Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de la Sociedad Entomológica Argentina
Print version ISSN 0373-5680On-line version ISSN 1851-7471
Rev. Soc. Entomol. Argent. vol.62 no.3-4 Mendoza Aug./Dec. 2003
Effects of some sesquiterpenes on the stored-product insect Tenebrio molitor (Coleoptera: Tenebrionidae)
Efectos de algunos sesquiterpenos sobre el insecto de productos almacenados, Tenebrio molitor (Coleoptera: Tenebrionidae)
García, Matías *,**, Marta E. Sosa*, Osvaldo J. Donadel**, Oscar S. Giordano** and Carlos E. Tonn**
*Área de Zoología. **INTEQUI-CONICET. Facultad de Química, Bioquímica y Farmacia. Universidad Nacional de San Luis. Chacabuco y Pedernera, 5700, San Luis. Argentina; e-mail: molitor@unsl.edu.ar
ABSTRACT. In order to evaluate the allelochemical activity of some sesquiterpenes isolated from the native plant Tessaria absinthioides (Hook. et Arn.) DC, and some semi synthetic derivatives against Tenebrio molitor L. larvae, we have developed bioassays directed to quantify repellency, larval mortality, and its effects on the development. Although costic aldehyde caused the maximum repellent effect, all the compounds showed a significant effect at some dose or time, indicating behavioral avoidance. The topical application of costic aldehyde produced the largest increase on the duration of the pupal stage. Tessaric acid exhibited the highest toxicity by topical application at the experiment closure. Both eremophilane–1(10),2,11(13)–triene–12–oic, and g-costic acids induced some morphological deformities.
KEY WORDS. Sesquiterpenes. Tenebrio molitor. Repellency. Development. Mortality.
RESUMEN. Con el objeto de evaluar sesquiterpenos aislados de la planta nativa Tessaria absinthioides (Hook et Arn) y algunos derivados semisintéticos frente a larvas de Tenebrio molitor L., se desarrollaron bioensayos orientados a la cuantificación de la repelencia, mortalidad de larvas y efectos sobre el desarrollo. Aldehído cóstico produjo el mayor incremento en la duración del estado pupal por aplicación tópica. Acido tessárico exhibió el más alto porcentaje de mortalidad al finalizar el período de experimentación. Los productos eremophilan-1(10),2, 11(13)–trien–12–oico y ácido g–cóstico dieron lugar al mayor número de malformaciones. Si bien aldehído cóstico mostró la máxima actividad de repelencia, todos los compuestos evaluados produjeron efectos significativos en el ensayo de elección.
PALABRAS CLAVE. Sesquiterpenos. Tenebrio molitor. Repelencia. Desarrollo. Mortalidad.
INTRODUCTION
It is well accepted that natural products from plants may constitute new sources of insect pest control. In this regard, a group of alellochemicals with potential value is the terpenoids. The activities of these compounds have been studied against stored-product insects (Harmatha & Nawrot, 1984; Daniewsky et al., 1993, 1995; González Coloma et al., 1995; Phillips et al., 1993, 1995, Phillips, 1997; Huang & Ho, 1998; Ortego et al., 1999). For sesquiterpenes, their biological activities are many and varied, from plant growth regulation to interference with insect metamorphosis (Nawrot et al., 1984; Schoonhoven & Fu-shun, 1989; Harborne, 1993).
Tenebrio molitor L. is a serious stored-product insect pest with a world–wide distribution (Soltani et al., 2002). The effects of secondary compounds on it have been shown in several studies (Pascual et al., 1990; Enriz et al., 1994, 2000; Luco et al., 1994; Sosa et al., 1994, 2000; Carrizo et al., 1998). In this work we have evaluated the bioactivity of sesquiterpenes isolated from plants growing in the Cuyo region (Argentina) and some semisynthetic derivatives, against T. molitor larvae.
MATERIAL AND METHODS
Chemicals. Tessaric acid (1) and ilicic acid (2) (Fig. 1) were isolated from aerial parts of Tessaria absinthioides (Hook. et Arn.) DC, as previously reported (Kurina Sanz et al., 1997). Compound 3 was obtained from 2 by dehydration (Donadel et al., 1998). Compound 4 was obtained from 1 by reduction using alane (LiAlH4/AlCl3) in THF (Brown & Krishnamurthy, 1979). Derivatives 5 and 6 were prepared by reduction and dehydration reactions of compound 2 (Guerreiro et al., 1979). Identities of all compounds were confirmed by 1H NMR (200.13 MHz), 13C NMR (50.23 MHz), EIMS, and IR spectral data, as well as optical rotation.

Fig. 1. Structure of compounds 1-6.
Insects. Experiments were conducted in the laboratory using established colonies of T. molitor. Larvae were reared on wheat brain at 25 ± 1°C, 65 % relative humidity, and a 16:8 (L:D) photoperiod.
Laboratory assays.
Topical application. Recently ecdysed fifth instar larvae, of T. molitor were randomly selected, and starved for 24 hs. Two acetone solutions of each compound (20 and 40 µg/µl) were prepared. Test solutions were topically applied to the ventral surface of the thoracic segments with a Hamilton micro syringe (2 µl/larvae). Controls were treated with the solvent alone. There were two replicates of 15 larvae each, for each individual compound tested. After treatment, insects were placed into plastic vials (diameter 10 cm, height 7 cm) containing food and held at 25 ± 1°C with a 16:8 (L:D) photoperiod, and examined daily for 60 days (García et al., 2003). The duration (days) of the pupal stage was recorded, as well as the inhibition of the imaginal moult. Moult inhibition was classified according to the morphofunctional criteria reported by Pascual et al. (1990), using four basic categories (see description in Table I). This classification represents an arbitrary categorization of a continuum from normal adults, to inhibitory effects simply affecting the ecdysis and the pre–ecdysial cuticle deposition process.
Table I. Response of T. molitor larvae to sesquiterpene compounds in a topical bioassays. Effects on the duration (days ± SEM) of pupal instar and different degrees of imaginal moult inhibition

Insects mortality was recorded 1, 2, 3, and 60 days (experiment closure) after treatment. The mortality percentage was calculated by the equation [1-A*B/X*D]*100, where A represents the number of treated larvae that remain alive at the end of the experiment; B represents the number of untreated larvae at the beginning of the experiment; X represent the number of treated larvae at the beginning of the experiment; and D is the number of untreated larvae living at the end of the experiment (Henderson & Tilton, 1955).
Contact toxicity. An aliquot of 20 µl of each solution with 20 and 40 µg/µl concentration was diluted into 2 ml of acetone. The solution was applied to the surface of a glass Petri dish (diameter 9 cm, height 1.5 cm) and homogeneously dispersed. Controls were treated with solvent alone (García et al., 2003). The solvent was allowed to evaporate for two hours prior to the introduction of 10 larvae of the last instar of T. molitor. Treated Petri dishes were kept at 25 ± 1°C with a 16:8 (L:D) photoperiod. Each treatment was replicated three times. Insect mortality was recorded after 1, 2, and 3 days, and mortality (%) was calculated using the Henderson & Tilton (1955) equation.
Choice bioassay. The experiment utilized a two-choice arena. Two glass erlenmeyer flasks (100 ml) with a glass tube (diameter 0.5 cm; length 2 cm) added to its base each were joined together by a short rubber tube. A 1 ml dose of test solution was applied to reach 10, 20, 40 and 80 mg/cm2 concentration. Each solution was applied to the treated erlenmeyer as uniformly as possible with a pipette. The control side (the other erlenmeyer) was treated with acetone alone. Erlenmeyers were air dried to evaporate the solvent completely. Once the erlenmeyers were dry, they were joined by a rubber tube. Each rubber tube has a hole (diameter 0.4 cm) that make possible to introduce 10 last instar larvae of T. molitor. Once the larvae were introduced the hole in the rubber was covered with sello-tape. Each treatment was replicated ten times. Bioassays were conducted in complete darkness at 25± 1°C and 65% humidity. After 30 minutes, a Response Index (RI) for larvae in the two–choice bioassay was calculated as RI = (T-C/Tot)*100 (Phillips et al., 1993), where T is the number of larvae responding to the treatment; C is the number responding to the control, and Tot is the total number of insects released. Positive RIs indicate attraction to the treatment and negative RIs indicate repellency; values could theoretically range from –100 for a complete repellency, to +100 for complete attraction.
Statistical Analysis. Growth assays were analyzed using Kruskal - Wallis test; Dunn's Multiple Comparisons test at the P < 0.05 level. Toxicity by topical application was analyzed by x2. For contact toxicity, data were analyzed by Kruskal - Wallis test; Dunn's Multiple Comparisons test at the P < 0.05 level. In Response Index assays numbers responding to treatments and controls were subjected to ANOVA test, and Least Significant Difference test at the P < 0.05 level. PriProbit analysis (Sakuma, 1998) was used to determine ED50, ED90, and the corresponding 95% confidence intervals. A relation between the ED50 and the time of exposure was measured by a simple regression analysis.
RESULTS AND DISCUSSION
The sesquiterpenes evaluated herein possess topical toxicity, growth alteration effects, as well as repellent activities to T. molitor larvae. Tessaric acid (1), and costic aldehyde (6) increased pupal stage duration. Eremophilan-1(10),2,11(13)-trien-12-oic (4) and g-costic (3) acids produced the major number of morphological abnormalities. Nevertheless, all the compounds produced some effects upon development (Table I).
Although none of the tested compounds produced a significant mortality within the first 3 days after starting the experiment, tessaric acid (1) was toxic at 40 mg/ml dose after 60 days of treatment (Table II), and increased pupal stage duration when was applied topically at 20 µg/µl dose (Table I). These results suggest that compound 1 could be acting in a toxic but delayed way. Similar effects have been seen on larvae of Tribolium castaneum (Herbst) treated with this compound (García et al., 2003). When products were applied in a contact surface biotest they did not cause significant mortality at any time. This could be related with the fact that the topical application is a more closer and intrusive method.
Table II. Percent mortality of T. molitor larvae treated with sesquiterpene compounds in a topical bioassays at the end point of the experiment.

Mean ± SEM followed by b,c,d (P< 0.05; P<0.01; P<0.001. by c2 test) are significantly different from the control.
Experiments displayed repellency at some doses (Tables III, IV, V, VI, VII, VIII)
Table III. Mean Response Index of T. molitor larvae to compound 1at different doses in two-choice bioassays.
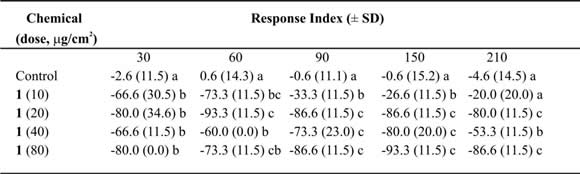
Means followed by different letters indicate significant difference between those responding to treatment and those responding to control (ANOVA test; Least Significant Differences test). N = 10 bioassays at each doses.
Table IV. Mean Response Index of T. molitor larvae to compound 2 at different doses in two-choice bioassays.
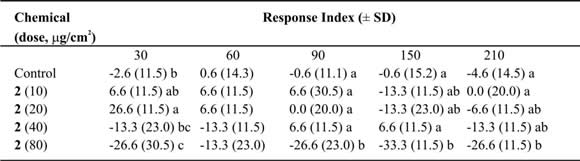
Means followed by different letters indicate significant difference between those responding to treatment and
those responding to control (ANOVA test; Least Significant Differences test). N = 10 bioassays at each doses.
Table V. Mean Response Index of T. molitor larvae to compound 3 at different doses in two-choice bioassays.
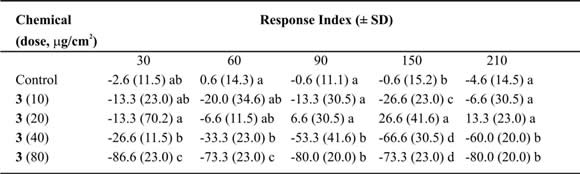
Means followed by different letters indicate significant difference between those responding to treatment and
those responding to control (ANOVA test; Least Significant Differences test). N = 10 bioassays at each doses.
Table VI. Mean Response Index of T. molitor larvae to compound 4 at different doses in two-choice bioassays.
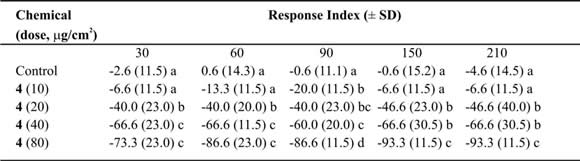
Means followed by different letters indicate significant difference between those responding to treatment and those responding to control (ANOVA test; Least Significant Differences test). N = 10 bioassays at each doses.
Table VII. Mean Response Index of T. molitor larvae to compound 5 at different doses in two-choice bioassays.

Means followed by different letters indicate significant difference between those responding to treatment and those responding to control (ANOVA test; Least Significant Differences test). N = 10 bioassays at each doses.
Table VIII. Mean Response Index of T. molitor larvae to compound 6 at different doses in two-choice bioassays.
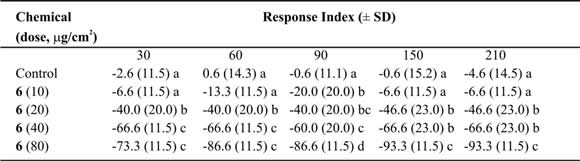
Means followed by different letters indicate significant difference between those responding to treatment and those responding to control (ANOVA test; Least Significant Differences test). N = 10 bioassays at each doses.
Tessaric acid (1) caused an important repellent action not always related with the concentration (Table III). Due to this we could not calculate the ED50 value, nevertheless an acute repellent action of this compound on T. molitor larvae can not be denied. Additionally, g-Costic acid (3) reached RI values higher than 73% at 80 µg/cm2 dose (Table V). ED50 values were similar throughout 210 minutes of treatment (Table IX).
Table IX. Response (ED50, ED90 and 95% Confidence Limits) of sesquiterpenes on T. molitor larvae.
The relation between the time of exposure and the ED50 found was weak (r2 = 0.55; p > 0.005). The absence of variation through time would indicate the continue need of the same dose in this lap of time. Nevertheless, g-Costic acid (3) shows a slow tendency to reduce with time (slope = -0.04 ± 0.02).
Eremophilan-1(10),2,11(13)-trien-12-oic acid (4) was repellent against T. molitor larvae. It produced one of the highest values of RI (Table VI) and showed low values of ED50 after 150 and 210 minutes of treatments (Table IX). At the same time, a relation between the experimental time and the ED50 was found (r2 =0.72; slope = –0.2 ± 0.07; p<0.05) (Fig. 2).

Fig. 2. Simple regression analysis between the ED50 and the time of exposure (diamonds = compound 4; squares = compound 6).
This relation could be based on the time necessary to be received by the chemoreceptors of the larvae and the physico-chemical behavior of the compound. As long as the time pass, the same repellent action will be reached with less amount of compound. We can postulate that at a first time, the compound (4) need top reach a certain concentration to activate certain sensillia receptors located on the insect body. Once the receptors are activated this could elicit a cascade of electrochemical events that reduce the threshold needed for sensorial elements activation. (Wojtasek, 2002).
Ilicic aldehyde (5) showed an acute repellent action (Table VII) and the lowest ED50 value (ED50 =17.26 µg/cm2), after 30 minutes of treatment. After this time the repellent action decreased slightly with time. It is possible that the compound lost its influence on the larval receptor due to a saturation mechanism. A potential degradation of the assayed compound with the time could be an alternative interpretation. Additionally, no relationship was found between the time of exposure and the ED50 value (r2 = 0.29; p>0.005).
Costic aldehyde (6) produced a significant increase on the duration of the pupal stage when was apply at 40 µg/µl dose (Table I). Compound 6 caused the appearance of adults with morphological deformities. The oenocytes are responsible for the deposition of postecdysial pupal-adult cuticle (Glhito et al., 1978; Quennedey et al., 1983; Delbecque et al., 1986). Probably, the normal function of the oenocytes or the epidermis itself, is affected by costic aldehyde (6). Similar results were found by Bowers & Thompson (1963), Thompson & Uebel (1966), Low et al. (1968) on Oncopeltus fasciatus (Dallas) and T. molitor specimens treated with a variety of alcohols, a sesquiterpenoid ester, and nonsesquiterpenoids. This kind of results were studied by Pascual et al. (1990) and Quennedey (1998) when the response of T. molitor specimens treated with azadarichtin and 20-hydroxyecdysone were evaluated, and by Soltani et al. (1996) using triflumuron. These authors have shown how the evaluated compounds were acting at endocrine level and producing morphological abnormalities. According to these results, we can postulate that costic aldehyde (6) could be affecting the deposition of postecdysial pupal adult cuticle (Harbone, 1993; Carrizo et al., 1998). When this compound was applied topically it produced intermediate toxicity at 20 µg/µl dose. Possibly a topical application allows an inner contact with the ventral segments of the abdomen, leading to a wide penetration through the cuticle. Similar results were found with treated adults of T. castaneum with eugenol (Obeng-Ofori & Reichmuth, 1997).
In two-choice assays, costic aldehyde (6) reached values up to 83% (Table VIII) at 80 µg/µl dose. The repellent activity could be related with the possession of an aldehyde group and its action on taste receptors. In certain lepidopteran larvae, this substance block the stimulatory effects of glucose, sucrose and inositol on chemosensory receptor cells located in the mouthparts. Therefore, some investigators have proposed that there is a sulphydril group at the receptor site that binds covalently to the double bond of the a, b-unsaturated moiety via Michael addition (Gershenzon & Croteau, 1999). When the ED50 was calculated, compound (6) showed values that ranged from 29 to 24 µg/cm2 (Table IX). Some relationship between time of exposure and effective dose was found (r2=0.74; slope = -0.03 ± 0.01; p < 0.05) (Fig. 2), indicating, as in the eremophilan-1(10),2,11(13)-trien-12-oic acid, a linear relationship among the dose needed to repel the 50% of the larvae and the lap time required for activation of the receptor. Nevertheless, the action of costic aldehyde is very different from the eremophilan-1(10),2,11(13)-trien-12-oic acid (4) because it has a smoother line. This possibly indicates that the compound needs to be all the time at the same concentration to reach the 50% of repellency. It is possible that the action against the receptor decreases with time, or this compound is metabolized much more quickly than the other compound.
CONCLUSIONS
We conclude that the six compounds showed an acute activity in the choice bioassay, indicating behavioral avoidance. The appearance of adults with morphological deformities could be indicating that eremophilan-1(10), 2,11(13)-trien-12-oic (4) and g-costic (3) acids affect the normal deposition of postecdysial cuticle. This data provide an important preliminary information, that should be fully explored for using in stored production protection. More research is needed in order to understand the specific mechanism of action each compounds.
ACKNOWLEDGMENTS
We thank CONICET, CYTED (IV 13) and UNSL for financial support. This paper is a part of the Doctoral thesis of M. G.
LITERATURE CITED
1. BOWERS, W. S. & M. THOMPSON. J. 1963. Juvenile hormone activity: effects of isoprenoid and straigth-chain alcohols on insects. Science 142: 1469-1470. [ Links ]
2. BROWN, H. C. & KRISHNAMURTHY, S. 1979. Forty years of hydride reductions. Tetrahedron 35: 567-607. [ Links ]
3. CARRIZO, F. R., M. E. SOSA, S. FAVIER, F. PENNA, E. GUERREIRO, O. S. GIORDANO & C. E. TONN. 1998. Growth–inhibitory activities of benzofuran and chromene derivates toward Tenebrio molitor L. J. Nat. Prod. 61: 1209-1211. [ Links ]
4. DANIEWSKY, M., M. GUMULKA, K. PTASZYNSKA, P. SKIBICKI, E. BLOSZYK, B. DROZDZ, S. STROMBERG, T. NORIN & M. HOLUB. 1993. Antifeedant activity of some sesquiterpenoids of the genus Lactarius (Agaricales: Russulaceae). Eur. J. Entomol. 90: 65-70. [ Links ]
5. DANIEWSKY, M., W. ANCZEWSKI, E. BLOSZYK, B. DROZDZ, U. JACOBSON, & T. NORIN. 1995. 3-Ethoxy-9-epi (trans fused) lactarane sesquiterpenes of Lactarius origin. Antifeedant activity. Pol. J. Chem. 69: 1697-1683. [ Links ]
6. DELBEQUE, J, M. MEISTER & A. QUENNEDEY. 1986. Conversion of radiolabelled 2,22,25-trideoxyecdysone in Tenebrio pupae. Ins. Biochem. 16, (1): 57-63. [ Links ]
7. DONADEL, O., E. GARCÍA, E. GUERREIRO & C. E. TONN. 1998. Easy preparation of Bioactives eudesman-12 (5b)-olide derivatives from ilicic acid. An. Asoc. Quim. Argent. 86: 90-93. [ Links ]
8. ENRIZ, R. D., H. BALDONI, E. JÁUREGUI, M. E. SOSA, C. E. TONN & O. GIORDANO. 1994. Structure activity relationships of clerodane diterpenoids acting as antifeedant agents. Agric. Food. Chem. 42: 2958-2963. [ Links ]
9. ENRIZ, R. D, H. A. BALDONI, M., M. A. ZAMORA, M. E. SOSA, C. E. TONN, J. M. LUCO & M. GORDALIZA. 2000. Structure-antifeedant activity relationship of clerodane diterpenoids. A comparative study with withanolides and azadirachtin. Agric. Food. Chem. 48: 1384 -1392. [ Links ]
10. GARCÍA, M., M. E. SOSA, O. J. DONADEL, O. S. GIORDANO & C. E. TONN. 2003. Allelochemical effects of eudesmane and eremophilane toward Tribolium castaneum larvae. J. Chem. Ecol. 29 (1): 175-189. [ Links ]
11. GERSHENZON, J. & CROTEAU, R. 1999. Terpenoids. In Rosenthal, G. A. & M. R. Berenbaum (eds.). Herbivores: Their interactions with secondary plant metabolites Volume 1. Academic Press, San Diego, California. pp. 165-170. [ Links ]
12. GLHITO, I., J. DELBECQUE & J. DELACHAMBRE. 1978. Prothoracic gland involution related to moulting hormone levels during metamorpfhosis of Tenebrio molitor L. J. Insect Physiol. 25: 187-191. [ Links ]
13. GONZALEZ-COLOMA, A., M. REINA, R. CABRERA, P. CASTAÑERA & C. GUTIERREZ. 1995. Antifeedant and toxic effects of sesquiterpenes from Senecio palmensis to Colorado potato beetle. J. Chem. Ecol. 21: 1255-1269. [ Links ]
14. GUERREIRO, E., J. KAVKA, O. GIORDANO & E. GROS. 1979. Sesquiterpenoids and flavonoids form Flourensia oolepis. Phytochemistry. 18: 1235-1237. [ Links ]
15. HARBONE, J. B. 1993. Advances in Chemical Ecology. Nat. Prod. Rep. 10: 327-348. [ Links ]
16. HARMATHA, J. & NAWROT, J. 1984. Comparison of the feeding deterrent activity of some sesquiterpene lactones and a lignan lactone towards selected insect storage pests. Biochem. Syst. Ecol. 12 (1): 95-98. [ Links ]
17. HENDERSON, C. F. & E. W. TILTON. 1955. Test with acaricides against the brown wheat mite. J. Econ. Entomol. 48: 157-161. [ Links ]
18. HUANG, Y. & S. HO. 1998. Toxicity and antifeedant activities of cinnamaldehyde against the grain storage insects, Tribolium castaneum (Herbst) and Sitophilus zeamais Motsch. J. Stored Prod. Res. 34 (1): 11-17. [ Links ]
19. KURINA SANZ, M., O. DONADEL, P. ROSSOMANDO, C. E. TONN & E. GUERREIRO. 1997. Sesquiterpenes from Tessaria absinthioides. Phytochemistry. 44 (5): 897-900. [ Links ]
20. LOW, P. F., DAVEY, B. G., LEE, K. W. & BAKER, D. E. 1968. Juvenile Hormone: Activity of Natural and Synthetic Synergists. Science 161: 895-897. [ Links ]
21. LUCO, J. C., M. E SOSA, J. C. CESCO, C. E TONN & O. S. GIORDANO. 1994. Molecular connectivity and hydrophobicity in the study of antifeedant activity of clerodane diterpenoids. Pestic. Sci. 41: 1-6. [ Links ]
22. NAWROT, J., J. HARMATA & L. NOVOTNY. 1984. Insect feeding deterrent activity of bisabolangelone and some sesquiterpenes of eremophilane type. Bioch. Syst. Ecol. 12 (1): 99-101. [ Links ]
23. OBENG–OFORI, D. & REICHMUTH, CH. 1997. Bioactivity of eugenol, a major component of essential oil of Ocimum suave (Wild.) against four species of stored–product Coleoptera. Int. J. Pest Manag. 43 (1): 89-94. [ Links ]
24. ORTEGO, F., J. LÓPEZ-OLGUÍN, M. RUÍZ & P. CASTAÑERA. 1999. Effects of toxic and deterrent terpenoids on digestive protease and detoxication enzyme activities of colorado potato beetle larvae. Pestic. Biochem. Physiol. 63: 76-84. [ Links ]
25. PASCUAL, N., M. P. MARCO & X. BELLÉS. 1990. Azadirachtin induced imaginal moult deficiencies in Tenebrio molitor L. (Coleoptera: Tenebrionidae). J. Stored Prod. Res. 26 (1): 53-57. [ Links ]
26. PHILLIPS, T. W. 1997. Semiochemicals of stored product insects: research and applications. J. Stored Prod. Res. 33 (1): 17-30. [ Links ]
27. PHILLIPS, T. W., X. JIANG, W. BURKHOLDER, J. PHILLIPS & H. TRAN. 1993. Behavioral responses to food volatiles by two species of stored-product coleoptera, Sitophilus oryzae (Curculionidae) and Tribolium castaneum (Tenebrionidae). J. Chem. Ecol. 19 (4): 723-733. [ Links ]
28. PHILLIPS, T. W., M. PARAJULEE & D. WEAVER. 1995. Toxicity of terpenes secreted by the predator Xylocoris flavipes (Reuter) to Tribolium castaneum (Herbst) and Oryzaephilus surinamensis (L.). J. Stored Prod. Res. 31 (2): 131-138. [ Links ]
29. QUENNEDEY, A., B. QUENNEDEY, J. DELBECQUE & P. DELACHAMBRE, 1983. The in vitro development of the pupal integument and the effects of ecdysteroids in Tenebrio molitor (Insecta: Coleoptera). Cell Tissue Res, 232: 493-511. [ Links ]
30. QUENNEDEY, A. 1998. Insect epidermal glands cells: Ultrastructure and Morphogenesis. Microscopic Anatomy of Invertebrates. Chapter 8, II A. Insecta: 177-207. [ Links ]
31. SAKUMA, M. 1998. Probit analysis of preference data. Appl. Entomol. Zool. 33 (3): 339-347. [ Links ]
32. SCHOONHOVEN, L. M. & Y. FU-SHUN. 1989. Interference with normal chemoreceptor activity by some sesquiterpenoid antifeedants in an herbivorous insect, Pieris brassicae. J. Insect Physiol. 35 (9): 725-728. [ Links ]
33. SOLTANI, N., N. SOLTANI-MAZOUNI & J. DELACHAMBRE. 1996. Evaluation of triflumuron, a benzoylphenylurea derivative, on Tenebrio molitor pupae (Coleoptera: Tenebrionidae): effects on cuticle. J. Appl. Ent. 120: 1-3. [ Links ]
34. SOLTANI, N., N. ARIBI, H. BERGHICHE, S. LAKBAR, & G. SMAGGHE. 2002. Activity of RH-0345 on ecdysteroid production and cuticle secretion in Tenebrio molitor pupae i n vivo and in vitro. Pestic. Biochem. Physiol. 72: 83-90. [ Links ]
35. SOSA, M. E., C. E. TONN & O. S. GIORDANO. 1994. Insect antifeedant activity of clerodanes diterpenoids. J. Nat. Prod. 57 (9): 1262-1265. [ Links ]
36. SOSA, M. E., C. E. TONN, E. GUERREIRO & O. S. GIORDANO. 2000. Bioactividad de flavonoides sobre larvas de Tenebrio molitor (Coleoptera: Tenebrionidae). Rev. Soc. Entomol Argent. 59 (1-4): 179-184. [ Links ]
37. THOMPSON, M. J. & UEBEL, E. C. 1966. Juvenile Hormone: Identification of an Active Compound from Balsam Fir. Science 154: 1020-1021. [ Links ]
38. WOJTASEK, H. 2002. Pheromone Perception In The Olfactory Sensillum. In Nicieja Slawomir (ed.). Molecular mechanisms of insect chemical communication: from pheromone biosynthesis to perception and degradation. Uniwersytet Opolski, Opole. pp. 39. [ Links ]
Recibido: 14-III-2003
Aceptado: 10-VII-2003












