Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Biocell
Print version ISSN 0327-9545
Biocell vol.37 no.2 Mendoza Aug. 2013
ORIGINAL ARTICLES
Age related interaction of dopamine and serotonin synthesis in striatal synaptosomes
Manoochehr Messripour1 and Azadeh Mesripour2
1Department of Medical Biochemistry, Khorasgan Branch, Islamic Azad University, Isfahan, Iran
2Medical Plant Research Center, Shahre Kord University of Medical Sciences, Iran
*Address correspondence to:
Manoochehr Messripour. Department of Medical Biochemistry, Khorasgan Branch, Islamic Azad University, Isfahan, IRAN. E-mail: m_messripour@yahoo.com
Received: June 14, 2012.
Revised version received: August 14, 2013.
Accepted: August 18, 2013.
ABSTRACT: Tyrosine hydroxylase and tryptophan hydroxylase are key rate limiting enzymes in the biosyn-thesis of dopamine and serotonin, respectively. Since both enzymes are active in striatum, and affected by age, this study was undertaken to investigate interaction between dopamine and serotonin synthesis in brain striatal synaptosomes of aging rat. Male Wistar rats (3 and 30 month old) were killed by decapitation and brain striatal synaptosomes were prepared by discontinuous Ficoll/sucrose gradient technique. Synaptosomes were incubated in the presence of added pargiline (monoamineoxidase inhibitor), dopamine or serotonin synthesized during 25 min was measured by HPLC, employing electrochemical detection. Dopamine synthesis in synaptosomes prepared from young animals was markedly inhibited by addition of 5 µM serotonin concentrations (30%) and increasing serotonin concentrations up to 50 µM caused only a smaller additional inhibition. Dopamine synthesis in synaptosomes obtained from old rats was significantly lower than that of youg animals and addition of serotonin concentrations up to 50 µM had little effect on these preparations. In case of serotonin synthesis, exogenously added 5 µM dopamine inhibited serotonin synthesis in the synaptosomes of both ages by about 40%, whereas with higher concentration of dopamine (10-50 µM) the rate of inhibition was highly pronounced in old rats as compared to that of young animals. It is concluded that dopamine and serotonin interaction may be significant, and that these should be considered in long-term treatments of Parkinson's disease with L-DOPA.
Keywords: Aging; Nerve ending; Neurotransmitter; Tyrosine hydroxylase; Tryptophan hydroxylase
Introduction
Tyrosine hydroxylase (TH, EC 1.14.16.2) and tryptophan hydroxylase (TPH, 1.14.16.4) are key rate limiting enzymes in the biosynthesis of dopamine and serotonin respectively (Nagatsu et al., 1964; Lovenberg et al., 1968). A number of investigations on partially purified cell free preparations of TH and TPH have indicated that these enzymes interact with several different molecules, such as non-heme iron (II), molecular oxygen and reduced pteridine cofactor BH4 to catalyze the conversion of tyrosine to dihydroxyphenylalanine and 5-hydroxytryptophan (Hamon et al., 1981; Andersson et al., 1992). They utilize molecular oxygen to oxidize the reduced pteridine cofactor BH4. Based on kinetic studies of rat TH-catalyzed reaction, a sequential reaction mechanism has been proposed with an ordered binding of BH4 as the first substrate, followed by dioxygen and tyrosine which is subjected to feedback inhibition by catecholamines (Flatmark et al., 1999). This inhibition is suggested to be competitive with BH4 and not with tyrosine (Flatmark et al., 1999). Similar studies on a possible serotonin TPH feedback loop have been complicated by difficulty of preparing adequate of purified enzyme of sufficient stability (Vitto and Mandell, 1982). However, a number of reports indicated that both enzymes are tightly regulated by inhibitory feedback via autoreceptors, a phenomenon which has been termed "receptor mediated feedback inhibition"(el Mestikawy and Hamon, 1986; Mestikawy et al., 1986). Moreover, it is now well established that serotonin and catecholamines inhibit TH and TPH activities respectively (Devau et al., 1987; Johnson et al., 1993).
Synaptosomes retain the natural pteridine cofactor necessary for tyrosine and tryptophan hydroxylation and also the ability to respond to exogenously applied substances in a manner similar to that reported for in vivo experiments (Devau et al., 1987; Johnson et al., 1993; Makoto et al., 1994; Lladó et al., 1996). Since dopamine and serotonin share a common synthetic pathway and the synthesis of striatal neurons transmitter is influenced by age (Ponzio et al., 1982; Hashiguti et al., 1993) this study was undertaken to investigate the interaction between dopamine and serotonin synthesis in brain striatal synaptosomes in the aging rat.
Materials and Methods
Animals and chemicals
Male Wistar rats were housed in a temperature-controlled room at 20-24ºC and maintained in a 12-h light/dark cycle. Food and water were provided ad libitum. 'Young rats' were 3 months old and weighed 200-250 g, while 'old rats' were 30 months old and weighed 650-720 g. All procedures were approved by the Ethical Committee of the Isfahan University of Medical Sciences, and conducted in accordance with the 'Principles of Laboratory Animal Care' (National Institutes of Health publication no. 86-23, revised 1985).
All chemicals were of reagent grade and obtained from Sigma Chemical Company (Germany). Deionized water was used throughout this study.
Preparation of synaptosomes
Rats were killed by decapitation between 8 to 9 AM and the brain was dissected on ice by the method of Glowinski and Iversen (1966). Synaptosomes were prepared from the dissected striatum essentially as described by Booth and Clark (1978). Briefly, the dissected brain striatum from 6 young or 4 old rats were dropped into ice-cold isolation medium (0.32M-sucrose/L mM-potassium EDTA/l0 mM-Tris HCl, pH7.4) and chopped into small pieces with scissors. Blood and other debris were washed out and the chopped tissue was then homogenized in a glass homogenizer using a glass pestle with 0.1 mm clearance. This homogenate was diluted to 60 mL with isolation medium and centrifuged at 1200 gfor 3 min at 4ºC. The supernatant was then centrifuged at 16000 g for 10 min, producing the crude mitochondrial/synaptosomal pellet. This pellet was resuspended in 30 mL of Ficoll/sucrose medium [12% (w/w) Ficoll, 0.32 M sucrose, 50 M potassium EDTA, pH 7.4] and homogenized. The suspention was transferred into a centrifuge tube and above this 5 mL of 7.5% Ficoll/ sucrose medium [7.5% (w/w) Ficoll, 0.32M-sucrose, 50 pM-potassium EDTA, pH 7.4] was carefully layered. Finally, 5 mL of isolation medium was also carefully layered on top. Then the tubes were centrifuged at 70000 gfor 40 min at 4ºC. Synaptosomes were gently sucked off from the the second interphase and resuspended in 10 mL of cooled incubation medium (125 mM NaCI, 5 mM KCl, 1 mM CaCl2, 1 mM MgCl2, 10 mM glucose, 1mM ascorbic acid, and 15 mM sodium phosphate buffer) and centrifuged at 5500 g for 10 min. The pellet was resuspended in incubation medium to give a protein concentration of 8 to 10 mg/mL (as determined by the Lowry's method, after lysing synaptosomal membranes in a 2%, w/v, Na-deoxycholate solution). The optimal pHs for dopamine (Patrick and Barchas, 1974) and serotonin synthesis (Karobath, 1972) were adjusted by using different concentrations of Na2HPO4 or NaH2PO4 at 6.1 and 7.4 respectively. All procedures were performed at 4ºC.
Lactate dehydrogenase assay
The integrity of the synaptosomal membrane was assessed by lactate dehydrogenase (LDH) activity in the presence and absence of 0.1 % Triton X-100 (Booth and Clark, 1978). The assay was performed in the same incubation media with the optimal pHs used for determination of tyrosine hydroxylase (pH 6.1) and tryptophan hydroxylase (pH 7.4) plus NADH (0.2 mM) at 25ºC using a Pye Unican spectrophotometer (SP 8199). The reaction was started by addition of pyruvate (0.2 mM) and oxidation of NADH (0.2 mM) at 340 nm was monitored against a blank containing all compounds except pyruvate.
Measurement of dopamine and serotonin synthesis
The methods involved determination of the levels of dopamine or serotonin by HPLC before and after incubation of synaptosomes, in the presence of a monoamine oxidase inhibitor, pargyline (Messripour and Clark, 1982; Rastegar et al., 1993). Briefly, 200 µL (2 mg protein) aliquots of resuspended synaptosomes were incubated in the presence of either 40 µM tyrosine or tryptophan and 100 µM pargyline. The incubations were carried out at pHs of 6.1 or 7.4 for 15 min at 37ºC and the reactions were stopped by freezing the samples over solid CO2. The concentration of the amines in non incubated samples (blanks) were subtracted from the amine content of the incubated samples to calculate the net rate of dopamine or serotonin formed in an appropriate incubation period. The results are expressed as the amount of the amine formed/min/mg protein.
High performance liquid chromatography
Dopamine and serotonin were extracted from samples as previously described (Messripour and Clark, 1982; Rastegar et al., 1993) and HPLC assays of the amine content of the extracts were carried out on an Altex 15cm X 4.6mm I.D. Ultrasphere-IP column, using an Altex model 110A pump and LC-4 amperometric controller with a TL-4 detector compartment (Bioanalytical systems) and linked to a Hewlett-Packard integrator recorder (HP 3380A). The mobile phase for chromatography of dopamine was composed of 90% 0.1M potassium dihydrogen orthophosphate, 0.l mM potassium EDTA, 0.3 mM sodium octyl sulphate and 10% HPLC grade methanol (final pH 3.0). The same mobile phase was used for estimation of serotonin except that a lower (0.3 mM) concentration of sodium octyl sulphate was used.
Statistical analysis
Results were expressed as mean±SD. The inter-group variations were evaluated by one way analysis of variance (ANOVA) followed by Tukey's test, using SPSS (Statistical Package for the Social Sciences. Statistical level of significance was set at P < 0.05.
Results
The LDH latency (the ratio between apparent LDH activity in the presence or absence of Triton X-l00) showed no significant differences at pH 6.1 (17.7 ± 0.5, n=6) and 7.4 (17.6 ± 0.6, n=6). However, the synthesis of dopamine and serotonin by striatal synaptosomes was affected by pH, being high for dopamine and low for serotonin at pH 6.1, and high for serotonin and low for dopamine at pH 7.4.
The rates of dopamine synthesis by striatal synaptosomal preparations from young and old rats were 14.55 ± 1.3 and 10.79 ± 0.66 pmol/min/mg protein, respectively. The differences were statistically significant. However, the rate of serotonin synthesis by synaptosomes was not significantly different between young and old rats (2.48± 0.33 and 2.63± 0.29 pmol/ min/mg protein, respectively).
Figure 1 shows the effect of a range of serotonin concentrations on dopamine synthesis by striatal synaptosomes prepared from young and old rats. Dopamine synthesis by synaptosomes from young rats was markedly inhibited (~30 %) by the addition of 5-50 µM serotonin. However, these concentrations of serotonin had no effect on dopamine synthesis by synaptosomes from old rats.
Figure 2 shows the inhibitory effect of a range of dopamine concentrations on serotonin synthesis by striatal synaptosomes from both young and old rats. Synthesis was inhibited by the synaptosomes at both ages by about 40% (P<0.005) whereas with higher concentration of dopamine (10-50 µM), the rate of inhibition was highly pronounced in the synaptosomes of old rats as compared to those of young animals.The dose response curve was a non linear fashion for both ages, but the pattern of inhibition was different to that seen for dopamine synthesis. The effect is more marked in synaptosomes from old than from young rats.
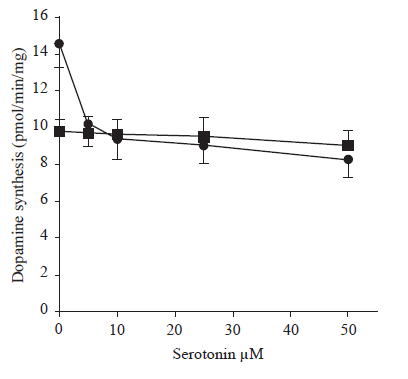
FIGURE 1. The effect of different concentration of serotonin on dopamine synthesis in young and old rats. The striatal synaptosomal preparations from 3 months (.-.) and 30 months (¦-¦) old were incubaed in different concentrations of serotonin (0 to 50 µM), and the rate of dopamine synthsis were measured in the presence of tyrosine (40 mM) and pargyline (100 mM) by HPLC method. Each point represents the mean ± SD of 6 separate experiments performed in triplicate. Differences between young and old rats in the absence of serotonin (0µM) is statisticaly significant.
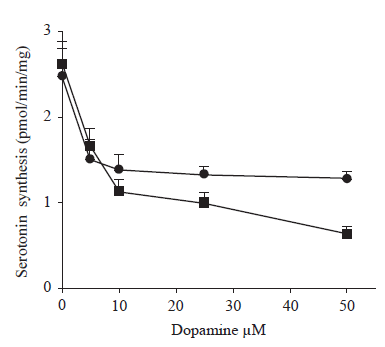
FIGURE 2. The effect of different concentration of dopamine on serotonin synthesis in young and old rats. The striatal synaptosomal preparations from 3 months (.-.) and 30 months (¦-¦) old were incubated in different concentrations of dopamine (0 to 50 µM), and the rate of serotonin synthesis was measured in the presence of tryptophan (40 mM) and pargyline (100 mM) by HPLC. Each point represents the mean± SD of 6 separate experiments performed in triplicate. Differences between young and old rats with dopamine concentrations of 25 and 50 µM are statistically significant.
Discussion
Membrane integrity of synaptosomes is believed to be an important factor for the maintenance of the enzymes and the reduced pteridine cofactor at near optimal levels for dopamine and serotonin synthesis (Karobath, 1972; Patrick and Barchas, 1974). As has been indicated previously (Booth and Clark, 1978; Messripour and Clark, 1982) the measurement of dopamine and serotonin synthesis in purified synaptosomes in presence of a monoamine oxidase inhibitor gives rise to the optimal rate at which the neurotransmitters can be synthesized at the synapses from their precursors tyrosine and tryptophan respectively. The procedure for synaptosomal preparation may have important implications for the final metabolic and membrane integrity of the obtained synaptosomes and hence on its suitability as a model of the nerve ending for studying neurotransmitters interaction at the synaptic region of the neurons of the brain. The either strong or weak responses of synaptosomes to low (up to 5 µM) and high (5-50 µM) concentrations of the exogenously added neurotransmitters (Figs. 1 and 2) is supposed to be effected by at least 2 mechanisms; (a) by uptake of the serotonin or dopamine from the assay mixture thus increasing intramembrane concentrations of the neurotransmitter and causing inhibition of the hydroxylase enzymes and (b) by presyn-aptic receptors, which may influence neurotransmitter release and hence intraterminal neurotransmitters via Ca2+ and cyclic AMP dependent mechanisms cause regulation of the enzyme protein itself. This study confirms the hypothesis of an inhibitory control by serotonin on TH activity in the dopaminergic neurons of the sriatum, indicating that serotonin could regulate the activity of the enzyme through specific serotoninergic receptor (De Deurwaerdère et al., 2004) . However, the results of this study indicate (Fig. 1) that dopamine synthesis in the synaptosomal preparations of old rat striatum was not affected by addition of serotonin. It is suggested that dopaminergic modulatory mechanisms on the presynaptic regions of striatal neurons are not functioning in old rats as they are doing in the neurons of young animals. Conversely, the synthesis of serotonin in the synaptosomal preparation of old animals was more sensitive to the addition of dopamine as compared to that of young animals (Fig. 2). It appears that dopamine/ serotonin "cross talk"regulates the neurotransmitter levels at synapses is significantly modulated during the aging process. Such interaction might be noteworthy, where longterm treatment with the dopamine precursor, L-DOPA of patients suffering from Parkinson's disease, renders the drug less effective and patients experience fluctuations in response and even psychiatric problems (Rascol et al., 2000) which might be mediated by decreased serotonin levels.
Acknowledgements
This work was supported by the Research Council of Khorasgan Branch, Islamic Azad University, Isfahan.
References
1. Andersson K, Vassort C, Brennan B, Que Jr L, Haavik J, Flatmark T, Gros F, Thibault J (1992). Purification and characterization of the blue-green rat phaeochromocytoma (PC12) tyrosine hydroxylase with a dopamine-Fe (III) complex. Reversal of the endogenous feedback inhibition by phosphorylation of serine-40. Biochemical Journal 284: 687-695. [ Links ]
2. Booth RF, Clark JB (1978). A rapid method for the preparation of relatively pure metabolically competent synaptosomes from rat brain. Biochemical Journal 176: 365-370. [ Links ]
3. De Deurwaerdère P, Navailles S, Berg KA, Clarke WP, Spampinato U (2004). Constitutive activity of the serotonin2C receptor inhibits in vivo dopamine release in the rat striatum and nucleus accumbens. Journal of Neuroscience 24: 3235-3241. [ Links ]
4. Devau G, Multon MF, Pujol JF, Buda M (1987). Inhibition of tyro-sine hydroxylase activity by serotonin in explants of newborn rat locus ceruleus. Journal of Neurochemistry 49: 665-670. [ Links ]
5. el Mestikawy S, Hamon M (1986). Is dopamine-induced inhibition of adenylate cyclase involved in the autoreceptor-mediated negative control of tyrosine hydroxylase in striatal dopaminergic terminals? Journal of Neurochemistry 47: 1425-1433. [ Links ]
6. Flatmark T, Almås B, Knappskog PM, Berge SV, Svebak RM, Chehin R, Muga A, Martínez A (1999). Tyrosine hydroxylase binds tetrahydrobiopterin cofactor with negative cooperativity, as shown by kinetic analyses and surface plasmon resonance detection. European Journal of Biochemistry 262: 840-849. [ Links ]
7. Glowinski J, Iversen LL (1966). Regional studies of catecholamines in the rat brain. I. The disposition of [3H]norepinephrine, [3H]dopamine and [3H]dopa in various regions of the brain. Journal of Neurochemistry 13: 655-669. [ Links ]
8. Hamon M, Bourgoin S, Artaud F, El Mestikawy S (1981). The respective roles of tryptophan uptake and tryptophan hydroxylase in the regulation of serotonin synthesis in the central nervous system. Journal de Physiologie 77: 269-279. [ Links ]
9. Hashiguti H, Nakahara D, Maruyama W, Naoi M, Ikeda T (1993). Simultaneous determination of in vivo hydroxylation of tyro-sine and tryptophan in rat striatum by microdialysis-HPLC: relationship between dopamine and serotonin biosynthesis. Journal of Neural Transmission - General Section 93: 213-223. [ Links ]
10. Johnson EA, Tsai CE, Shahan YH, Azzaro AJ (1993). Serotonin 5-HT1A receptors mediate inhibition of tyrosine hydroxylation in rat striatum. Journal of Pharmacology and Experimental Therapeutics 266: 133-141. [ Links ]
11. Karobath M (1972). Serotonin synthesis with rat brain synaptosomes. Effects of serotonin and monoamineoxidase inhibitors. Biochemical pharmacology 21: 1253-1263. [ Links ]
12. Lladó J, Esteban S, García-Sevilla JA (1996). The [alpha] 2-adreno-ceptor antagonist idazoxan is an agonist at 5-HT1A autoreceptors modulating serotonin synthesis in the rat brain in vivo. Neuroscience letters 218: 111-114. [ Links ]
13. Lovenberg W, Jequier E, Sjoerdsma A (1968). Tryptophan hydroxylation in mammalian systems. Advances in Pharmacology 6: 21-36. [ Links ]
14. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry 193: 265-275. [ Links ]
15. Makoto N, Wakako M, Tsutomu T, Miyuki O, Kasan P (1994). Inhibition of tryptophan hydroxylase by dopamine and the precursor amino acids. Biochemical Pharmacology 48: 207-210. [ Links ]
16. Messripour M, Clark JB (1982). Tyrosine hydroxylase activity in rat brain synaptosomes: direct measurement using high performance liquid chromatography. Journal of Neurochemistry 38: 1139-1143. [ Links ]
17. Mestikawy S, Glowinski J, Hamon M (1986). Presynaptic dopa-mine autoreceptors control tyrosine hydroxylase activation in depolarized striatal dopaminergic terminals. Journal of Neurochemistry 46: 12-22. [ Links ]
18. Nagatsu T, Levitt M, Udenfriend S (1964). Tyrosine Hydroxylase. The Initial Step in Norepinephrine Biosynthesis. Journal of Biological Chemistry 239: 2910-2917. [ Links ]
19. Patrick R, Barchas J (1974). Regulation of catecholamine synthesis in rat brain synaptosomes. Journal of Neurochemistry 23: 7-15. [ Links ]
20. Ponzio F, Calderini G, Lomuscio G, Vantini G, Toffano G, Algeri S (1982). Changes in monamines and their metabolite levels in some brain regions of aged rats. Neurobiology of Aging 3: 23-29. [ Links ]
21. Rascol O, Brooks DJ, Korczyn AD, De Deyn PP, Clarke CE, Lang AE (2000). A five-year study of the incidence of dyskinesia in patients with early Parkinson's disease who were treated with ropinirole or levodopa. New England Journal of Medicine 342: 1484-1491. [ Links ]
22. Rastegar A, Ciesielski L, Simler S, Messripour M, Mandel P (1993). Brain monoamines following castration of aggressive muricidal rats. Neurochemical Research 18: 471-477. [ Links ]
23. Vitto A, Mandell AJ (1982). Stability properties of activated trypto-phan hydroxylase from rat midbrain. Journal of Neurochemistry 37: 601-607. [ Links ]














