Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de la Sociedad Entomológica Argentina
Print version ISSN 0373-5680
Rev. Soc. Entomol. Argent. vol.70 no.3-4 Mendoza July/Dec. 2011
NOTA CIENTÍFICA
Phenotypic plasticity for Drosophila melanogaster (Diptera: Drosophilidae) larval olfactory behaviour in response to whole fruit olfactory stimuli
Plasticidad fenotípica para el comportamiento olfativo larval de Drosophila melanogaster (Diptera: Drosophilidae) como respuesta a estímulos olfativos de frutos enteros
Lavagnino, Nicolás J. and Juan J. Fanara
Laboratorio de Evolución. Departamento de Ecología, Genética y Evolución. Facultad de Ciencias Exactas y Naturales. Universidad de Buenos Aires. Pabellón II. Ciudad Universitaria. C1428HA. Buenos Aires, Argentina; e-mail: nlavagnino@ege.fcen.uba.ar
Recibido: 12-V-2011
Aceptado: 12-VII-2011
ABSTRACT. Drosophila melanogaster Meigen 1830 (Diptera: Drosophilidae) is the quintessential insect model organism. However, with a few exceptions, ecological features of this species have been poorly investigated. In the present work we describe a behavioural assay to quantify olfactory behaviour of D. melanogaster larvae in response to complex olfactory stimuli that are present in the natural environments, i. e.: rotten fruits that act as hosts in nature. Results obtained using this assay reveal that there is intra-population genetic variation and phenotypic plasticity for the character in a natural population from west-central Argentina.
KEY WORDS. Olfactory behaviour; Natural variation; Phenotypic plasticity; Drosophila melanogaster; Behavioural assay.
RESUMEN. Drosophila melanogaster Meigen 1830 (Diptera: Drosophilidae) es el organismo modelo por excelencia en insectos. Sin embargo, salvo algunas excepciones, los aspectos ecológicos de esta especie han sido poco estudiados. En el presente trabajo, se describe un ensayo comportamental para cuantificar la conducta olfativa de larvas de D. melanogaster en respuesta a estímulos olfativos complejos que se encuentran en los ambientes naturales, i. e.: frutos en descomposición que son hospedadores en la naturaleza. Los resultados obtenidos, utilizando este ensayo, revelaron que existe variabilidad genética intra-poblacional y plasticidad fenotípica para el carácter en una población natural del centro oeste de Argentina.
PALABRAS CLAVE. Comportamiento olfativo; Variabilidad natural; Plasticidad fenotípica; Drosophila melanogaster; Ensayo comportamental.
Drosophila melanogaster Meigen 1830 (Diptera: Drosophilidae) is the quintessential insect model organism for genetic, physiological or developmental research.
However, with a few exceptions (Sokolowski et al., 1986; Hoffman & O' Donnell, 1990; Medina-Muños & Godoy-Herrera, 2005; Reaume & Sokolowski, 2006), the ecological features of this species have been poorly investigated. In this sense, further research is needed, both in situ in natural environments and in the lab using methods designed to recreate natural situations or scenarios. In this study, we present a behavioural assay to quantify olfactory behaviour of D. melanogaster larvae in response to natural host fruit stimuli and the utilization of this assay to ask whether there is intra-population genetic variation and phenotypic plasticity
- defined as the property of individual genotypes to produce different phenotypes when exposed to different environmental conditions (Schlichting & Pigliucci, 1998)
- for larval olfactory behaviour (LOB) in a natural population from west-central Argentina.
To test this hypothesis we first collected 20 to 30 gravid D. melanogaster females from a natural population near the locality called Lavalle (Province of Mendoza, Argentina, Latitude 32º 50' S, Longitude 68º 28' W, Altitude 647 m). Then we set up a single strain by mass culture using the offspring of each single gravid female collected. This strain, that represents a sample of the natural genetic variation in Lavalle population, was used to quantify larval olfactory response to different rotten fruits that act as hosts for D. melanogaster larvae in nature. Host fruit used were Vitis vinifera Linnaei ("grape") a natural host of D. melanogaster larvae in Lavalle, and Prunus persica Linnaei ("peach") an alien fruit to larvae derived from Lavalle used in this study (Lavagnino et al., 2008). The strain was maintained by full-sib mating for 10 generations on cornmeal-molasses-agar medium under standard conditions of 25 ± 1 ºC, 70% humidity and a 12-h light : 12-h dark cycle, and was never exposed to the host fruit used as stimuli until the initiation of the behavioural assay.
The behavioural assay used was modified from the classical assay of Aceves-Piña & Quinn (1979). Adult females from the Lavalle strain were allowed to lay eggs for 8 hours on an agar medium with commercial yeast paste. Larvae were tested after 36 h. Before being tested they were washed from the yeast paste. Between 10 to 30 larvae were placed in the center of a 10 cm Petri dish containing 10 ml of 2,5% agar. 300 μl of a preparation consisting of whole rotten fruit ("grape" and "peach") diluted in distilled water (6 g / ml) and 300 μl of distilled water (H2Od) were placed on filter paper discs on opposite ends of the Petri dish. To prevent diffusion of odorants through the agar and to eliminate larval gustatory responses, the filter paper discs containing the rotten fruit stimulus or H2Od were placed on inverted lids cut off from 15 ml Falcon tubes. Lids sidewalls where covered with black tape to avoid phototaxis. Seven minutes after the introduction of the larvae, the number of individuals within a 30 mm radius from each filter disc and the larvae that remained between both 30 mm radius were counted. A larval response index (LRI) was calculated for each Petri dish as:
LRI = [(nfruit - nwater)/ ntotal] x 100
Where "n" designates the number of larvae and the subscripts indicate the sides of the Petri dish containing rotten fruit or distilled water (H2Od), respectively. This index varies between -100 (total repulsion) and +100 (total attraction). An RI = 0 indicates indifferent behaviour. When tested in groups, individual larvae respond to stimuli independent of other individuals (Monte et al., 1989). We ran 9 replicates of the "peach" test and 10 of the "grape" test. All behavioural tests were performed between 2 pm and 4 pm under controlled temperature (25 ± 1 º C), light (5,4 ± 0,2 x 105 lx) and humidity (42 ± 5 %). Finally, it was not necessary to turn the plates to avoid positional bias. In fact, a control test performed with distilled water as stimuli on both sides of the Petri dish showed no significant results in a T-test for dependent samples, indicating the absence of side effects (Table I).
Table I. Description of statistical analysis results of larval olfactory response to a recreated natural scenario. Side effect experiment: T-test for dependent samples results between the mean number of larvae within a 30 mm radius of the stimuli when H2Od was used in both opposite sides of the Petri dish. Stimulus experiment: result of the comparison by a one fixed factor ANOVA of mean LRI to "grape" and "peach" (sources of variation were: stimulus with two levels -"grape" and "peach"- and the error term; with 1 and 17 D.F., respectively). Significant p-value is in bold. On the left of the table, sketches of the test plates and indications of comparisons carried out in each case are shown.

Our study determined that LRI are significantly different when the stimuli are "grape" or "peach" (Table I), indicating that LOB exhibit phenotypic plasticity when both natural resources were used as olfactory stimulus. Although mean LRI determine that "grape" and "peach" are both attractive stimulus because they showed positive values, "grape" (mean LRI = 34,53) produced a significantly larger attraction than "peach" (mean LRI =15,69) (ANOVA, p = 0,042; Table I; Fig. 1). Variance of LRI was 448,51 when the stimulus was "grape" and 235,86 when it was "peach". Considering that the strain used represents a sample of the pool of genotypes in the natural population, the presence of a different from zero variance suggests that this population harbours natural genetic variation for LOB.
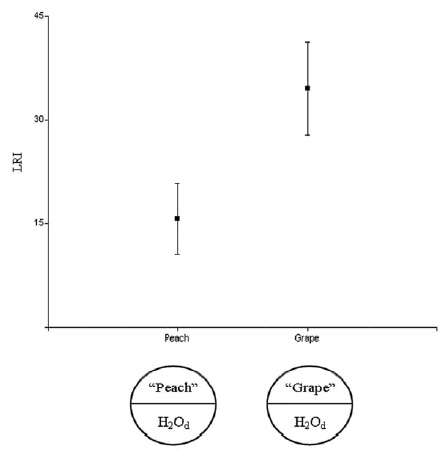
Fig. 1. Mean olfactory response (LRI) to extracts of rotten "peaches" and "grapes" of D. melanogaster larvae derived from Lavalle natural population. A sketch of the test plates used in each experiment is shown below the graphic. We ran 9 replicates of the "peach" test and 10 of the "grape" test. From 10 to 30 larvae were used in each replicate. Error bars are standard errors.
In conclusion, we present here a behavioural assay to quantify D. melanogaster larval olfactory response to complex olfactory stimuli that are present in the natural environments, i. e.: rotten fruits that act as hosts in nature (the assay could also be used for larvae of closely related species). Results obtained using this assay suggest that this population harbours natural genetic variation for LOB. Also that wild derived larvae from a natural population located in Lavalle present phenotypic plasticity for olfactory behaviour with stronger attractive response to a known host fruit ("grape") than to an alien one ("peach"). Phenotypic plasticity is a key step in the process of phenotypic evolution and could participate in the maintenance of genetic variation (Schlichting & Pigliucci, 1998; West-Eberhard, 2003) within natural populations, then assessing its occurrence becomes pertinent to deepen the comprehension of these evolutionary and ecological issues in this species.
ACKNOWLEDGEMENTS
This work was supported by grants from Universidad de Buenos Aires, CONICET and Agencia Nacional de Promoción Científica y Técnica. NJL is a recipient of a post-doctoral scholarship of CONICET (Argentina) and JJF is a member of Carrera del Investigador Científico of CONICET (Argentina).
LITERATURE CITED
1. ACEVES-PIÑA, F. & W. QUINN. 1979. Learning in normal and mutant Drosophila larvae. Science 206: 93-96. [ Links ]
2. HOFFMANN, A. A. & S. O'DONNELL.1990. Heritable variation in resource use in Drosophila in the field. In: Barker, J. S. F., W. T. Starmer & R. McIntyre (Eds.). Ecological and Evolutionary Genetics of Drosophila, Plenum Press, New York. [ Links ]
3. LAVAGNINO, N. J., R. R. H. ANHOLT & J. J. FANARA. 2008. Variation in genetic architecture of olfactory behaviour among wild-derived populations of Drosophila melanogaster. J. Evolution. Biol. 21: 988-996. [ Links ]
4. MEDINA-MUÑOZ, M. C. & R. GODOY-HERRERA. 2005. Dispersal and prepupation behaviour of Chilean sympatric Drosophila species that breed in the same site in nature. Behav. Ecol. 16: 316-22. [ Links ]
5. MEIGEN, J. W. 1830. Systematische Beschreibung der Bekannten Europaischen Zweiflugeligen Insekten. Vol. 6. Theil Schulze, Vienna. [ Links ]
6. MONTE, P., C. WOODAD, R. AYER, M. LILLY, H. SUN & J. CARLSON. 1989. Characterization of the larval olfactory response in Drosophila and its genetic basis. Behav. Genet. 19: 267-283. [ Links ]
7. REAUME, C. J. & M. B. SOKOLOWSKI. 2006. The nature of Drosophila melanogaster. Curr. Biol.16: R623-R628. [ Links ]
8. SCHLICHTING, C. D. & M. PIGLIUCCI. 1998. Phenotypic Evolution: A Reaction Norm Perspective. Sinauer Associates, Sunderland, MA. [ Links ]
9. SOKOLOWSKI, M. B., S. J. BAUCR, V. WAI-PING, L. RODRIGUEZ, J. L. WONG & C. KENT. 1986. Ecological genetics and behaviour of Drosophila melanogaster larvae in nature. Anim. Behav. 32: 403-408. [ Links ]
10. WEST-EBERHARD, M. J. 2003. Developmental Plasticity and Evolution. Oxford University Press, New York. [ Links ]














