Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista de la Sociedad Entomológica Argentina
versión impresa ISSN 0373-5680
Rev. Soc. Entomol. Argent. vol.74 no.1-2 La Plata jun. 2015
TRABAJO CIENTÍFICO
Spatial distribution, feeding and length-mass relationships of Diamphipnopsis samali (Plecoptera, Diamphipnoidae) in a North Patagonia Andean stream, Argentina
Distribución espacial, alimentación y relaciones largo peso de Diamphipnopsis samali (Plecoptera, Diamphipnoidae) en un arroyo andino del Norte de Patagonia, Argentina
Omad, Guillermo H., Pablo Pessacq & Luis B. Epele
Centro de Investigación Esquel de Montaña y Estepa Patagónica (CIEMEP), CONICET-UNPSJB. Roca 780, 9200. Esquel, Chubut, Argentina. E-mail: guillermoomad178@hotmail.com, pablopessacq@yahoo.com.ar, luisbepele@hotmail.com
Recibido: 23-II-2015
Aceptado: 21-IV-2015
ABSTRACT. Some aspects of the biology and ecology (distribution pattern, feeding habits and length-mass relationship) of a population of Diamphipnopsis samali are studied in a first order stream surrounded by exotic forest located in North Patagonia, Chubut province, Argentina. Two samplings were performed during high and low water periods. All information was treated with fixed-effects 2-factor ANOVA models to assess significant differences. Mouth parts analysis of D. samali indicates that it corresponds to an intermediate type between carnivorous and herbivorous-detritivorous type. The guts content shows that the species specially fed on wood and fungal hyphae. No significant difference on the consumed food between sampling periods was found, even though there were significant differences between food content on large and small larvae. Regarding the habitat there was a clear preference for habitats waterfall and log and the difference between run and pool was also significant. There were no significant differences in substrate preference between high and low water periods. This Patagonian endemic species seems to be well adapted to the presence of exotic riparian forest using fallen logs for shelter and food.
KEY WORDS: Habitat use; Larval feeding; Exotic forest.
RESUMEN. Fueron estudiados algunos aspectos de la biología y ecología (patrones de distribución espacial, hábitos alimenticios y relación largo-masa) de una población de Diamphipnopsis samali en un arroyo de primer orden que atraviesa un bosque exótico de pino en el centro-norte de Patagonia (Chubut, Argentina). Dos series de muestreos fueron realizados durante períodos de aguas altas y aguas bajas. Toda la información fue tratada con ANOVAS de dos factores y efectos fijos para evaluar diferencias significativas. El análisis de las piezas bucales de D. samali indica que corresponden a un tipo intermedio entre el tipo carnívoro y herbívoro-detritívoro. El contenido estomacal muestra que esta especie se alimenta de hifas fúngicas y madera, aunque en los estadios tempranos se encuentran restos de insectos. No se hallaron diferencias significativas en la alimentación entre períodos de aguas altas y aguas bajas, sin embargo, existieron diferencias significativas entre la dieta de larvas grandes y pequeñas. Fue clara la preferencia por el sustrato tronco y cascada, y también significativamente diferente entre los tipos pozón y corredera. No hubo diferencias significativas en la elección de hábitat entre los períodos de aguas bajas y aguas altas. Esta especie endémica de Patagonia parece estar bien adaptada a la presencia de un bosque ripario exótico, utilizando los troncos caídos como alimento y refugio.
PALABRAS CLAVE: Uso de hábitat; Alimentación larval; Bosque exótico.
INTRODUCTION
A stream is a heterogeneous combination of factors such as substrate size, flow velocity, organic matter of different nature and substrate persistence, among others (Paul & Meyer, 2001; Merritt et al., 2008). All these factors create a great heterogeneity, which is usually positively correlated with insect species richness, by providing a wide range of habitats (Ward, 1989). Some of these factors, like substrate size, flow velocity and organic matter are usually used to define habitat type.
Plecoptera (stoneflies) are an important component of the macroinvertebrate fauna in cold, pristine freshwater streams; they inhabit these environments as larvae (Zwick, 1980; Allan, 1995) and play an important role in food webs, recycling nutrients as primary or secondary consumers (Lamberti & Moore, 1984; Peckarsky, 1984). This order includes species of all functional feeding groups such as periphyton scrapers, collectors, exclusive shredders and predators (Allan, 1995; Stewart & Stark, 1993). Plecoptera larvae exhibit a marked sensitivity to environmental changes and they are usually used in assessing water quality conditions (Rosenberg & Resh, 1993).
Streams in Patagonian mountains are subjected to different land use practices; the most common are pasture conversions, exotic plantations, and wood collection/deforestation (Miserendino et al., 2011). The consequences that these activities are having on functional structure of the local, highly endemic biota are not explicitly known (Miserendino & Masi, 2010). Benthic macroinvertebrates, including stoneflies, have developed different mechanisms to exploit food resources, which reflects the heterogeneity of environmental characteristics of the watercourses, but these mechanisms can be affected by human activities in or close to the watercourses, forcing the biota to adapt to the new conditions or disappear (Masi, 2006).
Habitat selection is probably one of the most important selective responses that affect Plecoptera survival; many species are found in certain kind of streams and others only in specific microhabitats (Hynes, 1976). Particular association of several stonefly species with coarse particulate organic matter (CPOM) or fine particulate organic matter (FPOM) and substrate type have been found (Albariño, 2001; Ernst & Stewart, 1986; Krzysztof & Stewart, 1987; Velásquez & Miserendino, 2003a, b; Derka et al., 2004; Merritt et al., 2008) and such partitioning of habitat is expected for many stoneflies species (Stewart & Stark, 1993).
Many studies on Patagonian streams refer to taxonomic and biogeographic aspects of Plecoptera (i.e. Illies, 1963; McLellan et al., 2006; Vera, 2006, 2008; Pessacq, 2009), and in recent years the interest about different aspects of the group has rapidly increased (e.g. Albariño & Balseiro, 1998; Miserendino, 2000; Velásquez & Miserendino, 2003a; Miserendino, 2006; Hollmann & Miserendino, 2008; Epele & Miserendino, 2011). However, these studies cover a small proportion of the biological and ecological diversity of these highly endemic and vulnerable to habitat changes species (Miserendino, 2000).
A few studies of habitat type selection including Plecoptera have been conducted in the Patagonian region; Velásquez & Miserendino (2003a, b) found that some scraper stoneflies (e.g. Chilenoperla sp., Klapopteryx kuscheli Illies) prefer cobbles and pebbles substrate in riffles. On the other hand Epele & Miserendino (2011) found that Notoperla magnaspina McLellan and N. fasciata McLellan, did not show any substrate preferences.
Other studies were focused on the effect of land use in macroinvertebrate communities including stoneflies; Miserendino & Masi (2010) found that in streams with native riparian vegetation the macroinvertebrate community (especially shredder) was more diverse compared with streams running through exotic vegetation. Albariño & Balseiro (2002) noticed that shredders such as large stoneflies (e. g. Klapopteryx kuscheli) and caddisflies shredders (Parasericostoma cristatum Flint) fed only on Nothofagus pumilio and not on Pinus ponderosa leaf packs in Patagonian streams.
To perform this study, we selected Diamphipnopsis samali Illies, a representative of Diamphipnoidae, a little diverse family endemic of the subantartic forest that includes six species in two genera (Illies, 1960).
The aim of this contribution is to investigate the habitat and feeding preferences of Diamphipnopsis samali present in a stream in an exotic forest of Pinus radiata, as well as to ascertain length mass relationship, contributing to the overall knowledge of Patagonian Plecoptera. Biological and ecological studies on Diamphipnoidae are still limited. There are a few studies that show data of species distribution and some ecological aspects (Valdovinos et al., 2010; Figueroa et al., 2007; Palma and Figueroa, 2008).
MATERIALS AND METHODS
Study area
This study was carried out in a nameless first order stream tributary of Lake Epuyen, belonging to the river Puelo basin in northern Chubut Province (Patagonia, Argentina). The study site (42°08'17"S; 71°31'56"W) has an elevation of 495 m.a.s.l. and is surrounded by a riparian forest composed mainly of the exotic species Pinus radiata. The climate in the region is cold temperate to cold moist. There is a strong west-east gradient in rainfall and snow precipitations, with values ranging from 3000 mm/year in the mountain to 150 mm/year on the east plateau. Summers are warm and the probability of frost is recorded throughout the year (121.4 day/year with frost). The average temperature of the warm season does not exceed 20 ºC and the average is around 9 ºC (Coronato & del Valle, 1988).
Habitat sampling
The study involved two sampling dates, the first was coincident with the high water period (November 2007), and the second with the low water period (May 2008). On each sampling date, water temperature, conductivity, pH and dissolved oxygen were measured with a Hach sension 156 multiparameter probe.
On each sampling date organic detritus and macroinvertebrates were collected with a Surber sampler of 0.09 m2 and 250 µm mesh size, and fixed with 4 % formalin. Four different habitats were selected and defined as run, log, pool and fall. At each habitat four replicate samples were collected (n=32). In qualitative samplings conducted by the junior author on the collecting site and others before this study, many specimens of Diamphipnoidae were collected in small waterfalls and clinging under logs. This previous information leads us to include this habitat types in the sampling. As log and fall are not usually included in other studies, some comments are needed. The first habitat is composed of small falls in the stream, with a width of 40-60 cm and a height of 30-50 cm. The surber sampler was located just beneath the water fall, where big to medium size stones are located, among which pine needles and woody material are trapped. Only the horizontal section of the fall was included, while the vertical section was not touched. In log habitat, small logs of Pinus radiata located in riffles, with a similar surface to surber sampler (in some cases two or more small logs) were located and the sampler was applied over them, all insects in the log were removed. Most of the logs were old and partially rotten, and the insects were found on the underside of the logs. In the laboratory, Diamphipnopsis samali individuals were sorted under a magnifying lens with 5 x magnification. Organic matter from each sample was separated in eight fractions using a series of sieves (1000-250 µm): fine particulate organic matter (FPOM), coarse particulate organic matter (CPOM), was composed of unidentified organic material, pine needles, woody material, herbaceous, fungal hyphae, leaves and fruits and seeds. These components were dried at 105 ºC for up to 4 h in an oven and weighed on an electronic balance with 0.5 mg accuracy.
Mouthparts morphology, gut contents and food categories
Heads and mouthparts of D. samali were dissected, observed under 100 x microscope and drawn (Fig. 1). Foregut contents of 10 individuals (five small and five large) collected in different periods (high water and low water periods, 10 insects each) and different habitats were analyzed following Palmer et al. (1993). The foregut content was homogenized over a slide in glycerin and 5 randomly selected fields were counted under a direct microscope at 400 x magnification. Five food categories were identified: 1) wood, 2) fungi, 3) algae, 4) invertebrates and 5) others. The area covered by each item was measured with a graduated eyepiece. Quantification of each item was based on the fraction of area covered relative to the total area covered by the guts contents. Food categories were identified under microscope when needed, the category others was composed of unidentified organic and inorganic matter.
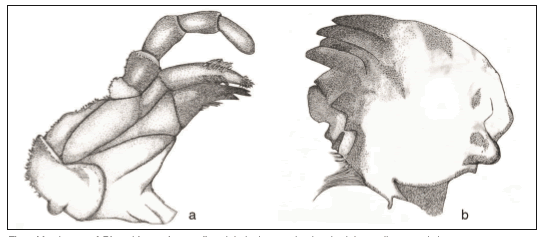
Fig. 1. Mouthparts of Diamphipnopsis samali. a, right lacinea and galae; b, right maxila, ventral view.
Length - mass relationship
To obtain biomass data we performed length-mass regression models. Thus, we measured total body length (TL, measured from labrum to distal tergum) as the distance between the anterior part of the head and the posterior part of the last abdominal segment, the pronotum width (PW), and the head capsule width (HCW) to the nearest 0.05 mm (Benke et al., 1999). Measurements were taken at 8 - 32 x magnification (depending on the size of the individuals) with a linear eyepiece micrometer inserted in a stereomicroscope (Snellen & Stewart, 1979; Short & Ward, 1980; Richardson, 2001).
Body mass relationships can be described by the general power equation DM = a Lb, or after logarithmic transformation ln DM = ln a + b ln L (where: a, b = regression constants, DM = dry mass, and L = length parameters) (Epele & Miserendino, 2011). For dry mass determinations, each specimen was individually transferred to a piece of aluminum foil. Drying was performed at 105° C for up to 4 h (Smock, 1980). The stoneflies were weighed on an electronic balance with 0.5 mg accuracy. In the procedure, 41 specimens of D. samali were examined and measured. Different regression models were evaluated to fit the data points to suitable regression equations. In general, linear regression models were appropriate.
Data analyses
Prior to analysis, data were checked for normality and homogeneity of variances using Kolmogorov-Smirnov and Levene's test, respectively, and transformed as log (x + 1) when appropriate (Gotelli & Ellison, 2004). Fixed effects two way Analysis of variance models (ANOVA) were used to assess significant differences in total density among habitats (n = 4), and between seasons (n = 2). The interaction between habitat and season was also assessed. A posteriori, comparisons among habitats were performed with Tukey's (HSD) test (p < 0.05). Additionally, we studied the differences between small and large larvae gut content and organic fractions in benthic samples.
RESULTS
Environmental features
Water temperature ranged from 4.7 (May) to 12 ºC (November). The oxygen content was 10.7 mg.l-1 at low water period and 8.27 mg.l-1 during the high water period, whereas the percentage of saturation was 104 % and 65 % at low and high water periods, respectively. The pH was stable on both periods of study and was near 7.4. Minimum water depth was 5 cm at run habitats and the maximum was 40 cm at pool habitats. Stream width varied from 0.45 to 2.2 m during low and high water periods, respectively. The substrate of the stream bottom was dominated by gravel, pebble, and sand.
Mouth parts analysis and guts contents
The analysis of D. samali mouth parts under microscope showed well-developed and sclerotized mandibles and maxillae. The mandible (Fig. 1) is quite short, and left and right mandibles are asymmetrical, it possesses five sharp teeth and one flat wide tooth, there is a fringe of hair-like setae on the inner margin and no prostheca. On the maxillae, the galea ends with a dense and fine hair brush and the hairy lacinia ends on three sharp and well-sclerotised teeth (Fig. 1). Maxillary palps are long and strong. The labium has long palps, the glossa and paraglossa are well developed, but the glossa is smaller than the paraglossa. Specimens from the different habitats were used to analyze gut content, in D. samali large individuals, wood was the predominant item (50%), fungi was the second most abundant item (30%), followed by others (13%) and algae (7%). The item others was composed of unidentified organic and inorganic matter. The item invertebrate was absent in the diet of large individuals (Fig. 2), and when present, it was apparently composed by mayflies, identified by some head and leg pieces. For small individuals, fungi, others and wood where the most representative items (32.5, 30 and 27.5% respectively), followed by invertebrates (9%) and algae (1%). The non-parametric analysis showed that there were not significant differences between the type of food consumed between periods (low and high water periods) (Kruskal-Wallis; p>0.05), instead large larvae consumed significantly more wood and algae that small ones (Kruskal-Wallis; p<0.05).

Fig. 2. Food item proportions in the guts contents of larval instars of Diamphipnopsis samali.
Larval density, biomass, and organic matter
Body length (L) was selected to estimate biomass because the regression models obtained showed the best fit (Table I). The highest density was found during high water period at fall habitat (133.3 ind.m-2), while the smallest values were found in the same period for run habitat (13.88 ind.m-2) (Fig. 3). In terms of biomass, the highest value (1.82 g.m-2) was also found in fall habitat during high water period, while the smallest value was found in low water period on habitat run (0.09 g.m-2) (Fig. 3). Late instar larvae were recorded during high water period (mostly at log habitat); meanwhile the recruitment larvae dominated the low water period (Fig. 3). No specimens were found at pool habitats in any of the periods considered.
Table I. Values for the constants ln a and b, obtained for dry weight (W, g) and body total length (TL, mm), (HCW, mm) head capsule width and pronotum width (PW, mm) for D. samali, (n) number of individuals used to calculate the regression.

Fig. 3. Mean and standard deviation of density (a) and biomass (b) of Diamphipnopsis samali in a nameless stream tributary of Lake Epuyen, Chubut Province, Argentina, during high (November 2007) and low (May 2008) water periods.
Woody material was the most abundant fraction of organic matter in log and fall habitats (Fig. 4). The FPOM values ranged from 10 to 52 g.DM.m-2 in the entire study. The highest value of CPOM was found at log habitat (17.16 g.DM.m-2) in low water period and the smallest value was found during high water period for run habitat (4.96 g.DM.m-2). Concerning other items in CPOM, pine needles showed the highest values at log habitat during high water period, and the other values were lower than 25 g.DM.m-2 in the other habitats considered. Herbaceous, fungal hyphae and fruits and seeds were the fraction that showed the lowest values for both periods and were not included in the figures.

Fig. 4. Mean and standard deviation values of unidentified Coarse Particulate Organic Matter (CPOM), Woody material, Pine needles and Leaves, Fine Particulate Organic Matter (FPOM). All values are in grams of Dry Mass per square meter (g DM m-2). For all habitats considered in High (black diamond) and Low (white diamond) water periods.
Larval distribution
Diamphipnopsis samali density values were significantly higher at fall and log habitat than at pools and runs (ANOVA, p < 0.01). Also density values were significantly different between run and pool, with a preference for the former compared to the last. No difference was found in density or biomass values between high and low water periods (ANOVA, p = 0.84 and p = 0.34; respectively).
DISCUSSION
Diamphipnopsis samali mouth parts, as described in results, do not fit a carnivorous type, or a completely herbivorous-detritivorous type either. Stewart & Stark (2002) classify mouthparts of North American stoneflies in two types: carnivorous and herbivorous-detritivorous. Sephton and Hynes (1983) also mention a few omnivorous species, with intermediate characteristics in mouthpart morphology. Even though the overall aspects of mouthparts are those of an herbivorous-detritivorous, as described in results, they do not fit completely with this type of mouthparts (but clearly neither with a carnivorous type), so they could be classified with intermediate characteristics. The mandibles possess sharp teeth, an inner fringe of hair-like setae and lack a prostheca, characteristic of carnivorous species although they are short and asymmetrical, typical features of herbivorous-detritivorous species. The labium is longer than in an herbivorous-detritivorous, but not as long as in a carnivorous species. After analyzing the stomach contents, we can state that the larvae of D. samali are primarily wood shredders and might be feeding from fungal hyphae present in rotten wood or less likely, wood itself. Wood and fungi compose 60-80% of gut contents depending on larval size (it must be added, that large food pieces such as wood are frequently underestimated compared with smaller pieces such as fungi or algae, due to the very large volume of the first). The morphology of mouthparts may be linked to the particular and unusual diet of D. samali, and the mandible agrees with a cutter-shredder morphology. In the stream under study, the riparian corridor is almost completely replaced by Pinus radiata, an exotic pine tree. Whiles and Wallace (1997) stated that conversion of forest to pine monoculture can negatively influence benthic taxonomic diversity. However, D. samali shows to be well adapted to the presence of exotic riparian forest, using the fallen logs within the water as shelter and probably as food and showing a high density of individuals. In contrast with our results, Miserendino & Masi (2010) found in the same stream that the richness of macroinvertebrates, including stoneflies shredders was lower compared to places with native riparian forest and harvest forest. Even though there are no studies on larva density in other rivers for this species, our results seem to show that D. samali is well represented on the stream, showing a high density values for such big insects.
The occurrence of small fragments of invertebrates, a smaller proportion of wood, and a higher proportion of other items on the diet of small larvae, could indicate that juvenile stages might be feeding in an omnivorous way, consuming less wood and fungi and more animal matter than larger larvae. These results are comparable to those found by Azzouz & Sanchez-Ortega (2000), who studied the stomach contents of nine Plecoptera species of Africa. These authors found that different larval instars could feed in a similar way but with some differences, increasing or reducing the percentage of the different fractions of vegetables, fine and coarse organic matter, and invertebrates as the energy requirements changes. The only two studies of feeding habits in Patagonian Plecoptera (Díaz Villanueva & Albariño, 1999; Epele & Miserendino, 2011), conducted on species belonging to the genus Notoperla (N. archiplatae (Illies), N. fasciata and N. magnaspina) show a phytophagous diet specialized in diatoms of the periphyton. Their mandibles are adapted for a scraping-grinding function, possessing an apical wide scoop-like tooth, sharp teeth and reduced molar area, the galea possesses an apical dense crown of bristles and the laciniae apical rounded teeth.
Stewart & Stark (1993) mention that even when each species can be included in a feeding category according to the major fraction represented on the intestinal contents, a species cannot be confined exclusively to one category alone, since the presence of different types of food indicates that no single feeding mechanism is involved. Cummins (1973) also mentions that the general trend among aquatic insects is to be polyphagous without specialization in food intake.
Log and fall were positively selected by D. samali as type habitat. Regarding log habitat, organic substrata, such as submerged woody debris, are an important habitat for insects (Ernst & Stewart, 1986); insects that live in such substrata use it as a stable substrate, and those who directly feed on wood are the exception (Merritt et al., 2008). In the case of D. samali, the positive selection of log habitat can be explained by its feeding behavior, the gut contents are composed mainly of wood and fungi that grow in rotting wood; whether wood is digested or just ingested in order to include fungi, is not known. This could also explain the selection of fall, because loose woody material was the highest fraction, even though significant differences were not observed between the amount of woody material and type habitat. Additionally, fall habitat was composed by small to large cobbles (70-200 mm), with wide interstitial space that could provide refuge from current and predators (Hauer & Resh, 1996). On the other hand, runs were composed of small-medium size gravel (mainly 10-60 mm) that could prevent that large stoneflies (20-32 mm) properly use interstitial space, either to have access to woody material accumulated or refuge from current and predators (Brown & Brussock, 1991). The dimension of interstitial space could be important for stoneflies habitat selection, and undoubtedly to control access to mineral and vegetable substrate (Stewart & Stark, 1993). The habitat log could also provide refuge from current and predators, as all the specimens hang on the inferior side of the logs, between the wood and the river bottom (Allan, 1995). It must be addressed that, as explained in materials and methods, log habitat was composed of logs located at runs.
Diamphipnopsis samali was not present in pools, in accordance with other studies where insects in general are poorly represented in this habitat (e.g. Doeg & Milledge, 1991; Buffagni & Comin, 2000; Velásquez & Miserendino, 2003a, b). This can be explained by the instability of substrata and higher predation by trout (Allan, 1995). Studies in Patagonian plecopterans show that they are virtually absent in pools with sand substrata, but they are abundant and diverse in runs-riffles with cobble and pebble substrate (Miserendino, 2000; Velásquez & Miserendino, 2003a, b). Thus, the negative selection of runs observed in this study is somehow an exception. In conclusion, spatial distribution in D. samali could be affected by the kind of substrata and the kind of organic matter, with a negative selection for run and pool habitat, which could be explained by the size of the interstitial space, not big enough to provide access for food and shelter for these big insects.
ACKNOWLEDGEMENTS
The authors would like to thank Dr. Laura Miserendino and Dr. Cecilia Brand for the critical reading of the manuscript and language review. This work was partially funded by CONICET.
LITERATURE CITED
1. ALBARIÑO, R. J. 2001. Food habit and mouthpart morphology of a South Andes population of Klapopteryx kuscheli (Plecoptera: Austroperlidae). Aquatic Insects 23: 171-181. [ Links ]
2. ALBARIÑO, R. & E. BALSEIRO. 1998. Larval size and leaf conditioning in the breakdown of Nothofagus pumilio leaves by Klapopteryx kuscheli (Insecta, Plecoptera) in a South Andean stream. International Review of Hydrobiology 83: 397-404. [ Links ]
3. ALBARIÑO, R. & E. BALSEIRO. 2002. Leaf litter breakdown in Patagonian streams: native versus exotic trees and the effect of invertebrate size. Aquatic Conservation: Marine and Freshwater Ecosystem 12: 181-192. [ Links ]
4. ALLAN, D. 1995. Stream Ecology. Structure and Function of Running Waters. Chapman & Hall. [ Links ]
5. AZZOUZ, M. & A. SANCHEZ-ORTEGA. 2000. Feeding of the nymphs of nine stonefly species (Insecta: Plecoptera) from North Africa (Rif Mountains, Morocco). Zoologica Baetica 11: 35-50. [ Links ]
6. BENKE, A. C., A. D. HURYN, L. A. SMOCK & J. B. WALLACE. 1999. Length-mass relationships for freshwater macro-invertebrates in North America with particular reference to the southeastern United States. Journal of the North American Benthological Society 18: 308-343. [ Links ]
7. BROWN, A. V. & P. P. BRUSSOCK. 1991. Comparison of benthic invertebrates between riffles and pools. Hydrobiologia 220: 99-108. [ Links ]
8, BUFFAGNI, A. & E. COMIN. 2000. Secondary production of benthic communities at the habitat scale as a tool to assess ecological integrity in mountain streams. Hydrobiologia 422: 183-195. [ Links ]
9. CORONATO, F. R. & DEL VALLE, H. F. 1988. Caracterización hídrica de las cuencas hidrográficas de la provincia del Chubut. Publicación Técnica. Cenpat-Conicet. Puerto Madryn. Chubut. Argentina, 183 pp. [ Links ]
10. CUMMINS, K. W. 1973. Trophic relations of aquatic insects. Annual Review of Entomology 18: 183-206. [ Links ]
11. DERKA, T., J. M. TIERNO DE FIGUEROA & I. KRNO. 2004. Life Cycle, Feeding and Production of Isoptena serricornis (Pictet, 1841) (Plecoptera, Chloroperlidae). International Review of Hydrobiology 89: 165-174. [ Links ]
12. DÍAZ VILLANUEVA, V & R. ALBARIÑO. 1999. Feeding habit of Notoperla archiplatae (Plecoptera) larvae in a North Patagonia Andean stream, Argentina. Hydrobiologia 412: 43-52. [ Links ]
13. DOEG, T. J. & G. A. MILLEDGE. 1991. Effects on experimentally increasing concentrations of suspended sediment on macroinvertebrate drift. Australian Journal of Marine and Freshwater Research 42: 519-526. [ Links ]
14. ERNST, M. R. & K. W. STEWART. 1986. Microdistribution of stoneflies (Plecoptera) in relation to organic matter in an Ozark foothills stream. Aquatic Insects 8: 237-254. [ Links ]
15. EPELE, L. B. & M. L. MISERENDINO. 2011. Life cycle, production and habitat selection of Notoperla fasciata and N. magnaspina (Plecoptera: Gripopterygidae) in a headwater Patagonian stream. Fundamental and Applied Limnology 178(3): 219-229. [ Links ]
16. FIGUEROA R., A. PALMA, V. RUIZ & X. NIELL. 2007 Análisis comparativo de índices bióticos utilizados en la evaluación de la calidad de las aguas en un río mediterráneo de Chile: río Chillán, VIII Región. Revista Chilena de Historia Natural 80: 225-242. [ Links ]
17. GOTELLI, N. J. & ELLISON A. M. 2004. A primer of ecological statistics. Sinauer Associates Inc. publishers. Sunderland, Massachusetts. [ Links ]
18. HAUER, F. R. & V. H. RESH. 1996. Benthic Macroinvertebrates. In: Hauer F. R & Lamberti G. A. (eds.), Methods in Stream Ecology, Academy Press, New York, pp. 229-269. [ Links ]
19. HYNES, H. B. N. 1976. Biology of Plecoptera. Annual Review of Entomology 21: 135-153. [ Links ]
20. HOLLMANN, M. E. T. & M. L. MISERENDINO. 2008. Life history and emergence patterns of stoneflies species in mountain streams of the Futaleufú basin, Patagonia (Argentina). Annales de Limnologie 44(2): 135-144. [ Links ]
21. ILLIES, J. 1960. Archiperlaria, eine neue Unterordnung der Plecopteren. Beiträge zur Entomologie 10: 661-697. [ Links ]
22. ILLIES, J. 1963. Revision der südamerikanische Gripopterygidae (Plecoptera). Mitteilungen der Schweizerischen Entomologischen Gesellschaft 36: 145-248. [ Links ]
23. KRZYSZTOF, M. J. & K. W. STEWART. 1987. Annual Stonefly (Plecotera) production in a second order Oklahoma Ozark stream. Journal of North American Benthological Society 6(1): 26-34. [ Links ]
24. LAMBERTI, G. A. & J. W. MOORE. 1984. Aquatic Insects and primary consumers. In: Resh V. H., Rosenberg D. M. (eds.), The Ecology of Aquatic Insects, Praeger Publishers, New York, pp. 164-195. [ Links ]
25. MASSI, C. I. 2006. Organización funcional de la comunidad de macroinvertebrados de ríos cordilleranos sometidos a distintos usos de la tierra en el noroeste de Chubut. Seminario de Licenciatura para acceder al Título de Licenciada en Ciencia Biológicas. Universidad Nacional de la Patagonia San Juan Bosco. Facultad de Ciencias Naturales. 58 pp. [ Links ]
26. MC LELLAN, I. D., M. L. MISERENDINO & M. E. T. HOLLMANN. 2006. Two new species of Notoperla (Plecoptera: Gripopterygidae) and a redescription of Notoperlopsis femina Illies. Zootaxa 1140: 63-68. [ Links ]
27. MERRITT, R. W., K. W. CUMMINS & M. B. BERG. 2008. An Introduction to the Aquatic Insects of North America. 4th edition. Kendall/Hunt Publishing Company. [ Links ]
28. MISERENDINO, M. L. 2000. Distribución y variación estacional de Plecoptera en un sistema fluvial de la cordillera patagónica. Revista de la Sociedad Entomológica Argentina 59: 149-160. [ Links ]
29. MISERENDINO, M. L. 2006. Seasonal and spatial distribution of stoneflies in the Chubut River (Patagonia, Argentina). Hydrobiologia 568: 263-274. [ Links ]
30. MISERENDINO, M. L. & C. I. MASSI. 2010. The effects of land use on environmental features and functional organization of macroinvertebrate communities in Patagonian low order streams. Ecological Indicators 10: 311-319. [ Links ]
31. MISERENDINO, M. L., R. CASAUX, M. ARCHANGELSKY, C. DI PRINZIO, C. BRAND & A. M. KUTSCHKER. 2011. Assessing land-use effects on water quality, in-stream habitat, riparian ecosystems and biodiversity in Patagonian northwest streams. The Science of the Total Environment 409(3): 612-624. [ Links ]
32. PALMA, A & R. FIGUEROA. 2008. Latitudinal diversity of Plecoptera (Insecta) on local and global scales. Illiesia 4: 81-90. [ Links ]
33. PALMER, C., J. O`KEEFE, A. PALMER, T. DUNNE & S. RADLOFF. 1993. Macroinvertebrate functional feeding groups in the middle and lower reaches of the Buffalo River, Eastern Cape, South Africa. I. Dietary variability. Freshwater Biology 29: 441-453. [ Links ]
34. PAUL, M. & J. MEYER. 2001. Stream in the urban landscape. Annual Review of Ecology and Systematics 32: 333-365. [ Links ]
35. PECKARSKY, B. L. 1984. Predator-prey interactions among aquatic insects. In: Resh V. H., Rosemberg, D. M. (eds.), The Ecology of Aquatic Insects, Praeger Publishers, New York, pp. 255-288. [ Links ]
36. PESSACQ, P. 2009. Estado de conocimiento del orden Plecoptera en la Patagonia. Revista de la Sociedad Entomológica Argentina 68(1-2): 169-175. [ Links ]
37. RICHARDSON, J. S. 2001. Life cycle and phenology of common detritivores from a temperate rain forest stream. Hydrobiologia 455: 87-95. [ Links ]
38. ROSENBERG, D. M. & V. H. RESH. 1993. Introduction to fresh-water biomonitoring and benthic macroinvertebrates. In: Rosenberg D. M., Resh V. H. (eds.), Freshwater biomonitoring and benthic macroinvertebrates, Chapman and Hall, New York, pp. 1-9. [ Links ]
39. SEPHTON, D. H. & H. B. HYNES. 1983. Food and Mouthpart Morphologyof the Nymphs of Several Australian Plecoptera. Australian Journal of Marine and Freshwater Research 34: 893-908. [ Links ]
40.SHORT, R. A. & J. V. WARD. 1980. Life cycle and production of Skwala parallela (Frison) (Plecoptera: Perlodidae) in a Colorado montane stream. Hydrobiologia 69: 173-175. [ Links ]
41. SMOCK, L. A. 1980. Relationships between body size and biomass of aquatic insects. Freshwater Biology 10: 375-383. [ Links ]
42. SNELLEN, R. K. & K. W. STEWART. 1979. The life cycle of Perlesta placida (Plecoptera: Perlidae) in an intermittent stream in northern Texas. Annals of the Entomological Society of America 72: 659-666. [ Links ]
43. STEWART, K. W. & B. P. STARK. 1993. Nymphs of North American Stonefly Genera (Plecoptera). The Tomas Say Foundation,Vol XII. Allen Press, Lawrence. [ Links ]
44. STEWART, K. W. & B. P. STARK. 2002. Nymphs of North American Stonefly genera (Plecoptera) 2nd edition. The Caddis Press, Columbus, Ohio. [ Links ]
45. VALDOVINOS, C., A. KIESSLING, M. MARDONES, C. MOYA, A. OYANEDEL, J. SALVO, V. OLMOS & O. PARRA. 2010. Distribución de macroinvertebrados (Plecoptera y Aeglidae) en ecosistemas fluviales de la Patagonia chilena: ¿Muestran señales biológicas de la evolución geomorfológica postglacial? Revista Chilena de Historia Natural 83: 267-287 [ Links ]
46. VELÁSQUEZ, S. M. & M. L. MISERENDINO. 2003a. Habitat type and macroinvertebrate assemblages in low order Patagonian streams. Archiv für Hydrobiologie 158: 461-483. [ Links ]
47. VELÁSQUEZ, S. M. & M. L. MISERENDINO. 2003b. Análisis de la materia orgánica alóctona y organización funcional de macroinvertebrados en relación con el tipo de hábitat en ríos de montaña de Patagonia. Ecología Austral 13: 67-82. [ Links ]
48. VERA, A. 2006. Una nueva especie de Gripopterygidae de Chile, Potamoperla testacea n. sp. (Insecta: Plecoptera). Gayana 70(2): 168-175. [ Links ]
49. VERA, A. 2008. Una nueva especie de Chilenoperla (Plecoptera: Gripopterygidae) y las consecuencias taxonómicas del descubrimiento de su ninfa. Gayana 72(2): 144-156. [ Links ]
50. WARD, J. V. 1989. The four dimensional nature of lotic ecosystem. Journal of North American Benthological Society 8(1): 2-8. [ Links ]
51. WHILES, M. R. & J. B. WALLACE. 1997. Leaf litter decomposition and macroinvertebrate communities in head water streams draining pine and hardwood catchments. Hydrobiologia 353: 107-119. [ Links ]
52. ZWICK, P. 1980. Plecoptera (Steinfliegen). Handbuch der Zoologie, 26. Walter de Gruyter. Berlin. [ Links ]













