anti-TRPC5 antibody inhibits store-operated calcium entry in freshly isolated pial arterioles. American Journal of Physiology (Heart and Circulatory Physiology) 291: H2653-2659. [ Links ]
Received: September 16, 2011.
Revised version received: March 13, 2012.
Accepted: July 14 , 2012.














