Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista de la Sociedad Entomológica Argentina
versión impresa ISSN 0373-5680versión On-line ISSN 1851-7471
Rev. Soc. Entomol. Argent. v.68 n.3-4 Mendoza jul./dic. 2009
NOTA CIENTÍFICA
Redescription of Lepidodexia (Asilidodexia) gaucha (Diptera: Sarcophagidae), and first record for Argentina
Redescripción de Lepidodexia (Asilidodexia) gaucha (Diptera: Sarcophagidae) y primer registro para Argentina
Mulieri, Pablo R. and Juan C. Mariluis
CONICET; ANLIS "Dr. Carlos G. Malbrán", Depto. Vectores. Av. Vélez Sársfield 563, (C1281AFR) Buenos Aires, Argentina; e-mail: vectores@anlis.gov.ar
ABSTRACT: The redescription of the male and the first description of the female of Lepidodexia (Asilidodexia) gaucha Lopes are presented on the basis of new material collected in Federación, Entre Ríos, Argentina. Detailed descriptions and illustrations of external morphology and terminalia of both sexes are provided. The specimens collected represent the first record of the species in Argentina.
KEY WORDS: Sarcophagidae; Sarcophaginae; Lepidodexia; Diptera;Taxonomy.
RESUMEN: Se presenta la redescripción del macho y la primera descripción de la hembra de Lepidodexia (Asilidodexia) gaucha Lopes, a partir de material colectado en Federación, Entre Ríos, Argentina. Se brindan descripciones detalladas e ilustraciones de la morfología externa y las estructuras genitales de ambos sexos. Los especímenes colectados representan el primer registro de la especie en Argentina.
PALABRAS CLAVE: Sarcophagidae; Sarcophaginae; Lepidodexia; Diptera; Taxonomía.
Recibido: 14-VII-2009;
Aceptado: 26-X-2009
Systematic relationships and generic concepts of several Sarcophaginae are still unresolved. The genus concept of Lepidodexia Brauer & Bergenstamm as defined by Pape (1996) currently includes more than 30 generic names synonymized as subgenera, based on similarities of the aedeagal morphology. The resultant genuscomplex includes 187 species, mostly Neotropical, which are much diversified in their external morphology (Pape, 1996).
Asilidodexia Townsend is a small group of sarcophagine flesh flies restricted to the Neotropical region. This taxon was originally described, as genus, by Townsend (1927) based on Asilidodexia asiliformis Townsend. More recently, Asilidodexia was considered as subgenus of Lepidodexia according to Pape (1996). Moreover, Asilidodexia was alternatively considered as member of the tribe Notochaetini (Lopes, 1969) and Johnsoniini (Verves, 1989), and closely related to Harpagopyga Aldrich, based on aedeagal morphology, reduced ocellar setae, and head shape (Lopes, 1938, 1992). Lopes (1992) provided a revision of Asilidodexia with a key to males, and four new species based on single male specimens. On the other hand, the females of this subgenus are poorly known, with a single species, L. (A.) wygodzinskyi Lopes, described from Paysandú, Uruguay (Lopes, 1982). At present, Asilidodexia is composed by seven species (Lopes, 1992).
Lepidodexia have not received much attention in previous taxonomic studies on the Argentinean sarcophagid fauna, with a total of 6 species listed for the local fauna, and L. (A.) proseni Lopes as the single member of the subgenus Asilidodexia (Pape, 1996).
Originally, Lepidodexia (Asilidodexia) gaucha Lopes was briefly described on the basis of a single male from Livramento, Rio Grande do Sul (Lopes, 1992). The aim of this work is to present the first record for Argentina, and to provide a redescription of L. (A.) gaucha based on male and female specimens collected in Federación, Entre Ríos.
The specimens were collected with a sweep net on the vegetation at the same site and date. The association of sexes was based on one couple collected while displaying pre-copulatory behavior. Phallic structures of the male were exposed using the technique described by Lopes (1973) and Dahlem & Naczi (2006). Thus, the identification of specimens was ensured by the examination of the male genitalia compared to original description provided by Lopes (1992).
Abdominal structures of female terminalia were extracted and cleared in 90 % lactic acid at ambient temperature (10 days), and mounted on a concave slide for study. The terminology used for descriptions of external morphology and for the female terminalia is that of McAlpine (1981). The terminology of phallic (or aedeagal) structures follows Lopes (1956), Pape (1994), and Mello-Patiu & Pape (2000). Illustrations were produced from photographs taken with a digital camera Olympus C-5050 Zoom mounted on a stereomicroscope. Illustrations of female sternites were made using a camera lucida attached to a stereomicroscope. The specimens are pinned and deposited in the collection of Departamento Vectores, ANLIS "Dr. Carlos G. Malbrán".
Acronyms used for collections are as follows:
ANLIS-DV Departamento Vectores,
ANLIS "Dr. Carlos G. Malbrán", Argentina;
MNRJ Museu Nacional, Río de Janeiro, Brazil.
Lepidodexia (Asilidodexia) gaucha Lopes, 1992 (Figs. 1-11)

Figs. 1-4. Lepidodexia (Asilidodexia) gaucha Lopes. 1, male head, left view; 2, right wing, dorsal view; 3, male abdomen, dorsal view; 4, female abdomen, dorsal view. Scale bars = 1 mm.
Asilidodexia gaucha Lopes, 1992: 126, 128 (as Asilodexia gaucha) (key; male, figs 11-15). Type-locality: Livramento, Rio Grande do Sul, Brazil. Holotype male (MNRJ).
Lepidodexia (Asilidodexia) gaucha: Pape, 1996: 229 (in catalog, stat. nov.).
Male. Total length = 6 mm.
Head. Head length at vibrissa shorter than at antennal base (Fig. 1). Parafacial plates with golden microtomentum; fronto orbital plate with golden microtomentum anteriorly, and gray microtomentum behind the posterior pair of frontal setae; parafacial and fronto-orbital plates bearing black and pale short setulae; post-cranium with gray microtomentum and black setulae (few pale setulae in its median portion); eyes reddish in live specimens; frontal vitta dark brown; frons at its narrowest point about 0.36 X head width; 5-6 frontal setae; reclinate orbital setae strong; proclinate fronto orbital setae small and weak; inner vertical setae strong and reclinate, outer vertical setae absent; ocellar triangle black, with one pair of proclinate ocellar setae; postocular setae black; genal groove and anterior half of genal dilation with golden microtomentum; posterior half of genal dilation and postgena with gray microtomentum and pale setae, gena with black setae; face black with gray microtomentum; facial ridge black with gray microtomentum, with setae and setulae close to vibrissa; 3-2 subvibrissal setae; antenna reddish, first flagellomere 1.25 X longer than length of pedicel with gray microtomentum in 2/3 apically, arista plumose on basal 1/2; palpus pale brown with some black long hairs apically.
Thorax. Black, with gray microtomentum; prescutum and scutum with intermediate and lateral stripes with gray microtomentum and three brown bands; scutellum with intermediate stripes with gray microtomentum interrupted in the middle by a brown oval spot; postpronotal lobe, notopleuron, katepisternum and anepimeron with gray microtomentum; anepisternum gray with a brown spot near to notopleuron; proepisternum gray, sometimes with few pale setulae on middle portion; two proepisternal setae strong, and one proepimeral setae, katepisternals 2, postalar wall bare. Chaetotaxy: acrostichals 0+1 (weak posterior pair), dorsocentrals 1+3 (spaced for 3), intra-alars 1+2 (anterior pair weak), supra-alars 1+3 (the middle pair stronger), anterior postpronotal 1, basal postpronotal 1, postalars 2, notopleurals 2 (plus one or two supplementary hairs). Scutellum with 2 pair of developed laterals. Wing hyaline, tegula brown, whitish basicosta and pale brown veins, R1 bare, R4+5 setulose in proximal 0.8 or less of distance to r-m, costal spine not differentiated, third costal sector with ventral setae, cell r4+5 closed at wing apex, calypteres whitish (Fig. 2). Legs with coxae and femora with gray microtomentum, trochanter and knees (basal femoral part) reddish with thin gray microtomentum, tibiae reddishbrown; middle femur without posteroventral ctenidium on its apical portion, 2 anterior setae, row of posteroventral setae on basal half; middle tibia with 2 anterdorsal setae, 1 anteroventral seta, 2 posterior setae; hind femur with rows of anterodorsal, anteroventral, and posteroventral setae; hind tibia with 2 or 3 anterodorsal setae, 2 anteroventral seta, and 2 posterodorsal setae; tarsi blackish.
Abdomen. Slender and elongate abdomen of dark brown ground color (Fig. 3); sternites covered by tergites, except ST 1 exposed; T 3-5 with median longitudinal dark brown vitta; T 1+2 covered of gray microtomentuum; two pairs of gray spots in T 3 and 4; T 5 reddish-brown covered with thin golden microtomentum on laterals; T 1+2 one pair of lateral marginal setae, T 3 with one pair of lateral marginal setae and one pair of median marginal setae, T 4 row of marginal setae (three pairs); T 5 with a complete row of marginal setae; ST 2-4 exposed only at apex; ST 5 V-shaped, reddish, with small hair like setae at posterior margin.
Terminalia. Syntergosternite 7+8 orange-reddish, with golden microtomentum apically, having a marginal row of three pairs of strong setae and some smaller basally black hair-like setae; epandrium orange reddish with black hair-like setae; cercus sligthly curved forward with pointed apex in profile (Fig. 5); cerci with cercal prong and cercal base of same length (Fig. 6); surstylus shining at apical portion with some hairlike setae and one stronger setae (Fig. 5); gonopod shiny reddish and curved (Fig. 7); paramere curved with a strong bristle (Fig. 8); aedeagus with vesica conspicuous, rounded and membranous, with a pair of small and slender sclerotizations (Fig. 9); juxta (or apical plate) sclerotized and rounded apically (lateral view) (Fig. 9); lateral styli not exposed (lateral view), and harpes composed by two small and well sclerotized plates (Fig. 9).

Figs. 5-9. Lepidodexia (Asilidodexia) gaucha Lopes, male. 5, cercus and surstylus, left view; 6, cerci, posterior view; 7, gonopod, left view; 8, paramere, left view; 9, aedeagus, left view; h: harpes; j: juxta (or apical plate); sur: surstylus; v: vesica (or ventralia). Scale bars = 0.1 mm.
Female. Total length = 4.5-5 mm.
Head. Differs from the male by possessing a wider frontalia, 0.42 X head width; one pair of strong fronto-orbital proclinate setae; antenna reddish with the first flagellomere covered by gray microtomentum in 3/4 apically.
Thorax. Similar to male. Female proepisternum bare.
Abdomen. Abdomen broader than in male specimen (Fig. 4). T3 without median marginal setae. ST 5 with row of 4-5 marginal setae (Fig. 11).
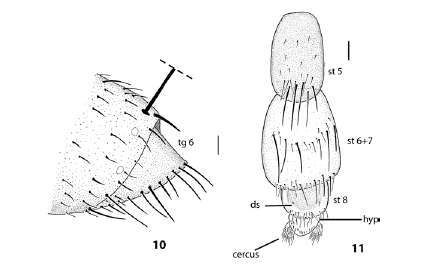
Figs. 10-11. Lepidodexia (Asilidodexia) gaucha Lopes, female. 10, female terminalia, right view; 11, dissected female terminalia, ventral view; ds: discal seta; hyp: hypoproct; st: sternite; tg: tergite. Scale bars = 0.1 mm.
Terminalia. Tergite 6 entire dorsally, large and pointed in lateral view (Fig. 10), covered with dense golden microtomentum, with a complete row of marginal setae; sternites covered by Tergite 6; ST 6+7 wider than ST 5, reddish with stronger row of setae at central line; ST 8 reddish, smaller, rounded borders and a slight concavity at basal part, covered with short setulae apically, and one pair of setulae at discal portion; hypoproct rounded covered with few short and stout setulae apically; cerci short and covered by setulae (Fig 11).
Ditribution. Argentina (Entre Ríos) and Brazil (Rio Grande do Sul).
Material examined. ARGENTINA, Entre Ríos: Federación, III-2009, 1 male, 2 females, P. R. Mulieri leg (ANLIS-DV).
Biology. Unknown.
Remarks. Lopes (1992) presented a key and established two groups of Asilidodexia species based on the male external morphology. The species of Asilidodexia recorded in Argentina, L. (A.) gaucha and L. (A.) proseni, can be distinguished by the following characters: wing cell r 4+5 closed at wing margin, more than one pair of marginal setae in the syntergosternite and one pair of mediam marginal setae in the third abdominal tergite. However, L. (A.) gaucha differs from L. (A.) proseni mainly by the presence of a single row composed by three pairs of marginal setae in the syntergosternite (four pairs in more than one row in L. (A.) proseni), the cerci slightly curved forward, surstyli with the presence of one stronger setae at apical portion, and the juxta (apical plate of Lopes) rounded apically.
The terminalia of the female of L. (A.) gaucha is very similar to the single previously known female of Asilidodexia, L. (A.) wygodzinskyi, based on their general appearance. Both species present the ST 8 rounded apically and concave at basal part, and the hypoproct bearing stout setae. However, L. (A.) gaucha possesses a pair of small discal setae on the ST 8, and a stronger row of marginal setae in the ST 5.
AKNOWLEDGEMENTS
The authors would like to thank Luciano D. Patitucci for his suggestions with the plates. Special thanks to Natalia Flores for her assistance during field work. We wish to express our acknowledgements to the anonymous referees for their useful comments and suggestions.
LITERATURE CITED
1. DAHLEM, G. A. & NACZI, R. F. C. 2006. Flesh flies (Diptera: Sarcophagidae) associated with North American pitcher plants (Sarraceniaceae), with descriptions of three news species. Ann. Entomol. Soc. Am. 99 (2): 218-240. [ Links ]
2. LOPES, H. S. 1938. Sobre um interessante novo genero de Sarcophagidae, que apresenta reducção dos esternitos abdominaes. Mem. Inst. O. Cruz 33 (3): 433-435. [ Links ]
3. LOPES, H. S. 1956. Sôbre a genitália masculina dos "Sarcophagidae". Rev. Brasil. Biol. 16: 59-64. [ Links ]
4. LOPES H. S. 1969. Family Sarcophagidae. In: Papavero, N. (ed.), A catalogue of the Diptera of the Americas south of the United States. Depto. Agricultura, São Paulo, pp.1-88. [ Links ]
5. LOPES H. S. 1973. Collecting and rearing sarcophagid flies (Diptera) in Brazil during forty years. An. Acad. Brasil. Ciénc. 45 (2): 279-291. [ Links ]
6. LOPES, H. S. 1982. Notes on American Sarcophagidae (Diptera) with descriptions of seven new species. Rev. Brasil. Biol. 42 (2): 285-294. [ Links ]
7. LOPES, H. S. 1992. On the genus Asilodexia (Diptera, Sarcophagidae) with description of four new species. Rev. Brasil. Biol. 51 (1): 125-132. [ Links ]
8. MCALPINE, J. F. 1981. Morphology and terminology-adults. In: McAlpine J. F., B. V. Peterson, G. E. Shewell, H. J. Teskey, J. R. Vockeroth & D. M. Wood (eds.), Manual of Nearctic Diptera. Vol 1, Research Branch, Agriculture Canada, Ottawa, pp. 9-63. [ Links ]
9. MELLO-PATIU, C. A. & T. PAPE. 2000. Definitions of Dexosarcophaga Townsend 1917 and Sarcofahrtiopsis Hall 1933, including two new species and a redescription of Sarcofahrtiopsis cuneata (Townsend 1935). Bol. Entomol. Venez. 15: 181-194. [ Links ]
10. PAPE, T. 1994. The world genus Blaesoxipha Loew, 1861 (Diptera: Sarcophagidae). Ent. Scand. Suppl. 45: 1-247. [ Links ]
11. PAPE, T. 1996. Catalogue of Sarcophagidae of the world (Insecta: Diptera). Mem. Entomol. Int. 8: 1-558. [ Links ]
12. TOWNSEND, C. H. T. 1927. New muscoid flies in the collection of the Deutsches Entomologisches Institut, in Berlin. Ent. Mitt. 16: 277-279. [ Links ]
13. VERVES, Y. G. 1989. Prof. Hugo de Souza Lopes and the modern system of Sarcophagidae (Diptera). Mem. Inst. O. Cruz 84 Supl. IV: 529-545. [ Links ]














