Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de la Sociedad Entomológica Argentina
Print version ISSN 0373-5680
Rev. Soc. Entomol. Argent. vol.70 no.3-4 Mendoza July/Dec. 2011
TRABAJOS CIENTÍFICOS
Factors affecting success of galls of Aditrochus coihuensis (Hymenoptera: Pteromalidae)
Factores que afectan el éxito de las agallas de Aditrochus coihuensis (Hymenoptera: Pteromalidae)
Nilsson, Michael*, Juan C. Corley** and Olle Anderbrant*
* Department of Biology, Lund University, Sölvegatan 37, SE-223 62 Lund, Sweden; e-mail: olle.anderbrant@biol.lu.se
** Grupo de Ecología de Insectos INTA EEA Bariloche, CC 277, (8400) Bariloche, Argentina; e-mail: jcorley@bariloche.inta.gov.ar
Recibido: 16-XII-2010
Aceptado: 21-X-2011
RESUMEN. Agallas provocadas por Aditrochus coihuensis (Hymenoptera: Pteromalidae) sobre su hospedero, Nothofagus dombeyi, fueron colectadas en la vecindad de San Carlos de Bariloche, Argentina, durante la primavera de 2007. Las mismas fueron examinadas y clasificadas como exitosas o no, sobre la base de restos de insectos dentro de ellas y el diámetro del agujero de emergencia. Distintas propiedades de las agallas (diámetro, grosor y dureza de la pared y diámetro de la cámara) fueron medidas y comparadas como exitosas y no exitosas (parasitadas). El diámetro de las agallas tuvo una fuerte correlación con el grosor de la pared, variable que se asocia con la protección contra los parasitoides. Los parasitoides (Torymidae), que fueron hallados dentro de las agallas, poseen un ovipositor más corto que el grosor de las paredes en aquellas plenamente desarrolladas. Sin embargo, es posible que los parasitoides ataquen las agallas por una ventana temporal, cuando las paredes son aún delgadas. La dureza de las mismas, por su parte, no influyó sobre el éxito de las agallas. También, se hallaron inquilinos (Curculionidae) dentro de ellas, en mayor frecuencia en las de mayor tamaño y con paredes más gruesas. Finalmente, se observaron evidencias de depredación, posiblemente por aves o avispas.
PALABRAS CLAVE. Nothofagus dombeyi; Cohiue; Patagonia; Insectos galígenos.
ABSTRACT. Galls of Aditrochus coihuensis (Hymenoptera: Pteromalidae) were collected from their host tree, the southern beech Nothofagus dombeyi, near San Carlos de Bariloche, Argentina, during the spring of 2007. Galls were carefully examined and classifi ed as successful or unsuccessful based on insect remains and emergence-hole diameter. Gall properties (diameter, wall thickness, chamber diameter, wall toughness) were measured and compared between successful galls and unsuccessful (parasitized) galls. Gall diameter and wall thickness were strongly correlated and thick walls seem to function as a protection against parasitoids. Parasitoids (Torymidae) that were found within some galls had an ovipositor length shorter than wall thickness of fully developed galls. Most likely, parasitoids attack galls only during a limited time when the wall is thin enough. Wall toughness did not seem to infl uence gall success. In total 67% of the galls suffered from parasitoids. Inquilines (Curculionidae) were found within some gall walls, with preference for larger galls and for those with thicker walls. Traces of potential bird or wasp predation were also found in a few specimens. Our present fi ndings contribute original descriptive information on these galls. Overall, our results lend further support on the potential importance of natural enemies on gall evolution.
KEY WORDS. Nothofagus dombeyi; Cohiue; Patagonia; Galling insects.
INTRODUCTION
Plant galls are formed subsequent to a parasitic attack, where the parasite (e.g. viruses, bacteria, fungi, nematodes or arthropods) hinders normal plant morphogenesis and causes the plant to differentiate into abnormal tissue (Meyer, 1987; Harper et al., 2004). In the case of insect induced galls, the tissue differentiates concentrically around the insect, into layers with protective and nutritive functions (Weis et al., 1988). Gall characteristics are partly dependent on the inducing species, as different species reorganise plant tissue in various ways, resulting in a multitude of gall appearances (Shorthouse & Rohfritsch, 1992), although characteristics are also dependent on the host plant (Weis et al., 1988).
There are a number of hypotheses to explain the evolutionary origin of galls. Some authors have argued that the adaptive value is in favour of the plant or in favour of both the plant and the herbivore; others have argued that galls function as a plant defence response or defence against insect attacks (Price et al., 1987). However, since a galling insect cannot survive without its gall and galls have been shown to incur a cost to the plant, such as delayed bud burst (Foss & Rieske, 2004) the evolutionary origin of galls should rather be sought among insect life history strategies instead of plant defence mechanisms (Price et al., 1987; Weis et al., 1988), and often this plant-gall inducer relationship is regarded as purely parasitic (Stone et al., 2002; Stone & Schönrogge 2003).
Price et al. (1987) suggested there are three major hypotheses that explain the nature of galls and the evolutionary pressures that may have led to gall evolution: (1) The nutrition hypothesis, suggesting that galls are an easy-accessible, high-nutrition storage site and a higher quality food source than ungalled tissue. (2) The enemy hypothesis, suggesting that galls provide physical protection against predators and parasites by having thick and/ or tough walls. (3) The microenvironment hypothesis, implying that galls give protection against the abiotic microenvironment (e.g. temperature and hygrotermal stress). Price et al. (1987) found strong evidence in favour of the nutrition hypothesis and mixed evidence for the enemy hypothesis. The microenvironment hypothesis was supported in a limited number of cases (Price et al., 1987), but still all three hypotheses receive attention from researchers (Stone et al., 2002; Stone & Schönrogge, 2003).
The purpose of this study is twofold. First, we provide a first detailed description of the globular stem galls caused by Aditrochus coihuensisOvruski (Hymenoptera: Pteromalidae), on the southern beech, Nothofagus dombeyi Blume. Aditrochus coihuensis adults are approximately 3.3 mm long and the species is virtually unstudied - its description by De Santis et al. (1993) is the only available scientific paper up to date, with exception of a recent record of the parasitoid Plectocynips pilosus Ros-Farré (Hymenoptera: Figitidae) from galls of A. coihuensis (Buffington & Nieves-Aldrey, 2011). We also report observations on parasitoids and inquilines associated with these galls, which might be of interest for future research. Secondly, we explore the enemy hypothesis in these galls, by focusing on the association of gall morphology and success of the gall inducer.
MATERIAL AND METHODS
Gall collection. All galls were collected in the vicinity of the city of San Carlos de Bariloche, Argentina (41º08'35"S, 71º18'44W). A small set of galls for preliminary measurements was collected on February 9, 2007. Thereafter, galls were collected between the 1st and 31st of March. The period of gall collection was late in the season and the vast majority of the gall inhabitants had already emerged. Three to four trees per site were sampled, at six sites (total n=20 trees and n=270 galls). Sites were chosen with respect to known N. dombeyi presence and were checked for gall-bearing individuals, of which the first 3-4 trees found were selected. A sampling area of 25% of the tree's height and canopy width was marked out at the tree's most sun-exposed side, which was browsed for all new galls. All branches containing galls were cut and placed in damp Oasis® foam in order to maintain gall freshness.
Gall measurements and classification. Emergence-hole diameter and chamber diameters were measured to establish whether these parameters differed between galls with and without parasitoids, and thus could be used to assess gall-success. Several gall morphological characteristics were studied to explore the enemy hypotheses. Specifically, we measured: (1) gall diameter, with a digital calliper; (2) gall toughness, with a Pesola® Medio-Line Pressure set (set as pressure dynamometer, modified with an attached 0.80 mm thick syringe, which was stung 3 mm into the gall), where the mean of five stings was used for analysis; (3) gall chamber-diameter; (4) emergence-hole diameter, both with a gauge ocular equipped stereoscope, where the mean of two perpendicular measurements was used for analysis; (5) gall wall thickness; calculated by subtracting chamber diameter from gall diameter and dividing by two and (6) insect presence as insect remains, abnormal chamber (non-circular or miscoloured) and/ or abnormal emergence-hole. The latter was noted for classification of galls as successful (gall-s) or unsuccessful (gall-us). Galls with traces of insect chitin skeleton and/ or a compressed mass of a black, adhesive substance attached to the chamber wall (plausible remains of A. coihuensis) were classified as successfully attacked by a parasitoid (i.e. gall-us). Emergence-hole diameter was also used for gall classification (see under Statistics). We also noted (7) the presence/absence of inquilines (henceforth inq-p/inq-a) and (8) the size of any parasitoid specimens found inside (we measured total body length, thorax laterally, thorax ventrally and length of ovipositors). A mean of thorax size (laterally and ventrally; i.e.: the thorax-mean) was also calculated, in order to compare with mean emergence-hole diameter. Insect remains suspected to belong to the gall inducer, A. coihuensis, were compared with the description and drawings by de Santis et al. (1993), and confirmed by colleagues (Dr. Marta Loiácono and Dr. Daniel Aquino of the Museum of La Plata, personal communication)
Statistical analysis. Standard statistical tests, t-test or Mann-Whitney U test, were used depending on normality and homogeneity of variances among samples (SPSS 11.0 for Windows). Toughness was statistically tested in two ways, either including all galls (tough-all) or excluding galls showing signs of degradation (miscoloured brown and/or internally disintegrated galls, tough-exclude). Also, Pearson regression analysis was conducted for a number of parameters, on all galls, gall-s only and gall-us only.
To determine whether galls had been subjected to parasitoid attack, they were classified according to the presence or absence of insect remains and abnormal chamber appearance (see above). All galls in which their appearance did not allow clear classification were excluded from analysis. A Mann-Whitney U test was conducted after classification, to test whether emergence-hole diameters differed between successful galls (gall-s) and unsuccessful galls (gall-us). A difference between groups was detected and a re-classification was carried out with respect to insect remains, abnormal chamber appearance and emergence-hole diameter. Emergence-hole diameter >1.1mm was classed as gall-s and <1.05mm as gall-us. The interval between 1.05 and 1.1mm was classed as unknown, unless insect remains were present.
RESULTS
Gall success. The first classification of gall success showed a difference between groups with respect to emergence-hole diameter (Mann-Whitney U, p<0.001). Successful galls had a mean emergence-hole diameter of 1.22 mm +0.098 SD while unsuccessful galls had a mean diameter of 0.86 mm +0.182 SD. In an attempt to elude misclassification, the limits were set to stricter values. Therefore, galls were re-classed, based on emergence-hole diameter, according to >1.1mm = gall-s, <1.05mm = gall-us and with values between 1.05 and 1.1 mm being excluded (Fig.1). After this re-classification, the difference between groups was still significant (Mann-Whitney U, p<0.001) and the mean emergence-hole diameter was 1.23mm (+0.08 SD) for gall-s and 0.83mm (±0.15 SD) for gall-us.
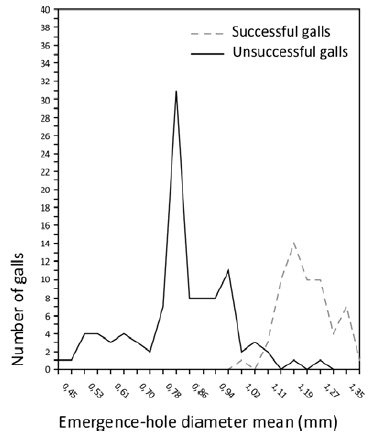
Fig. 1. Comparison of emergence-hole diameter between gall success classifications for A. cohiuensis (Mann-Whitney U, p<0.001) - successful galls (gall-s, mean diameter 1.23 mm ±0.082 SD) and unsuccessful galls (gall-us, mean diameter 0.83 mm ±0.151 SD).
Based on this classification 67% of galls caused by A. coihuensis suffered from parasitoid attacks (n: 182 total, 60 gall-s and 122 gall-us). Seven individuals belonged to A. coihuensis (Chalcidoidea: Pteromalidae, Fig. 2a, b) and nine specimens belonging to unidentified species of Torymidae (Chalcidoidea) were found within some galls (Fig. 2c). The mean thorax width was 0.55 mm for the Torymidae specimens and 0.87 mm for A. coihuensis, while mean length was 2.44 mm (Torymidae) and 3.05 mm (A. coihuensis). Three individuals (i.e., females) of the torymid wasps had ovipositors, spanning from 2.46-3.20 mm in length.
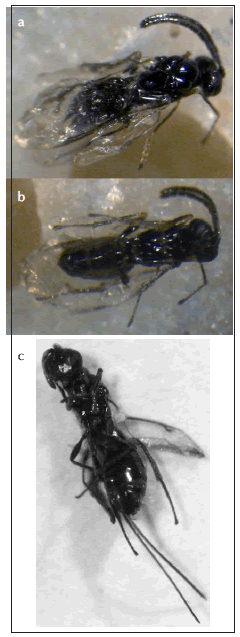
Fig. 2. (a) Insect identified as Aditrochus coihuensis (Hymenoptera: Pteromalidae) (mean length 3.05 mm), dorsal view; (b) ventral view; (c) Metallic green parasitoid (probably Hymenoptera: Torymidae) that was found within some galls (body mean length 2.44 mm, ovipositors 2.46-3.20 mm).
Gall morphometrics. A significant difference between successful and unsuccessful galls was found regarding gall diameter (t-test, p<0.001) and gall wall thickness (Mann-Whitney U, p<0.001) (Fig. 3 a, b). Gall-s had a larger diameter and thicker wall. Mean gall diameter was 13.2 mm (all galls), 15.9 mm (gall-s) and 12.5 mm (gall-us). Mean wall thickness was 5.0 mm (all galls), 6.3 mm (gall-s), 4.6 mm (gall-us). Also, gall-s had a slightly larger chamber diameter (mean 3.55 mm) than unsuccessful galls (mean 3.27 mm) (t-test, p<0.01; Fig. 3c). A very strong correlation was found between gall diameter and wall thickness, regardless of gall success (all galls: Pearson r=0.99 p<0.001, r2=0.98 (Fig. 4); gall-s: Pearson r=0.99 p<0.001, r2=0.98; gall-us: Pearson r=0.99 p<0.001, r2=0.99). Positive correlations were also found between gall diameter and chamber diameter for all galls (Pearson r=0.34 p<0.001, r2=0.11) and for gall-s (Pearson r=0.42 p<0.01, r2=0.18).
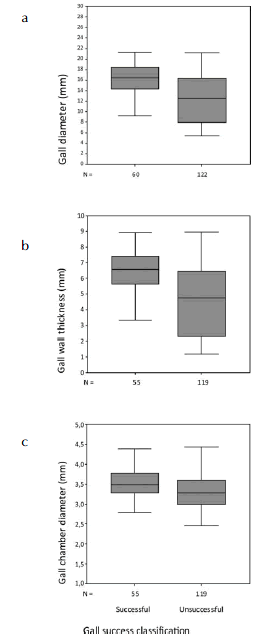
Fig. 3. (a) Gall diameter according to gall success classes (t-test, p<0.001; mean gall-s 15.9 mm, gall-us 12.5 mm). (b) Gall wall thickness compared between gall success classes (Mann-Whitney U, p<0.001; mean gall-s 6.3mm, gall-us 4.6mm). (c) Gall chamber diameter compared between gall success classes (t-test, p<0.01; mean gall-s 3.55 mm, gall-us 3.27 mm).
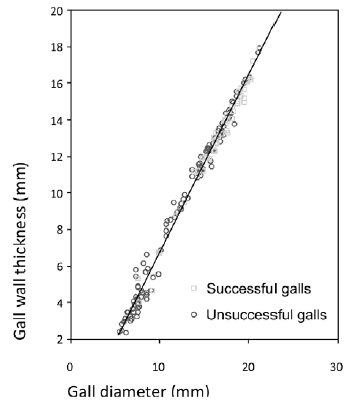
Fig. 4. Correlation between gall diameter and gall wall thickness when sorted by all galls (Pearson r = 0.991 p<0.001, r2=0.983).
Additional observations. A white unidentified larva was found within some galls (n=22). These galls lacked emergence-hole and had a circular chamber. Within these galls, remains were also found a probably exuviae from molting insect instars. Galls containing the larvae, had a significant smaller chamber diameter compared to gall-s (t-test, p<0.001) and gall-us (t-test, p<0.05). They also were smaller (gall diameter: t-test, p<0.001) and bore thinner walls (Mann-Whitney U, p<0.001) compared to gall-s, but not in comparison with unsuccessful galls (diameter: t-test, p>0.05; wall thickness: Mann-Whitney U, p>0.05).
An inquiline species of the family Curculionidae was found in several galls (n=48, 1-3 individuals per gall), either as larvae, pupae or even adults (Fig. 5a). Weevils dwell inside the gall wall, where individual larva creates a burrow and later perforates an emergence-tunnel. The larva does not pierce the gall surface, but the tunnel can be distinguished from the outside, as a circular mark, slightly darker than surrounding tissue (Fig. 5b). No evidence was found that these weevils penetrated the gall chamber and interacted with the host or parasitoid residing inside. Galls containing Curculionidae had a larger diameter (t-test, p<0.001) and thicker walls compared to galls without the inquiline (Mann-Whitney U, p<0.001). Further, there were no differences between inq-p and inq-a groups in chamber diameter (t-test, p>0.05) or emergence-hole diameter (Mann-Whitney U, p>0.05), irrespective of gall success.

Fig. 5. (a) Larva, pupae and adult of an inquiline (Curculionidae), found within the wall of some galls. (b) Inquiline larva (Curculionidae) and a burrow created by the larva (lower). The blue markings encircle where emergence-tunnels, perforated by larvae, abuts the epidermis. The tunnels can be distinguished as dots, slightly darker than surrounding tissue (upper). (c) Hollowed out craters in galls that reached as far in as to the chamber - possibly caused by bird or wasp predation.
A few of the collected galls (n=4, mean gall diameter 14.1mm) had large craters hollowed out, that reached as far as into the chamber (Fig. 5c). These craters might be from bird- and/or wasp-predation. Note that invading wasps have become a pest in Argentina in the last decades (Sackmann et al., 2008).
DISCUSSION
Emergence-hole diameter seems to be a valid indication of gall success. The larger emergence holes are more likely caused by A. coihuensis, given their larger mean thorax width, while the smaller exit holes are probably made by a parasitoid of the galler.
There was a strong correlation between wall thickness and gall diameter, and for both variables, highly significant differences were found between successful and unsuccessful galls. This finding suggests that wall thickness of fully developed galls, may favour escape from parasitoidism, as this exceeds the length of the ovipositor of potential parasitoids (Price & Clancy, 1986; Weis et al., 1988; Craig et al., 1990; Plantard & Hochberg, 1998). Of course, parasitoidism could occur at early stages of gall development (Washburn & Cornell, 1981; Stone & Schönrogge, 2002).
The chamber diameter was slightly smaller in failed galls, which suggests that A. coihuensis was killed in its larval stage. Thus, the smaller chamber diameter could be considered to be an indication of larval parasitoidism, presuming that the parasitoid is an idiobiont (i.e. prohibits further host-development after attack). Besides, galls with the unidentified larva had no emergence-hole, no insect remains (other than exuviae) and since they had smaller chamber diameter compared to both gall-s and gall-us, the larvae can be assumed to be A. coihuensis.
Toughness had no influence on gall success, according to the two toughness measurements. However, toughness might change with time and galls may have been softer in the beginning of their development, as in the case of the leaf-galling wasp Atrusca caprone Weld (Fernándes et al., 1999; see also Craig et al., 1990). Hence, if galls are attacked early in their development, as indicated by mean gall diameter and ovipositor length, gall toughness might have a significant influence on gall success, which would not be detected when toughness is measured in fully grown galls.
The unidentified curculionids seemed to prefer galls with large gall diameter and thicker walls. This seems logic, as the inquiline exploits the gall wall and has a certain requirement of space.
There were only few galls that showed signs of predation. Therefore, the level of predation might be too low to make an impact on gall-size. Note also that predation may have been mostly caused by recently invading non-native wasps (Sackmann et al., 2008). It is however conceivable that the gall is removed from the branch or entirely consumed in case of predation. In order to establish whether this is the case, it would be necessary to make observations over a longer period to determine predator behaviour.
In conclusion, gall success classification based on insect remains and emergence-hole diameter seems valid for A. coihuensis. The difference in gall success suggests that the gall wall thickness (but not gall toughness) affects gall survival. Our present findings contribute for the first time, descriptive information on gall morphometrics ecology in Nothofagus spp. in Patagonia. Our results, while limited in spatial and temporal scales, add to the existing knowledge on the importance of natural enemies on gall evolution.
ACKNOWLEDGEMENTS
We thank Dr. Marta Loiácono and Dr. Daniel Aquino of the Museum of La Plata, for their help with species identification and José Villacide for sharing his knowledge about galls. We also thank Juan Gowda, Ann-Mari Fransson, Karin Heinmann and Lucas Garibaldi for their help in practical matters regarding this study. This study was funded by the Department of Undergraduate Studies in Biology at Lund University and the Linneaus-Palme exchange program in cooperation with Laboratorio ECOTONO,CRUB-Universidad Nacional del Comahue and Grupo de Ecología de Insectos, INTA EEA Bariloche.
LITERATURE CITED
1. BUFFINGTON, M. L. & J. L. NIEVES-ALDREY. 2011. Revision of Plectocynipinae (Hymenoptera: Figitidae) with descriptions of a new genus and three new species from Chile. Proc. Entomol. Soc. Wash. 113: 91-108. [ Links ]
2. CRAIG, T. P., J. K. ITAMI & P. W. PRICE. 1990. The window of vulnerability of a shoot-galling sawfly to attack by a parasitoid. Ecology 71: 1471-1482. [ Links ]
3. FERNANDES, G. W., M. M. ESPIRITO-SANTO & L. M. FARIA. 1999. Cynipid gall growth dynamics and enemy attack: The effect of gall size, toughness and thickness. An. Soc. Entomol. Bras. 28: 211-218. [ Links ]
4. FOSS, L. K. & L. K. RIESKE. 2004. Stem galls affect oak foliage with potential consequences for herbivory. Ecol. Entomol. 29: 273-280. [ Links ]
5. HARPER, L. J., K. SCHÖNROGGE, K. Y. LIM, P. FRANCIS & C. P. LICHTENSTEIN. 2004. Cynipid galls: insect-induced modifications of plant development creates novel plant orgns. Plant, Cell Environ 27: 327-335. [ Links ]
6. MEYER, J. 1987. Plant galls and gall inducers. Gebrüder Borntraeger. Berlin, Stuttgart. [ Links ]
7. PLANTARD, O. & M. E. HOCHBERG. 1998. Factors affecting parasitism in the oak-galler Neuroterus quercusbaccarum (Hymenoptera: Cynipidae). Oikos 81: 289-298. [ Links ]
8. PRICE, W. P. & K. M. CLANCY. 1986. Interactions among three trophic levels: Gall size and parasitoid attack. Ecology 67: 1593-1600. [ Links ]
9. PRICE, W. P., G. W. FERNANDES & G. L. WARING. 1987. Adaptive nature of insect galls. Environ. Entomol. 16: 15-24. [ Links ]
10. SACKMANN, P., A. G. FARJI-BRENER & J. C. CORLEY. 2008. The impact of an exotic social wasp (Vespula germanica) on the native arthropod community of NW Patagonia, Argentina: An experimental study. Ecol. Entomol. 33: 213-224. [ Links ]
11. DE SANTIS, L., P. FIDALGO & S. OVRUSKI. 1993. Himenópteros parasitoides de los géneros Aditrochus Ruebsaamen y Espinosa Gahan (Insecta, Hymenoptera, Pteromalidae) asociados a agallas en Nothofagus (Fagaceae) del sur de Argentina y Chile. Acta Entomol. Chilena 18: 134-146. [ Links ]
12. SHORTHOUSE, J. D. & O. ROHFRITSCH (eds.). 1992. Biology of insect-induced galls. Oxford University press, New York. [ Links ]
13. STONE, G. N. & K. SCHÖNROGGE. 2003. The adaptive significance of insect gall morphology. Trends Ecol. Evol. 18: 512-522. [ Links ]
14. STONE, G. N., K. SCHÖNROGGE, R. J. ATKINSON, D. BELLIDO & J. PUJADE-VILLAR. 2002. The population biology of oak gall wasps (Hymenoptera: Cynipidae). Annu. Rev. Entomol. 47: 633-668. [ Links ]
15. WEIS, A. E., R. WALTON & C. L. CREGO. 1988. Reactive tissue sites and the population biology of gall makers. Annu. Rev. Entomol. 33: 467-486. [ Links ]
16. VEBLEN, T. T., R. S. HILL & J. READ (eds.). 1996. The ecology and biogeography of Nothofagus forest. Yale University press. New Have and London. [ Links ]














