The genus Meromacrus Rondani (Syrphidae: Eristalinae) has 44 species from the Southern United States to Northern Argentina and Chile (Ricarte et al., 2020; Barahona-Segovia et al., 2021). Adults visit flowers, for example, of Sapium glandulosum (L.) Morong (Euphorbiaceae) (Blatch et al., 2003) and mango, Mangifera indica L. (Anacardiaceae) (Jirón & Hedstrom, 1985; Blatch etal., 2003), pollinating flowers of this latter species (Jirón & Hedstrom, 1985; Castillo-Carrillo, 2013) as well as those of Miconia tonduziiCogn. var. tonduzii (Melastomataceae) (Kriebel & Zumbado, 2014). The above-mentioned flower visitation data come from observations of Meromacrus melansoni Blatch in Blatch et al. (2003), the only species for which there are data available at this regard.
Larvae of Meromacrus are saprophagous in various breeding sites of plants, from decaying cacti to tree rot-holes, where they may coexist with other congeneric larvae or even with those of other eristalines (Blatch et al., 2003; Pérez-Bañón et al., 2003; Ricarte et al., 2020). The only evidence of Meromacrus breeding in decaying cacti is that of Meromacrus cactorum Ricarte et al. in Espostoa melanostele (Cactaceae), from an arid cactus-dominated habitat of Peru (Ricarte et al., 2020).

Fig. 1: Meromacrus cactorum Ricarte et al. a. female, lateral view. b. female, dorsal view. c. female visiting flower of Browningia candelaris. d. habitat, near Parca village, Tarapacá region, Chile. Scale bars = 1 mm
The presence of Meromacrus in Chile has just been confirmed with specimens of Meromacrus laconicus (Walker) from fruit croplands of the transversal valleys (Barahona-Segovia et al., 2021). Recent examination of museum specimens, personal observations of Rodrigo Barahona-Segovia (RBS), and ongoing studies on the pollination of Browningia candelaris (Meyen) Britton & Rose (Cactaceae) (Humana et al., unpublished data) have revealed the presence of a second Meromacrus species from Chile, M. cactorum (Fig. 1a-b). The general aim of this work is to improve the knowledge on the recently described M. cactorum by providing new faunistic and biological data from Chile.
Diagnosis. Body coloration black. Antenna orange with pedicelum slightly longer than flagellomeres. Face yellow with a black vitta. Occiput with yellow pilosity. Transverse sutures, notopleuron, posterior anepimeron, and posterior margin with yellow pilosity. Posterior margin with two maculae with sparse yellow pilosity. Wings hyaline. Metafemur swollen as broad as the width of tergum 4. Metatibia curved. Abdomen enlarged; in males with scarce yellow pilosity near anterior margin on tergum 2 and 3. Females with maculae more notorious in tergites 2, 3, and 4, each near the anterior margin.
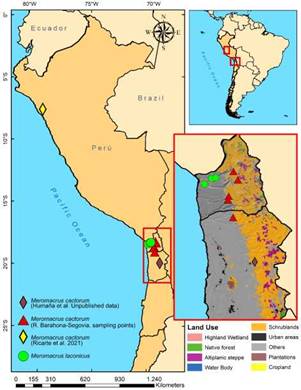
Examined material. CHILE: Arica y Parinacota: 1 $, Socoroma, -18.267752 S, -69.597317 W, 2,128 m, 28.iii.2018, RBS leg. (133SYR/PCRBS); 2 ££, Timar, -18.750301 S, -69.69506 W, 2,362 m, 15.x.1976, N. Hichins leg. (01SYR/UTA, 02SYR/UTA); 1 & and 1 ¦?, same locality as latter 2 ££, 16.viii.1976, G. Díaz P. leg. (03SYR/UTA, 04SYR/UTA); 1 ¦?, same locality as latter two specimens, 6.xi.1980, 1,800 m, N. Hichins leg. (125SYR/PCRBS); 1 ¦?, Huañacahua, -18.819161 S, -69.711639 W, 6.xii.1972, A. Aguilera leg. (05SYR/UTA). Tarapacá: 1 ¦?, Miñe-Miñe to Miñita, -19.163972 S, -69.608156 W, 2,100 m, 26.ii.1976, Héctor Vargas leg. (06SYR/UTA); near Parca village, Panoya ravine, -20.028240 S, -69.199947 W, 2,855 m, June-July 2019; acronyms: PCRBS (personal collection of RBS), UTA (Universidad de Tarapacá) (Fig. 2).
Meromacrus cactorum was observed and collected (with hand net) along random 50 m transects in two different scrubland habitats: (1) inland spiny tropical forest dominated by Geoffroea decorticans (Gillies ex Hook. & Arn.) Burkart and Prosopis alba Griseb. (Fabaceae), typical of deep ravines, from the see level were recorded mainly in ravines at intermediate altitudes with dominant xerophytic vegetation, there were also some records in the tropical Andean scrubs, at higher altitudes (above 3,000 m). The reported range of habitats suggests that adults might be more generalist in their preferred environments than expected, having the option to exploit a wider range of floral resources.
The flower visits reported for B. candelaris indícate that M. cactorum may have a role in the pollination of this vulnerable cactus [VU B1ab(iii) + 2ab(iii), DS41/ MMA2011; (MMA, 2020)], which is threatened by illegal logging (González & Molina, 2017; Humaña et al., unpublished data). In addition, M. cactorum was also reported from fruit croplands of rural areas, where visits to flowers of some cultivated plants were also observed (e. g. prickly pear). up to 1,500 m, and heavily influenced by agricultural activity; (2) Andean tropical forest dominated by B. candelaris and Corryocactus brevistylus (K. Schum. ex Vaupel) Britton & Rose (Cactaceae), between 2,000-2,800 m (Fig. 1d; González & Molina 2017). Flower visitations were reported from highlands with low Andean tropical scrubs of Parastrephia spp. (Asteraceace), 2,500-3,500 m (González & Molina, 2017).
Two females of M. cactorum were observed on flowers of prickly pear, Opuntia ficus-indica (L.) Mill. (Cactaceae) during 43-75 sec, in Socoroma village. A third female was observed on three different flowers of Cumulopuntia sphaerica (C. F Forst.) E. F Anderson (Cactaceae) near Pachama. For this third female, duration of the visit at each individual flower was 34 sec, 42 sec and 12 sec, respectively. Some other specimens were observed visiting 2-3 flowers of two different individuals of B. candelaris cacti (each cactus had 8-10 flowers available). The observed MI. cactorum entered the flowers and spent a few seconds inside, most likely pollinating (Fig. 1c).
The new records of MI. cactorum from Chile extend its known range for more than 1,500 km (in straight line) further southeast of the type locality in Peru (Ricarte et al., 2020; Fig. 2), indicating that it might be present all along the Peruvian coast. Although Chilean specimens
The apparently large distribution of M. cactorum, together with its broad spectrum of flowers visited, make the network of interactions of this syrphid with plants far to be fully understood. To which extent the pollination of certain plants, either wild or cultivated, depends on M. cactorum is another important aspect to be tested in future studies.
ACKNOWLEDGMENTS
We thank Dante Bobadilla and Héctor Vargas, as well as Hugo Benítez for giving us access to the Universidad de Tarapacá (UTA). RBS thanks Vicente Valdés Guzmán for the lab photos, the Agencia Nacional de Investigación y Desarrollo (ANID) for his PhD doctoral scholarship (CONICYT 21160404) and postdoctoral grant ANID-FONDECYT 3200817, as well as Instituto de Ecología y Biodiversidad de Chile (IEB), for the grant FB170008. Antonio Ricarte’s position at the University of Alicante is funded by the ‘Vicerrectorado de Investigación y Transferencia de Conocimiento’ (Ref. UATAL05).












 uBio
uBio