Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Agriscientia
versión On-line ISSN 1668-298X
Agriscientia v.24 n.2 Córdoba jul./dic. 2007
Vertical variation of atrazine mineralization capacity in soils
Hang, S.; S. Houot and E. Barriuso
Hang, S. Facultad de Ciencias Agropecuarias U. N. C., CC 509, 5000 Córdoba, Argentina. S. Houot and E. Barriuso. National Institute of Agronomical Research, Environment and Arable Crops, BP 01, 78850 Thiverval-Grignon, Francia. Correspondence to: shang@agro.uncor.edu
SUMMARY
The variation of atrazine mineralization capacity was investigated in two soil profiles selected with (adapted soil) and without (non-adapted soil) atrazine application history in Córdoba, Argentina. Atrazine evolution was characterized by balance of 14C-U-ring atrazine radioactivity between mineralized, extractable and non-extractable fractions by means of incubations under laboratory conditions in two soil situations: with dry heat treatment (T105) and without heat treatment (Control). The strong decrease of atrazine mineralization after microbial inactivation during all the incubation period in adapted soil suggests that the atrazine degraders were distributed throughout the entire adapted soil profile and were responsible for atrazine mineralization in subsurface horizons. In the non-adapted soil profile, the cometabolic process of mineralizing atrazine was corroborated. The atrazine mineralization capacity of subsurface horizons, where herbicide accumulation increases, should be considered and stimulated as a mechanism for the natural bioremediation of soils with frequent atrazine application.
Key words: Atrazine mineralization; Adapted sub-soil; Microbial inactivation; Bioremediation.
Hang, S.; S. Houot y E. Barriuso, 2007. Variación vertical de la capacidad de mineralización de atrazina en suelos. Agriscientia XXIV (2): 87-95
RESUMEN
Se estudió la variación de la capacidad de mineralización de atrazina en dos suelos de la provincia de Córdoba, Argentina. Se seleccionaron dos perfiles de suelos con (suelo adaptado) y sin (suelo no adaptado) historia de aplicación de atrazina. La evolución de atrazina fue caracterizada mediante el balance de la radioactividad del 14C del anillo triazínico identificando las fracciones mineralizada, extractable y no extractable, durante incubaciones en condiciones de laboratorio con suelo tratado con calor (T105) y sin él (Control). La disminución de la capacidad de degradación de atrazina después de la inactivación microbiana y la limitada recuperación de esa capacidad observada en el perfil de suelo adaptado sugirió que los microorganismos degradadores de atrazina estarían distribuidos en todo el perfil siendo los responsables de la mineralización de atrazina en los horizontes subsuperficiales. Contrariamente, en el perfil de suelo no adaptado, la recuperación de la mineralización de atrazina a niveles similares o ligeramente superiores a los del Control corroboró la vía cometabólica del proceso característica de suelos sin historia de uso de atrazina. La capacidad de mineralización de atrazina por los horizontes subsuperficiales, donde tiende a acumularse este herbicida, en suelos con aplicaciones frecuentes de atrazina debiera ser considerada y estimulada como un mecanismo de bioremediación natural.
Palabras clave: Mineralización de atrazina; Subsuelo adaptado; Inactivación microbiana; Biorremediación.
Fecha de recepción: 01/06/07; fecha de aceptación: 20/12/07
INTRODUCTION
Atrazine (6-chloro-N2-ethyl-N4-isopropyl-1,3,5-triazine-2,4-diamine) is a herbicide that has been banned from the phytosanitary market in some countries due to its pollution risks. However, in other countries such as Argentina and Brazil, it is still widely used (SAGPyA, 2001; Associação Nacional de Defesa Vegetal, 2003). Although atrazine behaviour in soil has been studied for the last 50 years, risks of pollution remain difficult to predict. As with other pesticides, the spatial and temporal variability of biodegradation in soils is probably the main obstacle to adjusting and validating prediction models.
During the last ten years, a great amount of experimental evidence has confirmed that some microorganisms or consortia can develop specific enzymatic systems able to mineralize the triazine ring (Mandelbaum et al., 1995; Topp et al., 2000) Identification of such populations both in pure cultures and in soil has led to a distinction between "adapted" and "nonadapted" soils, according to their capacity to mineralize atrazine (Barriuso & Houot, 1996). Currently, the occurrence of this phenomenon is well described (Stolpe & Shea, 1995; Shapir et al., 2000; Ofstrofsky et al., 2002; Smith et al., 2005), as well as the repeated use of atrazine as an initiator of the development of the adapted population (Barriuso & Houot, 1996; Hang et al., 2003). This adapted soil condition is a characteristiCOf the first centimetres of soil reached by herbicide; nevertheless the occurrence of this process has also been described in some subsoil layers (Vanderheyden et al., 1997; Jenks et al. , 1998; Sparling et al., 1998; Hang et al., 2005).
Larsen et al., (2000) consider that two factors determine potential subsurface water contamination by a pesticide: its transformation in soil and its ability to be transported through the soil profile. Moreover, Topp et al. (1995) consider that the destruction of pesticides migrating in soils requires microorganisms with suitable degradative capabilities, appropriate environmental conditions for the microorganisms to function, and sufficient residence time for biodegradation to occur.
Extractable residues of atrazine tend to accumulate in the first 35-50 cm of the soil profile (Clay et al. , 2000; Asare et al., 2001). Thus, to characterize and quantify the atrazine mineralization capacity of the horizons next to the surface, particularly in soils that receive this herbicide yearly, is an economical and useful tool for selecting agricultural practices that stimulate subsurface biodegradation, for estimating the risk of transport to groundwater and for selecting bioremediation practices.
The aim of this work was to evaluate vertical variation of atrazine mineralization capacity in an adapted soil profile with atrazine application history. The occurrence of atrazine-degraders in the soil profile was evaluated by means of soil microbial activity inactivation. The behaviour of 14C-atrazine was studied in laboratory incubations by following the distribution of radioactivity between mineralized, extractable and non-extractable fractions. A nonadapted soil profile was included as a soil control without accelerated atrazine mineralization.
MATERIALS AND METHODS
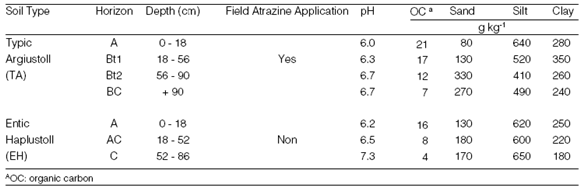
This work was carried out in two soil profiles clasified as Typic Argiustoll (TA) and Entic Haplustoll (EH) according to the Soil Taxonomy Classification System (Soil Survey Staff, 1998). Sampling was performed by horizons, avoiding mixing among them. After sampling, soils were air-dried and sieved to 2 mm. Particle size distribution into sand, silt and clay fractions was done by sieving and sedimentation. Soil pH was determined in water (soil/water, 1:1), and organic carbon (OC) content by dry combustion (Table 1). All techniques used are described in Sparks (1996).
Table 1. Location, use and some characteristics of the soils studied
Ring-U-labelled 14C-atrazine (radiopurity > 98%, specific activity: 7.77 107 Bq mmol-1) was purchased from Sigma. Isotopic dilution with unlabelled atrazine was made in water solution with a final concentration of 26.5 mg L-1 and 2.95 MBq L-1.
Microbial activity inactivation was carried out by means of dry heat sterilization. Ten grams of each horizon were heated at 105ºC for 24 h (T105 treatment). This treatment was made only once in order to keep a small percentage of survivors, <0.001% according to Lambert, 2003. All materials and solutions used in the T105 treatment were previously sterilized in autoclave.
The behaviour of 14C-atrazine was followed during laboratory incubations of 56 days at 28 ± 1°C in the dark in untreated (Control) and treated (T105) soils. Triplicate incubations were carried out in hermetically closed glass jars. One millilitre of 14C-atrazine solution was added to 10 g of dry soil. Soil water content was adjusted to water holding capacity with ultra-pure water (Milli-Q, Millipore) taking into account the volume of water added with the atrazine solution. 14C-CO2 released during the incubation was trapped in 2 mL of 2 M NaOH. Vials containing the NaOH were sampled and replaced after 28 and 56 days. Atrazine mineralization was determined by measuring 14C-CO2 in NaOH traps by liquid scintillation counting (Kontron Ins. Betamatic V Counter) using the Packard Ultima Gold XR as the scintillation cocktail.
After 28 and 56 days of incubation, soils with 14C-residues were extracted with 30 mL of methanol in glass centrifuge tubes. Tubes were agitated 12 h in a rotating agitator at room temperature, and then centrifuged for 15 min at 5000 g (Sorvall RC5B) and supernatants were recovered. This extraction procedure was repeated three times. All supernatants 14C content was measured on were pooled and their 1 mL by liquid scintillation counting as previously described.
After methanol extraction, soil pellets containing non-extractable 14C-atrazine residues (bound residues) were recovered and dried at 40ºC. Dry samples were ground in a mechanical agate mortar. Radioactivity was measured on three aliquots (100 to 200 mg) by liquid scintillation counting after combustion at 800ºC under oxygen flow in a Sampler Oxidizer (Packard) followed by 14C-CO2 trapping in 8 mL of Carbosorb E Packard, mixed with 12 mL of Permafluor E+Packard.
Methanol extracts were concentrated by evaporation near dryness under vacuum at 60ºC using a Rotavapor (Büchi); residue was dissolved in 0.5 mL of mobile phase used for HPLC analysis, and filtered through a 0.45 µm nylon filter. All concentrated extracts were then analysed by HPLCOn a Waters Nova-Pak C18 column (60Å, 4 µm, 4.6 x 250 mm) using a Waters appliance equipped with automatic injection and a 996-Photodiode array detector coupled on-line with a continuous flow radioactivity detector (Packard-Radioamatic Flo-One A-500). UltimaFloAP was used as a scintillation cocktail with a flow of 3 mL min-1. Mobile phase (1 mL min-1) was solvent A (water, sodium dodecyl sulphate, SDS, 5 mM, pH adjusted to 2.8 with HCl); and solvent B (water/methanol 1/9 v/v, SDS 5 mM, pH adjusted to 2.8 with HCl) as described by Loiseau et al., (2000) . Analytical standards of de-alkylated (deethyl-atrazine, DEA; deisopropyl-atrazine, DIA and deethyl-deisopropyl-atrazine, DEDIA) and hydroxylated (hydroxy-atrazine, HOAT; hydroxy-deethyl-atrazine, HODEA; hydroxy-deisopropyl-atrazine, HODIA and hydroxy-deethyl-deisopropyl-atrazine, HODEDIA) were used for metabolite identification in extracts.
The effect of microbial inactivation treatment (T105) on atrazine mineralization was estimated by the ratio:
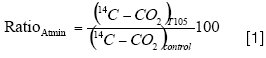
where(14C-CO2)control is the percentage of 14COf total initial applied that was released during Control treatment incubation, and (14C-CO2)control is the percentage of 14C of total initial applied that was released during T105 treatment incubation.
Analysis of variance (ANOVA) and the Tukey test were made between treatments for each horizon. The software used was InfoStat (2003).
RESULTS
The distribution of 14C-atrazine in mineralized (Atmin) extractable residues (ER) and non-extractable residues (NER) during 56 days of incubation, expressed as a percentage of initial applied radioactivity, is shown in Table 2. The ratio between the percentages of 14C-CO2 released after two treatments (T105, Control) according to [1] was calculated and is shown in Fig. 1 (a, b).
Table 2: Evolution of 14C-atrazine distribution into mineralized (Atmin), extractable (ER) and non-extractable residues (NER) during incubation in two soil profiles with (T105) and without (Control) microbial activity inactivation.
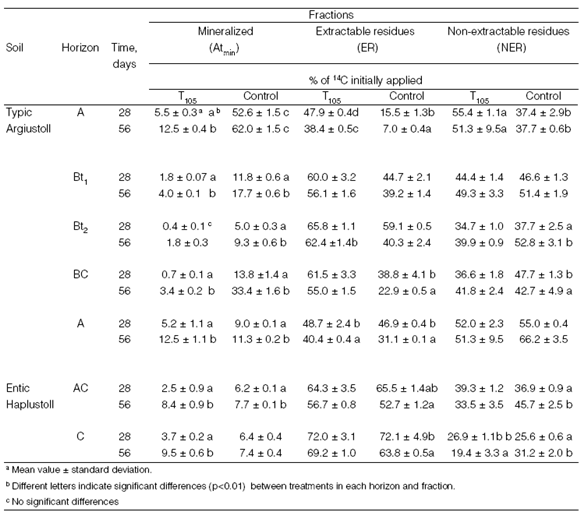
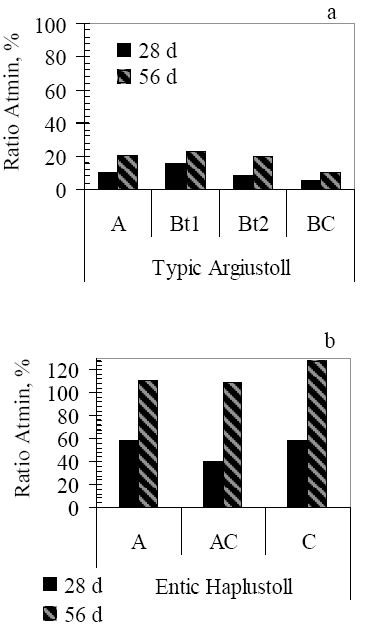
Figura 1. Atrazine mineralization decrease due to microbial activity inactivation after two incubation periods (28 and 56d) expressed as a ratio (Ratio Atmin) between atrazine mineralization (AT min) after T105 and Control treatments.![]() 100
100
In the adapted soil profile (TA), with previous atrazine application, the percentage of 14C-CO2 released after T105 treatment and 28 days incubation, was from 5 to 15% of the Control (i.e. 85-95% decrease). At the end of the incubation period (56 days), the percentage of 14C-CO2 released in T105 treatment compared to the Control was similar to those obtained after 28 days.
In the non-adapted soil profile (EH), without previous atrazine application, the percentage of 14C-CO2 released after T105 treatment and 28 days ranged from 40 to 58% (55-42% decrease); at the end of the incubation period, 14C-CO2 percentages were very close and even higher in T105 treatment compared to Control.
In the TA soil profile, ER were higher in T105 treatment than in the Control treatment (P<0.01) during all experiments. Table 3 shows the ER composition (atrazine and metabolites). The ER proportion decreased when the mineralization fraction increased. On the other hand, ER evolution did not show a behaviour pattern associated with soil depth but rather with atrazine mineralization. The atrazine proportion in ER after T105 treatment ranged between 33.6 and 57%, whereas that in the Control treatment ranged between 4.5 and 32.8%. The ER composition in the Control treatment, after 28 days incubation, showed high heterogeneity in relation to dealkylated and hydroxylated metabolites in both soil profiles. The metabolites were DEA, DIA, DEDIA, OHAT, OHDEA and traces of OHDIA, OHDEDIA and other more polar metabolites and represented up to 53% of the ER fraction. Higher metabolite diversification in ER is considered to be characteristiCOf systems with accelerated mineralization (Houot et al., 1998). After T105 treatment the diversification decreased, with DEA, OHAT and OHDEA being the most frequently detected metabolites.
Table 3. Composition of the extractable radioactivity (a Detected compounds).
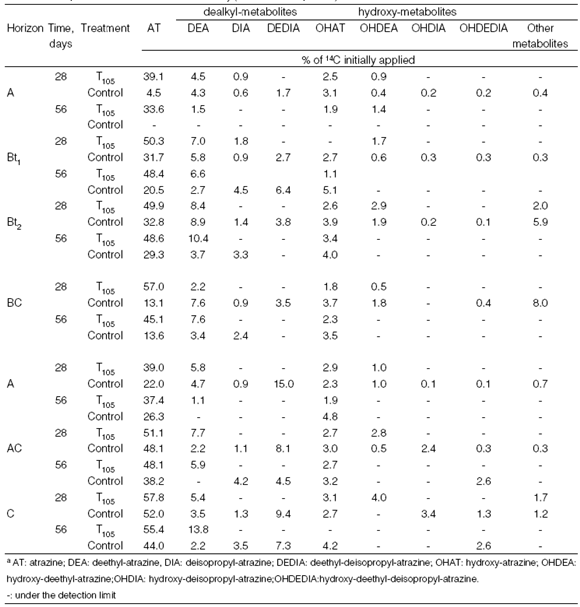
In the TA soil profile, significant differences (P<0.05) were detected in the atrazine proportion between Control and T105 treatments. Whereas in Control soils atrazine ranged from 4.5 to 32.8% (28 d) and from "not detected" to 29.3% (56 d), in the T105 treatment the range was 39 to 54% (28 d) and 33.6 to 48.6% (56 d). The atrazine proportion in the ER fraction was an average of 47% (± 24) in the TA Control and increased to 83% (±5) in the T105 treatment. A strong decrease in the atrazine proportion in ER residues was noted in adapted soils (Barriuso and Houot, 1996).
In the non-adapted soil profile (EH), where atrazine degradation has been demonstrated to be carried out by a cometabolic population (Bollag and Liu, 1990), ER increased with depth and decreased with incubation time. This behaviour is associated to the ER fraction being a bioavailable fraction (Carter, 2000) and to total soil microbial activity decreasing with depth (Stolpe and Shea, 1995). ER composition changes were small between treatments. Atrazine was up to 70% (± 12) of the ER fraction in the Control treatment and 83% (±5) in the T105 treatment, i.e. the metabolite fraction in non-adapted soil represented only 17% on average of the ER fraction. NER evolution (Table 2) in the TA Control treatment was related to the evolution of atrazine mineralization. While horizons with the highest atrazine mineralization (A and BC) showed the lowest NER percentages, in Bt 1 and Bt 2 horizons NER increased with incubation time.
In non-adapted soil (EH), in the Control treatment NER decreased with depth and increased with the incubation time (P<0.01), while in the T105 treatment, NER decreased with depth and incubation time.
DISCUSSION
Occurrence of an adapted microflora in the upper horizon of the TA profile has been confirmed in a previous work (Hang et al., 2003). In the Control treatment, atrazine mineralization was ? 50% in a period less than 28 d during laboratory incubations. It has been demonstrated that previous atrazine applications stimulate the development of a soil microbial community capable of using atrazine as a source of C, N, and energy (Barriuso & Houot, 1996) and this is called accelerated atrazine mineralization. The significant decrease in 14C-CO2 released in the T105 treatment compared to the Control treatment (85-95%) during all the experiment period showed that atrazine degraders were eliminated with T105 treatment and that this specific microbial population was not recovered during a second month of incubation. Atrazine mineralization in subsurface horizons of TA profile showed similar behaviour as in the A horizon. The proportion of 14C-CO2 reduction after the T105 treatment compared to the Control was similar to that observed in the A horizon (85-95% decrease). This behaviour suggests that microbial population responsible for atrazine mineralization in subsurface horizons is similar to that in the upper horizon. Atrazine degraders have been reported in some specific subsoil horizons (Vanderheyden et al., 1997; Jenks et al., 1998; Di et al., 1998). Kördell et al.(1995), working with chlorotoluron and simazine, pointed out that within one soil profile an identical metabolism occurs along different horizons. Our results suggest there was an atrazine degradation pattern in the adapted soil profile due to specific microflora being distributed throughout the adapted soil profile.
On the other hand, there was a similar behaviour between the adapted A-horizon and the other subsuperficial horizons in relation to ER composition changes after T105 treatment. The higher proportion of ER in the T105 treatment than in the Control treatment (P<0.01) during all the experiment is associated to microbial inhibition. However, significant differences (P<0.05) in atrazine proportion between Control and T105 treatments were detected. Whereas in Control soils atrazine ranged from 4.5 to 32.8% (28 d) and from "not detected" to 29.3% (56 d), in the T105 treatment the range was 39 to 54% (28 d) and 33.6 to 48.6% (56 d). The atrazine proportion in the ER fraction was on average 47% (± 24) in the TA Control and increased to 83% (±5) in the T105 treatment. A strong decrease in atrazine proportions in ER residues has been pointed out for adapted soils (Barriuso & Houot, 1996). However, future studies will be made to identify atrazine-degraders in the subsoil TA profile.
The lowest atrazine mineralization in the Control treatment was observed in the argillic horizon ( Bt1+ Bt2). The intensity of atrazine mineralization depends on the number of degraders (Sparling et al., 1998). Preferential flux could partially explain atrazine behaviour in the Bt horizon. The cracks developed during wetting-drying cycles favour the fast circulation of soil solution. It is considered that the degradation of pesticides migrating from soils requires microorganisms with suitable degradative capabilities, appropriate environmental conditions for microorganisms to function and sufficient residence time for biodegradation to occur (Topp et al., 1995). Preferential flux is a very frequent phenomenon in clayey horizons (Flury & Flühler, 1994). Bundt et al., (2001) point out that an important characteristic of preferential flow is that solutes bypass a large part of the soil matrix. These processes could partly explain the decrease in atrazine mineralization in the argillic horizon and its concomitant increase in the BC horizon.
Atrazine evolution in non-adapted soil (EH), where we detected a similar behaviour patttern of atrazine evolution to that of total microbial activity (Hang et al., 2003) corroborated differences in atrazine mineralization capacities between the two soil profiles. Both soil profiles showed differences in edaphic properties, and so it may be considered that differences in atrazine mineralization capacity could be explained by these soil properties. However, it has been demonstrated that there is no direct link between rates of mineralization and any soil properties, including the size of the total microbial biomass (Yassir et al., 1999).
After T105 treatment, a high recovery of atrazine mineralization capacity was observed during the second month of the incubation period in all horizons. This suggests that atrazine mineralization was carried out in this soil by a microbial population that recovered in the second month of incubation. Several works have demonstrated that in soils without prior applications of atrazine, mineralization was carried out by a cometabolic, non-specific and competitive population (Bollag & Liu, 1990; Barriuso & Houot, 1996).
The NER of the Control treatment decreased with depth and increased with incubation time (P<0.01), while in the T105 treatment, NER decreased with depth and incubation time. Benoit et al. (1999) have pointed out that the herbicide fraction retained could be biologically degraded. On the other hand, the stimulus of atrazine mineralization observed in T105 after 56 days in this profile could be explained, in part, by a high extractable atrazine proportion or bioavailable fraction (Johnson et al., 1999) remaining during the first incubation period (28d). Microbial recovery caused an increase of atrazine mineralization and a decrease of NER formation, according to Nam & Alexander (2001), who determined an inverse relation between the proportion of non-extractable residues of a compound and its mineralization rate.
CONCLUSIONS
Atrazine evolution was evaluated in two soil profiles and the results suggested that each soil profile had similar processes along different horizons for atrazine mineralization to occur.
Our results suggest that atrazine-degraders were distributed through all the adapted soil profile and were responsible for atrazine mineralization in subsurface horizons.
In soils with high and frequent atrazine application, the atrazine mineralization capacity of the subsurface horizons should be considered and stimulated as a mechanism of natural bioremediation.
ACKNOWLEDGMENTS
This work received a grant from the "Programa de Cooperación Franco-Argentino ECOS Sud-SETCYP, A00U01".
REFERENCES
1. Asare D.K.; T.W. Sammis., D. Smeal., H. Zhang and D.O. Sitze, 2001. Modeling an irrigation management strategy for minimizing the leaching of atrazine. Agr. Water Manage. 48:225-238. [ Links ]
2. Associação Nacional de Defesa Vegetal, 2003. [Online]. [In Portuguese.] Available at: http://www.sindag.com.br/new/setor/interna.php?cod=12. Verified: (03/06/04). [ Links ]
3. Barriuso, E. and S. Houot, 1996. Rapid mineralization of the s-triazine ring of atrazine in soils in relation to soil management. Soil Biol. Biochem. 28:1341-1348. [ Links ]
4. Benoit, P.; E. Barriuso and G. Soulas, 1999. Degradation of 2,4 D; and 2,4 dichlorophenol and 4-chlorophenol in soil after sorption on humified and nonhumified organic matter. J. Environ. Qual., 28:1127-1135. [ Links ]
5. Bollag, J.M. and S.Y. Liu, 1990. Biological transformation processes of pesticides. In: Cheng H.H (Ed.), Pesticides in the soil environment: Processes, impact, and modeling. SSSA Book Ser. 2, Madison. 169-211 pp. [ Links ]
6. Bundt, M.; F. Widmer., M. Pesaro., J. Zeyer and P. Blaser, 2001. Preferential low paths: Biological "hot spots" in soils. Soil Biol. Biochem. 33:729-738. [ Links ]
7. Carter, A., 2000. Herbicide movement, in soils: Principles, pathways, and processes. Weed Research, 40:113:122. [ Links ]
8. Clay, S.A.; R.H. Dowdy., J.A. Lamb., J.L. Anderson., B. Lowery., R.E. Knighton and D.E. Clay, 2000. Herbicide movement and dissipation at four Midwestern sites. J. Environ. Sci. Health, B35 (3), 259-278. [ Links ]
9. Di, H.J.; A.G. Aylmore and R.S. Kookana, 1998. Degradation rates of eight pesticides in surface and subsurface soils under laboratory and field conditions. Soil Sci. 163:404-411. [ Links ]
10. Flury, M. and H. Flühler, 1994. Susceptibility of soils to preferential flow of water: a field study.Water Resources Research 30:1945-1954. [ Links ]
11. Hang, S.; E. Barriuso and S. Houot, 2003. Behavior of 14C-Atrazine in Topsoils of the Argentinean Semi-arid Pampa under Different Cropping Managements. J. Environ. Qual., 32: 2216-2222. [ Links ]
12. Hang, S.; E. Barriuso and S. Houot, 2005. Atrazine behavior in the different pedological horizons of two Argentinean non-till soil profiles. Weed Research, 45:130-139. [ Links ]
13. Hang, S.; S. Houot and E.Barriuso, 2007. Mineralization of 14C-atrazine in a Typic Haplustoll in three winter weed control managements. Soil Till. Research, 96: 234-242 [ Links ]
14. Houot, S.; E. Barriuso and V. Bergheaud, 1998. Modifications to atrazine degradation pathways in a loamy soil after addition of organic amendments. Soil Biol. Biochem. 30:2147-2157. [ Links ]
15. InfoStat. 2003. InfoStat v.1.6,p. Grupo Infostat. FCA. Universidad Nacional de Córdoba, Argentina. [ Links ]
16. Jenks, B.; F. Roeth and A. Martin, 1998. Influence of surface and subsurface soil properties on atrazine sorption and degradation. Weed Sci. 46:132-138. [ Links ]
17. Johnson, S.; J. Herman., A. Mills and G. Hornberger, 1999. Bioavailability and desorption characteristics of aged, non-extractable atrazine in soil. Environ. Tox. Chem. 18:1747-1754. [ Links ]
18. Kördel W.; U. Wahle., H. Knoche and K. Hund, 1995. Degradation capacities of chlorotoluron and simazine in subsoil horizons. The Sci. Total Environ.171:43-50. [ Links ]
19. Larsen, L.; S. Sorensen and J. Aamand, 2000. Mecoprop, isoproturon, and atrazine in and above a sandy aquifer: Vertical Distribution of mineralization potential. Environ. Sci. Technol. 34:2426-2430. [ Links ]
20. Lambert, R.J.W., 2003. A model for the thermal inactivation of micro-organisms. J. Appl. Microbiol., 95:500-507. [ Links ]
21. Loiseau, L. ; E. Barriuso., Y. Zegouagh., C. Largeau and A. Mariotti, 2000. Release of the atrazine non-extractable (bound) residues of two soils using degradative techniques. Agronomie 20:513-524. [ Links ]
22. Mandelbaum, R.M.; L.P. Wackett and D.L. Allen, 1995. Isolation and characterization of a Pseudomonas sp. that mineralizes the s-triazine herbicide atrazine. Appl. Environ. Microbiol. 61:1451-1457. [ Links ]
23. Nam, K. and M. Alexander, 2001. Relationship between biodegradation rate and percentage of a compound that becomes sequestered in soil. Soil Biol.Biochem. 33:787-792. [ Links ]
24. Ostrofsky, E.; J. Robinson., S. Traina and O. Tuovinen, 2002. Analysis of atrazine-degrading microbial communities in soils using most-probable-number enumeration, DNA hybridization, and inhibitors. Soil Biol. Biochem. 34:1449-1459. [ Links ]
25. SAGPyA, 2001. Estimaciones Agrícolas. Bs. As. Argentina. Available at: http://www.sagpya.mecon.gov.ar/scripts/0-2/icereal.idc. Verified: (03/06/04). [ Links ]
26. Shapir, N.; S. Goux., R. Mandelbaum and L. Pussemier, 2000. The potential of soil to mineralize atrazine as predicted by MCH-PCR followed by nested PCR. Can. J. Microbiol. 46:425-432. [ Links ]
27. Smith, D.; S. Alvey and D. Crowley, 2005. Cooperative catabolic pathways within an atrazine-degrading enrichment culture isolated from soil. FEMS Microbiology Ecology. 53:493-495. [ Links ]
28. Soil Survey Staff, 1998. Keys to Soil Taxonomy 8th ed. USDA Natural Resour. Conserv. Serv., Washington, DC. [ Links ]
29. Sparks, D., (Editor), 1996. Methods of Soil Analysis. Part 3- Chemical Methods. ASA, SSSA, CSSA, Madison, WI, USA. [ Links ]
30. Sparling, M.; R. Dragten., J. Aislabie and R. Fraser, 1998. Atrazine mineralization in New Zealand topsoils and subsoils: influence of edaphic factors and numbers of atrazine-degrading microbes. Aust. J. Soil Res. 36:557-570. [ Links ]
31. Stolpe, N.B. and P.J. Shea, 1995. Alachlor and atrazine degradation in a Nebraska soil and underlying sediments. Soil Sci. 160:359-370. [ Links ]
32. Topp, E.; D. Gutzman., B. Bourgoin., J. Millette and D. Gamble, 1995. Rapid Mineralization of the Herbicide Atrazine in Alluvial Sediments and Enrichment Cultures. Environ. Tox. Chem. 14:743-747. [ Links ]
33. Topp, E.; H. Zhu., S.M. Nour., S. Houot., M. Lewis and D. Cuppels, 2000. Characterization of an atrazine degrading Pseudominobacter sp. isolated from Canadian and French agricultural soils. Appl. Environ. Microbiol. 66:2773-2782. [ Links ]
34. Vanderheyden, V.; P. Debongnir and L. Pussemier, 1997. Accelerated degradation and mineralization of atrazine in surface and subsurface soil materials. Pestic. Sci. 49:237-242. [ Links ]
35. Yassir, A.; Lagacherie, B., Houot, S., Soulas, G., 1999. Microbial aspects of atrazine biodegradation in relation to history of soil treatment. Pestic. Sci. 55, 799-809. [ Links ]














