Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista de la Sociedad Entomológica Argentina
versión impresa ISSN 0373-5680versión On-line ISSN 1851-7471
Rev. Soc. Entomol. Argent. v.67 n.1-2 Mendoza ene./jun. 2008
Calliphoridae (Diptera) from wild, suburban, and urban sites at three Southeast Patagonian localities
Calliphoridae (Diptera) de ambientes no habitados, suburbanos y urbanos en tres localidades del sudeste patagónico
Mariluis, Juan C. */**, Juan A. Schnack */***, Pablo P. Mulieri, */** and Luciano D. Patitucci */**
* Consejo Nacional de Investigaciones Científicas y Técnicas, Buenos Aires, Argentina.
** ANLIS «Dr. C. Malbrán», Departamento Vectores. Av. Vélez Sarsfield 563, 1281 Buenos Aires, Argentina; e-mail: jcmariluis@yahoo.com.ar , jcmariluis@anlis.gov.ar , mulierii@yahoo.com , lpatitu@yahoo.com.ar
*** División Entomología, Facultad de Ciencias Naturales y Museo, Universidad Nacional de La Plata, Paseo del bosque s/nº, 1900 La Plata, Buenos Aires, Argentina; email: js@netverk.com.ar
ABSTRACT. Species composition, relative abundance, sex ratio and habitat preference of blowflies (Diptera: Calliphoridae) from Caleta Olivia, Puerto Deseado, and Puerto San Julián (Santa Cruz Province, Argentina) were studied during late spring and summer in 2004-2005. Results showed a higher prevalence of the exotic species, Calliphora vicina (Robineau-Desvoidy) and Phaenicia sericata (Meigen) at urban sites over the natives, Compsomyops fulvicrura (Robineau-Desvoidy) and Sarconesia chlorogaster (Wiedemann), which shows a strong preference for those sampling sites either not inhabited or less influenced by human activities. Sex ratio was female biased for all species, except for Sarconesia chlorogaster, which exhibited a male prevalence over females.
KEY WORDS. Calliphoridae; Species composition; Relative abundance; Synanthropy; Southeastern Patagonia.
RESUMEN. Durante fines de la primavera-verano de 2004-2005, se analizó la composición, abundancia relativa y proporción de sexos de especies de Calliphoridae (Diptera) en las localidades de Caleta Olivia, Puerto Deseado y Puerto San Julián (Provincia de Santa Cruz, Argentina). Tres sitios de muestreo afectados diferencialmente por la intervención humana fueron seleccionados en cada localidad. En las áreas urbanas, las especies exóticas Calliphora vicina (Robineau-Desvoidy) y Phaenicia sericata (Meigen) fueron dominantes. Por el contrario, las nativas Compsomyiops fulvicrura (Robineau-Desvoidy) y Sarconesia chlorogaster (Wiedemann) predominaron tanto en áreas inhabitadas como en aquellas con bajo impacto humano. En los cebos utilizados para las capturas, se observó mayor proporción de hembras en todas las especies, excepto Sarconesia chlorogaster que mostró mayor proporción de machos.
PALABRAS CLAVE. Calliphoridae; Composición específica; Abundancia relativa. Sinantropía; Sudeste Patagonia.
INTRODUCTION
Knowledge of the taxonomy and ecology of blowfly species is important not only for basic entomology, but also for other fields of science and its applications, especially sanitary biology. This is due to their role as pathogen vectors (Greenberg, 1971; Graczyk et al., 2001), indicators for forensic studies (Byrd & Castner, 2001) and myiasis producers (Zumpt, 1965; Hall & Wall, 1995).
Many insect species are associated with environmental conditions created by man (synanthropy), especially muscoid flies (e.g., Nuorteva, 1963). Habitat preferences of blowfly species and the structure of local communities may exhibit significant geographical variation (Nuorteva, 1963). Comparative studies among wild, suburban, and urban environments housing calliphorid species have been conducted in several geographic locations in Argentina, including northeastern subtropics (Mariluis et al., 1990), the suburbs of Buenos Aires city (Schnack et al., 1995), northwest (Mariluis & Schnack, 1996) and mid-west Patagonia (Schnack et al.,1998), southeastern Santa Cruz Province and mid-eastern Tierra del Fuego (Schnack & Mariluis, 2004).
According to findings from previous works, similar values of specific composition to the ones already registered in other localities of the Patagonian steppe are expected to be found. We hypothesize that differences in the species relative abundance among different sites, with variable degrees of urbanization, will occur. We expect to find a higher relative abundance of cosmopolitan species among urban sectors of the coastal localities of Patagonia, Argentina.
The use of carrion baits is crucial for the study of blowfly populations (Hall, 1995). The majority of Calliphoridae females used them as protein sources for eggs maturation as well as for egg-laying substrate (Erzinçlioglu, 1996). Thus, we expect a bias toward females during capture. We will compare sex ratio in order to quantitative describe how species will respond.
The aim of this work was to analyze blowfly species composition, relative abundance and sex ratio, and the main factors governing them, considering sites differently affected by human influence in three localities along the Atlantic Patagonian coast.
MATERIAL AND METHODS
Study sites comprised urban (U), isolated dwelling (ID) and wild (W) habitats at three localities in northeastern Santa Cruz province: Caleta Olivia (46º 26' 17'' S, 67º 32' 05'' W), Puerto Deseado (47º 44' 00'' S, 65º 54' 01'' W), and Puerto San Julián (49º 17' 60'' S, 67º 43' 00'' W). At each studied location, the U sites were located in the geographical center of the town, the ID sites were at the city edge and the W sites were in the steppe, 5-8 km away from the urban limits.
The study sites lie in the «Provincia Patagónica», where two main districts may be distinguished: the Central-Patagonian (Puerto San Julián) and the San Jorge Gulf (Caleta Olivia and Puerto Deseado). The former is mainly represented by a shrubby steppe dominated by Chuquiraga avellanedae Lorentz (Asteraceae), Nassauvia glomerulosa (Lag. Ex Pindl) and Stipa spp. (Poaceae); the latter is dominated by Trevoa patagonica Speg (Rhamnaceae) and Colliguaya integerrima Gill. et Hook. (Euphorbiaceae) (Cabrera, 1971).
Blowflies are small and very mobile organisms, which makes them difficult to sample at randomly-selected locations. Thus, sampling methods are mostly based on estimates or counts on aggregation sites, such as fly-attracting baits (Muirhead-Thomson, 1968). Samples were taken during the late spring and summer (November 2004- February 2005), once a month per site. Seven hourly captures of adult flies (10:00 am - 04:00 pm) were taken with a hand net on a bait composed of 150 g of rotten cow meat (five days old). The baits were exposed for 15 minutes every hour. The sampling effort was the same at each site and consisted of one bait per site and seven netting events per date. Blowflies were killed in glass vials with carbon tetrachloride and stored in the field as described elsewhere (Schnack & Mariluis, 2004); once in the laboratory, they were identified and counted using the key of Mariluis & Schnack (2002).
When calliphorid samplings are obtained in several places at different dates, raw counts reflect the local sampling milieu at the particular site and date, rather than any fundamental differences among assemblages (McLeod & Donnelly, 1962). For this reason our hypotheses are based on relative proportions. Raw data is given as complementary informative purpose.
We took into account the proportion of males and females in order to describe the response to the used bait. Thus, a Friedman's analysis of variance by ranks was applied using the species as treatments and sites as blocks, and Dunn's procedure was performed for multiple comparisons (Zar, 1996).
Hypothesis of increasing proportions of exotic species with the urbanization, and the opposite trend for the natives was assessed by the use of test for independent proportions qualitatively ordered (Fleiss, 1981).These analyses were performed for each species at each locality. For this purpose we use numbers of flies of a given species over the total number of flies per site.
The relative abundance of native species among different habitats was compared by using the non-parametric Mann-Whitney U test for two independent samples (Zar, 1996). For this analysis, sites belonging to the same habitat type were used as replicates.
A correspondence analysis (CA) was used to summarize the relationships between species and sites. This is a statistical visualization method for illustrating the associations between the levels of a two-way contingency table (Kent & Coker, 1992). For this analysis raw number of flies gives asymmetric scaling on the configuration of species and sites. To avoid this potential effect we give the same weight to all species. This was done by converting numbers collected per site to percentage of the total yield per species (total number of given species for all sites) (Pollet, 2001).
RESULTS
A total of 2905 specimens were captured at the three surveyed localities; they included four species: the natives, Compsomyiops fulvicrura (Robineau-Desvoidy) and Sarconesia chlorogaster (Wiedemann), and the exotics, Phaenicia sericata (Meigen) and Calliphora vicina (Robineau-Desvoidy).
Males and females of the recorded species showed significant differences in their response to the bait used (ANOVA Chi SquareN=7, df=3 = 13.29, P < 0.005). Due to female proportion, sex ratio was female biased for Calliphora vicina (max-min range values: 1.00-0.70), Phaenicia sericata (0.87- 0.72) and Compsomyiops fulvicrura (0.93- 0.66). On the other hand, Sarconesia chlorogaster showed a male biased sex ratio according to its lower female proportion (0.67-0.13) (Fig. 1).
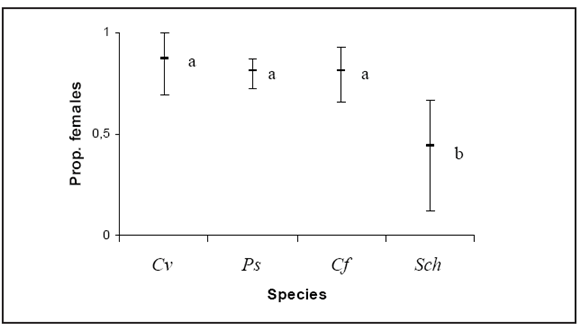
Fig. 1. Proportion of females of blowfly species obtained from the nine capture sites (lowest and highest bars represent minimum and maximum values, respectively; middle bar refers to mean values). Data labeled with different letters are significantly different according to Dunn's test (P < 0.05). Abbreviated names of species: Cv = Calliphora vicina; Ps = Phaenicia sericata; Cf = Compsomyiops fulvicrura; Sch = Sarconesia chlorogaster.
The exotic, Phaenicia sericata and Calliphora vicina showed a higher proportion of captures at the U sites in all studied localities, being the former species significantly dominant. Their species relative abundance decreased significantly towards the W sites (Table I). On the contrary, the native, Compsomyiops fulvicrura and Sarconesia chlorogaster showed in most of the samples a high relative abundance in W sites decreasing significantly in U sites (Table II). Nevertheless, this decrease from W to U sites was not accounted for the few specimens of Sarconesia chlorogaster sampled at Caleta Olivia. The CA ordination diagram shows this trend with the arrangement of species and sites along the main axis (Fig. 2). The eigenvalues of axis 1 and 2 were 0.49 and 0.05, respectively, the first expressing 83.32 % of the data variability. According to the first axis, the two native blowfly species recorded were located within the positive right half of the diagram containing most of the W and ID sites, showing the ID site of Caleta Olivia an intermediate position. On the other hand, the exotic species were located on the left half. While Calliphora vicina proportion was higher at U sites in Puerto San Julian, Phaenicia sericata shows the same trend in Caleta Olivia and Puerto Deseado. On the other hand, axis two divides the urban sites in two major groups according to the occurrence of two dominant species. The first one, Phaenicia sericata in Caleta Olivia and the second one, Calliphora vicina in Puerto Deseado and San Julian. In the remaining habitats (sites ID and W) the homogeneity is influenced by Compsomyiops fulvicrura (Fig. 2).
Table I. Proportion and number of exotic calliphorid species, Calliphora vicina and Phaenicia sericata, obtained from each sampling site. P values account for differences among the proportions for the site types.
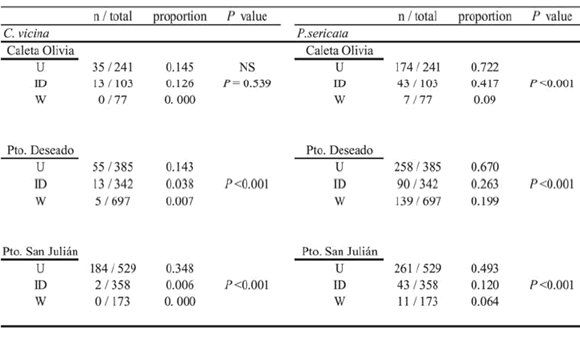
Table II. Proportion and number of native calliphorid species, Compsomyiops fulvicrura and Sarconesia chlorogaster, obtained from each sampling site. P values account for differences among the proportions for the site types.

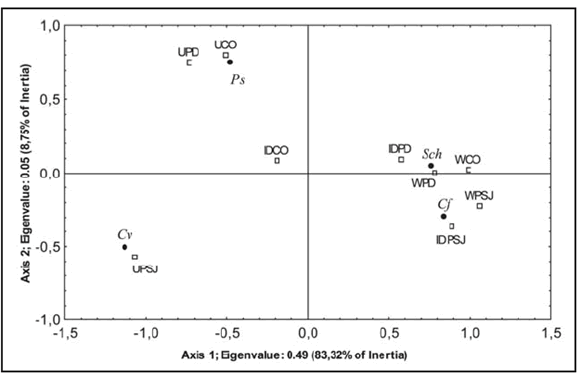
Fig. 2. CA ordination diagram of sampling sites and localities (white squares) and calliphorid's species (black dots) collected in Caleta Olivia (CO), Puerto Deseado (PD) and Puerto San Julián (PSJ). Site types, U: Urban, ID: Isolated Dwellings, W: Wild. Species abbreviations as in Fig. 1.
From the ordination diagram, two main groups of sites can be considered according to the relative abundance of two types of blowflies: native and exotic species. The first group included the six sites belonging to W and ID habitats, while the second, all the three U sites. Native species are significantly more abundant in ID-W sites (Mann-Whitney P < 0.05) (Fig. 3).

Fig. 3. Relative abundance of native blowflies in two groups of habitats: Urban (U) (n = 3) and Isolate Dwelling plus Wild Sites (ID-W) (n = 6). Columns represent median value and error bars represent first and third quartile. Columns labeled with different letters are significantly different (Mann-Whitney test P < 0.05).
DISCUSSION
The higher prevalence of Phaenicia sericata and Calliphora vicina in U environments and that of Compsomyiops fulvicrura and, secondarily of Sarconesia chlorogaster, in W and ID sites in all studied localities suggest that native and exotic species differ markedly in their habitat selection. The high synanthropy displayed by the exotic species recorded may express the ecological disorder caused by urbanization, which usually favors the settlement of invasive species (Nuorteva, 1971; Schnack & Spinelli, 2000).
From a sanitary viewpoint, U sites provided useful information given that among the recorded species, the dominant, Phaenicia sericata and Calliphora vicina, show high sanitary importance. Phaenicia sericata, a nearly worldwide species, would be the most medically important blowfly. This endophylic and highly synanthropic species is one of the primary myiasis producers in Europe, and to a lesser extent in Sudan, South Africa, and Australia (Baumgartner & Greenberg, 1985). Moreover, cases of human myiasis have been recorded in hospitals of Chicago, Illinois, USA (Greenberg, 1984) and of Buenos Aires, Argentina (Mariluis & Guarnera, 1983). Calliphora vicina exhibits endophilic and communicative habits in urban settlements, and capacity to produce myiasis on man and animals (Delhaes et al., 2001; Schnack & Mariluis, 2004). Even though, Calliphora vicina was not the most abundant species at U sites, it showed preference for urban habitats. Nevertheless, during a survey carried out in Buenos Aires outskirts (30º 35´ 28" S, 64º 12´ 22" W) (Schnack et al., 1995) a synanthropic index (Nuorteva, 1963) calculated for this species was negative, suggesting its preference for the wildness. On the other hand, this species was formerly regarded as synanthropic in Hungary (Mihalyí, 1967). Although these observations seem contradictory, synanthropy may be an adequate approach at a local population scale instead of being at the species level (Schnack et al., 1998).
Female sex ratio was found for three species. On the other hand, we cannot explain why Sarconesia chlorogaster sampled from the baits used were male biased as it was also observed in former studies (e.g., Mariluis & Schnack, 1989; Mulieri et al., 2006). It would be possible that these species' females are autogenic. Like most of blowfly species, if their eggs reached maturity without the need of a protein meal, they would not be compelled to feed on the bait. The attracting baits may indeed distort the actual sex ratio, regardless of their nature. However, they do not necessarily alter species numerical trends. In a recent study carried out in the coastline of Buenos Aires city, changes of blowfly species abundance displayed similar trends when captured either on cow liver or on feces, being sex ratio female biased for most of the species, except Sarconesia chlorogaster, whose females and males were similarly represented (Mulieri et al., 2006).
According to the data gathered in this study, the low species richness of all study sites is remarkable. This is in agreement to former observations: blowfly species richness shows a decrease as latitude increases as it was experienced in surveys previously undertaken in northeastern Argentina (14 species) (Mariluis et al., 1990), Buenos Aires (12 species) (Schnack et al., 1995), and western Patagonia (five species) (Mariluis & Schnack, 1996) and eastern Patagonia (six species) (Schnack & Mariluis, 2004).
In a broad sense, species richness in the study sites was similar to the one recorded in formerly studied locations of Santa Cruz and Tierra del Fuego provinces (Schnack & Mariluis, 2004). Nevertheless, some differences concerning species composition and relative abundance are worth being highlighted if both surveys are compared. One of the main differences is the presence of Protophormia terraenovae at higher latitudes (Schnack & Mariluis, 2004; Mariluis & Schnack, 2005), not recorded in this study. On the other hand, in the southern locations of Santa Cruz and Tierra del Fuego (50º-53º S) Calliphora vicina was significantly more abundant than Phaenicia sericata at U sites, while the opposite trend was recorded at ID sites. Moreover, whether all study sites are regarded within the above localities, Calliphora vicina and Phaenicia sericata shared the role of co-dominants with the native, Compsomyiops fulvicrura (Schnack & Mariluis, 2004). At lower latitudes like those corresponding to this research (46º-49º S), Phaenicia sericata was significantly more abundant than Calliphora vicina at U sites and ID sites, and Compsomyiops fulvicrura, which showed marked preference for W sites, was the most abundant species if we gathered all study sites where this research was carried out. We speculate that the dominance of Calliphora vicina over Phaenicia sericata in the southernmost surveyed locations may be due to its higher tolerance to low temperatures. This is in agreement to former observations of Mariluis & Schnack (1986, 1989) in Buenos Aires where Calliphora vicina and Phaenicia sericata seasonally alternate their role of dominant species, the former being the most numerous in winter while the latter, in summer.
ACKNOWLEDGEMENTS
We are grateful to Roberto Petrusic and Santiago Ansín for their valuable help in the field work. We appreciate the critical comments and constructive suggestions made by Alejandro Farji-Brener, Diego Vázquez, and anonymous reviewers. This work was supported by a grant from the Technical and Scientific National Research Council of Argentina (CONICET, PIP # 02008).
LITERATURE CITED
1. BAUMGARTNER, D.L. & B. GREENBERG. 1985. Distribution and medical ecology of the blow flies (Diptera: Calliphoridae) of Perú. Ann. Entomol. Soc. Am. 78: 565-587.
2. BYRD, J. H. & J. L. CASTNER. 2001. Forensic entomology: The utility of arthropods in legal investigations. CRPress, London.
3. CABRERA, A. L. 1971. Fitogeografía de la República Argentina. Bol. Soc. Arg. Bot. 14 (1-2): 1-43.
4. ERZINÇLIOGLU, 1996. Blowflies. The Richmond Publishing Co. Cambridge.
5. DELHAES, M., B. BOUREL, L. SCALA, B. MUANZA, E. DUTOIT, F. WATTEL, D. GOSSET, D, CAMUS & E. DEI-CAS. 2001. Case report: recovery of Calliphora vicina first-instar larvae from a human traumatic wound associated with a progressive necrotizing bacterial infection. Am. J. Trop. Med. Hyg. 64(3-4): 159-161.
6. FLEISS, J. L. 1981. Statistical methods for rates and proportions. John Wiley & Sons, New York.
7. GRACZYK, T. K., R. KNIGHT, R. H. GILMAN & M. R. CRANFIELD. 2001. The role of non-biting flies in the epidemiology of human diseases. Microb. Infect.3: 231-235.
8. GREENBERG, B. 1971. Flies and Disease, Vol. 1. Princeton University Press, New Jersey.
9. GREENBERG, B. 1984. Two cases of human myiasis caused by Phaenicia sericata (Diptera: Calliphoridae) in Chicago area hospitals. J. Med. Entomol. 21(5): 615.
10. HALL, M.R. 1995. Trapping the flies that cause myiasis: their responses to host-stimuli. Ann. Trop. Med. Parasitol. 4: 333-357.
11. HALL, M. & R. WALL. 1995. Myiasis of human and domestic animals. Adv. Parasitol. 35: 257-334.
12. KENT, M. & P. COKER. 1992. Vegetation description and análisis. A practical approach. John Wiley & Sons, Chichester, New York.
13. MARILUIS, J. C. & E. A. GUARNERA. 1983. Miasis producida por Phaenicia sericata (Meigen, 1826) (Calliphorinae, Luciliini). Rev. Soc. Entomol. Argent. 42 (1-4): 143-147.
14. MARILUIS, J. C. & J. A. SCHNACK. 1986. Ecología de una taxocenosis de Calliphoridae del área platense (Prov. de Buenos Aires) (Insecta, Diptera). Ecosur 12/ 13 (23/24): 81-91.
15. MARILUIS, J. C. & J. A. SCHNACK. 1989. Ecology of the blow flies of an eusynanthropic habitat near Buenos Aires. Eos 65 (1): 93-101.
16. MARILUIS, J. C. & J. A. SCHNACK. 1996. Elenco específico y aspectos ecológicos de Calliphoridae (Insecta, Diptera) de San Carlos de Bariloche, Argentina. Bol. R. Soc. Esp. Hist. Nat. (Sec. Biol.) 92 (1-4): 203-213.
17. MARILUIS, J. C. & J. A. SCHNACK. 2002. Calliphoridae de la Argentina. Sistemática, ecología e importancia sanitaria (Diptera, Insecta). En: Salomón, O. S. (ed.) Actualizaciones en Artropodología Sanitaria Argentina, Fundación Mundo Sano, Buenos Aires, pp. 23-37.
18. MARILUIS, J. C. & J. A. SCHNACK. 2005. Introduction and spread of the Holartic blowfly Protophormia terranovae (Robineau-Desvoidy) (Diptera, Calliphoridae) in Patagonia and South Atlantic Islands. Biogeographica 80 (1-4): 35-42.
19. MARILUIS, J. C., J. A. SCHNACK, J. MUZÓN & G.R. SPINELLI. 1990. Moscas Calliphoridae y Mesembrinellidae de Puerto Iguazú. Composición específica y ecología (Insecta, Diptera). Graellsia 46: 7-18.
20. MCLEOD, J. & J. DONNELLY. 1962. Microgeographic aggregations in blowfly populations. J. Anim. Ecol. 31: 542-543.
21. MIHALYÍ, F. 1967. The danger index for synanthropic flies. Acta Zool. Hung. 13: 373-377.
22. MUIRHEAD-THOMSON, R. C. 1968. Ecology of insect vector populations. Academic Press, London and New York.
23. MULIERI, P. R., J. P. TORRETTA, J. A. SCHNACK & J. C. MARILUIS. 2006. Calliphoridae (Diptera) of the coastline of Buenos Aires, Argentina: species composition, numerical trends, and bait's preferences. Entomol. News 117(2): 139-147.
24. NUORTEVA, P. 1963. Synanthropy of Blowflies (Dipt., Calliphoridae) in Finland. Ann. Ent. Fenn. 29: 1-49.
25. NUORTEVA, P. 1971. The synanthropy of birds as an expression of the ecological disorder caused by urbanization. Ann. Zool. Fenn. 8: 547-553.
26. POLLET, M. 2001. Dolichopodid biodiversity and site quality assessment of reed marshes and grasslands in Belgium (Diptera: Dolichopodidae). J. Insect Conserv. 5: 99-116.
27. SCHNACK, J. A., J. C. MARILUIS, N. CENTENO & J. MUZÓN. 1995. Composición específica, ecología y sinantropía de Calliphoridae (Insecta: Diptera) en el Gran Buenos Aires. Rev. Soc. Entomol. Argent. 54 (1-4): 161-171.
28. SCHNACK, J. A., J. C. MARILUIS, G. R. SPINELLI & J. MUZÓN. 1998. Ecological aspects on urban blowflies in midwest Argentinean Patagonia (Diptera: Calliphoridae). Rev. Soc. Entomol. Argent. 57 (1-4): 127-130.
29. SCHNACK, J.A. & G.R. SPINELLI. 2000. Vectors and parasites introductions into the New World. Their potential sanitary impact in Argentina. Three case studies, pp. 36-48. In: S.S. Caglar, B. Alten and N. Özer (Eds.). Proceedings of the 13th European SOVE Meetings. Society for Vector Ecology, September 2000. Published and Printed by DTO, Ankara, Tutkey, pp 275.
30. SCHNACK, J. A. & J. C. MARILUIS. 2004. Calliphoridae (Diptera) from Southeastern Argentinean Patagonia: species composition and abundance. Rev. Soc. Entomol. Argent. 63 (1-2): 85-91.
31. ZAR, J.H. 1996. Biostatistical Analysis. Prentice Hall, New Jersey.
32. ZUMPT, F. 1965. Myiasis in man and animals in the Old World. Butterworths, London. [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ] [ Links ]
Recibido: 27-04-2007;
Aceptado: 29-02-2008














