Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Revista de la Sociedad Entomológica Argentina
versión impresa ISSN 0373-5680versión On-line ISSN 1851-7471
Rev. Soc. Entomol. Argent. vol.78 no.3 La Plata set. 2019
Artículo-Article
Effect of gibberellic acid on the queen cell production and queen performance in colonies of Apis mellifera L. (Hymenoptera: Apidae)
Efecto del ácido giberélico en la producción de celdas reales y el rendimiento de la reina en colonias de Apis mellifera L. (Hymenoptera: Apidae)
SZAWARSKI, Nicolás1,2,*, REVAINERA, Pablo1,2, MITTON, Giulia1,2, GIMENEZ-MARTÍNEZ, Pablo1,3, BRASESCO, Constanza1,2, RAMOS, Facundo1, DE FEUDIS, Leonardo1, FUSELLI, Sandra1,4, EGUARAS, Martin1,2, LAMATTINA, Lorenzo2,5 & MAGGI, Matías1,2
1 Centro de Investigación en Abejas Sociales (CIAS). Instituto de Investigaciones en Producción Sanidad y Ambiente (IIPROSAM). Universidad Nacional de Mar del Plata (UNMdP). Mar del Plata, Argentina.
* E-mail: n.szawarski@gmail.com
2 Consejo Nacional de Investigaciones Científicas y Técnicas. Buenos Aires. Argentina.
3 Agencia Nacional de Promoción Científica y Tecnológica. Buenos Aires, Argentina.
4 Comisión Investigaciones Científicas de la Provincia de Buenos Aires (CICPBA). La Plata, Argentina.
5 Instituto de Investigaciones Biológicas, Universidad Nacional de Mar del Plata (UNMdP). Mar del Plata, Argentina.
Received 17 - I - 2019 | Accepted 01 - VII - 2019 | Published 26 - IX - 2019 https://doi.org/10.25085/rsea.780303
ABSTRACT. For honey bees (Apis mellifera L.), the reward for pollination service is obtaining food: nectar, a sugar solution; or pollen, which contains a high percentage of proteins and lipids. There are also other floral rewards ecologically relevant for pollinators, like resins, and/or minor components, such as phytohormones and polyphenols. When rearing A. mellifera queens, several factors affect grafting success: age of the grafted larvae, design and position of the artificial queen cells, priming queen cells with royal jelly before grafting, or nutrition of the queen rearing colony. This study aimed to evaluate the effect of two phytohormones: gibberellic acid (GA) and indole acetic acid (IAA) on queen cell production, the development of hypopharyngeal glands of nurse bees, and the performance of queen bees in colonies of A. mellifera. The results indicate that GA positively infuences queens production efficiency in vertical orphaned colonies and their reproductive performances in honey bee colonies. These results support the potential of plant metabolites as dietary supplements to improve queen bee production methods and their performance in colonies as well.
KEYWORDS. Honey bee. Phytomolecules. Queen rearing. Supplemental feeding.
RESUMEN. Para la abeja melífera (Apis mellifera L.), la recompensa por la polinización es la obtención de alimento: néctar, una solución azucarada, o polen, que contiene un alto porcentaje de proteínas y lípidos. También hay otras recompensas florales ecológicamente relevantes, las resinas, y/o componentes minoritarios, como las fitohormonas y polifenoles. En la cría de reinas de A. mellifera diversos factores afectan el éxito de los traslarves: la edad de las larvas, el diseño y la posición de las celdas reales artificiales, el cebado de las cúpulas con jalea real o la alimentación de la colonia que criará las reinas. El objetivo de nuestro estudio fue evaluar el efecto de dos fitohormonas, ácido giberélico (GA) y ácido indoalacético (IAA) en colonias de A. mellifera sobre la producción de celdas reales, desarrollo de las glándulas hipofaríngeas de abejas nodrizas y desempeño de abejas reinas. Los resultados indican que el tratamiento con GA en colonias sometidas al método de semiorfandad para la cría de reinas, mejoró la eficiencia de la producción de las mismas y su rendimiento reproductivo en colonias de A. mellifera. Estos resultados apoyan el potencial de utilizar metabolitos vegetales como suplementos dietarios para mejorar los métodos de producción de abejas reinas y su desempeño en las colonias.
PALABRAS CLAVE. Abeja melífera. Cría de reinas. Fitomoléculas. Suplemento alimentario.
INTRODUCTION
In natural environments, production of new queen bees can be driven by the aging of the old queen, its poor quality, its accidental death/loss by the beekeeper when managing the colonies, or the creation of new colonies (swarming). In the parthenogenetic life cycle o f Apis mellifera, each fertilized egg can turn into a potential queen if, throughout its life, larvae are raised entirely on royal jelly secreted by the hypopharyngeal and mandibular glands of nurse bees (Deseyn & Billen, 2005; Spannhoff et al., 2011). This natural behavior allows beekeepers to rear queens from worker-laid larvae in initial stage.
Beekeepers have developed techniques to rear large numbers of queen bees for various practical purposes, such as altering certain genetic characteristics of the colonies, starting new colonies, increasing brood and honey production, etc. (Büchler et al., 2013). Therefore, several tools and methods are currently available for rearing queen bees, being the Doolittle Method the most popular among queen breeders. This method consists in grafting newly-hatched larvae (stage L1) selected from a mother colony and then introduced into the orphan colony for the production of potential queens (Büchler et al., 2013).
Grafting success is often modulated by four main factors: priming queen cells with royal jelly before grafting (Pickard & Kither, 1983), the design and position of the artificial queen cells (Al-Fattah et al., 2011), the age of the grafted larvae (Mahbobi et al., 2012) and/ or the nutrition of the queen rearing colony (mother colonies) (Gençer et al., 2000; Mahbobi et al., 2012). For example, in colonies destined to queen rearing, feeding with a mixture of vitamins or pollen dissolved in sugar syrup positively affected the cell acceptance rate of grafted larvae for queen cell production (Gençer et al., 2000). On the other hand, a good performance of a bee colony is determined by the quality of the queen, which can be evaluated based on several characteristics, such as weight, number of ovarioles, size of the spermatheca, fecundity rate (number of eggs laid perday), as well as brood quality (Kovacic & Puskadija, 2016).
Nurse bees determine the fate of sibling female larvae by controlling food quantity and quality, resulting in two distinct phenotypes: queens and workers (Sagili et al., 2018). These worker bees have hypopharyngeal and mandibular glands that produce and secrete royal jelly, a complex mixture for the growth and development of young bees (Deseyn & Billen, 2005; Spannhoff et al., 2011). It contains mainly water (~65%), crude protein (~12%), and monosaccharides (glucose and fructose) (~10%). The remainder is composed of an ether-soluble fraction of fatty acids, primarily (E)-10-hydroxy-2-decenoic acid (10HDA), a compound associated epigenetic regulation of queen-bee development (Spannhoff et al., 2011; Wang et al., 2015). For pollinators such as honey bees the reward for pollination service is, in most cases, obtaining food in the form of nectar, sugar solution, or pollen, which contains a high percentage of proteins and lipids (Chalcoff et al., 2014). There are also other less frequent foral rewards, though ecologically relevant for pollinators, like resins, and/or minor components, such as phytohormones and polyphenols (Mao et al., 2013; Negri et al., 2015; Wang et al., 2017). In recent years, focus has been placed on the study of this type of molecules which have shown to be beneficial in bee immune system (Mao et al., 2013; Negri et al., 2015; Richardson et al., 2015). This study aimed to evaluate the effect of two phytohormones: gibberellic acid (GA) and indoleacetic acid (IAA) on queen cell production, the development of hypopharyngeal glands of nurse bees, and the performance of queen bees in colonies of A. mellifera.
MATERIAL AND METHODS
Biological material and characterization of bee ecotype
Field experiments were carried out in the experimental apiary of the Social Bees Research Center located in Santa Paula, National Route 226, Km 10, Mar del Plata city (-37.9348798, -57.682817), Buenos Aires, Argentina. The experiments were conducted between September 2016 and January 2017.
Field research was carried out with a local A. mellifera ecotype (A. mellifera carnica x A. mellifera ligustica) . In order to determine bee ecotype, we performed a morphometric analysis of the experimental apiary using geometric morphometrics (Gimenez-Martínez et al., 2017). Twenty individuals were removed per colony, whose right posterior wing was individually excised. Photographs were taken using the Leica DM2500 optical microscope with a 4x magnification and DFC295 camera. Images were saved as .JPG fles for later analysis. All the software necessary for the morphometric analysis is available on the Internet (http://life.bio.sunysb.edu/morph/), and is of free use. A fle was created with the images using tpsUTIL version 1.33 (Adams et al., 2004) for their analysis. Afterward, tpsDIG2 was used to manually digitalize 16 homologous landmarks (Francoy et al., 2008). For statistical analysis of the populations, a canonical variate analysis was performed in MorphoJ software (Klingenberg, 2011). Subsequently, the statistics software package PAST (version 2.17c, http://folk.uio.no/ohammer/past) (Hammer & Harper, 2013) was used to calculate the squared Mahalanobis distance; and distances within clusters were determined with Ward's method.
Field Trials
Trial A
We worked with three colonies of A. mellifera housed in Langstroth beehives with double brood boxes (two standard hives separated by a queen excluder) for queen cells production, applying the semi-orphaned method (Laidlaw & Page, 1997). This method consists in isolating queen bees in the lower hive box and keeping in the upper box sealed and open worker bee brood. The queen bee is isolated from the box above by placing a queen excluder that prevents the queen from passing through while allowing the passage of worker bees. In the middle of the upper box, there is a graft frame that holds the plastic queen cups where the 12 to 24-hour-old larvae of worker bees are grafted. Since the queen is prevented from reaching the upper box to oviposit eggs, a vertical orphaned condition is created that stimulates nurse bees to accept grafted larvae and produce queen cells. The three colonies (so-called "starter colonies") were equally managed by the same beekeeper during the three months that Trial A lasted.
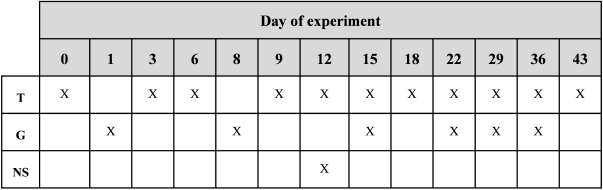
A specific treatment was assigned to each starter colony: a) starter colony 1 (i1) was the control group, and was provided with 200 ml of 66% sugar syrup; b) starter colony 2 (i2) received 200 uM of indole acetic acid (Sigma Aldrich) in 200 ml of 66% sugar syrup; c) and starter colony 3 (i3) was provided with 25 uM of gibberellic acid (Sigma Aldrich) in 200 ml of 66% sugar syrup. The start of the trial was defned as day 0 (D0). Each starter colony received 10 applications of each treatment that were called Tn, being n the number of applications and T the treatment given. The frst eight applications were made every three days and the last two on a weekly basis due to the adverse climate conditions that limited the visits to the experimental apiary (Table I).
Grafting Efficiency. The day after the frst treatment was applied (Table I), each colony received 24 grafted larvae in the upper box (semi-orphaned colony). The graft consisted in placing a 12 to 24-hour-old worker bee larva in a plastic bee cup that worked as a receptor for potential queen larvae. All the larvae used for this trial came from a fourth colony defned as the mother
colony. The next graftings were systematically carried out every seven days. A total of seven graftings of 24 larvae each were performed in each colony. Five days after each grafting, the total number of accepted larvae was recorded (Table I), defned as the number of bee cups with a sealed queen cell. Percentage was calculated according to the following formula:
Percentage grafting efficiency in = (Bee cups with sealed queen cells/24) * 100
To analyze the percentage of grafting efficiency, Kruskal Wallis trial (α = 0.05) was used.
Effect of diet on the development of the hypopharyngeal glands of nurse bees. Development of the hypopharyngeal glands is associated with the diameter of the acini (cluster of gland cells), and refects the number of proteins produced (Deseyn & Billen, 2005). A simple method positively correlates the hypopharyngeal gland acini size to the head fresh weight of worker bees (Damiani et al., 2017). To estimate the development of the hypopharyngeal glands, on day fve of treatment (13 days from the start of the trial) (Table I), nurse bees that were feeding/attending to open queen cells were sampled at each starter colony. Approximately 250 nurse bees were sampled per colony. In this way, bees that were seen to be attending to the queen were sampled. Samples were stored in a freezer (-20 ºC) until processing. Then, a sub-sample of 50 bees was randomly chosen from each sample in the laboratory. Each bee was identifed according to the treatment applied, and their head was cut with a scalpel. Heads were individually weighed in an analytical balance. The Kruskal Wallis trial (α = 0.05) was used to analyze if there were statistical differences in the head weights sampled between treatments.
Trial B
The last grafting was used to produce virgin queens fed with 25 uM gibberellic acid (GA) in 66% sugar syrup and 66% sugar syrup only (control). Nine (9) nuclei without queens of four frames each (two with sealed cells, one with open brood, and one with honey and pollen) were prepared for this purpose. They remained queenless for three days. The queen cells from the starter control colony (i1) were randomly inserted into three (3) nuclei; while those from the starter colony that had received the differential diet with 25 uM GA (i3) were randomly inserted into the other six (6) nuclei. The reason why only i3 was chosen, is that it differed in cell production with respect to the control (i1).
When the virgin queens were born, they were allowed to mate freely, and once egg laying was initiated in all the nuclei, population dynamics was registered fortnightly until one of the groups completed the brood chamber. The colony strength parameters were measured according to the subjective mode (Delaplane et al., 2013). Frames with adult bees, open brood areas, sealed brood areas, pollen areas and honey areas were recorded. The method consists in the visual estimation of the parameters defned above by two or more people as a percentage (%) of the covered area and then its conversion into units, i.e., the total number of adult bees and the number of cells covered with sealed brood, open brood, honey and pollen. Beehives were managed by the same two beekeepers during the months that Trial B lasted.
Table I. Experimental design on field showing the days of experiment: treatment application (T), grafts (G) and nurse bees sampling (NS)
Production of queen cells in starter colonies with a differential diet supplemented with gibberellic acid (GA) or indole acetic acid (IAA)
Regarding queen cell production in starter colonies with a differential diet, higher values were obtained in i3 colony supplemented with 25 uM GA (mean 78%), as compared to the control bee colonies (i1), which were provided only with a 66% sugar syrup solution (average 27%) (Kruskal-Wallis trial, p = 0.013), and to the colony supplemented with 200 uM IAA (i2) (approximated mean equal to 32%) (Kruskal-Wallis trial, p = 0.031). The i2 colony supplemented with 200 uM IAA did not show significant differences in terms of percentage of sealed queen cells with respect to control (Kruskal-Wallis trial, p = 1) (Fig. 1).
Development of the hypopharyngeal glands of worker bees that attended to the queen cells of the starter colonies i1, i2, and i3
The weight of head of the worker bees that attended to the queen larvae (indirect estimator of hypopharyngeal glands development) was higher in bees from the starter colony i3, which had received the supplemental diet with 25 uM GA, with respect to colonies i1 (Kruskal Wallis trial, p <0.001) and i2 (Kruskal Wallis trial, p <0.001). The weight of the worker bee heads from i2 colony, which received 200 uM IAA, did not show significant differences with respect to the bees from the control colony (Kruskal Wallis trial, p = 1) (Fig. 2).
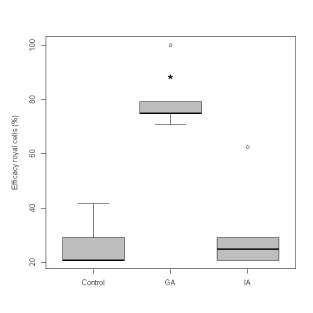
Fig. 1. Efficiency of sealed queen cells (%) in control starter colonies (sugar syrup only), supplemented with 25 uM gibberellic acid (GA) or 200 uM indole acetic acid (IA).
Asterisc (*) corresponds to the significant difference.
Development of bee colonies with queens produced in control starter colonies (i1) or supplemented with 25 uM gibberellic acid (GA) (i3)
The population dynamics of the bee colonies whose queens emerged from the colony supplemented with 25 uM GA (i3) differed with regard to control bee colonies.
Number of adult bees of the colonies with queens from mother colony i3 was higher compared with control on days 2 and 3, respectively (two-way ANOVA, p <0.05) (Fig.3 A). Number of bee brood was higher in colonies whose queens had emerged from the mother beehive i3 with regard to control colonies (ANOVA, p <0.001) (Fig. 3 B).
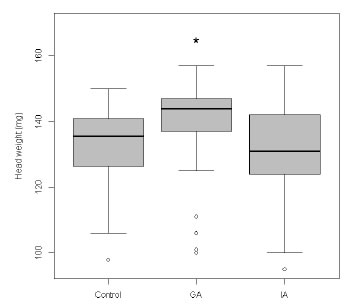
Fig. 2. Weight of head of the worker bees that attended the rearing of queens. Asterisc (*) corresponds to the significant difference.
As far as honey reserves are concerned, control colonies (queens from i1) ended (day 3) with a greater amount of honey as compared to those with queens from colony i3 (two-way ANOVA, p = 0.001) (Fig. 3 C). On the other hand, no significant differences were observed regarding pollen reserves between both groups of bee colonies (two-way ANOVA, p = 0.592) (Fig. 3 D).
DISCUSSION
To rear A. melliferaqueens, the number of accepted larvae depends on different factors such as quality, strength, and stage of development of the starter colonies, age of workers, age of grafted larvae, regulation of humidity, temperature in the colony and food supply (nectar fow, pollen and/or supplemental feeding) to the colonies that attend to the grafted queen cells (Büchler et al., 2013). Several studies have suggested that the supplemental feeding of the starter colonies is a key factor on the acceptance rate of the grafted larvae (Krol et al., 1992; Stace & White, 1994; Gençer et al., 2000; Büchler et al., 2013). The acceptance of queen cells after grafting and cell production per colony increased when queen rearing colonies were fed a defnite amount of iso-leucine in sugar syrup (Stace & White, 1994). In another study, a dietary supplement based on a mixture of vitamins or pollen (in sugar syrup) was tested in starter colonies, obtaining higher acceptance rates of grafted larvae for queen rearing (Gençer et al., 2000).
In our study, the frst objective was to evaluate the effect of two phytohormones found naturally in honey (Wang et al., 2017): gibberellic acid (GA) and indoleacetic acid (IAA) on queen cell production in colonies of A. mellifera.With this objective in mind, four colonies (one mother colony and three starter colonies) that had been analyzed by geometric morphometrics to identify bee ecotype were selected. Results indicated that the four colonies belonged to the same ecotype, a hybrid between A. m. ligustica and A. m. carnica. The production and quality of the queens (weight, egg laying capacity, etc.) are strongly infuenced by genetic factors (Büchler et al., 2013). Therefore, the fact that the ecotype of the bee colonies had previously been determined was important to guarantee uniform conditions. The breeding colony that received 25 uM GA as food supplement (mean = 78%) yielded the greatest efficiency in queen cell production as compared to the control starter colonies (mean = 27%) supplemented with 200 uM IAA (average = 32%).
There is no literature available on the effect of supplemental feeding with plant hormones on the acceptance rate and production of queen cells. There is the possibility that the concentration of IAA applied was not sufficient to visualize an effect at colony level. Therefore, further studies should be conducted with different concentrations of IAA as dietary supplement. The greater efficiency in queen cell production of the colony supplemented with GA could be explained by the fact that the queen-destined larvae received a better-quality diet than the other colonies did. This could be supported by another parameter also measured in the starter colonies for queen rearing: the head weight of the worker bees that attended to the young larvae. Heads of the nurse bees from the colony that received 25 uM GA were heavier, indirectly indicating a larger size of the hypopharyngeal glands (Damiani et al., 2017) of these bees compared to the bees coming from control colony to which 200 uM IAA were supplied. One hypothesis holds that the greater size of the hypopharyngeal glands is related to the quality/quantity of royal jelly, which enhances the acceptance rate of the queen-destined larvae. Several works have dealt with dietary vitamin E supplementation for queen rearing whose conclusions could support the above-raised hypothesis. For instance, weight of the pharyngeal glands of worker bees fed with vitamin E increased by more than 47% as compared to controls (only fed with syrup) (Cheng et al., 1993). A similar study proved that vitamin supplementation not only improved the percentage of acceptance of queen cells, but also increased the royal jelly production of the workers that attended to the larvae with respect to the controls (Sahinler et al., 2005). A recent work showed that the nutritional status of the larvae is meticulously assessed and used by workers in the selection of larvae for queen rearing (Sagili et al., 2018). Authors performed a series of experiments manipulating the nutritional status of one-day-old larvae by depriving them of brood food for a four-hour period. They observed that worker bees selected larvae for rearing queens from nutritionally non-deprived larvae during that time interval. This demonstrates that honey bees perceive the nutritional status of the larvae, and use that information when selecting those that will be destined for queen rearing.

Fig. 3. Development of bee colonies with queens produced in starter hives control and supplemented with 25 uM gibberellic acid (GA). A. number of adult bees; B. number of cells with breeding; C. number of cells with honey; D. number of cells with pollen, for days 1-2-3 corresponding to the three survey points. Significant differences are indicated by letters
Studies report that a supplemental diet in starter colonies improves the queen quality (Mahbobi et al., 2012) and heavier queens performed better in the colony, which is strongly linked to greater brood production (Gençer et al., 2000). For the second part of our study, we selected the queens reared in the colonies that had received 25 uM GA supplemental feeding (i3), due to the better responses obtained in the variables studied, along with the queens from the control colony (only fed with sugar syrup). Then, nuclei were organized with the selected queens standardized in population of adult bees, brood, and reserve in order to study queen performance in these new bee colonies. The population dynamics of the beehives with queens emerging from the starter colony and supplemented with GA showed significant differences relative to control colonies, mainly in terms of the number of adult bees and frames covered with brood. In this way, it was demonstrated that the performance of queens produced in colonies provided with GA was better than that of queens produced in control colonies.
The results indicate that gibberellic acid (GA) in colonies of A. melliferaunder a vertical orphaned colony method for queen rearing, would infuence queen production efficiency. This could be related to the differences found in the head weight of the nurse bees,
which could be ascribed to a greater development of the hypopharyngeal glands. We believe that the evidence presented here supports and reinforces the potential of plant metabolites as dietary supplements to improve queen bee production methods and their performance in honey bee colonies.
ACKNOWLEDGEMENTS
The authors would like to thank the National University of Mar del Plata (UNMdP), the Agencia Nacional de Promoción Científica y T ecnológica (ANPCYT) and the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) for fnancial support.
LITERATURE CITED
Adams, D.C., Rohlf, F .J., & Slice, D.E. (2004) Geometric morphometrics: Ten years of progress following the 'revolution'. Italian Journal of Zoology, 71, 5-16. [ Links ]
Al-Fattah, M.A.A., Mazeed, A.M., & Al-Hady, N.A. (2011) Quality and quantity of honeybee queens as affected by the number and distribution of queen cells within queen rearing colonies. Journal of Apicultural Science, 55(2), 31-43. [ Links ]
Büchler, R., Andonov, S., Bienefeld, K., Costa, C., Hatjina, F., Kezić, N., Kryger, P., Spivak, M., Uzunov, A., & Wilde, J. (2013) Standard methods for rearing and selection of Apis mellifera queens. Coloss beebook, Volume 1: standard methods for Apis mellifera research. Journal of Apicultural Research, 52(1). [ Links ]
SZAWARSKI, N. et al. Gibberellic acid effect on A. melliferaqueens rearing [ Links ]
Chalcoff, V.R., Morales, C.L., Aizen, M.A., Sasal, Y., Rovere, A.E., Sabatino, M., Quinteros, C., & Tadey, M. (2014) Interacciones plantas-animal, la polinización. Ecología e Historia Natural de la Patagonia Andina. Un cuarto de siglo de investigación en biogeografía, ecologíayconservación (ed. Raffaele, E., Torres Curth, M., Morales, C.L., & Kitzberger, T.), pp. 113-132. Fundación de Historia Natural Félix de Azara, C.A.B.A. [ Links ]
Cheng, C., Zhang, X., Shen, G., & Li, H. (1993) The effect of feeding syrup with vitamin E on the development of the pharyngeal gland of the worker bee. In: Abstracts book of the 33rd International Apicultural Congress of Apimondia,1993, Beijing, China. [ Links ]
Damiani, N., Porrini, M., Lancia, J.P., Álvarez, E., Garrido, M., Dominguez, E., Gende, L., & Eguaras, M. (2017) Effect of propolis oral intake on physiological condition of young worker honey bees, Apis melliferaL. Journal of Apicultural Science, 61(2), 193-202. [ Links ]
Delaplane, K., Steen, J., & Guzman-Novoa, E. (2013) Standard methods for estimating strength parameters of Apis mellifera colonies. Journal of Apicultural Research, 52(1), 1-12. [ Links ]
Deseyn, J., & Billen. J. (2005) Age-dependent morphology and ultrastructure of the hypopharyngeal gland of Apis mellifera workers (Hymenoptera, Apidae). Apidologie, 36(1), 49-57. [ Links ]
Francoy, T.M., Wittmann, D., Drauschke, M., Müller, S., Steinhage, V., Bezerra-Laure, M.A.F., De Jong, D., & Gonçalves, L.S. (2008) Identification of Africanized honey bees through wing morphometrics: two fast and efficient procedures. Apidologie, 39, 488-494.
Gençer, H.V., Shah, S.Q., & Firatli, Ç. (2000) Effects of supplemental feeding of queen rearing colonies and larval age on the acceptance of grafted larvae and queen traits. Pakistan Journal of Biological Sciences, 3(8), 1319-1322.
Gimenez-Martínez, P., Mendoza, Y., Invenizzi, C., Fuselli, S., Salces, R., Iriarte, P., & Maggi, M. (2017) Morphometric correlation between Apis mellifera morphotypes (Hymenoptera) and Varroa destructor(Acari) from Uruguay. Journal of Apicultural Research, 56(2), 122-129.
Hammer, Q., & Harper, D.A.T. (2013) PAST: Paleontological Statistics software package for education and data analysis. Paleontologia Electronica, 4(1), 9.
Klingenberg, C.P. (2011) MorphoJ: an integrated software package for geometric morphometrics. Molecular Ecology Resources, 11, 353-357.
Kovacic, M., & Puskadija, Z. (2016) Effect of queen cell preparation on larvae acceptance in starter honeybee colonies. In: Proceedings of 5th International Conference "Valis Aurea" focus on research and innovation, 2016, Pozega, Croatia. Pp: 229-233.
Krol, A., Hartwig A., & Topolska, G. (1992) Quality of queens reared in colonies receiving sugar supplemented with vitamin B1. Pszczelnicze Zeszyty Naukowe, 36, 32-40.
Laidlaw, H.H., & Page, R.E. (1997) Queen rearing and bee breeding.Wicwas Press, New York, USA.
Mahbobi, A., Farshineh-Adl, M., Woyke, J., & Abbasi, S. (2012) Effects of the Age of Grafted Larvae and the Effects of Supplemental Feeding on Some Morphological Characteristics of Iranian Queen Honey Bees (Apis mellifera meda Skorikov, 1929). Journal of Apicultural Science, 56(1), 93-98.
Mao, W., Schuler, M.A., & Berenbaum, M.R. (2013) Honey constituents up-regulate detoxification and immunity genes in the western honey bee Apis mellifera. Proceedings of the National Academy of Sciences, 110, 8842-8846.
Negri, P., Maggi, M., Ramirez, L., De Feudis, L., Szawarski, N., Quintana, S., Eguaras, M., & Lamattina, L. (2015) Abscisic acid enhances the immune response in Apis melliferaand contributes to the colony ftness. Apidologie, 46, 542-557.
Pickard, R.S., & Kither, G.Y. (1983) Acceptance of transplanted worker larvae by queen-cell starter colonies. Journal of Apicultural Research, 22(3), 169-174.
Richardson, L.L., Adler, L.S., Leonard, A.S., Andicoechea, J., Regan, K.H., Anthony W.E., Manson, J.S., & Irwin, R.E. (2015) Secondary metabolites in foral nectar reduce parasite infections in bumblebees. Proceedings Royal Society B: Biological Sciences, 282, 20142471.
Sagili, R., Metz, B., Lucas, H., Chakrabarti, P., & Breece, C. (2018) Honey bees consider larval nutritional status rather than genetic relatedness when selecting larvae for emergency queen rearing. Scientific Reports, 8, 7679.
Sahinler, N., Gül, A., & Şahin, A. (2005) Vitamin E supplement in honey bee colonies to increase cell acceptance rate and royal jelly production. Journal of Apicultural Research, 44(2), 58-60.
Spannhoff, A., Kim, Y.K., Raynal, N., Gharibyan, V., Su, M.B., Zhou, Y.Y., Li, J., Castellano, S., Sbardella, G., et al (2011) Histone deacetylase inhibitor activity in royal jelly might facilitate caste switching in bees. European Molecular Biology Organization, 12(3), 238-243.
Stace, P., & White, E. (1994) The use of Isoleucine as a supplement feed for honey bees (Apis mellifera) in Australia. Australasian Beekeeper, 96, 159-161.
Wang, Y., Ma, L., Zhang, W., Cui, X., Wang, H., & Xu, B. (2015) Comparison of the nutrient composition of royal jelly and worker jelly of honey bees (Apis mellifera). Apidologie, 47(1), 48-56.
Wang, Q., Cai, W.J., Yu, L., Ding, J., & Feng, Y.Q. (2017) Comprehensive Profling of Phytohormones in Honey by Sequential Liquid-Liquid Extraction Coupled with Liquid Chromatography-Mass Spectrometry. Journal of agricultural and food chemistry, 65(3), 575-585.














